Abstract
Objective:
The expression and significance of human epidermal growth factor receptor 2 (Her2) in upper tract urothelial carcinoma (UTUC) remains controversial. Thus, we aimed to systemically review the Her2 expression in UTUC patients and its relationship with pathological characters and clinical outcomes with meta-analysis.
Materials and methods:
A systematically computerized search in PubMed, Scopus, Embase and Cochrane was conducted. From a total of 454 related articles, 35 articles were finally reviewed and 16 papers were chosen for further analysis. Pathological characters included tumor stage, grade, lymph node metastasis (LNM) and lymphovascular invasion (LVI). The clinical outcomes included overall survival (OS), recurrence-free survival (RFS), cancer specific survival (CSS), metastatic-free survival (MFS) and progression-free survival (PFS). RevMan software was used for meta-analyses.
Results:
In total 16 studies from 1994 to 2020 were chosen, 14 studies used immunohistochemistry to assess the expression of Her2 and 5 studies used in situ hybridization, with a positive rate of 0 to 74.0% and 7.2 to 18.1%, respectively. Her2-positive was significantly associated with stage (pooled HR 1.86; 95 % CI 1.43–2.42), grade (pooled HR 2.81; 95 % CI 1.01–7.85) and LNM (pooled HR 1.93; 95 % CI 1.18–3.15). However, there was no statistically relationship between Her2-positive with LVI (pooled HR 1.48; 95 % CI 0.64–3.46) and RFS (pooled HR 1.41; 95 % CI 0.98–1.83).
Conclusions:
This review indicated that UTUC patients with Her2-positive tended to develop higher stage and grade tumors and LNM. The Her2 expression in UTUC patients deserves further investigation in the future.
Introduction
Upper tract urothelial carcinoma (UTUC) is an uncommon disease, accounting for 5–10% of urothelial carcinomas, and radical nephroureterectomy with bladder cuff excision is considered the gold standard surgery management of high-risk UTUC, while ureteroscopy and ureteroureterostomy are for low-risk UTUC.1,2 However, nearly 90% of patients would undergo bladder recurrence or distant metastasis, and the five-year cancer-specific survival (CSS) rate ranges from 30% to 60%.3-5 There are many situations that are beyond the scope of surgical intervention and consequently increasing attention is being paid to novel treatment concepts and drugs. For adjuvant therapy, cisplatin-based chemotherapy was recommended as the first-line treatment for high-risk UTUC by European Association of Urology. 2 However, for advanced or metastatic urothelial carcinoma patients, cisplatin-containing chemotherapy has not resulted in satisfying survival outcomes, with a median free-progression survival (PFS) time of only six months and overall survival (OS) time of one year.6-8 while the second-line choice, such as immunotherapy, showed worse efficacy.9-11 It poses a great challenge to manage such an embarrassed situation, and effective targeted drugs are urgently needed.
Her2, also named neu, c-erbB2 and EGFR2, is a transmembrane tyrosine kinase receptor and it plays fundamental roles in cell proliferation, differentiation, angiogenesis, and invasion as well as in the occurrence of malignant tumor events by activating the signaling pathways included PI3K, MAPK and PKA.12-17 The role of Her2 as a proto-oncogene has been well defined in breast cancer for nearly three decades and the first targeted monoclonal antibody, called Trastuzumab, was approved for metastatic breast cancer in 1998.18,19 With the application of the Her2 targeted therapy, the prognosis of patients with advanced breast cancer has improved greatly.
After comparing the Her2 expression in different tumor types in 37,992 patients, Yan et al. found that overexpressed Her2 was predominantly related with malignancies of epithelial origin, while tumors that originated from mesenchyme, neuroendocrine tissue, nerve tissue, and kidney tissue tended to be Her2-negative. 20 So Her2 might be a promising therapeutic target in urothelial carcinoma.
The role of Her2 in bladder cancer has been discussed in several reviews and relatively consistent conclusions have been drawn.21,22 However, only a few studies focus on the Her2 status in UTUC and its clinical significance remains controversial. In the present review, we summarized current evidence about the evaluation of Her2, and conducted a meta-analysis on the association between Her2 expression with pathological characters and the prognosis.
Methods
This study is conducted and reported according to the Meta-Analysis extension of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-NMA) reporting guidelines. 23
Search strategy and study selection criteria
The medical databases PubMed, Scopus, Embase and Cochrane were our main search databases. We searched and screened the titles and abstracts of the articles for the terms “UTUC,” “upper tract urothelial carcinoma,” “upper tract urinary carcinoma,” “urothelial carcinoma,” “renal pelvis and ureter,” “HER2,” “Neu,” “Erbb2,” “EGFR2,” and “human epithelial receptor 2”, and combined them using the Boolean operator (OR) in synonyms and the Boolean operator (AND) as the conjunction.
