Abstract
Respiratory monitoring is important in amyotrophic lateral sclerosis patients as progressive respiratory muscle weakness often leads to respiratory failure. We performed single-case feasibility evaluation of long-wave infrared imaging for noncontact respiratory rate estimation in a hospitalized amyotrophic lateral sclerosis patient and compared it with red–green–blue camera-based optical flow analysis and electrocardiogram-derived respiration. Respiratory rate was assessed using four methods: (a) manual counting as the ground truth; (b) long-wave infrared; (c) red–green–blue; and (d) single-lead electrocardiogram under three lighting conditions (regular, dim, and dark) with three 1-min trials per condition. Estimation accuracy was evaluated using mean absolute error relative to manual counting. Long-wave infrared imaging demonstrated the highest accuracy across all lighting conditions, with mean absolute errors of 1.67, 0.33, and 2.33 breaths per min, respectively. Red–green–blue-based estimation performed moderately well under regular and dim lighting but showed reduced accuracy in darkness, including one failed trial. Electrocardiogram-derived respiration showed the lowest accuracy and greatest variability across conditions. The limitations of the present study include its single-case design and absence of objective flow-based respiratory reference signals. However, this study demonstrates the feasibility and illumination robustness of long-wave infrared-based noncontact respiratory rate estimation in a hospital environment and supports further investigation of this approach for respiratory monitoring in amyotrophic lateral sclerosis patients.
Introduction
Respiratory rate (RR) is fundamental to respiratory assessment. 1 As one of the primary vital signs, alongside body temperature, blood pressure, and heart rate, it serves as a crucial indicator of a patient’s physiological status. 1 RR often reflects early signs of health deterioration, such as hypoxia, sepsis, and respiratory insufficiency, even before changes in oxygen saturation or blood pressure become apparent. 2
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease characterized by the loss of motor neurons in the brain and spinal cord. 3 Its estimated annual incidence is approximately 1–2 cases per 100,000 individuals worldwide, and the reported prevalence is 4–6 per 100,000 individuals. 3 It predominantly occurs during mid-to-late adulthood. As ALS advances, respiratory muscle weakness leads to ventilatory failure, which accounts for approximately 80% of deaths in patients with ALS. 4 Accordingly, monitoring respiratory function is central to ALS care. RR, in particular, is an accessible metric that can guide timely interventions and clinical decision-making. 5 Accurate RR measurement in ALS is therefore essential in clinical practice.
Visual inspection of chest wall motion is commonly performed to assess RR; however, this method is inaccurate and unsuitable for continuous bedside monitoring. Electrocardiogram (ECG) sensors can also be used to estimate RR. 6 Since respiration influences ECG waveforms, RR can be inferred from respiration-induced alterations; 6 however, this approach yields indirect estimates and often has limited accuracy. Imaging-based methods provide a direct alternative. Using red–green–blue (RGB) cameras, chest wall expansion and contraction can be tracked via optical flow to derive respiration patterns; however, performance is sensitive to illumination, especially under low-light or dark conditions. 7
Recent advances in the commercialization and accuracy of long-wave infrared (LWIR) imaging have mitigated RGB cameras’ sensitivity to variations in illumination, enabling more reliable RR estimation. Consequently, a growing body of work has investigated LWIR imaging as a robust tool for respiratory monitoring in clinical settings.8,9
This work aimed to explore the feasibility of LWIR imaging as a noncontact method for RR monitoring in a patient with ALS. Given the limitations of effort-dependent respiratory assessments and illumination-sensitive imaging techniques in advanced neuromuscular disease, this case report also aimed to examine whether LWIR imaging can yield stable RR measurements under typical inpatient lighting conditions and hence elucidate the potential role of contactless respiratory surveillance in ALS care.
