Abstract
Purpose:
Hippocampal sparing whole-brain radiotherapy (HS-WBRT) showed significantly lower long-term side effects compared to standard WBRT. Aim of this study is to describe a HS-WBRT real-world monoinstitutional experience within a retrospective cohort.
Methods:
Patients who completed HS-WBRT course, with Karnofsky Performance Status ⩾ 60 and radiological diagnosis of brain metastases (BMs) were enrolled. Treatment was performed using helical Tomotherapy scheduled in 30 Gy in 10 or 12 fractions or 25 Gy in 10 fractions. Oncological outcomes were clinically and radiologically assessed every three months. Toxicity was graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events 4.3.
Results:
One hundred and nineteen patients from 2016 to 2020 met inclusion criteria; after a median follow-up of 18 months, 29 patients were alive; 6- and 12-months overall survival rates were 66% and 41%, respectively. HS-WBRT response was assessed for 72 patients. Median time to any progression and intracranial failure (IF) was 4.5 and 13.7 months, respectively. The 6- and 12-month IF rates were 85% and 57%. Among 40 patients (34%) who experienced IF, 17 (42%) were oligometastatic, 23 (58%) polymetastatic and 15/40 developed IF within the hippocampi avoidance zone. No grade (G) ⩾ 2 acute toxicities were reported and one G2 (dizziness) late toxicity was described.
Conclusions:
HS-WBRT is well tolerated, and despite the hippocampal sparing region, the oncological control is satisfying. Further investigation is warranted to find patients who could most benefit from a HS-WBRT approach.
Introduction
Approximately 20% to 40% of all cancer patients develop brain metastases (BMs) and incidence rates are still increasing, primarily because of improved systemic options.1,2 In fact, oncological patients have an ever-increasing life expectancy, thanks to the introduction of more effective systemic treatments, such as biological-, target- and immuno-therapies. Therefore, it becomes important to improve strategies to achieve good local intracranial control while preserving the patient’s quality of life (QoL) by minimizing the onset of neurological deficits.
Regardless of the consolidative role assumed by stereotactic ablative radiation therapy (SABR) for limited BMs (usually less than 10 localizations), whole-brain radiotherapy (WBRT) still represents the standard of care in case of multiple BMs, due to its wide availability and effectiveness, both in neurological symptoms control and survival improvement.3,4
However, the WBRT approach is not free from severe side effects, especially in the long-term, such as decline in memory, poor attention concentration, visual spatial difficulties, complication in executive planning, poor fine motor control, and cognitive impairment.5 –8
Consequently, in recent years, clinical research has directed efforts to reduce RT-related neurotoxicities. Several preclinical and clinical investigations showed that neurological long-term adverse events are mainly related to radiation-induced cell damage to the hippocampus,9,10 a bilateral and symmetrical brain structure, containing the neuronal stem cells, fundamental for memory preservation and learning. Thus, reducing radiation dose to the hippocampus during WBRT, with the so-called hippocampal sparing (HS)-WBRT technique, has been proposed as a valuable strategy to mitigate neurocognitive impairment.11–15
In fact, improvements in modern precision RT techniques, such as intensity-modulated RT (IMRT), volumetric-modulated arc therapy (VMAT), and Helical Tomotherapy allow the administering of WBRT while sparing the hippocampi. Thanks to this procedure, it is possible to reduce the mean dose to the hippocampus by at least 80%, maintaining an acceptable coverage and dose homogeneity to the remaining parenchyma.1,2
A series of trials attempted to establish the role of HS-WBRT as the standard approach to preserve neurological functions without jeopardizing oncological patients’ outcomes.16–23
This study describes a single-center experience with HS-WBRT reporting the oncological outcomes of a retrospective series of patients harboring BMs. The aim of the present study is to report the feasibility and efficacy of the treatment with a particular focus on local disease control, and peri-hippocampal pattern of recurrence, adjusting for other oncological and physical variabilities.
Materials and methods
Patient selection
A retrospective series of patients treated with HS-WBRT at the Department of Radiation Oncology of the European Institute of Oncology IRCCS (IEO) were considered for the present analysis.
Eligibility criteria were the following: (1) age ⩾ 18 years, (2) any confirmed cancer histology, (3) radiological (computed tomography (CT) or magnetic resonance imaging (MRI) scan) diagnosis of BMs, (4) absence of peri and/or hippocampal BMs, (5) Karnofsky Performance Status (KPS) >=60, (6) available MRI, (7) completed RT schedule, (8) written informed consent signed for research purpose. Small Cell Lung Cancer (SCLC) patients undergoing prophylactic cranial irradiation (PCI) were not included in this study.
This work was approved by the Ethical Committee of Istituto Europeo di Oncologia (IEO) IRCSS and Centro Cardiologico Monzino IRCSS (notification nr. UID 2783).
Treatment planning and procedure
Target and organs at risk (OARs) were contoured on simulation CT (2.5 mm slice thickness), using a co-registered MRI image set to better identify the hippocampi. Planning target volume (PTV) was defined as the whole brain, with the exclusion of an avoidance area, a planning risk volume (PRV) represented by an isotropic expansion of 5 mm of the hippocampi. When simultaneous integrated boost (SIB) was indicated, its PTV was identified as the gross tumor volume (GTV) + 3 mm. OARs included hippocampi, lens, eyes, optic nerves, chiasm, cochleae, brainstem, and spinal cord; left and right hippocampi were contoured following Radiation Therapy Oncology Group (RTOG) atlas indications. 24 A representative IMRT plan and dose-volume histogram (DVH) for a patient treated with HS-WBRT is shown in Figure 1. All patients included in this study were treated with Helical Tomotherapy® (Accuray Inc., Sunnyvale, CA) and treatment planning was performed using Tomotherapy Planning System®. Plan optimization goals are listed in Table 1. Daily check of the setup accuracy was performed with the acquisition of a megavoltage CT.