Screening process and PICOS principle
We used the Population, Intervention, Comparison, Outcomes, and Study (PICOS) principle during the screening process to select the literature. 24 The principle was applied as follows: population (P)—the UTUC patients; intervention (I) —evaluation of HER2 status; comparison (C) —the patients in HER2-negative; outcomes (O) —the tumor stage, grade, lymph node metastasis (LNM), lymphovascular invasion (LVI), overall survival (OS), recurrence-free survival (RFS), CSS, metastatic-free survival (MFS) and progression-free survival (PFS); study designs (S)—retrospectively comparative studies.
Articles were excluded if they: (a) did not distinguish UTUC from urothelial carcinoma; (b) had no concrete information of HER2 status; or (c) were in the form of conference proceedings, letters or comments.
The specific screening flow chart is shown in Figure 1.
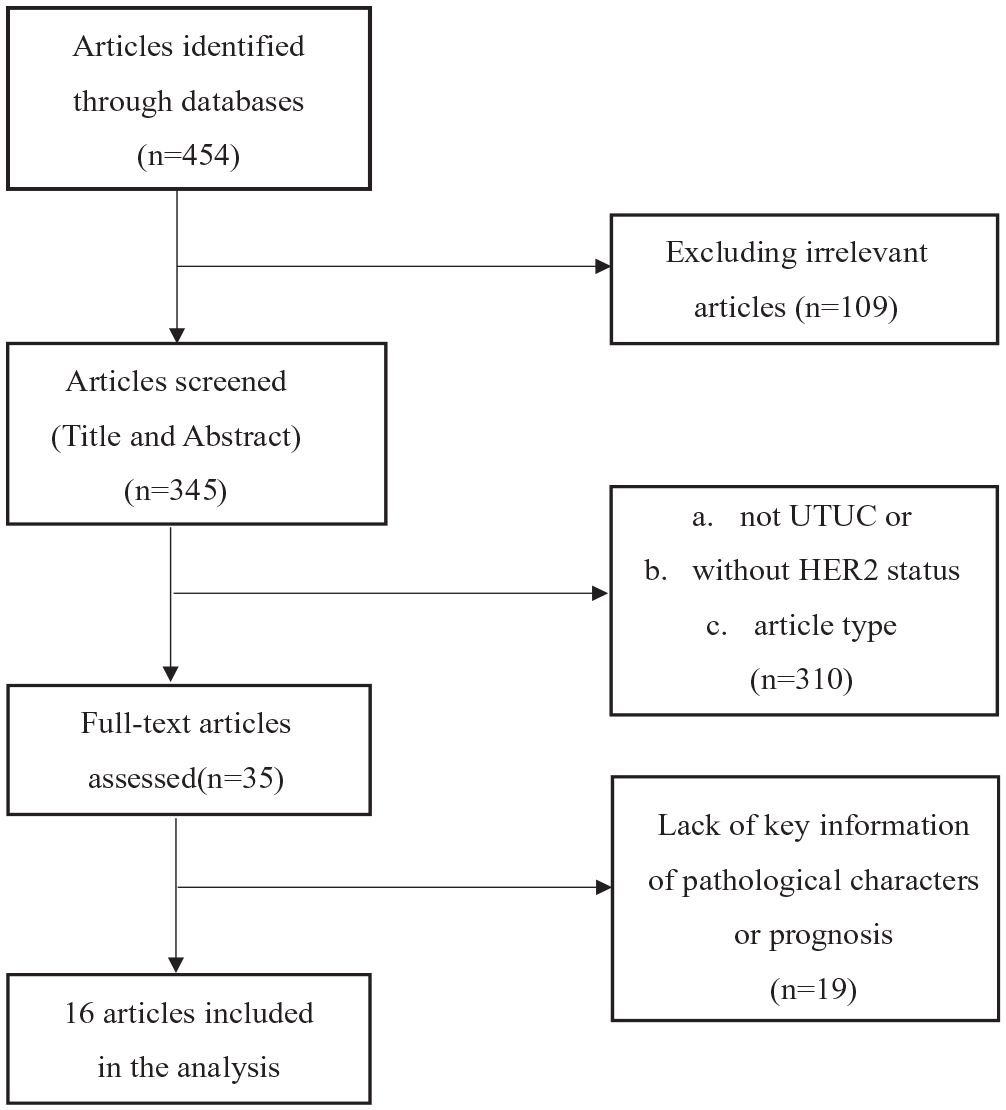
The specific screening flow chart of included studies.