Case report
Patient
We describe the case of a 60-year-old man who had been diagnosed with ALS 1 year ago. He did not have a significant medical history except ALS and was not on regular medication. Our study was approved by the local institutional review board of Yeungnam University Hospital, Yeungnam University, Daegu, South Korea (approval number 2025-03-062; approved 3 April 2025). Written informed consent for publication of this case report was obtained from the patient. The reporting of this study conforms to the Case Report (CARE) guidelines. 10
The patient provided informed consent for study participation and case publication. This work complies with the Declaration of Helsinki (1975, as revised in 2024) and was approved by the institutional review board. The patient was admitted to the Department of Rehabilitation Medicine at a university hospital for respiratory evaluation. His motor strength was 2/5 on the Medical Research Council scale in both upper and lower limbs. He required caregiver assistance for wheelchair mobility. A restrictive ventilatory pattern was detected on pulmonary function testing during hospitalization. His forced vital capacity (FVC) was 1.89 L (46% of predicted) at baseline and 1.75 L (43% of the predicted value) after bronchodilator administration. The forced expiratory volume in 1 s (FEV1) was 1.43 L (45% of predicted), with a preserved FEV1/FVC ratio of 0.88 (116% of predicted). The diffusing capacity for carbon monoxide was 12.82 mL/min/mmHg (60% of the predicted value).
Implemented sensors
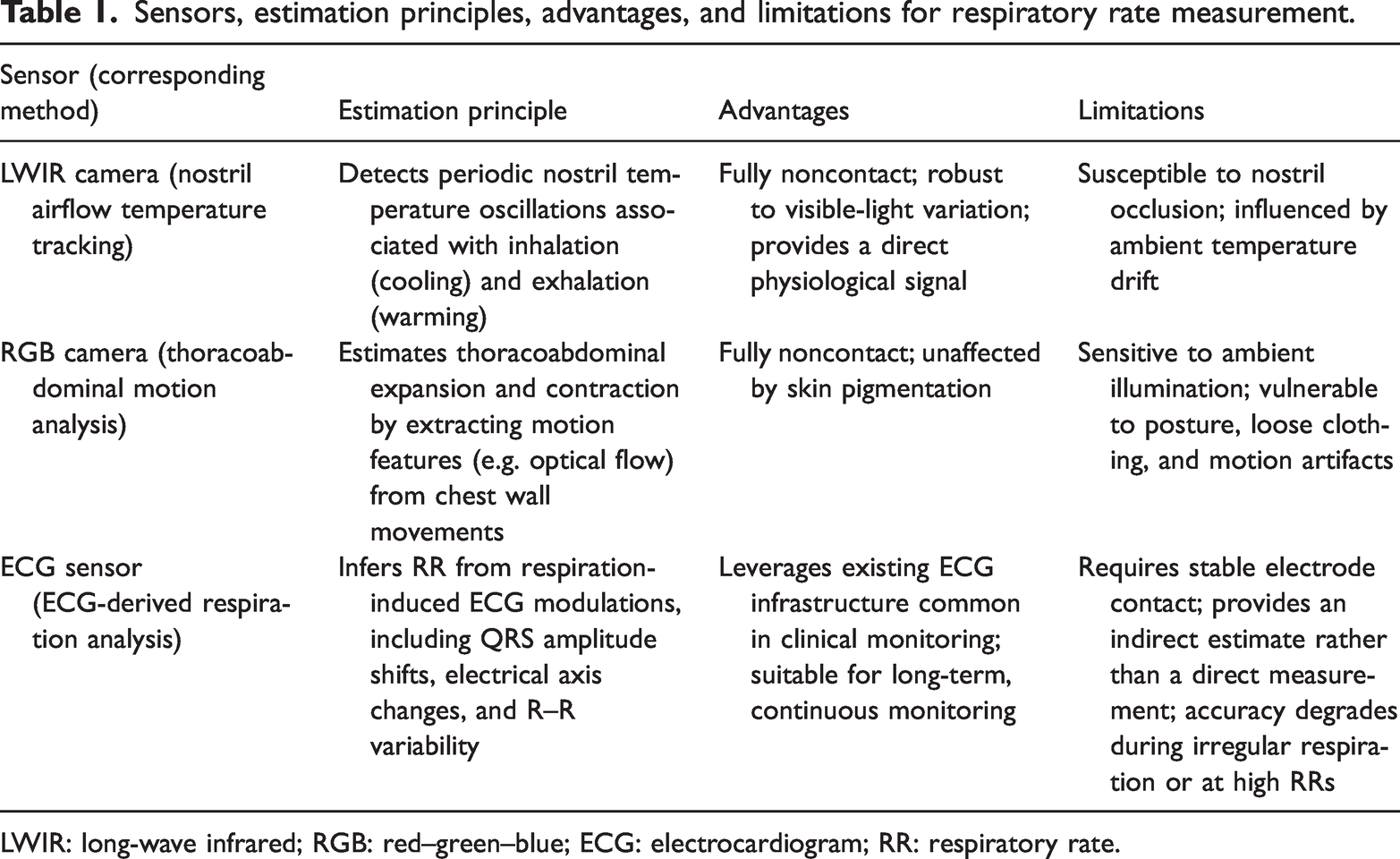
A single clinician obtained RR manually by counting chest wall expansions and contractions. In addition, three sensing modalities were used to estimate RR. All sensor recordings were manually synchronized. Estimation procedures for each modality are described below and summarized in Table 1.
