Abstract
Objective
To compare the efficacy of robot-assisted versus traditional core decompression combined with human umbilical cord-derived mesenchymal stem cell transplantation for osteonecrosis of the femoral head.
Methods
A total of thirty-eight patients were divided into two groups according to the surgical technique. The observation group (20 patients, 28 hips) underwent robot-assisted core decompression, while the control group (18 patients, 22 hips) underwent traditional decompression. Both groups underwent human umbilical cord-derived mesenchymal stem cell implantation. Relevant parameters were compared between the groups.
Results
At the final follow-up, the observation group showed lower visual analog scale scores and necrotic volume as well as higher Harris hip scores and femoral head survival rates than the control group; however, these differences were not statistically significant. The observation group required significantly fewer intraoperative fluoroscopies, experienced less intraoperative blood loss, and had a shorter operation time than the control group (all p < 0.01).
Conclusion
Our findings suggests that the robotic-assisted technique demonstrates comparable clinical and radiological outcomes as the traditional technique in the treatment of osteonecrosis of the femoral head. However, it may offer significant advantages in terms of surgical precision.
Keywords
Introduction
Osteonecrosis of the femoral head (ONFH) is a progressive and refractory disease. 1 The progression of the disease often leads to serious collapse of the subchondral bone and articular surface of the femoral head, even degenerative disease of the hip joint, and ultimately requires treatment involving osteotomy (OT), hemiarthroplasty (Hemi), or total hip arthroplasty (THA).2–4 The etiology and pathogenesis of avascular necrosis of the femoral head have always been under debate. There is considerable evidence stating that the increase in femoral head internal pressure plays an important role in the development of femoral head necrosis. 5
In 1985, Ficat designed the core decompression (CD) operation with open and closed medullary cavity. 6 Long-term clinical use has proved that it can effectively halt osteonecrosis progression. Traditional CD surgery requires complex hand–eye coordination, and surgeons need to manually control the guide needle, which may result in substantial deviations.7,8 Furthermore, clinical evidence has shown that excessive excision of necrotic tissue during core CD surgery may increase the risk of postoperative femoral head collapse. 9 With the rapid development of intelligent robots, robot-assisted surgical treatment is gradually being adopted in clinical practice, which can enable the performance of minimally invasive, accurate, and personalized surgeries to a greater extent.10,11 In addition to successfully completing thoracolumbar fracture surgery and atlantoaxial joint internal fixation surgery, TiRobotTM, the orthopedic navigation robot can be used to precisely implant different guide wires and screws in the proximal femur.12,13 The use of robot-assisted technology may reduce operation time, improve accuracy, and ensure the safety of surgeons and patients. 14 Considering these advantages of robot-assisted surgery, we planned this study to evaluate whether this method could reduce the number of intraoperative X-ray fluoroscopy examinations, intraoperative blood loss, and operation time.
Although some advantages were noted, CD did not fundamentally resolve the issues involved in repair of the necrotic bone.15,16 In recent years, the treatment of ONFH using stem cell transplantation has become an active research topic in many hip preservation therapies. 17 This method is based on the principle of promoting the regeneration of blood vessels and tissues in the necrotic region through the paracrine function and the proliferation and differentiation ability of stem cells. 18 Mesenchymal stem cells (MSCs) were isolated from Wharton’s jelly of the human umbilical cord-derived mesenchymal stem cell (hUC-MSC), and CD was used in combination with hUC-MSCs transplantation to treat early femoral head necrosis. Evidence suggests that hUC-MSCs mitigate necrosis and osteocyte apoptosis in GC-induced ONFH models by reducing the polarization of macrophages. 19
Considering the above-mentioned advantages of robot-assisted surgery and stem cell therapy for ONFH treatment, in the present study, we retrospectively analyzed the differences in treatment efficacy between robot-assisted CD combined with hUC-MSCs transplantation and traditional CD surgery combined with hUC-MSCs transplantation in ONFH treatment.
