Abstract
Introduction
Surgical resection of pulmonary metastases has been associated with increased survival at 5 years for osteosarcoma, but limited information is available on long-term outcome, role of repeated metastasectomies, and surgical sequelae in a pediatric population. We analyzed a consecutive series of children, adolescents, and young adults (AYA) treated by repeated lung metastasectomies during a period >40 years to estimate the clinical benefit and potential cure rate of salvage surgery.
Methods
All patients who underwent lung metastasectomy for osteosarcoma at the IRCCS Istituto Ortopedico Rizzoli of Bologna, University of Modena, and IRCCS Istituto Nazionale Tumori of Milan from May 1973 to January 2014 were included. Overall survival (OS) at 20 years from the first metastasectomy was calculated.
Results
A total of 463 consecutive children and AYA were analyzed. Median age was 15.9 years (range 0.2–23.2 years) and median follow-up 18.6 years. The 5- and 20-year OS were 34.0% and 29.7% (95% CI 25.5–34.0%). Among the 138 (29.8%) alive patients, 42 (30.4%) had disease recurrence cured by repeated metastasectomies. Disease-free interval from primary tumor, number of metastases, and complete resection were the most relevant survival predictors at multivariable model analysis.
Discussion
The extended follow-up of this consecutive series shows that repeated lung metastasectomy can achieve a permanent cure when offered to properly selected patients with metastases from osteosarcoma.
Introduction
Half a century ago, Edward Beattie and Ralph Marcove, thoracic and orthopedic surgeons at Memorial Sloan Kettering Cancer Center (MSKCC), published a report on 22 patients with osteogenic sarcoma, showing few long-term survivors after repeated lung metastasectomies. 1 At that time, 80% of children with osteosarcoma presented with or developed pulmonary metastases shortly after diagnosis, and all of them died of distant progression, often after limb amputation. 2 To circumvent such unfavorable conditions, Marcove and Gerry Rosen, a medical oncologist at MSKCC, designed a preoperative chemotherapy clinical trial aimed at reducing the risk of metastases, gaining time to assemble a custom-made bone prosthesis, and decreasing the amputation rate. 3
In 1982, a clinical trial was started at the Istituto Nazionale Tumori of Milan, combining neoadjuvant chemotherapy, primary tumor removal, and systematic resection of lung metastases in children with osteosarcoma. 4 All patients were approached by median sternotomy, with the aim of palpating bilateral nodules and resecting radiologically occult lesions, which were very frequent before the introduction of computed tomography staging. Repeated metastasectomies, whenever technically feasible, were undertaken. The study showed significant improvement of the 5-year overall survival (OS), from 35% in 72 children treated in 1970–1981 to 58% in 102 children treated in 1982–1988. 4
Six years later, the clinical benefit of systematic lung metastasectomy was confirmed by the results of the International Registry of Lung Metastases (IRLM), which included 5275 patients of all ages and various primary malignancies. 5 The IRLM provided a reliable classification system to predict the probability of survival at 5 and 10 years, based on the time interval from primary tumor, number of resected metastases, and absence of residual disease. 5
Nonetheless, many medical oncologists remained skeptical about the true curative potential of surgery for metastatic cancer, due to the lack of a control group and potential patient selection bias. 6
To address this criticism and provide more solid evidence on the curative potential of lung metastasectomy, we assessed the long-term survival (LTS) of a consecutive series of children and adolescents and young adults (AYA) treated by lung metastasectomy for osteosarcoma. The present analysis also includes previously reported patients.4,5,7–9
Methods
We analyzed all consecutive children and AYA with metastatic osteosarcoma treated by repeated lung metastasectomy in 3 Italian institutions (IRCCS Istituto Ortopedico Rizzoli of Bologna, University of Modena, and IRCCS Istituto Nazionale Tumori of Milan) from May 1973 to January 2014. According to European Society for Medical Oncology (ESMO) and European Society for Paediatric Oncology (SIOPE) guidelines, AYA were defined as patients ⩽24 years of age. All consecutive patients with a diagnosis of osteosarcoma, synchronous or metachronous operable lung metastases and available information on the treatment of primary tumor, and description of lung metastasectomy (date of surgery, size and number of resected metastases, resection volume, histology, and adjuvant therapy) and follow-up (site of recurrence, salvage therapy, and survival) were included. Exclusion criteria were disseminated disease or inoperable primary tumor. Preplanned bilateral thoracotomies were considered as one single intervention. A complete resection was defined as the surgical removal of all the macroscopic disease. If a complete resection was deemed impossible intraoperatively, the resection was classified as incomplete (R+).