Sensors, estimation principles, advantages, and limitations for respiratory rate measurement.
LWIR: long-wave infrared; RGB: red–green–blue; ECG: electrocardiogram; RR: respiratory rate.
LWIR camera (nostril airflow temperature tracking). The LWIR camera was aimed at the nostrils to capture cyclic respiration-related thermal fluctuations. Inspiration produced relatively cool airflow and expiration warm airflow, yielding a physiologically direct signal. 11 After basic denoising, temperature oscillations were extracted as a periodic waveform, and RR was computed as breaths per min (brpm). This method enabled contactless measurement and remained robust under dim or dark lighting conditions. However, accuracy can reduce due to partial nostril occlusion, and recordings can be influenced by ambient temperature drift. 12
RGB camera (thoracoabdominal motion analysis). Thoracoabdominal motion was monitored using an RGB camera by computing dense optical flow within a predefined chest–abdominal region of interest. The motion signal was postprocessed, and RR was derived from peak detection and conversion to brpm. This approach was fully contactless and unaffected by skin pigmentation, making it broadly applicable. However, it was highly sensitive to ambient illumination, with performance deteriorating under dim or dark lighting conditions. 7 In addition, motion artifacts from posture changes, loose clothing, or minor movements can distort the waveform and reduce reliability. 13
ECG sensor (ECG-derived respiration). Respiration was estimated from a single-lead ECG signal using the ECG-derived respiration method. R-peaks were identified, and three respiration-modulated features were extracted: (a) beat-to-beat QRS amplitude variation; (b) shifts in the electrical axis; and (c) R–R interval variability associated with respiratory sinus arrhythmia. The most strongly modulated feature was selected, and its dominant frequency was converted to brpm. This technique leveraged widely available ECG hardware, making it well suited for continuous, long-term monitoring. However, as an indirect measure of RR, accuracy could have decreased due to irregular breathing, and reliable measurements require stable chest electrode contact.
Experimental environment
All experiments were conducted in a patient ward at a university hospital. The participant laid supine and was instructed to breathe naturally while recordings were made under three illumination settings. For creating regular-light condition, all standard room lights were kept on, representing typical ward brightness. For creating dim light condition, the main room lights were switched off, and only a small caregiver night lamp remained, as is common in hospitals at nighttime. For creating dark condition, all the room lights were switched off, and illumination was limited to faint emissions from medical device power-indicator light-emitting diodes. To preserve relative brightness differences across conditions, RGB camera parameters, including ISO (sensor sensitivity) and exposure time, were fixed throughout the study period. With these settings, the average brightness in the dim light condition was approximately 75% of the regular-light condition and that of the dark condition was approximately 10%, as assessed using the mean pixel intensity of the recorded video. For each illumination condition, three independent trials were conducted, with the patient performing 1 min of natural breathing per trial.
The LWIR camera was directed at the face, focusing on the nostril area to track airflow-related temperature changes, and the RGB camera was oriented toward the thoracoabdominal region to capture respiratory motion. Both cameras were positioned approximately 2 m from the patient. The ECG device was attached to the chest with electrode patches and supplemented with a finger-contact sensor, displaying signals in real time. An overview of the experimental setup is shown in Figure 1, and sensor specifications are listed in Table 2.

