Abstract
Objective
This study aimed to identify the clinical risk factors for colorectal sessile serrated lesions and to develop a diagnostic model to facilitate clinical diagnosis.
Methods
Patients who underwent electronic colonoscopy during hospitalization between June 2021 and June 2024 were enrolled. General clinical data were collected for univariate and multivariate logistic regression analyses. A nomogram-based risk prediction model was developed using R language software.
Results
Compared with patients without abnormalities on colonoscopy, those with colorectal sessile serrated lesions showed statistically significant differences in male sex, history of smoking, history of hypertension, and higher body mass index (p < 0.05). Multivariate logistic regression analysis demonstrated that a history of smoking and hypertension were independent risk factors for colorectal sessile serrated lesions, with odds ratios of 3.761 (95% confidence interval: 1.795–7.880; p = 0.000) and 1.919 (95% confidence interval: 1.026–3.589; p = 0.041), respectively.
Conclusions
A history of smoking and hypertension were identified as independent risk factors for colorectal sessile serrated lesions. Increased attention should be given to screening for colorectal sessile serrated lesions in patients with these risk factors.
Introduction
The incidence of colorectal cancer (CRC) in China has shown a significant upward trend in recent years, ranking second among all malignant tumors. 1 A similar trend has been observed in the incidence of malignant tumors across Asia and globally,2,3 significantly affecting patients’ quality of life. Extensive studies on the incidence and progression of CRC indicate that, in addition to the adenoma–carcinoma pathway, CRC develops through the serrated pathway. The serrated pathway accounts for approximately 15%–30% of cases,4–10 with some studies reporting a prevalence exceeding 30%.9,11 Sessile serrated lesions (SSLs) and traditional serrated adenomas (TSAs) are precancerous lesions in the serrated carcinogenic pathway and are associated with a high malignancy rate. Particularly, some SSLs may rapidly progress to cellular dysplasia or invasive carcinoma.4,7,12 However, endoscopic diagnosis of SSLs is challenging because of their flat and sessile morphology, indistinct borders, and mucin cap, which make them difficult to distinguish from hyperplastic polyps and may result in incomplete resection. Additionally, its complexity makes diagnosis by endoscopy difficult.12–14 Therefore, early detection of SSLs is essential for timely diagnosis and treatment of CRC and for reducing mortality.13,15 This study retrospectively summarized the clinical characteristics of patients with colorectal SSLs and analyzed associated risk factors to improve gastroenterologists’ understanding and diagnostic ability regarding this disease.
Data and methods
General information
Data were collected from patients who underwent electronic colonoscopy during hospitalization in the Gastroenterology Department of Beijing Chuiyangliu Hospital between June 2021 and June 2024. Patients with a pathological diagnosis of SSLs were assigned to the case group (n = 99). To avoid dilution of the effect by including controls with other potentially confounding colorectal lesions (such as adenomas or hyperplastic polyps), patients with no detectable abnormalities on examination during the same period were randomly selected as the control group (n = 103). This study was approved by the Medical Ethics Committee of Beijing Chuiyangliu Hospital (Approval Number: 2025-019KY).
The inclusion criteria for the case group were as follows: (a) patients aged ≥18 years; (b) patients who underwent electronic colonoscopy in the Gastroenterology Department of Beijing Chuiyangliu Hospital with findings of colorectal polyps; and (c) patients with a pathological diagnosis of SSLs. SSLs were included regardless of size or location. The exclusion criteria were as follows: (a) patients with insufficient bowel preparation or incomplete colonoscopy for various reasons; (b) patients whose pathological diagnosis concurrently included adenomatous polyps; (c) patients with a history of intestinal diseases, including CRC, intestinal tuberculosis, Crohn’s disease, ulcerative colitis, familial adenomatous polyposis, or intestinal lymphoma; (d) patients with severe infection, organ failure, or other systemic diseases; (e) patients receiving hormones or immunosuppressants; and (f) patients with a history of hereditary CRC syndromes (e.g. familial adenomatous polyposis or Lynch syndrome).
