Abstract
Objective
This study aimed to evaluate the potential of hepatocyte-derived fibrinogen-related protein 1 (HFREP1) as a biomarker for community-acquired pneumonia and community-acquired pneumonia secondary to sepsis.
Method
This cross-sectional observational study included 124 patients with community-acquired pneumonia, 52 patients with community-acquired pneumonia–sepsis, and 123 healthy controls. Clinical data were collected, including procalcitonin and C-reactive protein levels. Serum HFREP1 concentrations were measured, and statistical analyses were performed.
Result
HFREP1 levels differed significantly among the healthy control, community-acquired pneumonia, and community-acquired pneumonia–sepsis groups (37.2 vs. 211.6 vs. 696.8 ng/mL). Binary logistic regression analysis identified procalcitonin and HFREP1 as independent predictors of community-acquired pneumonia–sepsis, while C-reactive protein was not an independent predictor. The area under the curve for HFREP1 in distinguishing community-acquired pneumonia patients from healthy controls was 0.8810. For differentiating community-acquired pneumonia–sepsis from community-acquired pneumonia, the area under the curves were 0.8814 for HFREP1, 0.8167 for C-reactive protein, and 0.8902 for procalcitonin.
Conclusion
HFREP1 may serve as a biomarker for community-acquired pneumonia and community-acquired pneumonia–sepsis. In community-acquired pneumonia–sepsis, HFREP1 was strongly correlated with C-reactive protein and procalcitonin, and its diagnostic performance was comparable to that of procalcitonin.
Keywords
Introduction
Community-acquired pneumonia (CAP) is an inflammation of the lung parenchyma that occurs outside the hospital, including cases that develop within the usual incubation period after admission due to infection with pathogens acquired in the community. CAP remains a leading cause of hospitalization and mortality worldwide. 1 The global incidence of CAP is estimated at 1.5–14 per 1000 population.2–3 The age-standardized incidence and mortality rates of lower respiratory tract infections, including pneumonia and bronchiolitis, are 6295 and 34.3 per 100,000 population, respectively. Lower respiratory tract infections affect approximately 489 million individuals worldwide and account for 2.6 million deaths per year, ranking as the fourth leading cause of death globally. 4 The diagnostic criteria for CAP recommended by the National Institute for Health and Clinical Excellence (NICE) include clinical features consistent with an acute lower respiratory tract infection, radiographic confirmation on chest X-ray with exclusion of alternative diagnoses, and onset in the community. 5 According to Sepsis-3.0, sepsis is defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection, with diagnosis based on evidence of infection-associated organ dysfunction. 6 More than one-third of patients with CAP develop severe sepsis early in the disease course. 7 Early identification of CAP and secondary sepsis enables timely treatment and may limit disease progression.
Inflammatory markers and peripheral blood biomarkers, such as procalcitonin (PCT) and C-reactive protein (CRP), play an important role in the early detection and assessment of CAP and sepsis.8–9 However, no single biomarker can independently establish the diagnosis of CAP. 10 Similarly, available biomarkers for sepsis have limitations, including high false-positive rates and limited value for early diagnosis. Hepatocyte-derived fibrinogen-related protein 1 (HFREP1), also known as FGL1 or hepassocin, is a hepatocyte-secreted protein with mitogenic activity. 11 Previous studies have shown that HFREP1 expression is upregulated in the serum of patients with non-small cell lung cancer, 12 and it has also been reported as a potential biomarker in benign conditions, including autoimmune diseases, infectious diseases, and acute inflammatory states. 13 Current clinical investigations of HFREP1 as a biomarker have mainly focused on cancer and autoimmune disorders, such as rheumatoid arthritis, hepatocellular carcinoma, and lung adenocarcinoma.14–16
This study aims to evaluate the potential of HFREP1 as a biomarker in CAP and CAP secondary to sepsis (CAP–sepsis) and to examine the associations among HFREP1, CRP, and PCT during the progression from CAP to sepsis. In addition, we assess the combined diagnostic efficacy of HFREP1, CRP, and PCT to improve the serological diagnosis of CAP–sepsis.
Methods
Ethical approval
This cross-sectional observational study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024). The study was approved by the Ethics Committee of the Affiliated Hospital of Nanjing University of Traditional Chinese Medicine (approval No. 2024NL-321-02; approved on 9 January 2025). Written informed consent was obtained from all participants, and all data were de-identified.