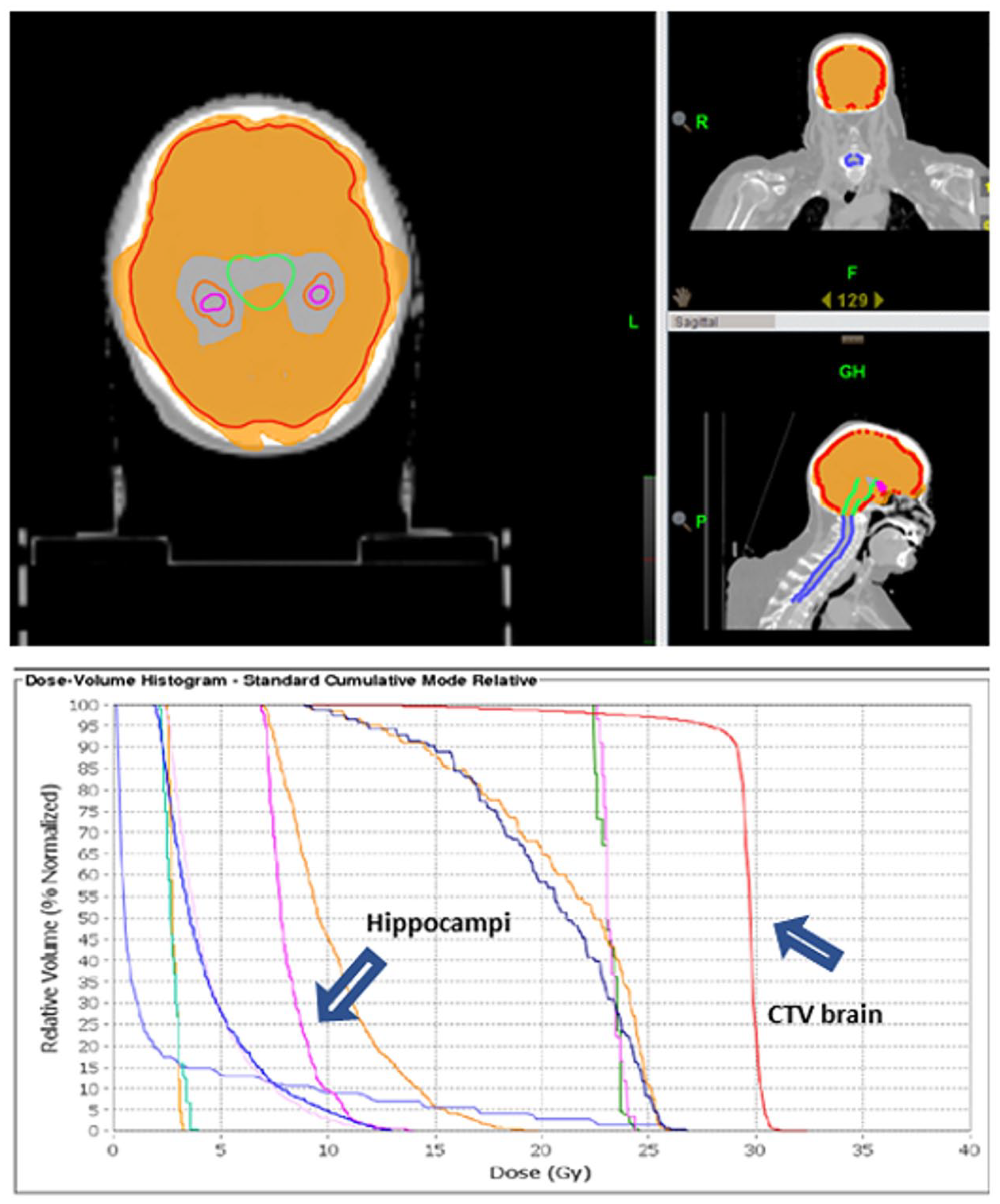
Example of dose distribution of a hippocampal sparing-whole brain radiotherapy plan (top) and the resulting dose-volume histograms (bottom).
Plan optimization objectives.

Dmax, maximum dose; Dmean, average dose; Dmedian, median dose, OARs, Organs at Risk; PTV, Planning Target Volume.
Response assessment and follow up procedure
Radiological response after RT was evaluated every three months after treatment by MRI and classified according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 25 An "in-field" relapse was considered as the finding of BMs on radiologic imaging within the treated brain that was not spared by RT in the treatment plan, i.e., the entire brain except the avoidance area of the PRV of the hippocampi.
Time-to-progression (progression-free survival, PFS) was defined as the time from the end of RT until the first occurrence of any of the following events: encephalic and/or systemic progression or death. Intracranial failure was defined as the time from HS-WBRT and the first brain relapse. Overall survival (OS) was calculated from the end of RT until the date of death from any cause, or the last contact date if alive. All patients alive or free of progression at the last follow-up date were considered right censored.
At the eventual subsequent intracranial progression, patients were divided into two groups: oligometastatic (⩽ 5 lesions) or polymetastatic; the relapse site was reported as close to hippocampal avoidance PRV or not.