Quality assessment
The basic information of the literature we included can be viewed in Table 1. The New Castle-Ottawa Scale (NOS) was used to evaluate the quality of non-randomized clinical trials. The evaluation process was completed by two authors (JJY and YQ). Any differences were settled through discussion, and a third person, one of the co-authors chosen randomly, was invited to judge if necessary.
The characteristics of the included studies.
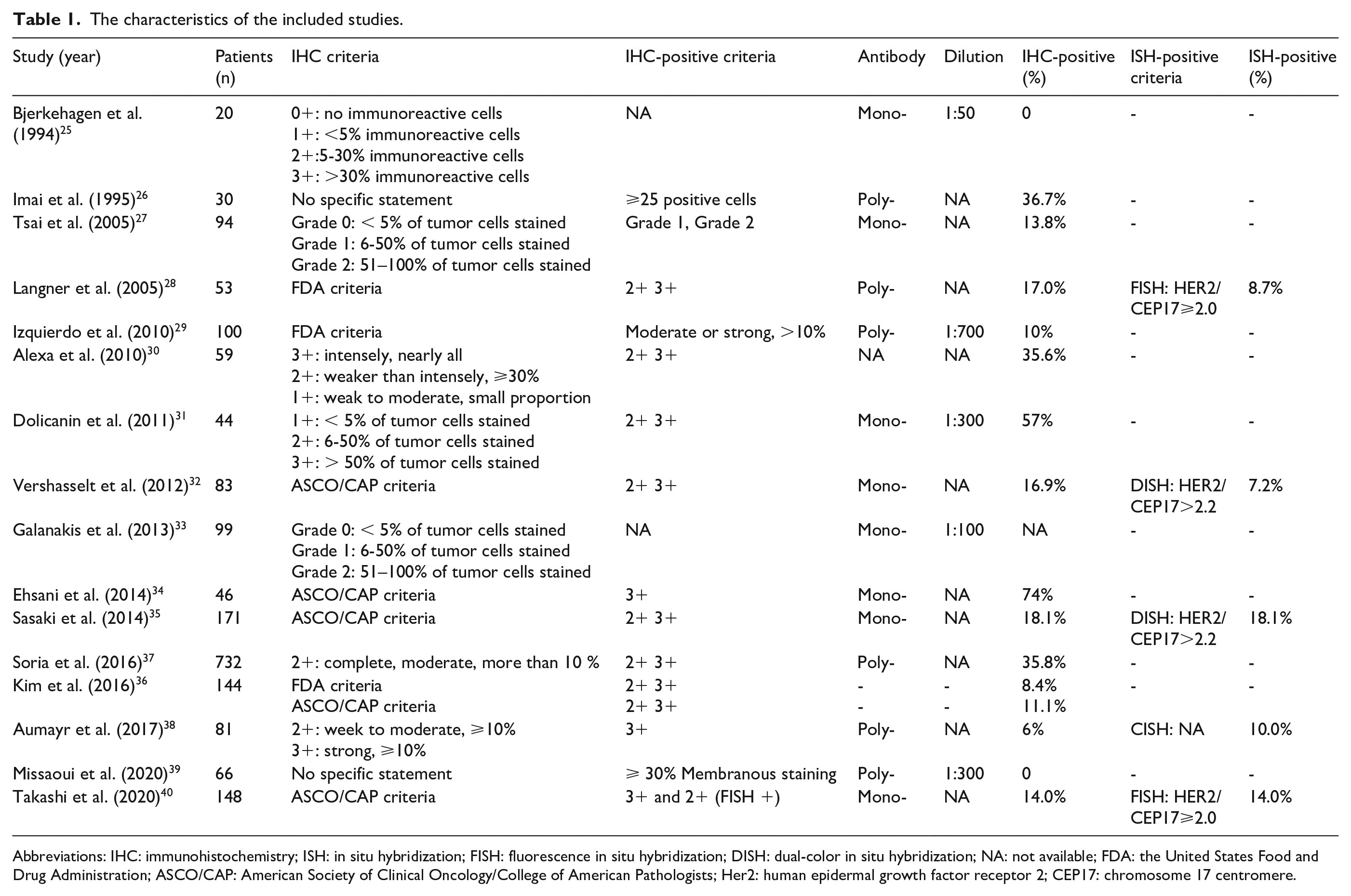
Abbreviations: IHC: immunohistochemistry; ISH: in situ hybridization; FISH: fluorescence in situ hybridization; DISH: dual-color in situ hybridization; NA: not available; FDA: the United States Food and Drug Administration; ASCO/CAP: American Society of Clinical Oncology/College of American Pathologists; Her2: human epidermal growth factor receptor 2; CEP17: chromosome 17 centromere.