Research objectives
A total of 124 patients with CAP, 52 patients with CAP secondary to sepsis (CAP–sepsis), and 123 healthy controls were consecutively recruited at Jiangsu Province Hospital of Chinese Medicine between January 2025 and March 2025.
Inclusion criteria
The inclusion criteria were as follows:
CAP group. Patients aged ≥18 years, regardless of sex, who met the diagnostic criteria for community-acquired pneumonia. CAP–sepsis group. Patients aged ≥18 years, regardless of sex, who met the diagnostic criteria for both CAP and sepsis. Healthy control group. Individuals aged ≥18 years, regardless of sex, without evidence of acute or chronic infection.
Exclusion criteria
The exclusion criteria were as follows:
Acute infection within the preceding month other than CAP; Chronic inflammatory or metabolic diseases (e.g. renal infection, pancreatic infection, diabetes mellitus); Other respiratory inflammatory diseases (e.g. bronchitis and asthma); Severe underlying diseases involving the cardiovascular, cerebrovascular, hepatic, renal, or hematological systems.
Because sepsis is often associated with acute multi-organ dysfunction, patients in the CAP–sepsis group could present with additional acute organ injury or organ failure.
Definitions
CAP. CAP was primarily characterized by cough accompanied by at least one additional lower respiratory tract symptom, such as fever, sputum production, dyspnea, wheezing, or chest discomfort/discomfort. The diagnosis was confirmed via chest X-ray, and the infection was acquired in the community.
5
Sepsis. Sepsis was diagnosed according to the 2016 International Guidelines for the Management of Sepsis and Septic Shock. Sepsis was defined as suspected or confirmed infection accompanied by a Sequential Organ Failure Assessment (SOFA) score ≥2.
6
Collection of clinical data and blood samples
Clinical data collection included the following: (a) Demographic information. Age, sex, height, weight, and contact information. (b) Medical history. Past medical history, treatment history, family history, and relevant comorbidities. (c) Clinical and laboratory data related to CAP and sepsis. Clinical manifestations, CRP and PCT levels, and etiological examination results. Disease-related laboratory indicators were collected within 7 days before hospital admission.
After screening and enrollment, a single venous blood sample (3.5 mL) was collected from each participant using a yellow-capped tube with a gel separator. The samples were allowed to stand at room temperature for 30 min and then centrifuged at 3000 × g for 10 min within 2 h of collection to separate serum. The serum was used for HFREP1 measurement. All HFREP1 samples were labeled and stored at −20°C, and concentration analysis was completed within 1 month. Before analysis, serum samples were centrifuged at 10,000 ×g for 1 min to remove visible precipitates. Samples with severe hemolysis or lipemia were excluded. Whole blood samples were gently inverted 4–5 times before assessing clot formation, and the sample volume met the minimum requirement for detection. Raw data were recorded and archived. Remaining serum samples were stored at −80°C for subsequent HFREP1 analysis.
HFREP1 protein detection
HFREP1 levels were measured using a Thermo microplate reader, a low-temperature refrigerator (Haier), a medical centrifuge (Baiyang), and a cold-chain storage system (Haier). Human serum HFREP1 levels were quantified via enzyme-linked immunosorbent assay (ELISA) using a double-antibody sandwich method. The assay was performed with a commercial HFREP1 ELISA kit (Jiangsu Pinming Medical Technology Co., Ltd.) according to the manufacturer’s instructions. The assay detection limit was ≤0.006 ng/mL, with a linear range of 0.008–6 ng/mL. The linear correlation coefficient (r) was ≥0.99. The intra-assay coefficient of variation (CV) was ≤10%, and the inter-assay CV was ≤15%. No adverse events occurred during sample collection or testing.