Patients and methods
Study design
This retrospective study was performed in accordance with the Helsinki Declaration of 1975, as revised in 2024, 20 and was approved by the Ethics Committee of Human Experimentation of our hospital. The study approval registration number is 2025-03-017-K01. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Inclusion and exclusion criteria
This retrospective analysis included all consecutive patients who had been diagnosed with ARCO stage II ONFH and had undergone robot-assisted or traditional CD combined with stem cell transplantation surgery at our hospital from January 2016 to June 2020.
The inclusion criteria were as follows: (a) age 18–60 years; (b) notable hip pain (visual analog scale (VAS) score ≥2); and (c) diagnosis of ONFH based on hip plain radiographs and magnetic resonance imaging (MRI).
The exclusion criteria were as follows: (a) systemic infection or local skin infection around the incision before operation; (b) history of steroid treatment in the past 6 months; (c) disease complicated with inflammatory arthritis; and (d) history of fracture or tumor in the proximal femur.
According to patients’ conditions and permissions, we performed surgeries using different methods. The participants were divided into two groups according to whether surgery was conducted using the TiRobotTM robot system. All the patients signed informed consent forms. We have deidentified all patient details. All surgeries were performed by the same group of senior surgeons.
Surgical technique
Robot component
Robot component
The orthopedic surgery robot TiRobotTM (TINAVI Medical Technologies Co., Ltd., Beijing, China) was used. This robot system consists of a workstation for surgical planning and control, an optical tracking device, a robot arm, and surgical instruments (Figure 1). 21
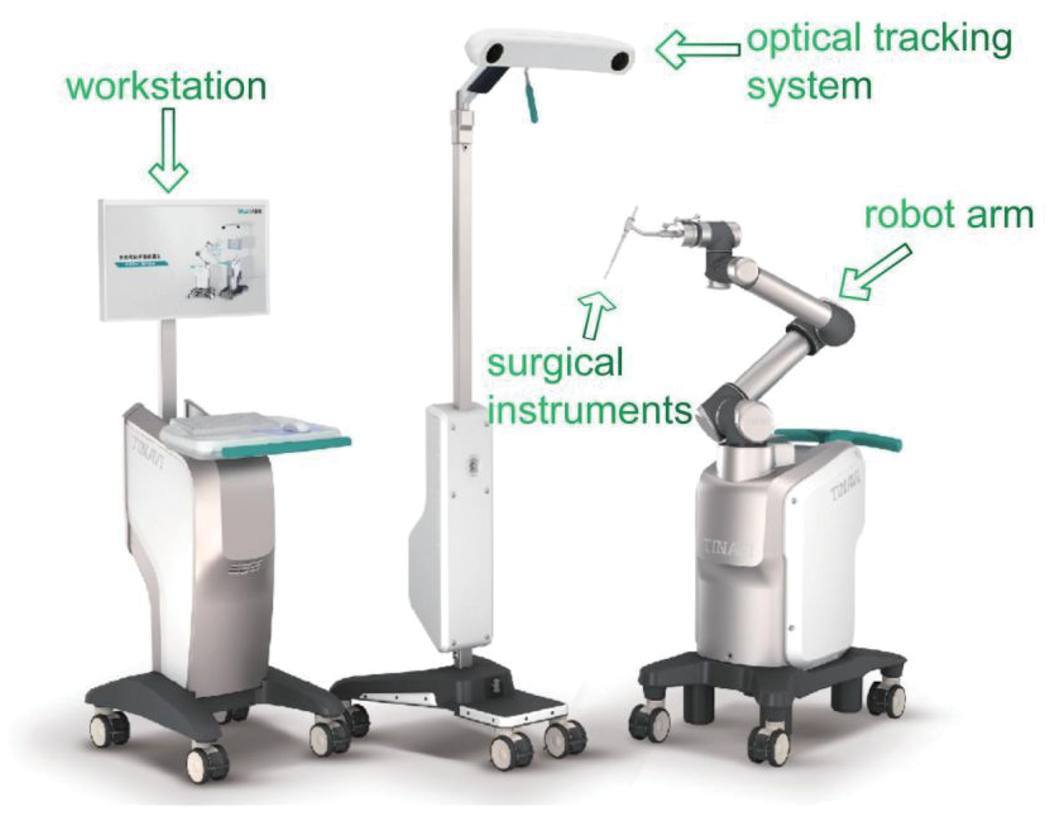
TiRobot, mainly including a workstation, an optical tracking system, a robotic arm and surgical instruments (photo provided by Tianzhihang Medical Technology, Beijing, China).