Data extraction
Two reviewers (JY and YQ) independently extracted data, and disagreements were resolved through consensus. The extracted data for each study included first author’s name, publication year, number of patients, detection methods, cut-off value, rate of Her2-positive expression, clinicopathological parameters (stage, grade, LNM and LVI), follow-up period and survival outcomes (OS, RFS, MFS CSS and PFS).
Data consolidation and analysis
We completed the complicated data synthesis operation process using RevMan 5.4.1 software (Cochrane). HRs and their corresponding 95 % CIs were obtained and pooled for survival outcome, and ORs and their CIs were used for pathological factors. As in other literature studies, a P value less than 0.05 was considered statistically significant. The I² value was used as the index to assess heterogeneity. If the index was more than 50%, it indicated that heterogeneity was significant. Heterogeneity was also evaluated with a random-effects. Publication bias was investigated using Egger’s and Begg’s graphical methods and p<0.05 was considered to be subject to publication bias.
Results
Study selection and characteristic
A total of 454 publications were initially identified through database searching, and 35 potentially relevant reports were confirmed. Among 35 full-text articles left for eligibility assessment, 17 studies were further excluded because they lacked some key information regarding pathological characters or prognosis. Finally, 16 eligible studies were identified (Figure 1). 25-40
The main characteristics of the included studies, which were all retrospective, are summarized in Table 1 and Table 2. Among 16 studies, a total of 2030 UTUC patients ranging from 20 to 732 patients per study were reported.
The pathological characters and clinical outcomes in included studies.
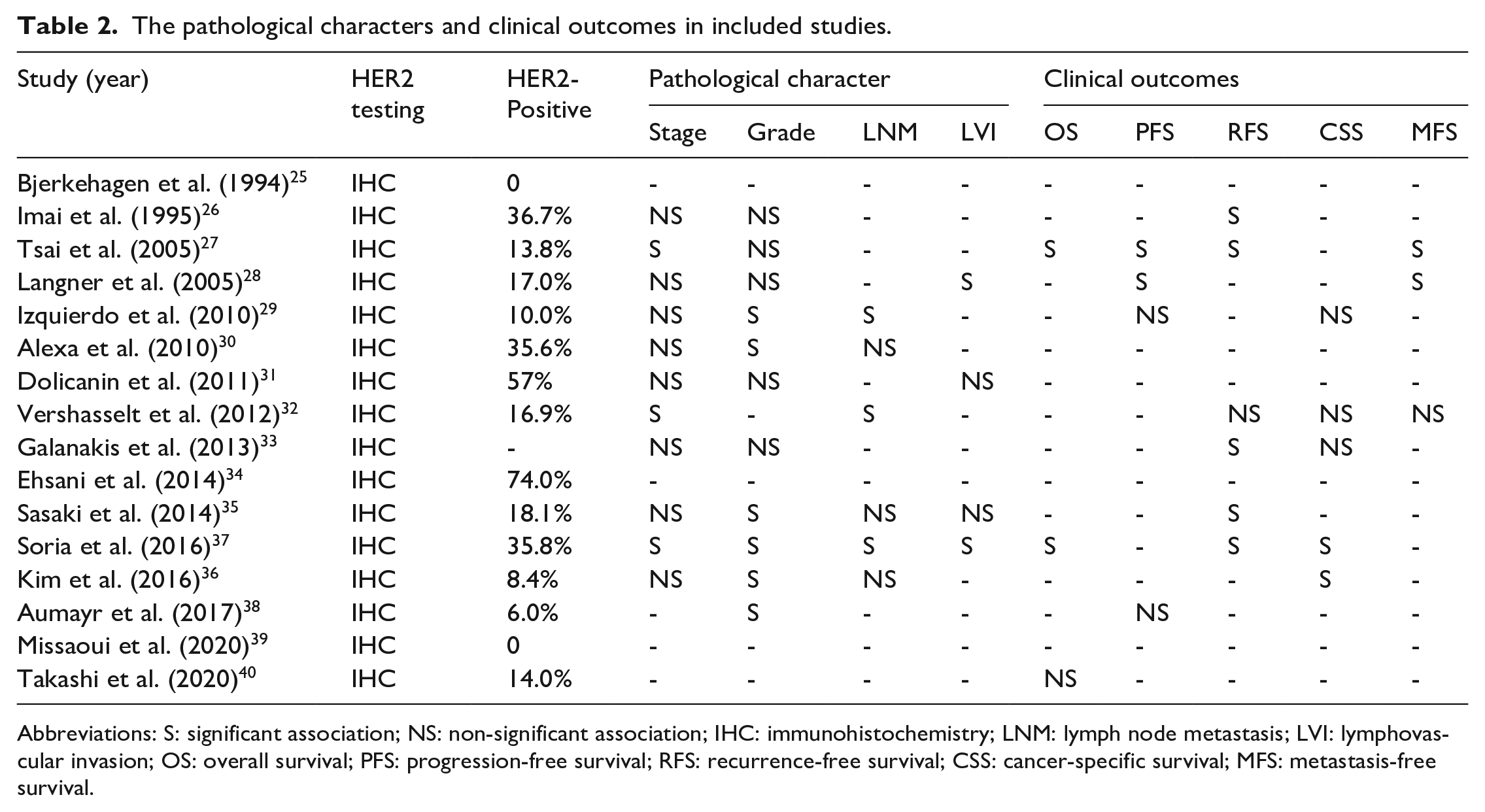
Abbreviations: S: significant association; NS: non-significant association; IHC: immunohistochemistry; LNM: lymph node metastasis; LVI: lymphovascular invasion; OS: overall survival; PFS: progression-free survival; RFS: recurrence-free survival; CSS: cancer-specific survival; MFS: metastasis-free survival.