Experimental setup and sensor locations. (a) The LWIR camera imaged the patient’s face, centered on the nostrils. (b) The RGB camera imaged the patient’s body, centered on the chest. (c) The ECG sensor was attached to the patient’s body; an auxiliary sensor was placed on the finger. LWIR: long-wave infrared; RGB: red–green–blue; ECG: electrocardiogram.
Devices and key technical specifications for sensors used to measure the respiratory rate.

LWIR: long-wave infrared; RGB: red–green–blue; ECG: electrocardiogram; EDR: ECG-derived respiration; VGA: video graphics array; NETD: noise-equivalent temperature difference; FHD: full high definition; MP: megapixel; BSI-CMOS: back-side-illuminated complementary metal–oxide–semiconductor; HDMI: high-definition multimedia interface; EVF: electronic viewfinder; TFT: thin-film transistor; NIBP: noninvasive blood pressure; fps: frames per second; USB: universal serial bus; SpO2: blood oxygen saturation; FLIR: forward looking infrared.
Evaluation metric
For the comparison of each estimation’s accuracy, manual counting was initially performed by a single trained medical staff member who visually observed the chest wall movement. However, respiratory counting by a single observer may introduce uncertainty, particularly under low-illumination or dark conditions, where visual assessment becomes more challenging. As such, the respiratory scenes simultaneously recorded by the RGB and LWIR cameras were retrospectively reviewed and manually re-analyzed by medical staff to mitigate this limitation and reduce potential counting errors. This secondary review was conducted independently of the automated algorithm outputs and specifically identified respiratory cycles based on visible thoracoabdominal motion (RGB) and airflow-related thermal oscillations at the nostrils (LWIR). A more reliable reference RR was determined for each trial by cross-validating respiratory motion in both imaging modalities. We used the mean absolute error (MAE), with manual counting as the ground truth (GT). MAE was defined as follows:

Results
In regular-light conditions, manual counting yielded RR values of 22, 18, and 16 brpm; LWIR imaging reported 21, 19, and 19 brpm; RGB imaging estimated 22, 18, and 26 brpm; and ECG recorded 24, 22, and 25 brpm. Among all methods, LWIR showed the smallest deviation from manual counting, RGB showed moderate agreement, and ECG showed the largest deviations. The MAE was 1.67 brpm for LWIR, 3.33 brpm for RGB, and 5.00 brpm for ECG, indicating that LWIR provided the most accurate estimates under regular-light conditions (Table 3).
RR measured using manual counting, LWIR camera, RGB camera, and ECG sensor under three illumination conditions. Relative light intensities for dim light and dark conditions were approximately 75% and 10% of regular light, respectively.

X: dim lighting but showed reduced accuracy in darkness, including one failed trial.
LWIR: long-wave infrared; RGB: red–green–blue; ECG: electrocardiogram; RR: respiratory rate; MAE: mean absolute error; brpm: beam rotations per min.
In dim light conditions, manual counting recorded RR values of 18, 23, and 22 brpm; LWIR imaging reported 19, 23, and 22 brpm; RGB imaging estimated 18, 22, and 22 brpm; and ECG measured 25, 22, and 22 brpm. Under dim light, both LWIR and RGB results were nearly identical to manual counts, demonstrating robustness despite reduced illumination. The corresponding MAE values were 0.33 brpm for LWIR, 0.33 brpm for RGB, and 2.67 brpm for ECG. These findings suggest that night time illumination reductions in hospital wards had little influence on camera-based accuracy, with performance close to the GT value (Table 3).
In dark conditions, manual counting yielded RR values of 17, 17, and 20 brpm, while LWIR maintained stable performance, reporting 21, 19, and 21 brpm. Only two RGB recordings produced valid values—9 and 16 brpm—because the remaining trial failed to detect the patient owing to insufficient image contrast. ECG varied more widely, yielding values of 12, 26, and 25 brpm. Thus, LWIR was least affected by the absence of visible light, whereas RGB performance degraded substantially in darkness. The MAE values were 2.33 brpm for LWIR, 4.50 brpm for RGB (two valid trials), and 6.33 brpm for ECG, underscoring the reliability and accuracy of LWIR measurements in dark environments (Table 3).