hUC-MSCs isolation and identification
hUC-MSCs were obtained from the clinical Stem Cell Center of the Affiliated Drum Tower Hospital of Nanjing University Medical School. 21 The expression of typical protein markers on the cell surface was detected using flow cytometry to identify the hUC-MSCs (Figure 2). 22
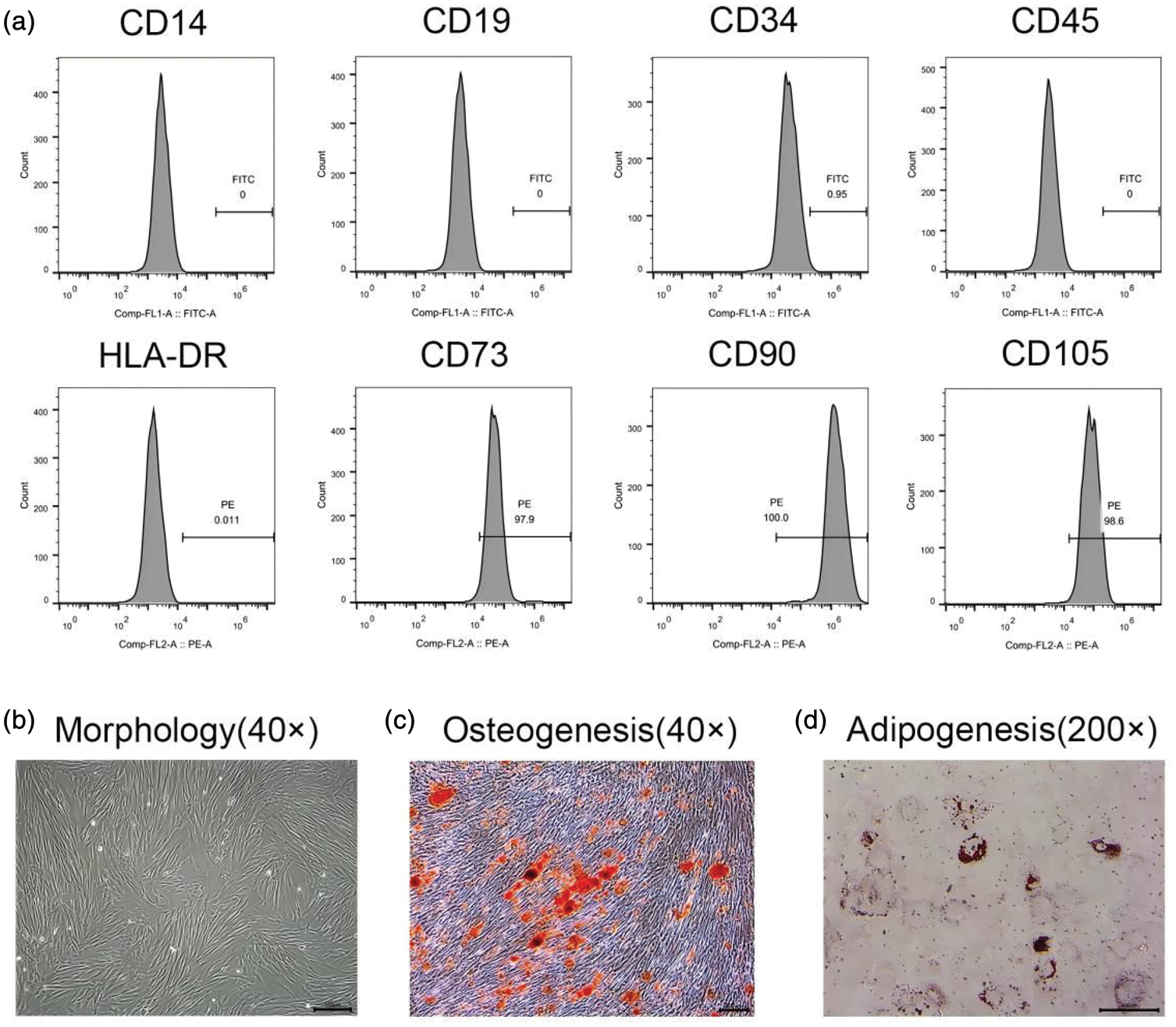
The characteristics of hUC-MSCs. (a) Fourth-passaged cells were analyzed for CD14, CD19, CD34, CD45, CD73, CD90, CD105, and HLA-DR using flow cytometry. (b) Fourth-passaged cells show typical fibroblast-shaped morphology. Alizarin Red S staining, and oil red O staining show that hUC-MSCs were induced into (c) osteogenic and (d) adipogenic lineages, respectively. hUC-MSCs: human umbilical cord-derived mesenchymal stem cells; HLA-DR: Human leukocyte antigen–DR isotype.