Study data included 20-year OS; proportion of LTS, defined as those patients alive at 5 years from the first lung metastasectomy; patient features associated with LTS; and predictive value of the classification system proposed by the IRLM. 5
The follow-up was closed on 31 December 2019.
Statistical methods
OS was defined as the time from the first lung metastasectomy to the time of death from any cause or last follow-up, whichever came first. Survival functions were estimated using the Kaplan-Meier method; median follow-up and its interquartile range (IQR) were estimated using the reverse Kaplan-Meier method. Disease-free interval (DFI) was defined as interval from primary tumor surgery and date of metastases occurrence. A Royston-Parmar model was used to estimate the mortality rate. 10 The average hazard of death per unit time was estimated as the observed number of deaths in that time interval divided by the total person-time of observation over the same time interval. The logistic regression model was used to identify survivors at 5 years (i.e. long-term survivors) to estimate the area under the curve statistic and the range of predictive probabilities of long-term survivors. For continuous predictors, classification and regression trees (CART) methodology 11 was used to identify the best thresholds for the OS endpoint. Restricted cubic splines were used to evaluate graphically and formally the linearity assumption for a continuous predictor in univariable Cox regression models. Further details of statistical analysis are reported in the Supplementary Data.
Results
Patient characteristics
Patient characteristics according to the length of survival are shown in Table 1. Overall, 274 (59%) were male and median age at the time of the first pulmonary resection was 15.9 years (range 0.2–23.2 years). In 315 (69%) cases, pulmonary metastases were monolateral. A single pathologically proven metastasis was present in 223 (48%) patients, and multiple metastases in 240 (52%). A total of 345 (74.5%) of the patients developed metachronous pulmonary metastases, with a median interval from treatment of primary osteosarcoma to lung metastases occurrence (DFI) of 18.0 months (1–77 months; missing data for one patient).
Patient features according to the length of survival.
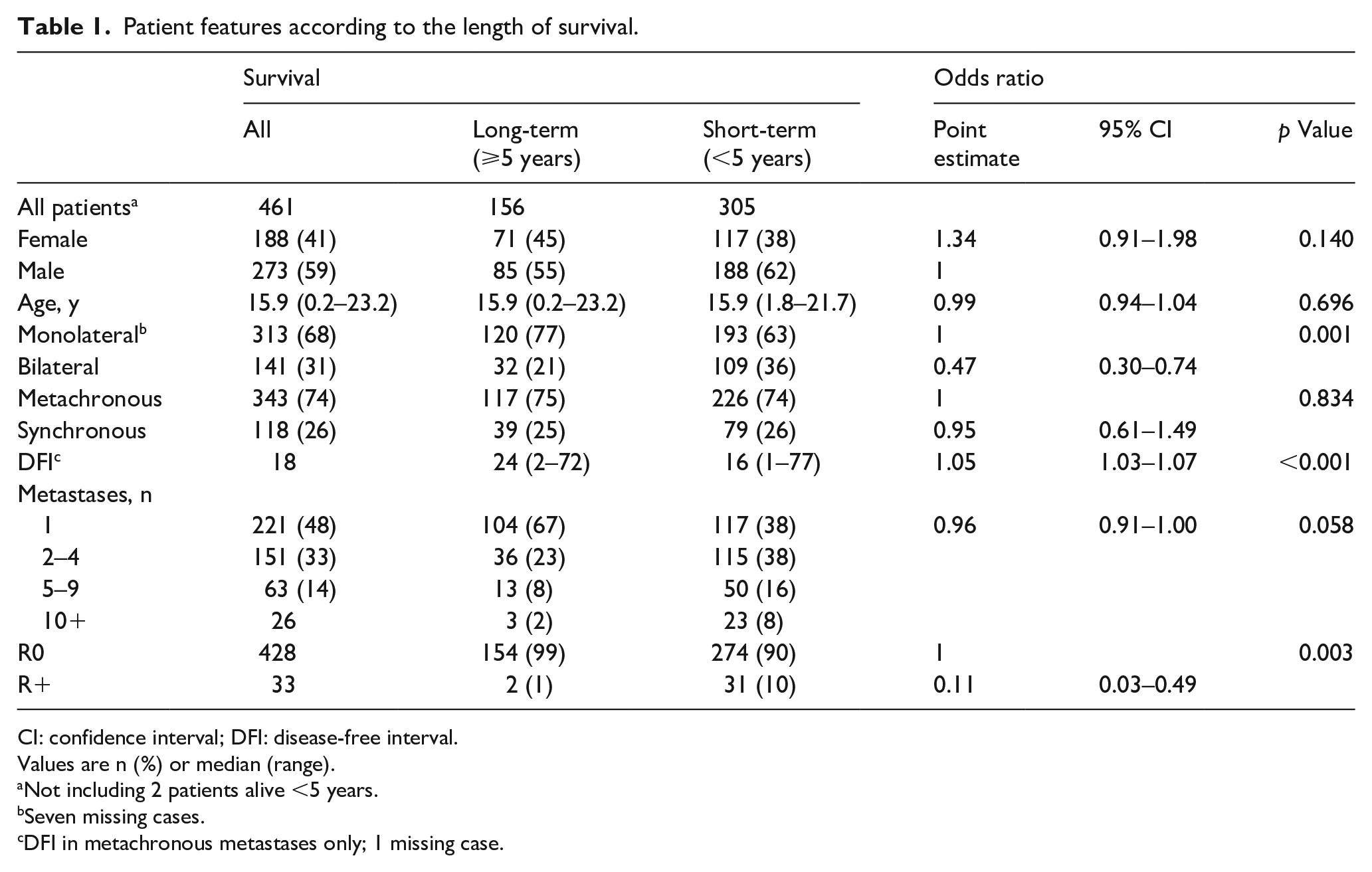
CI: confidence interval; DFI: disease-free interval.
Values are n (%) or median (range).
Not including 2 patients alive <5 years.
Seven missing cases.
DFI in metachronous metastases only; 1 missing case.
Patients with metachronous pulmonary metastases had 1 single nodule in 54.8% of the cases, more frequently as compared with patients with synchronous metastases (28.8%). Metachronous metastases were more often monolateral (75.4), as compared with synchronous metastases (50.8%). The surgical approach was monolateral in 305 (66.2%) cases, bilateral synchronous or staged (2-stage) thoracotomy in 87 (18.9%), sternotomy in 66 (14.3%) cases, and thoracoscopy in 3 (0.7%) cases. The usual surgical procedure was limited resection (wedge or precision) in 399 (86.2%) cases. Metastasectomy was considered complete (R0) in 430 (92.9%) cases. Chemotherapy was administered at the time of detection of pulmonary metastases to 448 patients (96.8%), radiotherapy to 10 (2.2%); metastasectomy was the only treatment in 9 (1.9%) cases. The proportion of patients who received multiple metastasectomies for lung relapse was 28.2% (44/156) in LTS and 38.7% (118/305) in short term survivors (STS) (odds ratio [OR] 0.62, 95% confidence interval [CI] 0.41–0.95).