Statistical methods
All statistical analyses were performed using SPSS version 27.0, Prism 10, and R software. Eighteen samples with missing PCT data and 12 samples with missing CRP data were imputed using linear regression interpolation. To avoid negative imputed values, all variables were first log-transformed before imputation and then back-transformed. Post hoc sensitivity analyses for missing data were performed, and the results were compared with those of a complete case analysis (including only participants with complete data). Logistic regression was used for hypothesis testing. Robustness was evaluated by comparing effect estimates (odds ratios), corresponding confidence intervals, and statistical significance obtained from both methods. The results were highly consistent with those of the complete case analysis (Tables S1 and S2). The kappa test was used to assess agreement between biomarkers and the reference standard. The relationship between two continuous variables was evaluated using Spearman’s rank correlation coefficient. Comparisons between two independent groups of continuous variables were performed using unpaired Student’s t-test for normally distributed data or the Mann–Whitney U test for non-normally distributed data. The Kruskal–Wallis test was applied to compare more than two independent groups with non-normal distribution. Differences in categorical data between groups were assessed using Pearson’s
Results
Baseline clinical characteristics of participants
Most participants were male, which may reflect the higher incidence of pneumonia in men. There were no statistically significant differences in sex, age, or body mass index (BMI) among the three groups, indicating comparability at baseline. White blood cell (WBC) counts in the CAP–sepsis group were significantly higher than those in the CAP and healthy control (HC) groups (p < 0.001), while WBC counts were similar between the HC and CAP groups (p = 0.266). Both leukocytosis and leukopenia were considered indicative of infection. CRP (p < 0.001) and PCT (p < 0.001) levels differed significantly between the CAP and CAP–sepsis groups. HFREP1 levels differed significantly among all three groups (p < 0.001) (Table 1). Pathogen identification was mainly performed using sputum culture and serological testing. Pathogens were detected in 28 patients (23%) in the CAP group and in 46 patients (88%) in the CAP–sepsis group. The Kruskal–Wallis test showed no significant difference in HFREP1 levels among gram-positive, gram-negative, and fungal infections (Table S3).
Participant characteristics.
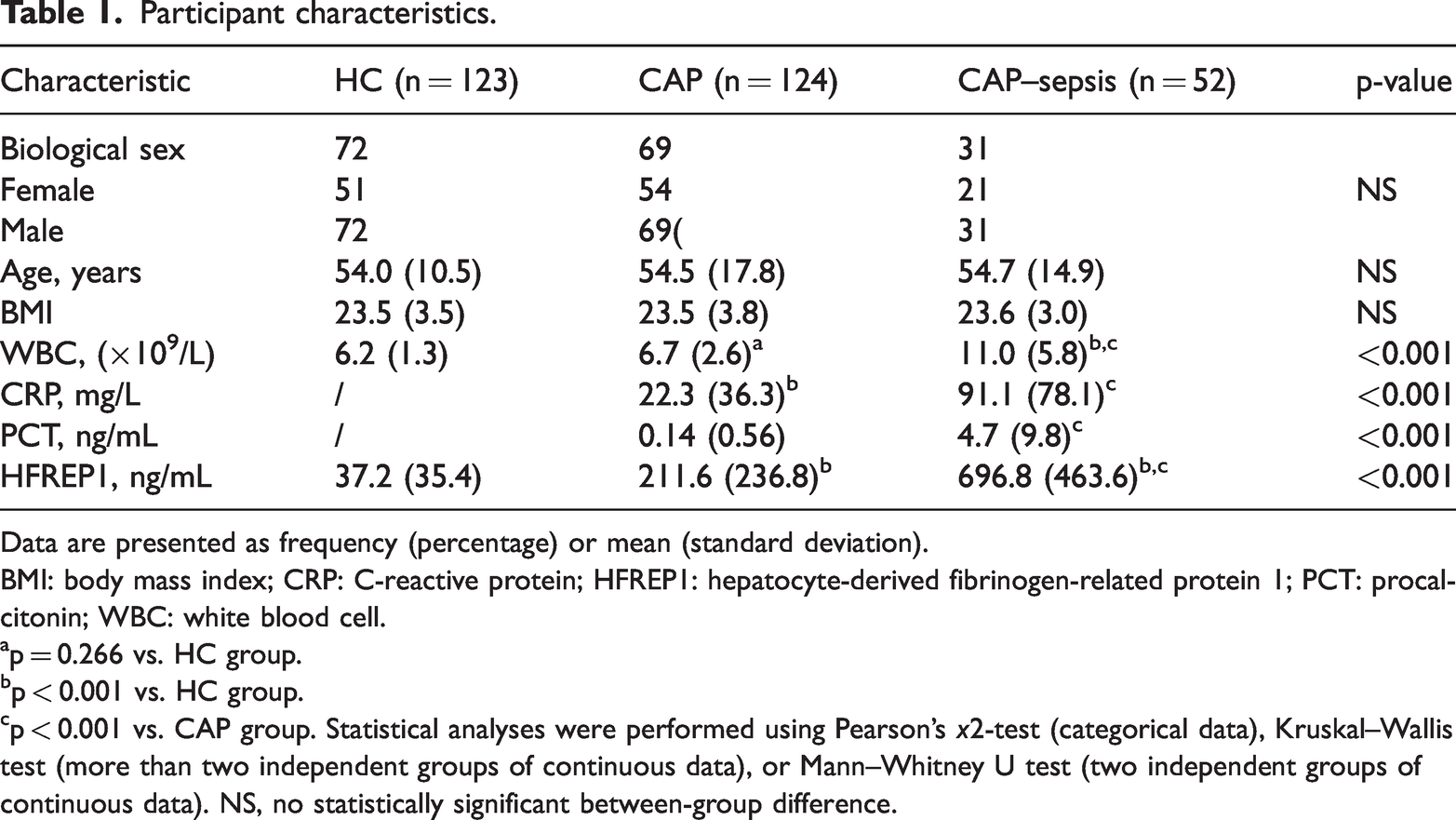
Data are presented as frequency (percentage) or mean (standard deviation).