Robot-assisted CD and hUC-MSCs transplantation
Surgical path planning
After administration of general anesthesia, the patient was placed in the supine position. Disinfection included the ipsilateral superior iliac spine where the tracer is placed. X-ray images of the hip joint in the upright and lateral positions were observed using C-arm fluoroscopy.
This information was imported into the workstation planning software, and registration points were marked according to the prompts on the software interface. It must be ensured that the X-ray image contains all ten anchor points on the locator ruler. According to the preoperative X-ray and MRI results, the orientation of the insertion needle from the lower part of the femur greater trochanter to the center of the necrotic area of the femoral head was planned, and then the appropriate insertion point, angle, and length were calculated automatically by the software.
CD and hUC-MSCs transplantation
After the surgical path was planned, the robotic arm was operated, which automatically moved the guide sleeve to the skin surface according to the planned needle direction and insertion point.
The guide needle was inserted through this point and in that direction. The surgeon made an incision approximately 1–2 cm into the skin where the needle was inserted. According to the position of the femoral head and necrotic lesion measured by the robot, a Kirschner wire of appropriate length was drilled; then, the robot was moved to insert the soft tissue protective sleeve along the guide wire. In the soft tissue sleeve, the guide needle is drilled into the trephine. The solid trephine was removed and replaced with a hollow trephine. The surgeon followed the needle into the neck of the femur and femoral head through the hardened area around the necrotic area and into the necrotic area until approximately 5 mm below the articular surface. The CD tube was curetted to remove the necrotic tissue. The extracted hUC-MSCs, autogenous cancellous bone particles, and a calcium sulfate (CaSO4)–calcium phosphate (CaPO4) bone graft replacement (PRO-DENSE®, Wright Medical Technology™, Inc. Arlington, TN, USA) were fully mixed and sent with special compression bone grafts into the decompression tunnel, and C-arm X-ray was performed to ensure they reached the decompression bone passage (Figure 3). The total number of hUC-MSCs transplanted into the necrotic area was approximately 2 × 107.

Illustration of the robot-assisted CD. (a, b) Anteroposterior and lateral intraoperative fluoroscopic images of the hip. (c, d) After fluoroscopic imaging information was imported into the workstation planning software, surgical trajectory for guide wire was planned. (e) Guide wire was inserted. (f) Solid trephine was inserted along the guide wire. (g, h) Hollow trephine was inserted along the guide wire into the necrotic area of the femoral head. (i) Impaction cancellous bone grafting. CD: core decompression.
Traditional CD surgery
The direction, position, and depth of drilling holes were designed in advance according to the X-ray image, computed tomography (CT), or MRI and preoperative C-arm X-ray fluoroscopy results. The rest of the procedure is similar to that for robot-assisted CD.
Postoperative management
After the effect of anesthesia wears off, the affected limb can move freely without weight bearing. Both groups of patients were instructed to refrain from performing weight bearing activities for 3 months, followed by partial weight bearing for 3 months. Six months postoperatively, full weight bearing was permitted if the patient’s radiographs showed that the bone tunnel had formed and healed.