OS and long-term outcome
The overall median follow-up was 18.6 years (IQR 13.4–26.6 years, Clark C index 94.2%), with 325 (70.2%) patients having died by December 31, 2019.
Median OS was 26.7 months (95% CI 23.0–29.6 months), 5-year OS 34.0% (95% CI 29.7–38.3%), 10-year OS 31.3% (95% CI 27.1–35.6%), and 20-year OS 29.7% (95% CI 25.5–34.0%) (Figure 1 [A]).
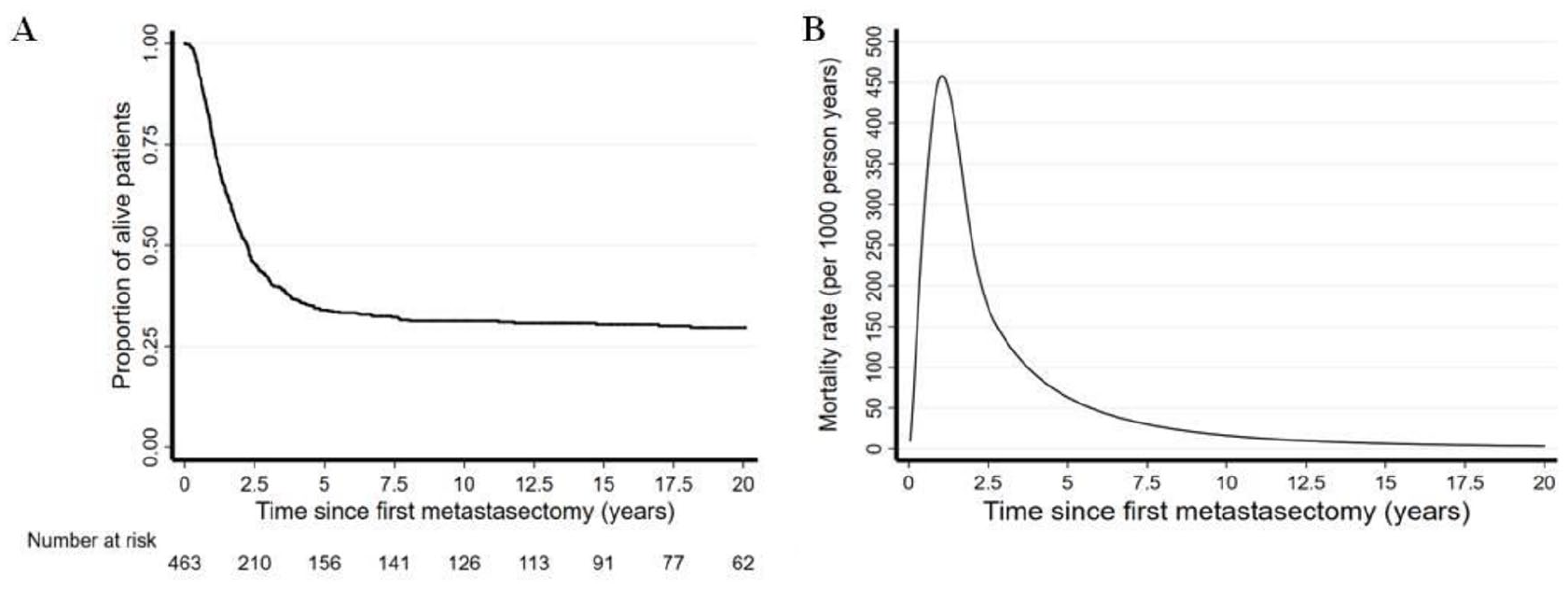
(A) Overall survival at 20 years in the whole population. (B) Smoothed mortality rate estimates.