HS-WBRT related toxicities were evaluated using Common Terminology Criteria for Adverse Events (CTCAE) version 4.03. 26
Statistical analysis
Descriptive statistics of patients' demographic and primary tumor features, as well as information about the encephalic metastasis status and treatments received before and during RT, was collected. OS and PFS curves were drawn using the Kaplan-Meier method and the log-rank test was used to assess survival differences among groups. Multivariate Cox proportional hazards regression models were used to assess the association between prognostic factors and clinical-pathological characteristics with OS and PFS adjusting for confounders. Fully adjusted risk estimates were presented as hazard ratio (HR) with 95% confidence intervals (CI).
Cumulative incidence of intracranial failure was estimated according to Kalbfleisch and Prentice, considering competing events such as systemic progression disease and death before re-evaluation. LR cumulative incidence was assessed from the end of RT to the day of the first recorded event, if any or to the last follow update. All patients alive at the last follow-up date were considered right censored. The Gray’s test was used to test the difference of the cumulative incidence between prognostic factors. Multivariate Fine and Gray Model was used to assess the association between prognostic factors and clinic-pathological characteristics with intracranial failure adjusting for confounders. The associations were quantified by HR with 95% CI.
Results
Study population
Between February 2016 and July 2020, 119 consecutive patients fulfilled the inclusion criteria. Patients’ characteristics are listed in Table 2. Median age at RT was 57.8 years (interquartile range (IQR) 51-66). The most represented primary cancer sites were breast (n = 63, 53%) and non-small cell lung cancer (NSCLC) (n = 46, 39%). Fifty-seven patients (49%) were diagnosed with synchronous metastases.
Patients’ baseline characteristics.
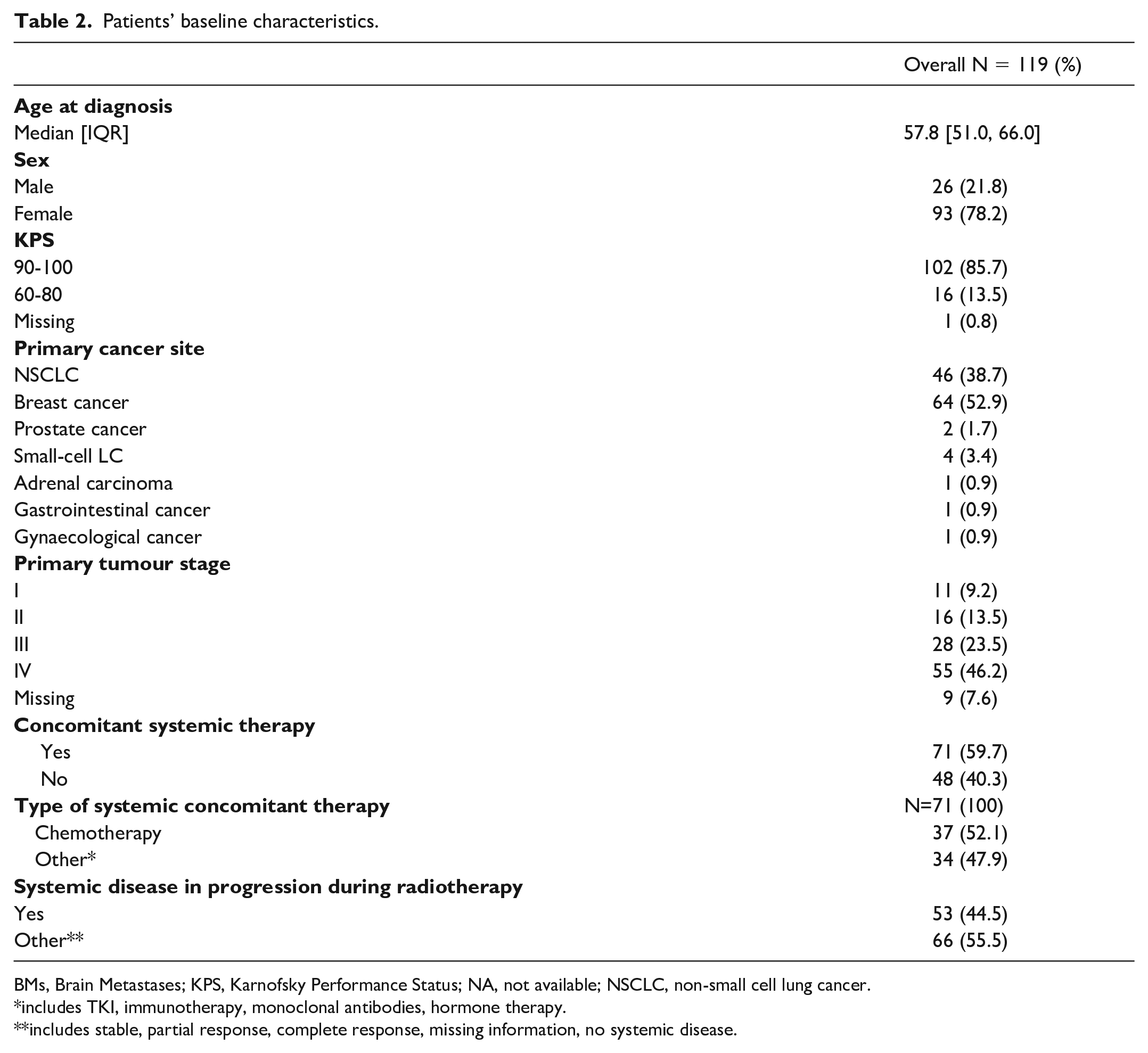
BMs, Brain Metastases; KPS, Karnofsky Performance Status; NA, not available; NSCLC, non-small cell lung cancer.
includes TKI, immunotherapy, monoclonal antibodies, hormone therapy.
includes stable, partial response, complete response, missing information, no systemic disease.