The inclusion criteria for the control group were as follows: (a) patients aged ≥18 years and (b) patients who underwent electronic colonoscopy in the Gastroenterology Department of Beijing Chuiyangliu Hospital during the same period, with no abnormalities detected, including polyps, inflammation, or other lesions. The exclusion criteria were as follows: (a) patients with insufficient bowel preparation or incomplete colonoscopy for various reasons; (b) patients with a previous colonoscopy history of intestinal diseases, including colon polyps, CRC, intestinal tuberculosis, Crohn’s disease, ulcerative colitis, familial adenomatous polyposis, or intestinal lymphoma; (c) patients with severe infection, organ failure, or other systemic diseases; (d) patients receiving hormones or immunosuppressants; (e) patients with a history of hereditary CRC syndromes (e.g. familial adenomatous polyposis or Lynch syndrome).
Research methods
After admission, data were collected from all patients, including sex, age, body mass index (BMI), and medical history such as hypertension, diabetes, arteriosclerosis, dyslipidemia, smoking history, alcohol consumption, cholecystectomy, appendectomy, and other basic information. Additional indicators were also recorded, including fasting blood glucose, glycated hemoglobin, serum uric acid, and lipid profile. Colonoscopy was performed by an experienced senior attending physician or above in the Gastroenterology Department. Concurrently, the pathological report was issued by an attending physician or above in the Pathology Department. All pathological diagnoses were independently established by at least two experienced gastrointestinal pathologists (attending physician level or higher) who were blinded to the clinical data. Diagnoses were made according to the 2019 World Health Organization (WHO) classification of digestive system tumors. In cases of diagnostic disagreement, a consensus was reached through joint review. This process was implemented to ensure diagnostic accuracy and consistency for SSL and to minimize misclassification with hyperplastic polyps.
Statistical analysis
Statistical analysis was performed using Statistical Package for the Social Sciences software, version 26.0. Measurement data were presented as mean ± SD for normally distributed data and as median (interquartile range) for non-normally distributed data. Counting data were expressed as the number of cases (percentage). Continuous variables between the two groups were analyzed using the independent-samples t-test or the Mann–Whitney U test. The presence of colorectal SSLs was defined as the value of the state variable. Lower test values indicated more definite tests. Univariate and multivariate logistic regression analyses were performed. A two-sided p value <0.05 was considered statistically significant. The predictive model was constructed using R software (version 4.5.0) to generate a visual nomogram.
Results
Comparison of general data between the case group and the control group
The general characteristics (sex, age, and BMI), past medical history (smoking history, alcohol consumption, hypertension, diabetes, arteriosclerosis, dyslipidemia, and Helicobacter pylori infection), and laboratory indicators (fasting blood glucose, glycated hemoglobin, serum uric acid, and lipid levels) were compared between the two groups. Statistically significant differences were observed in the case group with respect to sex, BMI, smoking history, and hypertension history (p < 0.05) (Table 1). The proportion of male patients was significantly higher in the case group than in the control group (45.5% vs. 25.2%, p = 0.003). BMI was also significantly higher in the case group (24.6 kg/m2 vs. 23.5 kg/m2, p = 0.039). In terms of medical history, the proportion of patients with a smoking history was significantly higher in the case group than in the control group (33.3% vs. 11.7%, p < 0.001), and the proportion of patients with hypertension was also higher in the case group (38.4% vs. 24.3%, p = 0.030).
Comparison of general information between case and control groups.