Smoothed mortality rate (hazard ratio [HR]) estimates are shown in Figure 1 (B). After the first pulmonary metastasectomy, HR estimates sharply increased in the first year, with a median HR of 54.9 deaths per 1000 person-years (95% CI 34.1–75.8 deaths) during the first 6 months, 102.2 deaths (95% CI 73.4–131.0 deaths) in the following 6 months, and 465 deaths (95% CI 388.1–558.5) at 1.0 year. The median HR monotonically decreased from 155.1 deaths (95% CI 117.2–193.1) in the second year to 107.5 deaths (95% CI 68.6–146.4) in the third year and only 6.2 deaths (95% CI 0–18.6 deaths) in the sixth year.
The cause of death was osteosarcoma recurrence in 283 (87%) patients, other diseases in 2 (0.7%), and unknown in 40 (12.3%). Among the 138 (29.8%) alive patients at the end of follow-up, 95 (68.8%) were free of disease from the first metastasectomy, 42 (30.4%) had disease recurrence cured by repeated metastasectomies, and 1 (0.7%) patient was alive with unresected recurrence. The disease status was not reported in 2 (1.4%), and one patient was lost to follow-up.
Predictors of LTS
Median survival was 31.6 months (24.6–42.8 months) in female patients and 24.0 months (20.5–28.2 months) in male patients. The 20-year OS was 33.2% (26.4–40.1%) and 27.2% (21.9–32.7%) in female and male patients, respectively.
HR was higher in bilateral vs monolateral disease (HR 1.59 [95% CI 1.27–2.00]; Figure 2 [A]), with a median survival of 20.4 months (95% CI 15.1–24.0) vs 29.6 months (95% CI 25.5–36.9), and a 20-year OS of 17.5% (11.6%–24.5%) vs 34.5% (29.2%–39.9%) in patients with bilateral as compared to those with monolateral metastases.
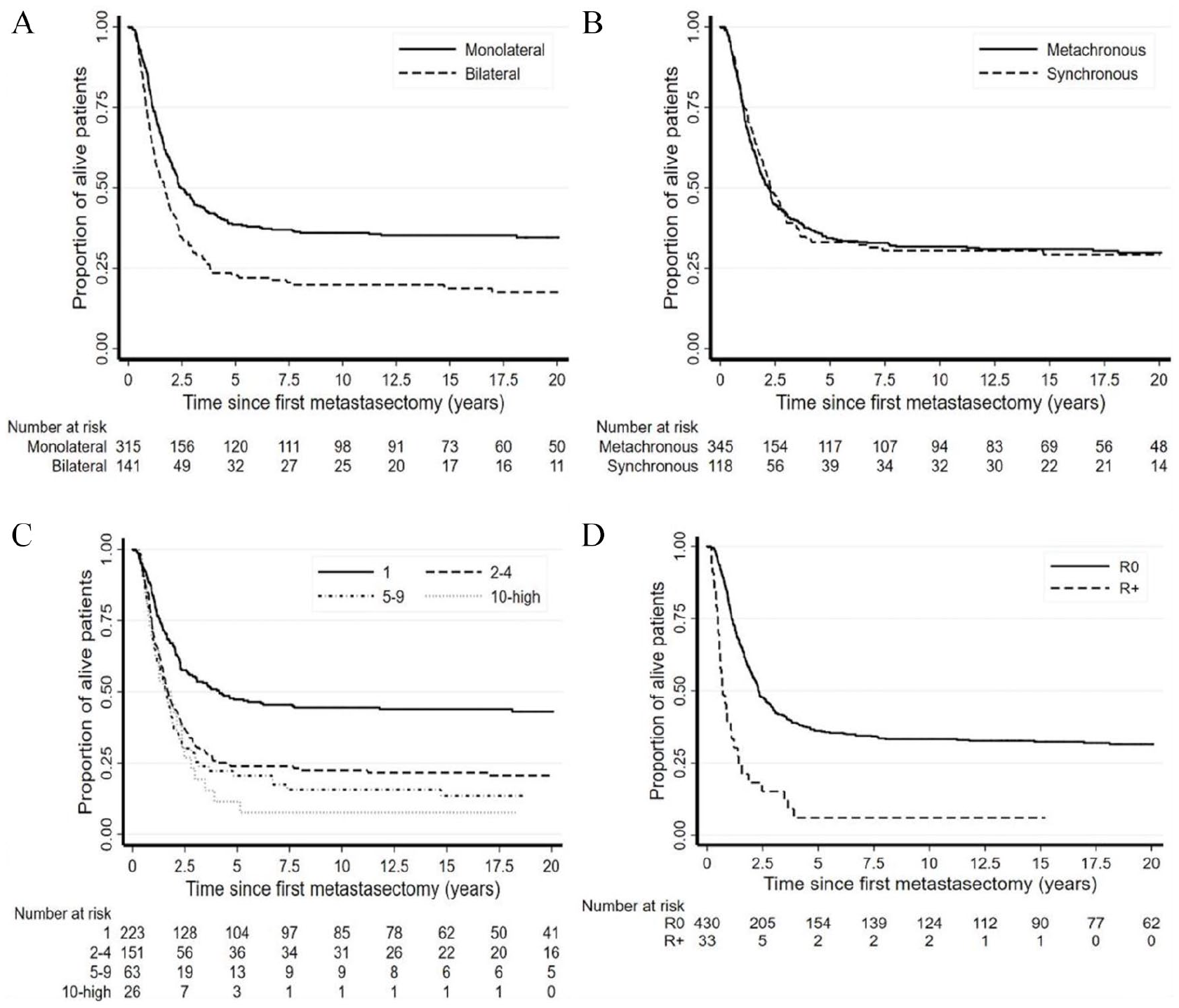