Observation indicators
Operation-related indexes
The two groups were compared in terms of operation time, number of guide wire attempts, number of intraoperative fluoroscopies, and intraoperative blood loss.
Efficacy evaluation
The VAS score and Harris hip score (HHS) were recorded preoperatively and at the final follow-up. All patients underwent at least two MRI examinations, one preoperatively and another at the final follow-up. The overall survival rate was calculated.
Measurement of the necrotic area
The necrotic lesion volume was measured using MRI. Images were acquired using MRI (Siemens MAGNETOM Prisma, static magnetic field strength 3.0 T) at an interval of 4 mm and a gap of 0.4 mm. The low-intensity area proximal to the band-like high-intensity region observed on the T2-weighted fat-suppressed coronal image was determined as the necrotic area (S) (Figure 4(a)), 23 which was measured in mm2 using the Picture Archiving and Communication System (PACS V6.1.4; manufactured by Chongqing Zhonglian Information Industry Co., Ltd.). Measurements were taken for the low-intensity areas on each slice (Sn mm2) (Figure 4(b)), and they were accumulated as the total volume (Vn, mm3). The volume of the necrotic lesion can be expressed using the following formula: Vn (mm3) = ∑(Sn × 4.4 mm). 24 All measurements were performed by two experienced physicians separately, and the average value was calculated.

Measure the necrotic area using MRI. (a) Coronal T2-weighted fat-suppressed MRI. (b) Measurement of the necrotic area of each layer using the software. MRI: magnetic resonance imaging.
Statistical analyses
All statistical analyses in this study were performed using Statistical Package for Social Sciences (SPSS) software (version 22.0, IBM SPSS, New York, NY, USA). Continuous variables were reported as mean and standard deviations values. Intergroup comparisons were performed using the unpaired t-test, while intragroup comparisons were performed using the paired t-test for normally distributed variables. Comparisons were performed for quantitative data using chi-square test analysis. p < 0.05 indicated statistical significance.
Results
The general characteristics of the two groups are presented in Table 1; no significant differences were observed.
Comparison of the general data between the two groups.

Values are reported as mean (standard deviation).
BMI: body mass index; ARCO: Association Research Circulation Osseous.
Operation-related indices are detailed in Table 2. The operation time in the observation group was 49.25 ± 2.21 min, while that in the control group was 65.83 ± 1.82 min. Compared with that in the control group, the operation time in the observation group was reduced by 25.19%, representing a statistically significant difference (p < 0.001). The guide wire insertion time in the observation group was 6.15 ± 0.57 min, while that in the control group was 23.5 ± 1.26 min; the difference between the two groups was statistically significant (p < 0.001). The number of intraoperative fluoroscopies was 13.25 ± 0.85 in the observation group and 23.78 ± 1.07 in the control group. This difference was statistically significant (p < 0.001). Intraoperative blood loss was 26.25 ± 2.11 mL in the observation group and 76.39 ± 3.74 mL in the control group. Compared with the control group, the intraoperative blood loss in the observation group was reduced by 65.64%. The difference between the two groups was statistically significant (p < 0.001).
Comparison of the operation-related indexes between the two groups.

Values are reported as mean (standard deviation).
Satisfactory outcomes were obtained in both groups. The mean HHS score of the control and observation groups increased significantly from 67.83 ± 6.55 and 68.95 ± 6.29 points, respectively, at baseline to 84.17 ± 5.09 and 87.9 ± 5.26 points, respectively, at the final follow-up (p < 0.05 and p < 0.05, respectively). The mean VAS score of the control and observation groups decreased significantly from 4.06 ± 0.62 and 4.25 ± 0.78 points, respectively, at baseline to 1.24 ± 0.48 and 1.23 ± 0.52 points, respectively, at the final follow-up (p < 0.05 and p < 0.05, respectively).