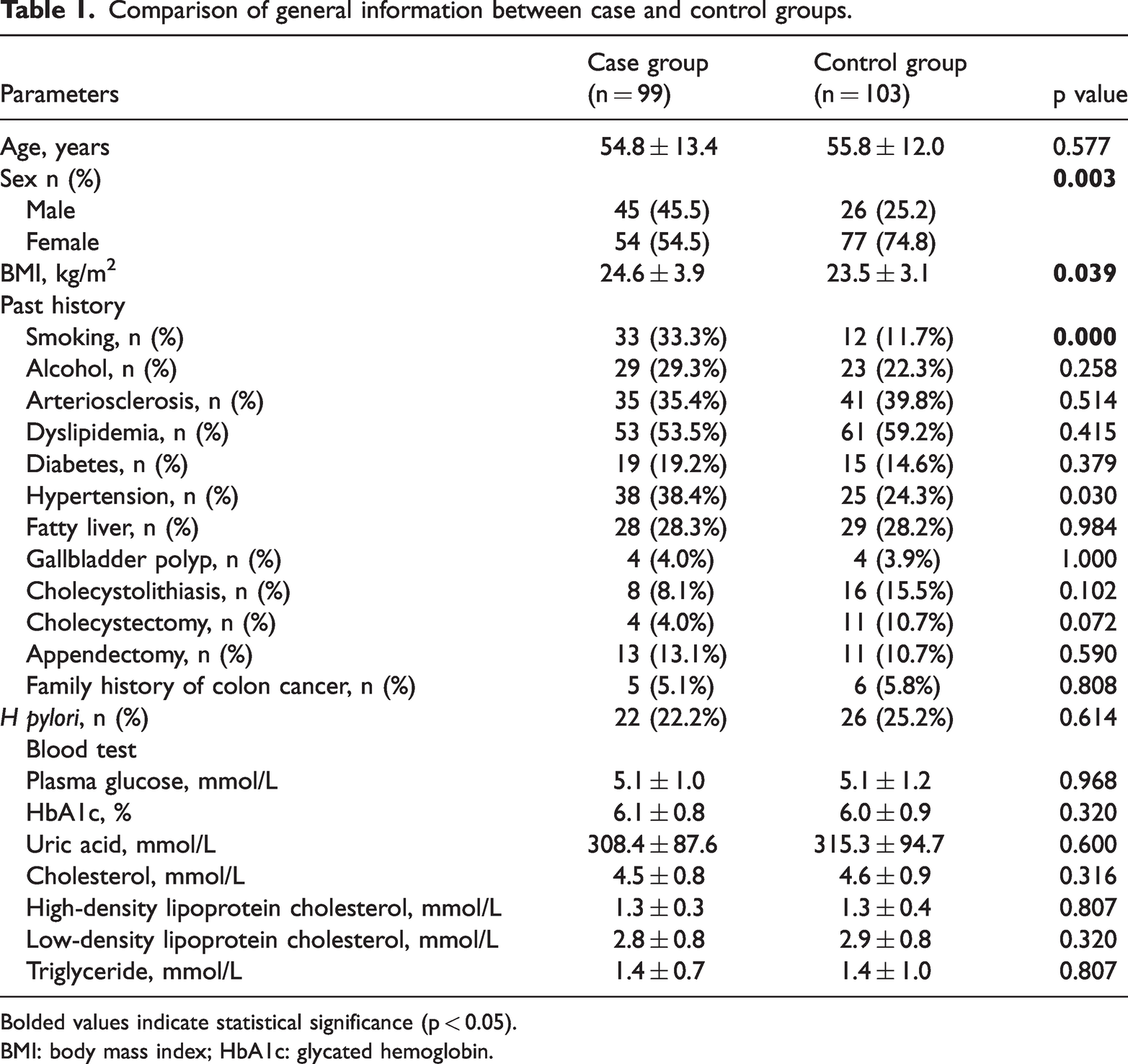
Bolded values indicate statistical significance (p < 0.05).
BMI: body mass index; HbA1c: glycated hemoglobin.
Analysis of risk factors for patients with SSLs
Differences in baseline characteristics were observed between the case and control groups. To account for these differences and control for potential confounding, multivariate logistic regression analysis was performed, incorporating variables that showed statistically significant differences in univariate analysis. Particularly, sex, smoking history, hypertension history, and BMI, factors with statistically significant differences (Table 1), were included as independent variables in the univariate and multivariate logistic regression analyses. The findings demonstrated that smoking history and hypertension history were independent risk factors for the development of colorectal SSLs, with odds ratios (OR) of 3.761 (95% confidence interval (CI): 1.795–7.880, p = 0.000) and 1.919 (95% CI: 1.026–3.589, p = 0.041), respectively (Table 2).
Univariate and multivariate logistic regression about analysis of risk factors for patients with SSLs.

BMI: body mass index; CI: confidence interval; OR: odds ratio; SSLs: sessile serrated lesions.