Overall survival according to disease extension: monolateral vs bilateral (A), disease-free interval: synchronous vs metachronous (B), number of resected metastases (C), and completeness of resection (D).
Patients with synchronous lung metastases at diagnosis had similar survival as patients with metachronous metastases (curve not shown), with a median survival of 27.7 months (95% CI 22.1–35.4) vs 26.2 months (95% CI 21.6–29.1), 20-year OS of 29.2% (21.2%–37.7%) vs 29.8% (24.9–34.8%), and HR of 0.98 (95% CI 0.77–1.26).
In cases with metachronous metastases, the DFI showed a significant impact on long-term survival, with a median survival of 13.0 months (95% CI 11.3–17.3) and 20-year OS of 16.0% (10.1%–23.1%) if DFI was below 14.5 months; a median survival of 27.5 months (95% CI 21.6–42.8) and 20-year OS of 27.8% (20.4%–35.7%) between 14.5 and 30.5 months; and a median survival of 352.3 months (95% CI 53.7–not defined) and 20-year OS of 56.0% (43.8%–66.5%) above 30.5 months (Figure 2 [B]).
Patients with a single metastasis had a median survival of 49.8 months (95% CI 31.4–217.4) and 20-year OS of 43.0% (36.3%–49.6%); with 2 to 4 metastases, a median survival of 20.5 months (17.0–26.2) and 20-year OS of 20.7% (14.5–27.6%); with 5 to 9 metastases, a median survival of 19.9 months (95% CI 13.1–22.8) and a 20-year OS of 13.5% (6.3%–23.5%); with >9 metastases, a median survival of 20.3 months (95% CI 10.6–28.4) and a 10-year OS of 7.7% (95% CI 1.3%–21.7%; Figure 2 [C]).
Patients achieving complete surgical remission (complete metastasectomy) had a median survival of 27.6 months (95% CI 24.6–33.6), with 20-year OS of 31.5% (27.0%–36.0%). After incomplete surgery (R+), the median survival was 8.4 months (95% CI 6.6–13.0), and the 10-year OS was 6.1% (95% CI 1.1%–17.6%; Figure 2 [D]).