Her2 evaluation
As shown in Table 1, from 1994 to 2020, IHC was used to assess the expression of Her2 in all 16 studies, with a positive rate of 0 to 74.0%, while ISH was in only four studies, with a positive rate of 7.2 to 18.1%. Furthermore, three studies tried to explore the relationship between IHC and ISH and finally found that Her2 overexpression and amplification were strongly correlated (p<0.0001).
For the scoring system of IHC, only three studies used FDA criteria and five studies used the ASCO/CAP criteria. In the remaining studies, 5%, 10%, 30% or 50% of immunoreactive cells with or without the membranous staining pattern and intensity were regarded as the cut-off value while two studies ignored the statement of the criterion.
The experimental methods and the positive judgment criteria were also various among studies. As Table 1 showed, eight studies used monoclonal antibody as primary antibodies, six studies used polyclonal antibodies and two studies did not describe the type of antibody employed even though nearly all the Her2 expression was quantified by HercepTest. At the same time, only five studies presented the dilution of antibody and the dilution ratios varied from each other.
Based on the Her2 expression score, Her2 expression was divided into Her2-positive and Her2-negative. However, among the 16 studies on UTUC, two studies did not elaborate the definition of Her2-positive in IHC and the remaining studies had inconsistent definitions, while the most commonly used one was Her2 2+ and 3+. In the five studies that used ISH, four only took the different HER2-to-CEP17 ratio with the ignorance of the gene copy number. Another one even did not state the value chosen in the study.
Her2 and pathological characters
As shown in Table 2, four studies were unable to evaluate the relationship between Her2 expression valued by IHC and pathological characters and in two of these it was due to lacking Her2-postive individuals. Among 12 studies, four studies found that there was no significant association between Her2 and tumor grade or stage, and the remaining successfully demonstrated at least one. Only six and four studies included LNM and LVI in their studies, respectively. UTUC patients with Her2-overexpressed were found to tend to develop high-grade tumors in six of these 11 studies, to develop high-stage tumors in three of these 11 studies, to have LNM in three of the six studies and LVI in two of the four studies, which were all statistically significant.
Seven studies reported concrete stage and grade data, four studies included LVI data and three studies involved LNM data. High heterogeneity (P = 0.008, I^2 = 65.7 % for grade and P = 0.021, I^2 = 69.1 % for LVI) and low heterogeneity (P = 0.081, I^2 = 46.7 % for stage and P = 0.728, I^2 = 0.00% for LNM) were determined among the studies. Thus, the random-effects models were used to pool the data for the pathological character about grade and LVI while fixed-effects models were used for stage and LNM. Meta-analysis showed that Her2-positive was significantly associated with stage (pooled HR 1.86; 95 % CI 1.43–2.42), grade (pooled HR 2.81; 95 % CI 1.01–7.85) and LNM (pooled HR 1.93; 95 % CI 1.18–3.15) (Figure 2a-c). Furthermore, there was no statistically relationship between LVI and Her2-positive (pooled HR 1.48; 95 % CI 0.64–3.46) (Figure 2d).