BMI: body mass index; CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; PCT: procalcitonin; WBC: white blood cell.
p = 0.266 vs. HC group.
p < 0.001 vs. HC group.
p < 0.001 vs. CAP group. Statistical analyses were performed using Pearson’s
Binary logistic regression
Binary logistic regression analysis comparing the CAP and CAP–sepsis groups showed that PCT (p = 0.049) and HFREP1 (p < 0.001) were independent predictors of CAP–sepsis, whereas CRP (p = 0.436) was not an independent predictor (Figure 1).
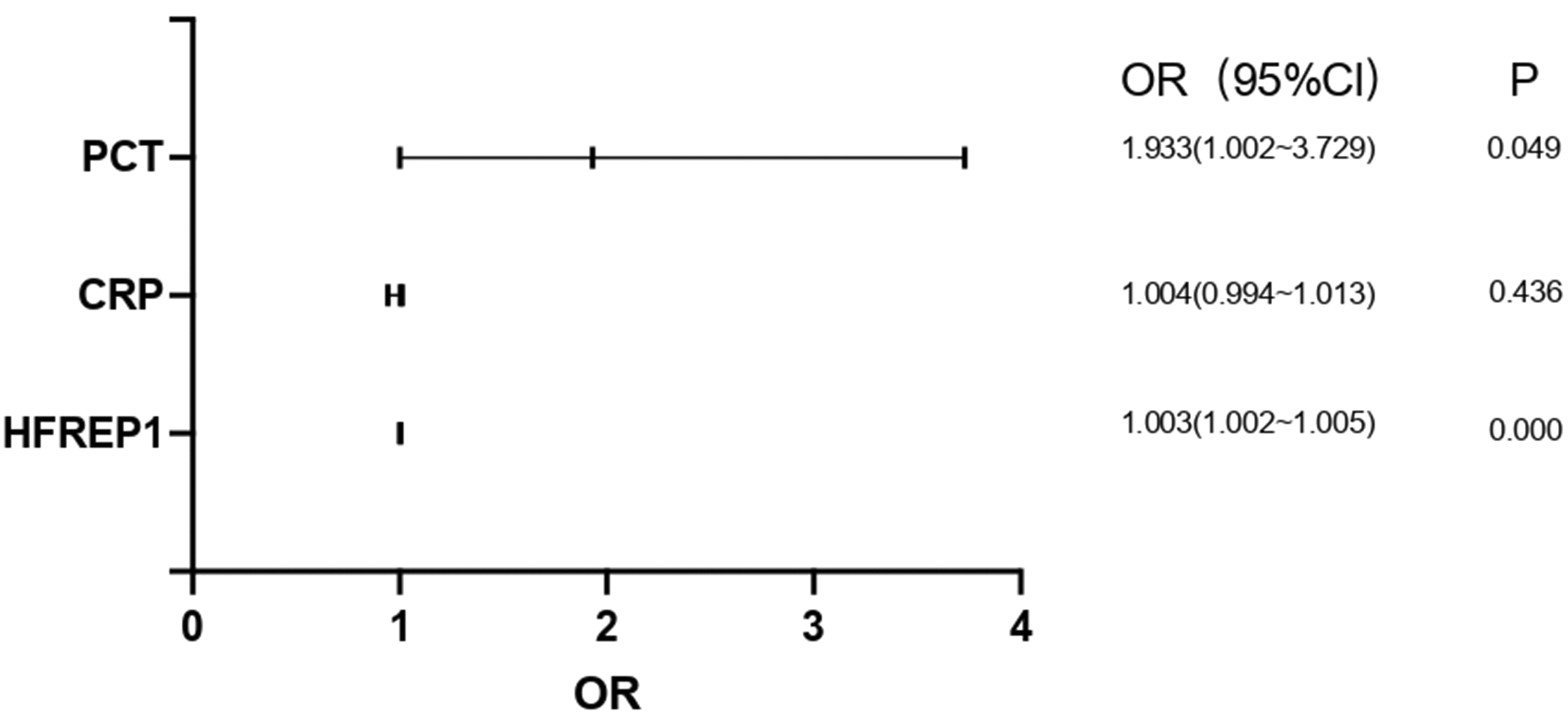
Binary logistic regression analysis of PCT, CRP, and HFREP1 for predicting CAP–sepsis. A p-value <0.05 indicates that the variable is an independent predictor. BMI: body mass index; CAP: community-acquired pneumonia; CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; OR: odds ratio; PCT: procalcitonin; WBC: white blood cell.
Correlation analysis
Correlations among serum PCT, CRP, and HFREP1 levels in the CAP and CAP–sepsis groups were evaluated. Because the data were not normally distributed, Spearman’s rank correlation coefficient was applied. Significant positive correlations were observed among the three biomarkers. The correlation coefficients between HFREP1 and PCT and between HFREP1 and CRP were 0.716 and 0.763, respectively, while the correlation coefficient between CRP and PCT was 0.768.
ROC
The ROC curve of HFREP1 for distinguishing healthy controls from patients with CAP showed an AUC of 0.8810 (95% confidence interval (CI): 0.8396–0.9224). The optimal cutoff value of HFREP1, determined by the maximum Youden index (0.628), was 65.125 ng/mL (Figure 2).

ROC curve of HFREP1 for distinguishing CAP from HC. CAP: community-acquired pneumonia; HC: healthy control; HFREP1: hepatocyte-derived fibrinogen-related protein 1; ROC: receiver operating characteristic.