The only 2 R+ patient alive at 5 years had presented with microscopic residual nodules at the first pulmonary metastasectomy, but no evidence of disease after the second (salvage) metastasectomy.
When we applied the IRLM system to this population, we could confirm the predictive value of IRLM classification for metachronous (Figure S2 [A]) as well as synchronous patients (Figure S2 [B]).
The results of multivariable model analysis (Table 2) confirmed DFI, number of metastases, and complete resection as the most relevant predictors for LTS.
Multivariable model on predictor variables of long-term survival.
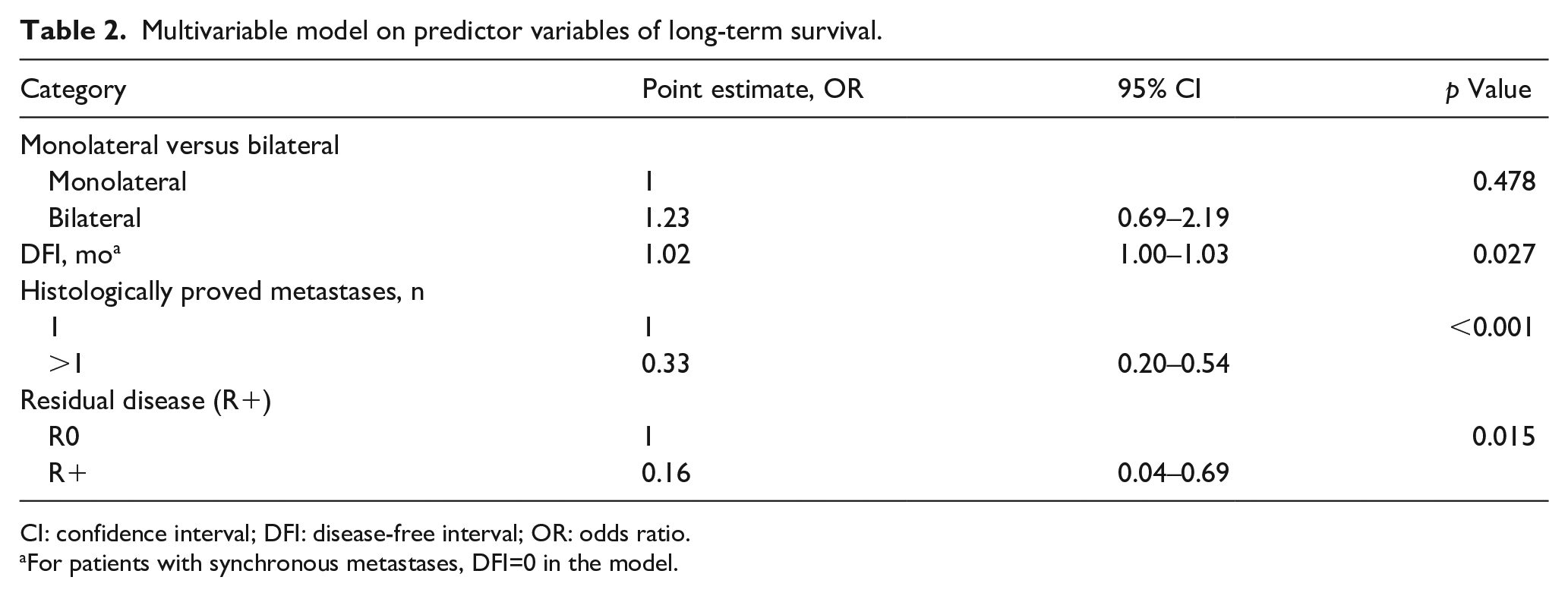
CI: confidence interval; DFI: disease-free interval; OR: odds ratio.
For patients with synchronous metastases, DFI=0 in the model.
The discrimination and calibration capabilities of the multivariable model were unable to identify patients with no prospective of long-term survival benefit, therefore unsuitable for surgical resection.