Discussion
This single-case feasibility study examined the feasibility of LWIR-based RR estimation in a hospitalized patient with ALS under varying illumination conditions and compared it with RGB-based imaging and ECG-derived respiration.
Under all illumination settings, LWIR-based RR estimation demonstrated more stable signal extraction and smaller absolute deviations from manual counting compared with RGB and ECG-derived approaches. By tracking cyclic temperature fluctuations associated with nasal airflow, 11 LWIR imaging provides a physiologically direct signal that is inherently independent of the illumination condition, a particularly relevant property, considering the dark condition used for evaluation in this study.
The RGB camera relied on optical flow tracking of thoracic wall motion, which requires good illumination, and therefore failed under dark conditions. 14 Moreover, in the third regular-light trial, the patient’s movement appeared to alter clothing arrangement, thereby disrupting chest-motion tracking and potentially leading to RR overestimation. 11
The ECG-derived approach estimates RR from respiration-modulated features in the ECG signal, enabling continuous monitoring with standard clinical hardware. However, its accuracy can decline with irregular breathing and it requires stable electrodes placed on the chest. In ALS patients, weakened respiratory muscles and irregular chest movements reduce signal reliability, while posture changes and variations in electrode–skin contact introduce additional instability. These limitations explain why the ECG-derived RR estimation was substantially less accurate and consistent than the imaging-based estimations.
To the best of our knowledge, this study represents the first feasibility evaluation of an inpatient with ALS in a real hospital ward that directly compared multiple RR estimation approaches, including LWIR-based imaging, RGB-based imaging, and ECG-derived respiration, within the same recording sessions. In particular, the inclusion of ECG-derived respiration obtained from routinely used bedside monitoring equipment provides a clinically relevant point of comparison, reflecting the most accessible respiratory surrogate available in everyday inpatient care.
This study was based on a single patient who had ALS and underwent a limited number of short recording sessions, which precluded statistical inference, generalizability, and subgroup analyses. The study was conducted as a feasibility evaluation with careful experimental control; however, its single-case design remained an inherent constraint. In addition, manual RR counting was performed by a single observer using objective respiratory reference standards; consequently, it likely introduced observer-related variability or counting errors. Although visual counting is commonly used in clinical practice, it remains an imperfect reference standard, particularly under low-light conditions. It is also restricted to MAE because the reference data consisted of discrete respiration counts rather than continuous respiratory waveforms suitable for agreement or variability analysis. Although MAE provides a straightforward and clinically interpretable measure of absolute differences in 1-min RR estimates, it does not capture respiratory waveform characteristics such as airflow dynamics and breath-to-breath variability.
Conclusion
In this single-case feasibility evaluation, LWIR-based imaging demonstrated technically more stable RR estimation in a hospitalized patient with ALS and maintained better performance in varying illumination conditions, including low-light and dark environments, compared with RGB-based imaging and ECG-derived respiration. Although the findings were limited by the single-case design, our findings suggest that LWIR imaging can contribute to the development of robust, noncontact respiratory monitoring technologies for patients with ALS in real-world inpatient settings. Further studies with larger cohorts and objective respiratory reference standards should be performed to extend and validate these preliminary observations.
Footnotes
Acknowledgments
None.
Author contributions
Jaeho Kim, Sungho Kim, Sreya Deb Sreshta, Uday Debnath, Yoo Jin Choo, and Min Cheol Chang conceived and designed the case report, collected and analyzed data, drafted the manuscript, and performed substantive revisions. All authors have read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed during the management of the patient reported herein are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare no conflicts of interest.
Ethics statement
The participant provided informed consent for participation and publication of this case report. This work was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the institutional review board.
Funding
This work was supported by a National Research Foundation of Korea grant funded by the Korean government (MSIT) (No. RS-2023-00219725).