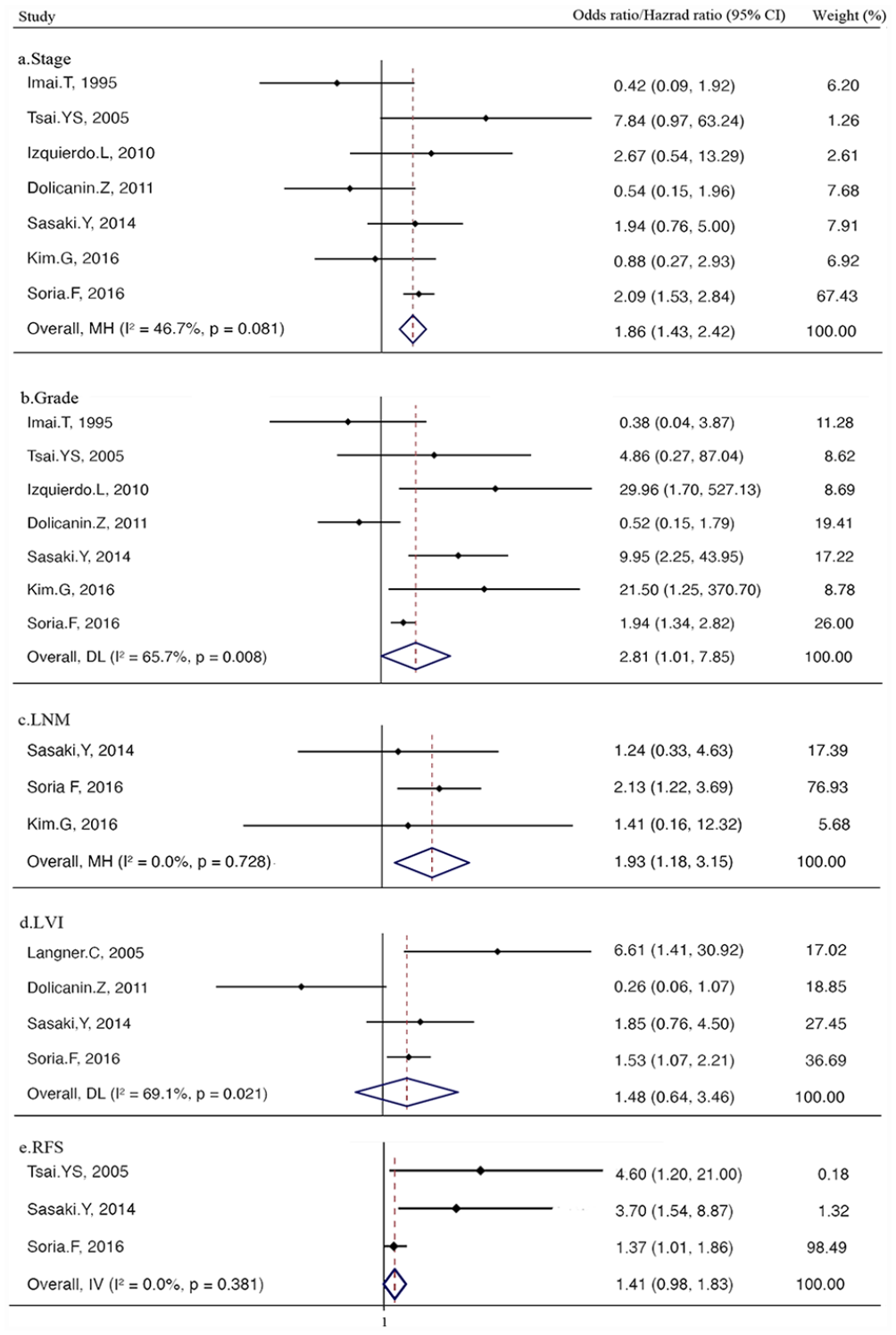
Forest plots for stage (a), grade (b), LNM (c), LVI (d) and RFS (e).
Her2 and prognosis
In UTUC, the association between Her2 and prognosis was further investigated in 10 of 16 studies (Table 2), and in these studies, prognostic indexes included OS, PFS, CSS, RFS and MFS. The most frequently used index was RFS which was used in six studies and the significant relationship with Her2 status was found in five studies. CSS and PFS were evaluated in five and four studies, respectively, and only two of each had the statistically significance with Her2 status.
However, detailed information of RFS, PFS and CSS were offered just in three, two and one studies, respectively. Low heterogeneity (P = 0.381, I^2= 0.00 %) of RFS was determined among three studies. Thus, the fixed-effects model was used to pool the data for RFS results and Meta-analysis showed that Her2-positive was not in significant relationship with RFS (pooled HR 1.41; 95 % CI 0.98–1.83) (Figure 2e).