Treatment
All constraints were respected, and doses prescribed for hippocampi avoidance are listed in Table 3. The majority of patients received 30 Gy, 104 (87%) in 10 fractions, and 13 (11%) in 12 fractions. Two patients underwent 25 Gy in 10 fractions. Nine patients (8%) were treated with SIB at 35 Gy dose.
Radiotherapy treatment characteristics.
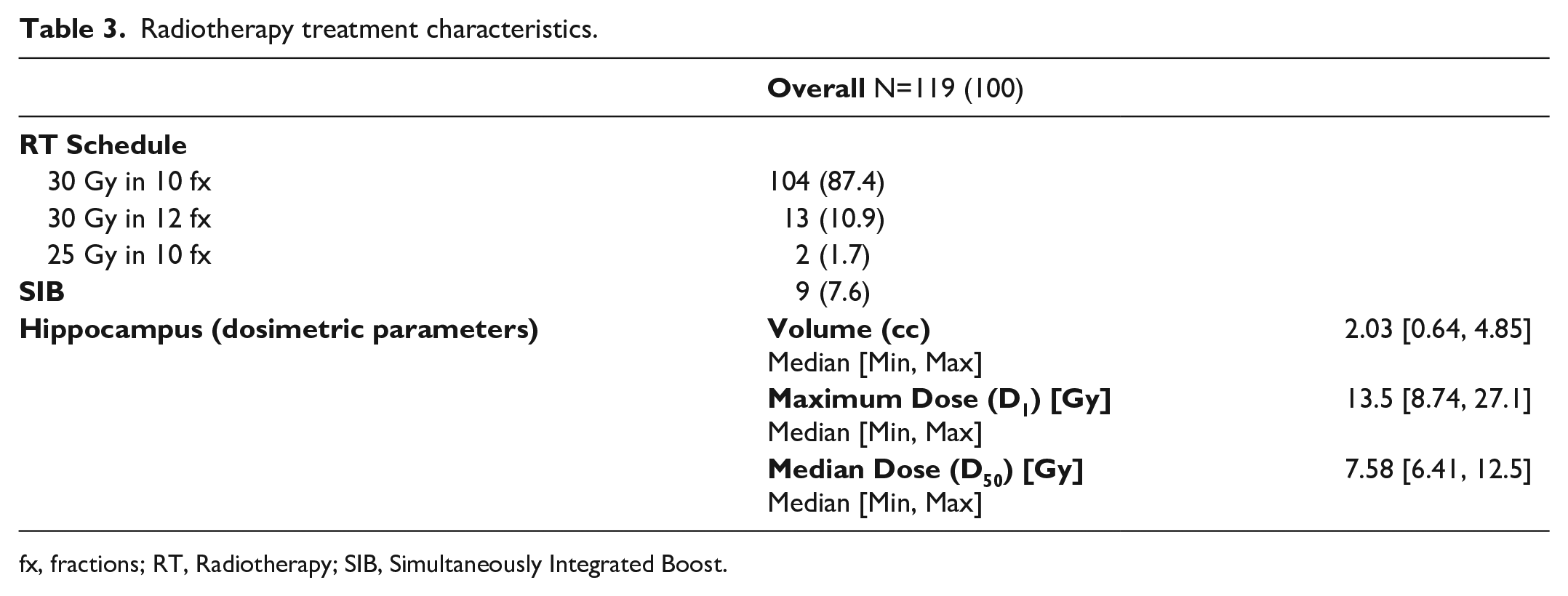
fx, fractions; RT, Radiotherapy; SIB, Simultaneously Integrated Boost.
Seventy-one patients (60%) received concomitant systemic therapy, 31% (n=37) of them received chemotherapy and the remaining 69% (n=34) target- or immuno-therapy.
Oncological outcomes
The median follow-up for patients alive at the time of the study was 18 months [95% CI: 12.2-30.9]. At the time of analysis, 29 patients (24%) were alive with clinical evidence of disease and 90 (76%) died of disease. The median OS was 9.7 months [95% CI: 7.5-11.5]. The 6- and 12-months OS rates were 66% (95% CI: 58-75) and 41% (95% CI: 33-51), respectively.
Analysis of HS-WBRT treatment response (Figure 2) was performed for 72 out of 119 patients (24 died before restaging and 23 were lost at follow-up; a summary of the cohort composition is provided in Supplementary materials S1). Median time to any progression was 4.5 months (95% CI: 3.3-5.6) with a median time to intracranial failure of 13.7 months (95% CI: 9.6-15.4). The 6- and 12-months intracranial PFS rates were 85% and 57%, respectively.