The AUCs for CRP, PCT, and HFREP1 for distinguishing CAP from CAP–sepsis were 0.8902 (95% CI: 0.8340–0.9464), 0.8167 (95% CI: 0.7469–0.8865), and 0.8814 (95% CI: 0.8326–0.9303), respectively (Figure 3). Delong’s test showed no statistically significant difference between the AUCs for PCT and HFREP1; both were significantly higher than that for CRP. The optimal cutoff values for PCT, CRP, and HFREP1 were 0.105 ng/mL, 31.360 mg/mL, and 277.340 ng/mL, respectively.

ROC curves of HFREP1, PCT, and CRP for distinguishing CAP from CAP–sepsis. CAP: community-acquired pneumonia; CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; PCT: procalcitonin; ROC: receiver operating characteristic.
The ROC analysis of the combined biomarkers showed that the combination of HFREP1 and PCT achieved the highest diagnostic efficacy, with an AUC of 0.9094 (95% CI: 0.8671–0.9518) (Figure 4). However, Delong’s test indicated that the addition of multiple biomarkers did not result in a statistically significant improvement in diagnostic efficacy.

ROC curve of the combination of HFREP1, PCT, and CRP for distinguishing CAP from CAP–sepsis. CAP: community-acquired pneumonia; CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; PCT: procalcitonin; ROC: receiver operating characteristic.
Correspondence test
The kappa test showed that PCT had the highest agreement with the Sepsis-3.0 criteria for distinguishing CAP from CAP–sepsis (κ = 0.620). The kappa value for HFREP1 was 0.593, slightly lower than that for PCT but higher than that for CRP (κ = 0.488) (Table 2).
The correspondence test of HFREP1, CRP, and PCT with Sepsis-3.0.

Kappa consistency test.
CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; PCT: procalcitonin.
Unassumed null hypothesis.