Publication bias
Due to the number restriction of included studies, Egger’ test and Begg’ test were used to evaluate the publication bias. And the results showed that there was no obvious publication bias with all the values of test more than 0.05 (0.764 and 0.380 of Egger’ test and Begg’ test for stage, 0.707 and 0.285 for grade, 1.000 and 0.334 for LNM, 0.734 and 0.956 for LVI, 0.296 and 0.268 for RFS).
Discussion
There is an increasing demand for novel and efficient therapies for advanced or metastatic UTUC patients. As a biomarker, Her2 has been well identified in breast and stomach carcinomas, and its targeted therapies were established clinical routines for Her2-positive ones, resulting in significantly increased disease control and survival benefit and providing another promising choice for those patients who were in resistance or void response to conventional treatment. To expand these findings into UC with the similar background of Her2 status, a series of studies were conducted and some are currently ongoing. In RC48-C005 and RC48-C009 trials, RC48, which consisted of anti-Her2 humanized monoclonal immunoglobulin-1 and antimitotic agent, showed an encouraging result for locally advanced or metastatic UC patients with ORR of 50.5% and Disease Control Rate (DCR) of 82.2%. 41 However, almost all the attention on Her2 in UC has been paid to bladder cancer, which accounts for the majority of UC, with only a few on UTUC.
Therefore, in the current study, we systematically reviewed the studies published in the past three decades on Her2 status in UTUC and try to explore relationship between Her2 with clinical pathological characters and prognosis with meta-analysis. The results showed that the Her2 expression varied substantially among studies and UTUC patients with Her2-positive tended to develop higher grade, higher stage and LNM which was statistically significant. However, there was no significant value in LVI and RFS. Given the considerable heterogeneity in the scoring system, positive judgment criteria, experimental method and sample size, most studies were considered to be of relatively poor quality, corresponding to the high heterogeneity in the results. And Egger’ test and Begg’ test reflected that there was no obvious publication bias.
The current scoring system was closely based on the criterion for breast cancer, it was well-known that scoring system was validated for breast cancer only and did not take account of histological differences between other different tissues. Furthermore, the molecular mechanisms involved in tumorigenesis and disease progression between BC and UTUC were reported to be different by comparing the receptor patterns.42,43 Thus, the clinical practice for Her2 in UTUC should not be duplicated in breast cancer or bladder cancer (BC), but rather unique model needs to be explored. In 2008, the investigators in the Trastuzumab for Gastric Cancer trial conducted a validation study to assess IHC and ISH protocols for testing Her2 status in advanced gastric cancer. By analyzing the inconsistency between IHC and ISH, they successfully modified the HercepTest criteria for gastric cancer and recommended it for clinical use. 44 It provided a good basis for the development of Her2 criteria in UTUC and in 2021, with reference to breast cancer, China formulated an expert consensus on Her2 detection in UC. This consensus tended to be easier to work with and proposed a new concept of low-copy amplification as one of the ISH results, which might lead to a more refined classification of Her2 and better comparison of the population in clinical trials for targeted therapies. This would then allow more patients to benefit from Her2 treatment. But its sensitivity and specificity remain unknown and needs further exploration with clinical practice.
In the context of breast cancer, as much as 20% of Her2 tests might perform inaccurately, as mentioned by ASCO/CAP.45, 46 It is important to understand that this occurred in breast cancer with relatively complete specifications, and for other tumors, the proportion of inaccuracy will be more at the rational speculation, which would greatly affect clinical judgement and practice. In addition to the antibody type and dilution ratio, the difference in the pre-analytical phrase of IHC, such as antigen retrieval, controls and blocking, might be the reason for false positive and false negative results to a certain extent, which directly leaded to the incomparability and increased the heterogeneity of all the studies. Additionally, there were three different kinds of ISH experiments among four studies 28 , 32 , 38 which might make the results variable. Thus, as the basis for evaluation and subsequent treatment reference, the standardization and unification of Her2 experimental methods are necessary for Her2 testing not only for IHC but also ISH.