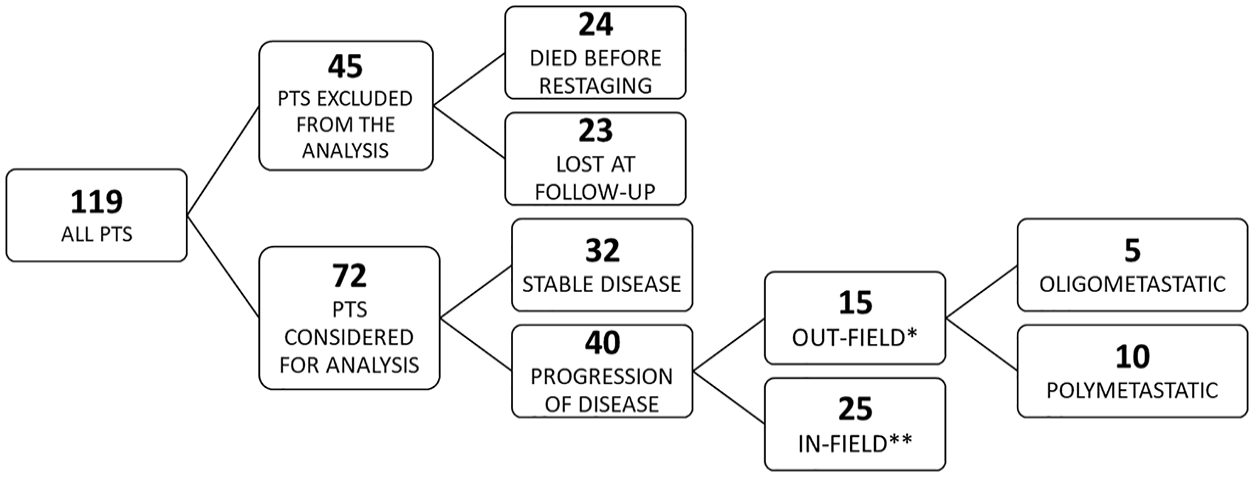
Summary of the cohort composition.
At the last follow-up, stable disease (SD) and progression of disease (PD) were observed in 32 (44%) and 40 (56%) patients, respectively. Among the 72 censored patients, intracranial failure was observed in 14 (20%) patients, whereas systemic progression was observed in 32 patients (44%); 26 patients (36%) had both.
Among patients with intracranial failure (n = 40), 17 (42%) were oligometastatic, while 23 (58%) experienced polymetastatic recurrence; 15 out of 40 developed intracranial failure within the hippocampi avoidance zone.
Considering only the oligorecurrences (n=17), in five cases hippocampi were involved, 11 cases were in-field PD, and one case was not reported.
At univariate and multivariate analysis, none of the considered clinical variables, namely age, KPS, concomitant therapies and use of chemo- vs target- or immuno-therapy, resulted significantly associated with intracranial failure.
As expected, systemic PD during RT course was found significantly associated with OS in multivariate analysis, with a nearly threefold increased risk of death (HR = 2.8, 95% CI: 1.78, 4.41, P < .0001, Figure 3). For patients undergoing concomitant systemic therapy during HS-WBRT, the type of administered agent (chemo vs others) was found to be associated with OS (P < .001, n = 71, Table 4). No prognostic factors were found to be significantly associated with OS, PFS and intracranial failure in patients not receiving concomitant therapy (n = 48, Table 5).
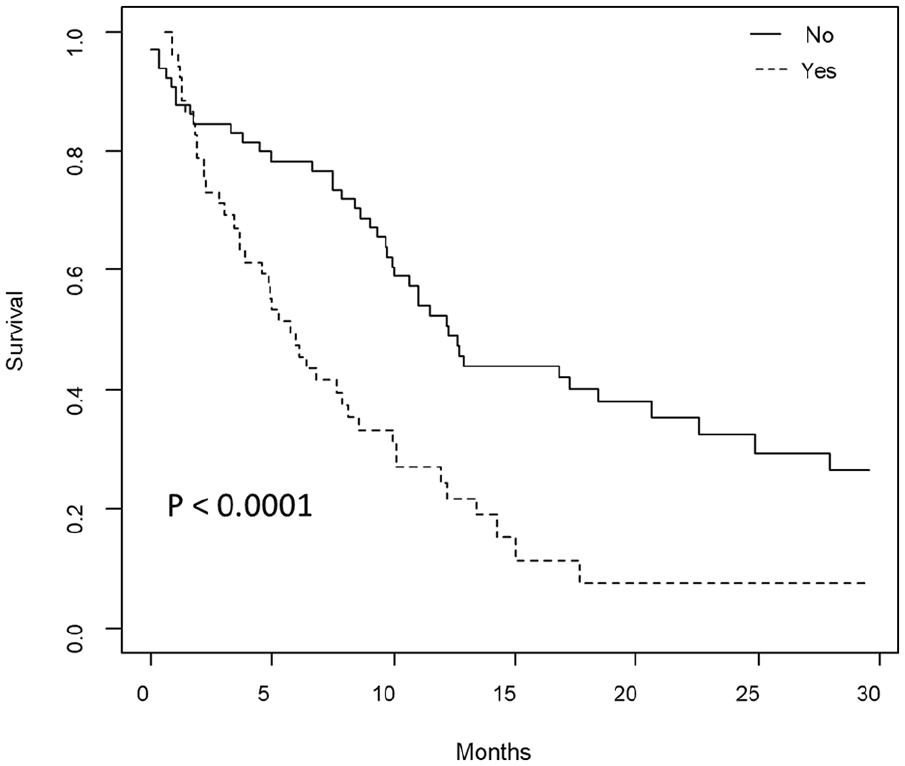
Overall survival stratified by systemic disease status (yes vs no) at the start of radiotherapy treatment.
Multivariate Cox models including patients with concomitant systemic treatment (n=71).