There were no significant differences in the preoperative HHS, VAS scores, and necrotic volume of femoral heads between the two groups. The HHS, VAS scores, and necrotic volume of femoral heads (Figure 5) at the final follow-up were also not significantly different between the groups. The effective rate in the observation group was 82.1%, which was higher than that in the control group (72.7%); however, this difference was not statistically significant (Table 3). We selected the conversion of THA as the final evaluation indicator, which represents an objective clinical failure state that warrants intervention treatment. 25

Regression of a necrotic lesion on MRI after robot-assisted CD combined with hUC-MSCs transplantation. (a) Preoperative MRI. (b) MRI at 12 months postoperatively. (c) X-ray taken at the final follow-up. CD: core decompression; hUC-MSCs: human umbilical cord-derived mesenchymal stem cells; MRI: magnetic resonance imaging.
Comparison of treatment efficacy between the two groups.

Values are reported as mean (standard deviation).
HHS: Harris hip score; VAS: visual analog scale.
Discussion
Once ONFH occurs, in most cases, the condition gradually deteriorates.25,26 There is no evidence that conservative treatment is effective in delaying ONFH progression; therefore, surgery to delay or even reverse disease progression has remained a topic of active research in the field of orthopedics.27,28 Arlet and Float first described CD, 29 which has proved to be an effective treatment for hip preservation.
Commonly, CD procedures include large incisions, tissue damage, and lengthy recovery duration. 30 The placement of a traditional CD guide needle mainly depends on intraoperative X-ray fluoroscopy, which often requires multiple adjustments to determine the direction and position of the guide needle, thereby increasing radiation exposure for both operator and patient as well as lengthening the operation time. 31 TiRobot™ was first introduced by Tian; 21 it is an orthopedic surgical robot that can be used to implant different guide wires and screws, especially for guide wire insertion of the proximal femur and spine. With the use of a robot-assisted system, the guide needle can be accurately placed into the necrotic area of the femoral head in a single attempt under robotic arm guidance, requiring only the anterior and lateral perspectives of the hip joint. Since the robotic system can provide the precise length of guide pin entry, it does not require repeated fluoroscopy to monitor the placement depth. 32 In our study, the operation time, blood loss, and number of intraoperative fluoroscopies in the observation group were less than those in the control group, and this difference was statistically significant.
During the initial use of orthopedic robots, there might have been some delay in preoperative planning; however, overall, the operation time was significantly reduced. 33 The operation time, number of intraoperative fluoroscopies, and blood loss in the robot-assisted surgery group were superior to those in the traditional surgery group. However, multiple fluoroscopies were still required during decompression and bone grafting because there was no reference point for determining the position of the drill and pressure bone graft in the femoral head. More importantly, the robot can determine the direction of the puncture and guide the surgeon precisely to the desired location. There was a correlation between the preoperative necrotic area of the femoral head and postoperative residual dead bone volume and failure rate. 34 However, it is not always better to remove as much dead bone tissue as possible during the surgery because it may cause postoperative femoral head collapse. Robot-assisted CD surgery can more accurately identify the depth and extent of necrotic areas. This helps preserve healthier bone tissue in the femoral head and reduces the possibility of femoral head collapse caused by excessive resection of necrotic lesions.
Robot-assisted CD has certain drawbacks. First, under the guidance of the robot arm, the number of X-ray fluoroscopy examinations was reduced during the placement of guide pins, however, during decompression and bone grafting, multiple fluoroscopies were still required to determine the position of the drill and pressure implant in the femoral head. Second, the preoperative necrotic area of the femoral head and residual necrotic bone volume were strongly correlated with the rate of surgical failure. Due to the defect of the decompression bit, there remained some necrotic areas for which effective intervention could not be performed.