Establishment of a risk prediction model for colorectal SSLs
The data of the identified variables, including smoking history and hypertension history, were plotted using the R language software to predict the occurrence of colorectal SSLs. Patients were assigned a score of 1 for the presence of smoking or hypertension history and 0 for absence. SSL occurrence was coded as 1, whereas nonoccurrence was coded as 0. A higher total score indicated a greater predicted risk of SSL occurrence (Figure 1).

Nomogram for predicting colorectal sessile serrated lesions (SSLs). The data, including smoking and hypertension history, were plotted using the R language software to predict the occurrence of colorectal SSLs. Patients were assigned a score of 1 for the presence of a history of smoking and hypertension and 0 for absence. SSLs occurrence was coded as 1, whereas nonoccurrence was coded as 0. A higher the total score indicated greater predicted risk of SSL occurrence.
Performance of the prediction model
The diagnostic model demonstrated fair discriminative ability, with an area under the receiver operating characteristic (ROC) curve (AUC) of 0.644 (95% CI: 0.578–0.707), as shown in Figure 2.
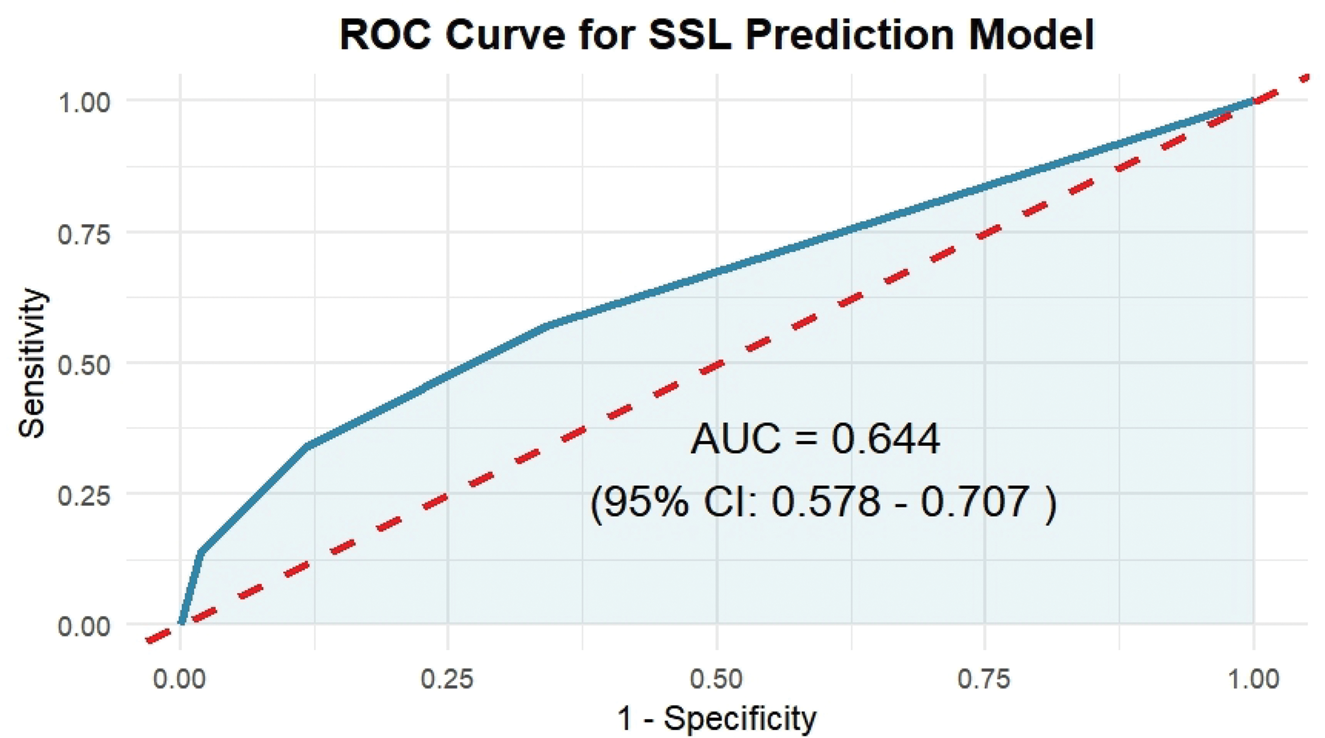
Receiver operating characteristic (ROC) curve of the diagnostic model for sessile serrated lesions (SSLs). The area under the ROC curve (AUC) was 0.644 (95% confidence interval: 0.578–0.707), indicating moderate discriminative ability. The dashed diagonal line represents the reference line for no discriminative ability (AUC = 0.5).