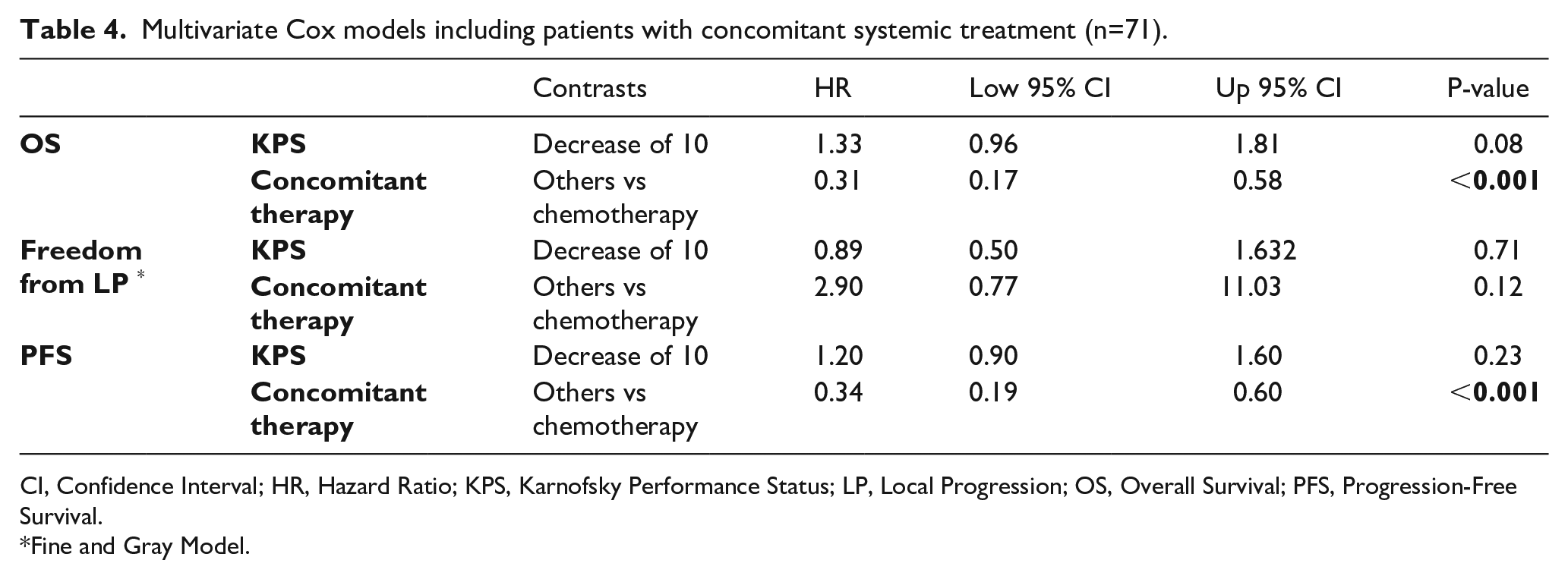
CI, Confidence Interval; HR, Hazard Ratio; KPS, Karnofsky Performance Status; LP, Local Progression; OS, Overall Survival; PFS, Progression-Free Survival.
Fine and Gray Model.
Multivariate Cox models including patients with no concomitant systemic treatment (n=48).
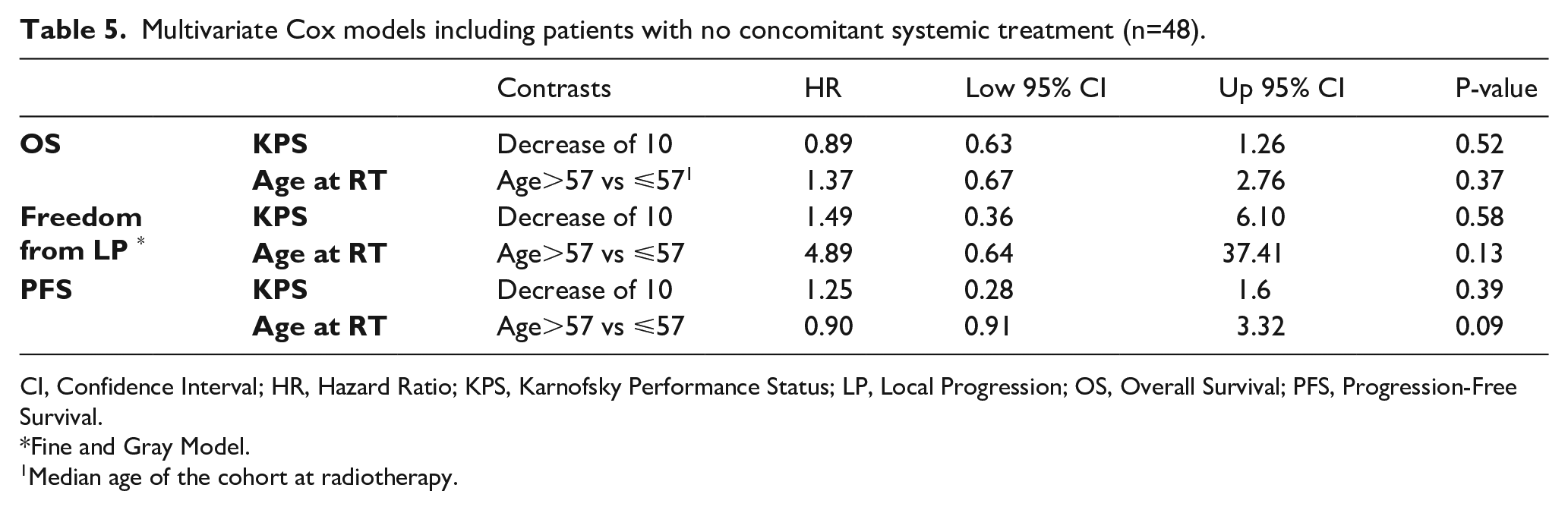
CI, Confidence Interval; HR, Hazard Ratio; KPS, Karnofsky Performance Status; LP, Local Progression; OS, Overall Survival; PFS, Progression-Free Survival.