Because of the regulation of signaling pathways, Her2-positive tumors can differ from negative tumors in terms of pathological characteristics. In breast cancer, Her2-positive was significantly associated with pathologic high-stage, number of axillary nodes invasion, in situ ductal carcinomas, and absence of estrogen or progesterone receptor. 47 Her2 staining intensity in gastric cancer was correlated with tumor size, serosal invasion and lymph node metastases. 48 Similar results were also found in the present study. Interestingly, the study, conducted by Langner et al, found that the correlation between Her2-overexpressed and amplified with tumor stage or grade was converse, where no IHC3+ cancer was found and the FISH-positive cancers were all low amplification, with Her2/CEP17 of 2.03, 2.77, 2.91, and 3.39. 28 In addition, there were three studies that applied the ISH to explore the gene status of Her2 and occasionally found a statistically significant relationship between tumor stage, tumor grade or distant metastasis with polysomy 17.28,34,38 The polysomy 17 in UC of bladder was found to be associated with recurrence. 49 But the impact of polysomy 17 in UTUC has not been well investigated. Furthermore, the impact of tumor location on Her2 status was observed only in a few studies. In Sasaki et al.’s study, 35 there was no Her2-positive in 76 renal pelvis UC and the same phenomenon was found in 20 renal pelvic by Bjerkehagen et al. in 1994. 25 Furthermore, Langner et al. reported that no strong overexpression of Her2 in IHC was observed in 53 patients with UTUC while 48 cases were in the renal pelvis and only five in the ureter. 26 However, it was noticed by Alexa et al. that most positive cases were localized in the renal calyxes or the basinet and all ureter tumors were considered negative. 30 But they failed to draw the conclusion about the relationship between Her2 status and tumor location. Until now, there was no study focused on the expression of Her2 status according to the location in UTUC.
In 2009, Jeffrey retrospectively analyzed 107 studies considering 39,370 breast cancer patients and found that on univariate or multivariate analysis, Her2 overexpression or amplification significantly correlated with the outcome in nearly 90% of studies. 50 Unlike breast cancer, the studies in UTUC to date have yielded inconsistent findings regarding the prognostic relevance of Her2. And the present meta-analysis had drawn a conclusion with no statistical significance between RFS, the only one that could be used for analysis, and Her2 status. What deserves noting is that in three studies where recurrence was confined to the bladder 27 , 35 , 37 only Imai et al. took no history of BC as the inclusion criterion. 26 In Sasaki et al.’s study, 35 31/173 individuals had a prior BC history or had BC at the time of diagnosis while 6/94 did in Tsai et al.’ study, 27 which might pose certain influence on the results and create great difficulties for drawing a conclusion between Her2 and RFS, especially for the intravesical recurrence. In general, the pure prognostic value of Her-2 overexpression is incompletely understood and doubtful although positive Her2 is a negative prognostic factor in most studies. It may be more meaningful and feasible to compare the prognosis of different Her2 scores rather than positivity or negativity. Furthermore, using Her2 to determine prognosis is not recommended by the ASCO expert panel.
As a potential therapeutic target, anti-HER2 therapy has been assessed in clinical trials as second-line treatment in patients with advanced or metastatic UC; whereas limited data existed with regard to UTUC due to the rarity. A phase II, multicenter study recruited 284 locally advanced or metastatic UC patients, including 145 UTUC patients, who previously failed at least one line of systemic chemotherapy, and yielded promising results about disitamab vedotin (RC48-ADC) with a combined overall response rate for patients with HER2 IHC 1+/2+/3+ of 54%. 51 More recently, single HER2-targeting agents, anti-HER2 agents in combination with cytotoxic chemotherapy, dual HER2 blockade, HER2-targeted antibody-drug conjugates (ADCs), and other novel therapeutic approaches is currently and positively under evaluation. 52
There were some limitations in the present review. The most obvious was the heterogeneity which was mostly from the Her2 status evaluation. The stratified analysis, aimed at reducing the heterogeneity, was unable to be implemented due to the inconsistent and un-unified backgrounds of these studies. Furthermore, the retrospective nature and small sample size limited the evidence level in the review.
Conclusions
In conclusion, we conducted a systematic review and meta-analysis on Her2 expression, its association with pathological characters or prognostic value in UTUC patients by comparing the results of 16 relative studies from 1994 to 2020. The results of Her2 expression varied between studies, and the scoring system, positive judgment criteria, and experimental methods should be further explored in UTUC. The association between Her2 and pathological characters or clinical prognosis is also controversial because of high heterogeneity. But it will remind surgeons to perform Her2 testing for patients who are in high-grade, stage or LMN, which may provide a choice of subsequent targeted therapies. For clinical outcomes, Her2 was not in significantly relationship with RFS, which deserves further investigation in the future.
Footnotes
Author contribution
(I) Conception and design: Jianjun Ye, Xinyang Liao, Qiang Wei and Yige Bao
(II) Administrative support: Yige Bao and Qiang Wei
(III) Collection and assembly of data: Xinyang Liao, Yu Qiu and Jianjun Ye
(IV) Data analysis and interpretation: Xinyang Liao, Yu Qiu and Jianjun Ye
(V) Manuscript writing: All authors.
This manuscript has been read and approved by all the authors, and each author believes that the manuscript represents honest work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant number: 81500522) and Science & Technology Department of Sichuan Province (Grant number: 2020YFS0090, 2020YFS0046).