Calibration plot of the nomogram for predicting SSLs
The calibration plot (Figure 3) showed deviation from ideal calibration (Hosmer–Lemeshow test, p < 0.05). However, the model maintained a strong monotonic relationship, with higher nomogram scores consistently associated with increased risk of SSL, supporting its potential value for risk stratification.

Calibration plot of the nomogram for predicting sessile serrated lesions. The solid black line represents the apparent calibration curve, the blue line represents the bootstrap bias-corrected calibration curve, and the dashed red line represents the ideal calibration.
Discussion
The incidence of CRC increases annually. 1 It is the most common malignant tumor of the digestive system and ranks second only to lung and breast cancers in the global incidence of malignant tumors. 2 In China, its incidence is second only to that of lung cancer. 1 However, in recent years, the widespread implementation of colonoscopy screening and the integration of prevention and treatment strategies have significantly improved the 5-year survival rate of patients with CRC worldwide and in China.16,17
Colorectal SSLs represent an important pathway in the development of CRC and are associated with a high rate of malignant transformation.4–7 Early intervention can significantly improve patient survival and quality of life. Additionally, it can alleviate pain in patients, shorten recovery time, and decrease the economic and psychological burden on families as well as the economic burden on society. 17
Colorectal serrated polyps were reclassified in 2019. 18 Therefore, we selected the reclassified medical records for statistical analysis. The indicators included in this study were chosen based on established and controversial factors reported in previous literature to identify variables associated with colorectal SSLs. Prior studies suggest that hypertension, diabetes, dyslipidemia, smoking history, arteriosclerosis, cholecystectomy, and appendectomy are risk factors for SSLs.14,15,19–26 Accordingly, these variables were included in this study for statistical analysis. Gallbladder stones, gallbladder polyps, fasting blood glucose, glycated hemoglobin, and serum uric acid, were incorporated as additional indicators.
Our study used a case–control design, which inherently resulted in differences between groups for several risk factors. The higher prevalence of smoking, hypertension, and male sex in the case group supports their association with SSL development. Importantly, multivariate analysis demonstrated that smoking and hypertension remained independently significant after adjustment for these baseline differences, thereby strengthening the validity of our findings.
The male predominance observed in our SSL cohort (45.5% vs 25.2% in controls) contrasts with patterns reported in some Western populations, where SSLs often show an equal sex distribution or female predominance.9,26 This discrepancy may reflect ethnic differences in the prevalence of risk factors, particularly the higher smoking rates among Chinese males, or differences in hormonal factors across populations. Our findings underscore the importance of developing population-specific risk stratification models.
The results demonstrated that a history of smoking (OR: 3.761, 95% CI: 1.795–7.880, p = 0.000) and a history of hypertension (OR: 1.919, 95% CI: 1.026–3.589, p = 0.041) were risk factors for colorectal SSLs, which differ from the findings of previous studies.
A study demonstrated that molecular pathological features, including microsatellite instability (MSI), CpG island methylator phenotype (CIMP), and DNA mutations such as BRAF and KRAS, are widely recognized as being associated with smoking and CRC. Given that MSI-high and CIMP-positive status are significant features of CRC arising through the serrated pathway, the strong association observed between smoking and SSLs is not unexpected. 27
The inclusion of hypertension in our prediction model warrants careful consideration. Although it emerged as a statistically significant factor in our cohort (OR: 1.919, 95% CI: 1.026–3.589), the literature presents inconsistent findings. Some studies have reported an association between hypertension and SSLs, 14 whereas others conducted in different populations have not confirmed this relationship. 24 This discrepancy suggests that hypertension may act as a marker of underlying metabolic syndrome or insulin resistance rather than as a direct causal factor in SSL pathogenesis. A study from Romania indicated that angiotensin-converting enzyme inhibitors and angiotensin II type 1 receptor blockers may slow tumor growth and tumor-associated angiogenesis; however, further research is needed to clarify their effects on SSLs. 14
At present, few risk prediction models are available for colorectal SSLs.14,15,19–26 In this study, independent risk factors identified through logistic regression analysis were used as predictors to construct a diagnostic model. Based on the nomogram, the total score for the predictors included in the multivariate analysis can be calculated, and the corresponding probability of colorectal SSL occurrence can then be estimated according to this total score.