CD can reduce the intraosseous pressure by decompression and open up the sclerosis zone that hinders osteonecrosis repair. 30 However, this repair is limited to a local area and does not comprehensively address the issue of femoral head repair. 35 Recent studies have shown that stem cells are capable of multiple differentiation and can be directed to differentiate into other cells under the action of specific cytokines, thereby promoting the repair of damaged tissues.36,37
MSCs are considered the perfect candidates for cell-based therapy and regenerative medicine.38,39 Some studies have pointed out that the use of autologous MSCs for patients with ONFH disease is feasible, safe in the long-term, and may improve survival.40,41 As an excellent representative of MSCs, hUC-MSCs have subomnipotent differentiation potential and can differentiate into several types of tissue cells such as the heart, liver, kidney, lung, pancreas, nerve, bone, muscle and fat, almost covering all the tissue cells in the human body. 42
In our study, hUC-MSCs were shown to differentiate into osteoblasts and adipocytes in vitro and be plastic-adherent under standard culture conditions. Flow cytometry confirmed the expression of CD105, CD73, and CD90 as well as the lack of expression of CD45, CD34, CD14, and human leukocyte antigen–DR isotype (HLA-DR) surface molecules. 32 Taken together, the hUC-MSCs in our study met the minimal criteria for cellular therapy. Intravenous delivery, targeted intra-arterial injection, and local injection are three methods for transplanting MSCs. Targeted intra-arterial injections appear to exert the optimal effect; however, it is clear that local injection offers maximum safety and tolerance. 43 Chen et al. 22 reported that intra-arterial infusion of hUC-MSCs is a feasible and relatively safe method for treating femoral head necrosis. In their study, the survival rate was 100% (5/5) in ARCO stage 2 patients and 75% (3/4) in ARCO stage 3 patients. Although the number of cases they reported was small, the results were encouraging. Notably, in our study, hUC-MSCs were not implanted through intra-arterial infusion as in their report. In our study, the survival rate in the observation group was 82.1% (23/28), which was lower than that in Chen et al.’s 22 report but slightly higher than that in the report by Bi, Zhang, and Zhao (75%, 12/16). 44 They also performed robot-assisted CD; however, no stem cells were implanted during the operation. However, owing to factors related to the study design, these results should be interpreted cautiously and regarded as observational rather than conclusive evidence. Due to the relatively small sample size and the fact that the two groups of patients were not at the same center, the role of stem cell implantation in the treatment of femoral head necrosis needs to be evaluated further. The number of stem cells implanted in the femoral head is an important factor that affects treatment efficacy. Based on current data, the injection of 106 to 109 cells may be reasonable. 18 However, it remains unclear how many stem cells should be implanted into the femoral head with varying degrees of injury for optimal results. Further research is needed to clarify this issue.
Limitations
Our study has certain limitations. First, the retrospective and nonrandom design of this study implies that the groups were formed based on clinical decisions rather than random allocation, which may have introduced unmeasurable confounding factors that could have influenced the results. 45 Therefore, these results should be interpreted cautiously. Second, the relatively small sample size lends limited statistical power to this study. Third, more groups should have been established to distinguish the roles of robot-assisted techniques and stem cell therapy in surgery. Finally, due to the heterogeneity in surgical techniques, even within the same group, minor differences in surgical techniques could not be completely avoided, which may account for part of the observed individual differences in therapeutic efficacy.
Conclusion
Although robot-assisted CD may offer advantages in terms of surgical precision, reduced trauma, and lower radiation exposure, it ultimately achieves clinical outcomes comparable with those of traditional surgery.
Footnotes
Acknowledgments
We are grateful to the operating room team at the Affiliated Jiangning Hospital with Nanjing Medical University for their collaboration and cooperation.
Author contributions
Chao Zhu and Xiaoli Fan designed the study; Xiaolong Jia, Lingyu Xin, Qin Hu, and Xin Zhou collected and analyzed the data; Xiaolong Jia, Qin Hu, Xudong Yan, and Hailong Zhang drafted the work; Xiaolong Jia and Lingyu Xin substantively revised the manuscript; Bin Wang was responsible for stem cell preparation; and Chao Zhu and Xiaoli Fan approved the final version of the manuscript. All the authors have read and approved the final manuscript.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
All authors declare they have no conflicts of interest.
Funding
This study was funded by grants from the Health Science and Technology development Special Fund Project of Nanjing, China (No. YKK20201), Youth Innovation and Research Fund of the Affiliated Jiangning Hospital with Nanjing Medical University (No. JNYYZXKY202410) and Nanjing Health Science and Technology Development Special Fund Project (No. ZKX22061).