Fine and Gray Model.
Median age of the cohort at radiotherapy.
Toxicity assessment
All patients received a pre-treatment with steroids to avoid acute HS-WBRT-related side effects. Acute grade (G)1 toxicities including vomiting, nausea, and cephalea were observed in 10 patients; no acute G ⩾ 2 toxicities were reported and 1 late G2 (dizziness) toxicity was described.
Discussion and conclusion
To date, the SABR represents the elective treatment for the treatment of a limited number of BMs, while the optimal therapeutic strategy for multiple BMs (>10) still represents an open question, as the balance between local control and long-term side effects is still being sought. Since the advent of new drugs (i.e. tyrosine kinase inhibitor (TKI) or immunotherapy) has extended the median life-expectancy, it becomes crucial to minimize late neurocognitive side effects, especially in long-surviving patients. Optimal OS rates for BMs treated with SABR were reported for a number of lesions up to 20. Nevertheless, local recurrence rates occur in up to 64% of the cases. 27 Based on these premises, WBRT remains the elective treatment in patients with more than 10 lesions.
While hippocampal metastases account for approximately 3% of all BMs, 14 a WBRT with hippocampal area sparing could both prevent overall cerebral atrophy and control the microscopic disease.
To the best of our knowledge, the present work is one of the largest studies reporting oncological outcomes on a cohort of 119 patients with BMs treated with HS-WBRT ± SIB. After a median follow-up of 18 months, it was reported a median OS of 9.7 months with a median intracranial failure time of 13.7 months.
None of clinical and tumoral variables was found associated with local control. Our results in terms of OS are in line with the published data in similar cohorts (Table 6). Gondi et al. 23 in 2014 in a cohort of 100 patients reported a median OS of 6.8 months, while a recent study by Popp et al. 28 reported a median OS of 9.9 months among 62 patients treated with a 42 Gy or 51 Gy SIB. On the other hand, a recent small cohort study (n = 32) by Lebow et al., 16 found a longer median OS of up to 19.6 months in patients treated with HS-WBRT and a 37.5 Gy SIB on the dominant lesion. Similarly, Oehlke et al. 17 in 2015 reported a median OS of 17.9 months in 20 patients undergoing HS-WBRT plus a 51 Gy SIB.
Summary of the studies on hippocampal sparing-whole brain radiotherapy.
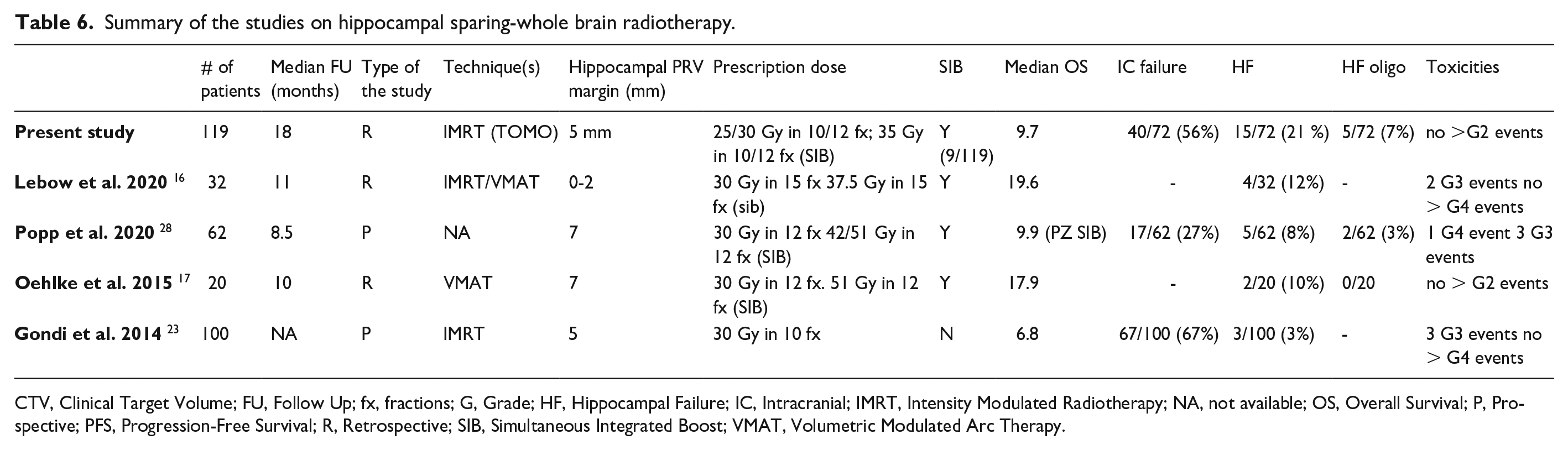
CTV, Clinical Target Volume; FU, Follow Up; fx, fractions; G, Grade; HF, Hippocampal Failure; IC, Intracranial; IMRT, Intensity Modulated Radiotherapy; NA, not available; OS, Overall Survival; P, Prospective; PFS, Progression-Free Survival; R, Retrospective; SIB, Simultaneous Integrated Boost; VMAT, Volumetric Modulated Arc Therapy.
The above-mentioned higher OS outcomes could be explained by several reasons: the small cohorts investigated, a > 35 Gy SIB performed on all included patients, whereas in the present study only nine out of 119 patients underwent a SIB, and the fact that our series includes negatively selected patients. In fact, as institutional policy, we propose SABR for all patients with < 6-10 BMs, with good performance status and favorable prognosis while WBRT ± HS is reserved to patients with multiple and/or large BMs and a worse prognosis.