Although our model demonstrated statistically significant miscalibration, its clinical value remains as a rapid screening tool. The nomogram effectively identifies patients who warrant more meticulous colonoscopic examination, particularly in the right colon, where SSLs are frequently missed. Future studies with larger cohorts may improve probability calibration and preserve the model’s practical simplicity. For example, in a patient with a history of smoking and hypertension, with scores of 100 and 50, respectively, and a total score of 150, the nomogram indicates that the probability of SSL occurrence is >60%, enabling clinicians to represent the risk of SSLs more intuitively.
This study suggests that, in clinical practice, patients with a history of smoking and hypertension should receive education and guidance to modify their lifestyles, including smoking cessation, blood pressure control, and completion of colonoscopy examinations. Notably, smoking and hypertension are also established risk factors for conventional colorectal adenomas. Therefore, our model helps identify patients at high risk for significant colorectal neoplasia overall, with particular attention to the often-missed SSLs. The clinical utility of the model lies in its ability to flag these high-risk patients for more meticulous colonoscopic evaluation, especially in the right colon, where SSLs are prevalent. Studies have shown that SSLs most commonly occur in the right colon.8,26 During colonoscopy, special attention should be directed toward detecting SSLs in this region. Early diagnosis and timely treatment are essential for improving the prognosis of CRC and reducing its disease burden. Although sedated (painless) colonoscopy is widely available, it remains an invasive procedure that requires bowel preparation, which many patients find burdensome. Therefore, many patients have concerns about this examination. Consequently, this study serves as an early warning regarding the occurrence of SSLs in the colorectal population, supports clinical risk assessment, and improves the precision of identifying high-risk groups for endoscopic screening.
This study has several limitations. First, it is a retrospective single-center study, which may affect the research results. Second, we did not further investigate the association between smoking quantity, smoking duration, control of hypertension, risk stratification, and the occurrence of colorectal SSLs. Future studies should incorporate quantitative indicators, such as smoking pack-years and detailed information on antihypertensive treatment, to enhance the model’s precision. Our model showed evidence of miscalibration, which limits the interpretation of absolute predicted probabilities. However, its ordinal risk stratification capability remains clinically valuable for identifying high-risk individuals. Therefore, future prospective, multicenter studies with larger sample sizes are warranted to validate and refine our model. Our model should be considered an initial framework, and future models should incorporate other established (e.g. BMI and age) and emerging risk factors (e.g. genetic markers) to improve predictive performance. Therefore, it is necessary to expand the sample size and formulate a prospective plan to further verify these findings and increase the predictive value of the risk model.
This study statistically analyzed the risk factors for colorectal SSLs and developed a noninvasive diagnostic model. This model guides physicians’ diagnosis and treatment in clinical practice, assists in clinical risk assessment, and enables more accurate endoscopic screening of high-risk populations. In addition, it provides individualized recommendations for high-risk patients, including completion of colonoscopy. Enhancing patient awareness of colorectal SSLs may help reduce the incidence and mortality of CRC, which is of importance to patients, their families, and society.
Footnotes
Acknowledgments
We thank Yanfei LI (China) for the kind assistance in providing statistical analysis.
Author contributions
WEI S and DONG LF designed the research; WEI S and Wang L wrote the paper; JIANG DJ analyzed the data; HU YJ contributed new analytic tools; LI HR performed the research. All authors have read and approved the final manuscript.
Data availability statement
Any additional data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
All the authors have read and agreed with the contents of the manuscript, and there are no conflicts of interest to disclose.
Supportive foundations
This study was supported by the Special Research Project of the Capital’s Health Development (No. 2024-3-7037).