Asymptotic standard error calculated under the original assumption.
Accuracy evaluation
Cross-tabulations of index test results against the reference standard are presented in Tables S4 to S6. For distinguishing CAP–sepsis from CAP, HFREP1 showed the highest sensitivity (86.54%; 95% CI: 73.60%–93.97%) among the three biomarkers, while its specificity was 79.03% (95% CI: 70.61%–85.61%), lower than that of PCT and CRP. PCT demonstrated the highest specificity (82.26%; 95% CI: 74.15%–88.32%), although the difference was not substantial compared with HFREP1 and CRP. The positive predictive values of all three biomarkers were suboptimal. However, all markers showed high negative predictive values, with the highest reaching 93.33% (95% CI: 86.28%–97.05%) (Table 3).
The accuracy evaluation of HFREP1, CRP, and PCT.

Sensitivity = a/(a + c); Specificity = d/(b + d); Positive predictive value = a/(a + b); Negative predictive value = d/(c + d). The values of a, b, c, and d are presented in Tables S1 to S3.
CI: confidence interval; CRP: C-reactive protein; HFREP1: hepatocyte-derived fibrinogen-related protein 1; PCT: procalcitonin.
Discussion
Current diagnostic guidelines for CAP do not incorporate hematological biomarkers. Computed tomography (CT), which is frequently used for diagnosis, involves radiation exposure and may not be acceptable to some patients; it is also contraindicated in certain populations, such as pregnant women. Therefore, identifying a serum biomarker to assist in the diagnosis of CAP has clinical relevance. PCT has been reported to help distinguish CAP from other respiratory diseases and to differentiate bacterial pneumonia from atypical or viral pneumonia. However, its clinical utility is limited by false-positive and false-negative results. 19 In a meta-analysis including 6599 patients with symptoms of acute respiratory infection or clinically suspected pneumonia, CRP showed the highest diagnostic accuracy (AUC = 0.802), followed by leukocytosis (0.777) and PCT (0.771). 20 Nevertheless, CRP can be elevated in various conditions, including acute infections, chronic inflammatory diseases, and trauma. 21 Pulmonary injury biomarkers such as surfactant protein D (SP-D), Krebs von den Lungen-6 (KL-6), and club cell secretory protein 16 (CC16) have shown limited diagnostic value for CAP, with AUCs ranging from 0.50 to 0.56. 22 In the present study, HFREP1 exhibits superior diagnostic capability for CAP, with an AUC of 0.881, suggesting its potential as a serum biomarker for CAP.
The diagnosis of sepsis currently relies on the Sepsis-3.0 criteria. However, detection of sepsis based solely on organ dysfunction may delay recognition. 23 From the standpoint of early identification and timely intervention to prevent complications such as organ failure, exclusive reliance on scoring systems alone may be insufficient. The identification of early biomarkers is therefore important. PCT is generally considered more accurate than CRP for the diagnosis of sepsis. A meta-analysis of 1368 patients reported that CRP had an AUC of 0.73 (95% CI: 0.69–0.77), with a sensitivity of 0.80 (95% CI: 0.63–0.90) and a specificity of 0.61 (95% CI: 0.50–0.72). 24 Three meta-analyses reported that the sensitivity and specificity of PCT for sepsis ranged from 77% to 85% and 75% to 83%, respectively,25–27 consistent with our findings. However, although PCT is useful in diagnosing sepsis with established organ dysfunction, its performance in early-stage or mild sepsis is limited, and it is not recommended for diagnosing ventilator-associated pneumonia.28–30 Recent studies have explored novel molecular biomarkers for early sepsis detection. For example, the expression profile of the Lnc-MALAT1/miR-125a axis has shown strong discriminative ability between sepsis patients and healthy controls (AUC = 0.931). 31 However, this approach has not been validated in comparisons between infected and septic patients, and its technical complexity may limit routine clinical application.
HFREP1 is a high-affinity ligand for lymphocyte activation gene 3 (LAG-3) and forms part of the HFREP1/LAG-3 immune checkpoint pathway. This pathway has been reported to inhibit T-cell function during tumor immune escape and to reduce the activity and proliferation of CD8+ T cells.13,32 In the early stage of CAP, an excessive inflammatory response is common; in severe cases, this may progress to T-cell exhaustion. Sepsis represents a dysregulated systemic immune response to infection, often characterized by early hyperinflammation followed by immune suppression. 33 T-cell exhaustion is considered an important contributor to secondary infection and mortality in sepsis. The elevated HFREP1 levels observed in pneumonia and sepsis may therefore be associated with its immunosuppressive effects. HFREP1 may participate in both modulation of inflammation and promotion of immune tolerance, although the underlying mechanisms require further investigation.