A median intracranial PFS of 13.7 months was found, accordingly with Popp et al. 28 findings (13.5 months). The other considered studies16,17,23 had worse outcomes (11.4, 10.0 and 5.9 months, respectively), despite the fact that two of them16,17 applied a > 35 Gy SIB on the major lesion(s).
In the present study, a total of 40 out of 72 (56%) patients experienced intracranial failure, a higher rate with respect to Popp et al. 28 (27%) although they applied a greater expansion to the hippocampal avoidance region (5 mm vs 7 mm). Of note is that this failure rate is comparable with the one of Gondi et al. 23 (67%), the only with a similar cohort size. Considering the total number of relapses in the hippocampal avoidance region reported in the present study, the risk of failure appears higher when compared with the available literature. However, analyzing in detail the burden of disease at recurrence, the real risk of failure could be more acceptable (five out of 15 recurrences were oligometastatic).
At multivariate analysis, as expected, both systemic disease progression and chemotherapy schemes administration were found to be negatively associated with OS. None of the considered studies (Table 6) performed a multivariate analysis. At univariate analysis, Lebow et al. 16 found a correlation between KPS at baseline and OS. In this investigation, KPS was not found to be associated with OS, however, it should be noted that, as shown in Table 2, the majority of patients (87%) had KPS > 80.
Regarding dosimetric comparison in the hippocampal avoidance region, in the current analysis a median D1 of 13.5 Gy and median D50 of 7.58 Gy were reported, doses close with the ones of Gondi et al. 23 (12.8 Gy and 5.5 Gy, respectively), who treated patients with Tomotherapy as well. In fact, it is well known that Tomotherapy in helical modality allows to better spare hippocampi with respect to IMRT and VMAT.29,30
The acute and late toxicity profiles were acceptable and no serious adverse events during the follow-up were reported across the available data (no G > 2 events). The lower toxicity rates of the present work with respect to the available literature (Table 6) can be ascribable to the pre-treatment steroid administration.
In a recent publication reporting the results from a phase III trial for patients with BMs, Brown and colleagues 31 concluded that HS-WBRT plus memantine should be considered as standard of care for patients with BMs, good performance status and no hippocampal metastases who are due to receive WBRT. Their results highlighted a lower risk of cognitive failure after HS-WBRT plus memantine versus WBRT plus memantine with no significative differences in OS, intracranial PFS, or toxicity.
The above-mentioned study reported the neurological impairment evaluation, that is lacking and outside the scope of the present analysis, given its retrospective nature and the absence of dedicated questionnaires for the assessment of cognitive function preservation. This certainly represents the main limitation of our study. Indeed, there is a rising interest in published studies reporting fewer neurological impairments after HS-WBRT, compared to conventional WBRT, justifying the increased use of this modern RT technique.32–35 Another limitation is represented by the fact that reported clinical outcome results come from a wide and heterogeneous series of negatively selected patients with several lines of previous treatments or synchronous polymetastatic disease. No calculation of the total number of brain metastases and volume was performed, representing an additional limitation of the study. Finally, 24 patients died before the first radiological re-evaluation and data for 23 patients were missing, making the results less robust but still comparable with the published series.
In conclusion, while WBRT remains the mainstay for patients with multiple BMs, the present study shows that HS-WBRT is feasible, well tolerated, and with satisfying oncological control. However, the non-negligible rate of intra-hippocampal failure in our real-world series warrants further investigations aiming at a more refined patient selection. Oncological outcomes after hippocampal region avoidance may be further improved by the addition of a SIB to the dominant lesions’ sites. Future studies should evaluate the HS-WBRT± SIB benefits compared to traditional WBRT in randomized prospective settings, evaluating cognitive preservation and intracranial disease control.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916231206926 – Supplemental material for Hippocampal region avoidance in whole brain radiotherapy in brain metastases: For all or for some? A real-world feasibility report
Supplemental material, sj-pdf-1-tmj-10.1177_03008916231206926 for Hippocampal region avoidance in whole brain radiotherapy in brain metastases: For all or for some? A real-world feasibility report by Giulia Corrao, Luca Bergamaschi, Vanessa Eleonora Pierini, Aurora Gaeta, Stefania Volpe, Matteo Pepa, Mattia Zaffaroni, Maria Giulia Vincini, Cristiana Iuliana Fodor, Gaia Piperno, Francesca Emiro, Annamaria Ferrari, Sara Gandini, Federica Cattani, Roberto Orecchia, Giulia Marvaso and Barbara Alicja Jereczek-Fossa in Tumori Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mattia Zaffaroni, Maria Giulia Vincini and Giulia Corrao received a research fellowship from the Associazione Italiana per la Ricerca sul Cancro (AIRC) entitled “Radioablation ± hormonotherapy for prostate cancer oligorecurrences (RADIOSA trial): potential of imaging and biology” outside the current study. Stefania Volpe is a PhD student within the European School of Molecular Medicine (SEMM), Milan, Italy. The institution of the authors (IEO, Milan) received a support by the Italian Ministry of Health with Ricerca Corrente and 5x1000 funds and by Accuray Inc. The sponsors did not play any role in the study design, collection, analysis and interpretation of data, nor in the writing of the manuscript, nor in the decision to submit the manuscript for publication.
Data availability statement
The dataset analyzed during the current study is not publicly accessible, however it could be accessible upon reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