Discussion
Osteosarcoma is the most common malignant bone tumor in children, although it represents <1% of all neoplasms. 12 Approximately 10%–20% of patients have metastases at the time of their diagnosis, lung being the most common site (85%–90% of all cases),13,14 while 30%–40% of patients with nonmetastatic osteosarcoma develop lung metastasis during their life.15–17 In the past 3 decades, the application of increasingly efficient multimodality therapies, combining more aggressive surgery and more effective chemotherapies, has improved the outcome of patients with nonmetastatic osteosarcoma (5-year OS from 60% to 70%).7,18,19 Despite these advances, the 5-year OS ranges from 10% to 30% when metastases occur.20–22
Children represent an ideal population to assess the curative potential of lung metastasectomy, because their life expectancy exceeds 50 years if cancer is cured, much longer than that expected in adult patients, where competing risks of death play a major role.
Comparing the results of 896 lung metastasectomies for osteosarcoma, published from 1989 to 2018 and summarized in Table S1, there is a wide range in resection rates (15%–100%), median follow-up (2–10 years), and 5-year survival (11%–68%).23–27 The present analysis provides a robust assessment of long-term outcome, well beyond the maximum length of follow-up reported in the literature.
In our experience, the risk of death sharply increased after metastasectomy, reaching its peak at 1 year (Figure 1 [B]), and in patients who did not achieve disease control (305 STS), median survival was less than 16 months. Such a poor outcome is in keeping with our prior observation that all children developing lung metastases from an unselected osteosarcoma cohort in 1970–1981, before systematic use of metastasectomy, died within 5 years. 3
Ifosfamide at standard or high dose, alone or in combination with etoposide, and gemcitabine/docetaxel have been employed for patients with osteosarcoma at the time of recurrence or metachronous metastases. 28
In this series, where chemotherapy was used in combination with salvage surgery, the majority of patients surviving 5 years after the first metastasectomy were still alive at 20 years, and a significant proportion of them were rescued by salvage resection after subsequent lung recurrence. We have also confirmed the predictive role of IRLM prognostic grouping in pediatric patients with metastatic osteosarcoma. We could not identify the combination of clinical features that might render the surgical salvage ineffective.
Our study may have some biases due to its retrospective analysis, long recruitment period, and heterogeneous treatments among different centers. In addition, individual tumor outcome depends on multiple genetic and molecular markers of cancer cells, as well as host-specific factors that could not be tested in a retrospective analysis. Continuing efforts to investigate prognostic factors related to the individual survival of patients with osteosarcoma are needed, particularly in the critical subset of AYA. 29
The extended follow-up of this consecutive series of children and AYA with metastatic osteosarcoma proves that with repeated lung metastasectomy and chemotherapy, a permanent cure can be achieved when offered to properly selected patients.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916211053048 – Supplemental material for Lung metastasectomy for osteosarcoma in children, adolescents, and young adults: proof of permanent cure
Supplemental material, sj-pdf-1-tmj-10.1177_03008916211053048 for Lung metastasectomy for osteosarcoma in children, adolescents, and young adults: proof of permanent cure by Ugo Pastorino, Emanuela Palmerini, Luca Porcu, Roberto Luksch, Paolo Scanagatta, Cristina Meazza, Giovanni Leuzzi, Maura Massimino and Piero Picci in Tumori Journal
Footnotes
Acknowledgements
The authors thank Dr. Francesca Fossati Bellani, Marco Gasparini, Gaetano Bacci, and Antonio Briccoli for their contributions to the initial study design and patient treatment.
Author contributions
U.P., R.L., and P.P. conceived the study. E.P., R.L., P.S., C.M., G.L., and M.M. participated in data collection. U.P., L.P., and P.P. performed the statistical analyses and contributed to interpreting the data. U.P., E.P., L.P., R.L., G.L., and P.P. participated in writing the manuscript. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