In this study, both PCT and HFREP1 were identified as independent predictors of CAP–sepsis and showed comparable performance in distinguishing CAP from CAP–sepsis, outperforming CRP. HFREP1 demonstrated distinct cutoff values for CAP and CAP–sepsis, suggesting potential utility across different stages of disease. In clinical research, multiple biomarkers are often combined to improve diagnostic performance. 34 The combined use of multiple biomarkers may better reflect the underlying pathophysiological processes of a disease and help compensate for the limited sensitivity or specificity of a single marker, thereby improving diagnostic accuracy and reliability. However, our analysis showed that the combination of PCT, CRP, and HFREP1 did not significantly improve diagnostic accuracy, which may be attributable to the strong correlations among these markers during the progression from CAP to secondary sepsis.
Importantly, we compared patients with confirmed infection (CAP) with those who developed sepsis (CAP–sepsis), rather than only comparing patients with healthy controls. This approach provides a more clinically relevant comparison, as sepsis typically occurs secondary to infection. Across the three study cohorts, our findings preliminarily support the value of HFREP1 in diagnosing CAP and CAP–sepsis. HFREP1 demonstrated better diagnostic performance than CRP for identifying sepsis. Its diagnostic profile across the transition from CAP to sepsis, together with its overall accuracy, may help address some limitations of existing clinical biomarkers.
Several limitations should be acknowledged. First, all patients in the CAP–sepsis group had established organ dysfunction, which limited evaluation of the early predictive value of HFREP1 and other biomarkers for early sepsis. Second, the lack of longitudinal follow-up hindered assessment of the prognostic value of HFREP1, PCT, and CRP for determining clinical outcomes in CAP–sepsis. Finally, the inclusion of only healthy controls, without other disease controls, may have restricted the evaluation of the specificity of HFREP1 in broader clinical contexts. In addition, the single-center, cross-sectional design may have restricted the generalizability of the findings.
Conclusion
In summary, HFREP1 may serve as a biomarker for CAP and CAP–sepsis. In patients with CAP–sepsis, HFREP1 was strongly correlated with CRP and PCT, and its diagnostic efficacy was comparable to that of PCT. Future large-scale, prospective studies—including patients with early-stage sepsis or systemic inflammatory response syndrome—are warranted to further clarify the role of HFREP1 in CAP and sepsis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261427147 - Supplemental material for Role of hepatocyte-derived fibrinogen-related protein 1 as a serum biomarker in the diagnosis of community-acquired pneumonia and sepsis: A cross-sectional study
Supplemental material, sj-pdf-1-imr-10.1177_03000605261427147 for Role of hepatocyte-derived fibrinogen-related protein 1 as a serum biomarker in the diagnosis of community-acquired pneumonia and sepsis: A cross-sectional study by Guoqing Wang, Liuliang Zhang, Xiaolan Zhu, Kai Qian, Lin Zhu, Boqian Chen, Wanjian Gu, Tao Guo and Shijia Liu in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank all study participants and the staff of the Department of Journal of International Medical Research for their support.
Authors’ contributions
Conceptualization: S.L.; methodology: T.G.; software: G.W.; validation: K.Q., G.W., L.Z., and B.C.; formal analysis: G.W.; investigation: L.Z. and X.Z.; resources: W.G.; data curation: L.Z.; writing—original draft preparation: G.W.; writing—review and editing: G.W.; visualization: G.W.; supervision: S.L.; project administration: S.L.; funding acquisition: S.L. All authors have read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be considered a potential conflict of interest.
Funding
This work was supported by the Talent Project established by Chinese Pharmaceutical Association Hospital Phamacy department (CPA-Z05-ZC-2023-003), the Project of Jiangsu Province Hospital of Traditional Chinese Medicine Peak Talent (No. k2026yrc20), and Special Fund for Science and Technology Plan of Jiangsu Province in 2023 (Key Research and Development Plan—Social Development Project; grant number BE2023607).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
