Abstract
Objective
This study was designed to examine the relationship between fecal incontinence and accelerated aging, determine its association with mortality risk, and evaluate whether accelerated aging mediates the link between fecal incontinence and mortality.
Methods
This cohort study used data from the National Health and Nutrition Examination Survey. Accelerated aging was quantified using phenotypic age acceleration. Multivariable linear regression models, Cox proportional hazards models, and mediation analysis were employed to assess the associations between fecal incontinence, phenotypic age acceleration, and mortality.
Results
Among 12,581 United States adults, the overall prevalence of fecal incontinence was 8.38%. Fecal incontinence was positively associated with phenotypic age acceleration (β: 0.98, 95% confidence interval: 0.43, 1.58) and increased risks of all-cause mortality (hazard ratio: 1.24, 95% confidence interval: 1.07, 1.44) and cardiovascular mortality (hazard ratio: 1.28, 95% confidence interval: 1.01, 1.63). Mediation analysis indicated that phenotypic age acceleration accounted for 9.56% of the effect of fecal incontinence on all-cause mortality.
Conclusion
Accelerated aging partially mediated the association between fecal incontinence and all-cause mortality in the United States population. These findings suggest new pathways for mitigating the broader health impacts of fecal incontinence.
Keywords
Introduction
Fecal incontinence (FI) is a prevalent condition affecting an estimated 7%–15% of the United States population.1,2 Beyond its substantial impact on quality of life, FI imposes a considerable burden on healthcare systems.3,4 Nevertheless, the true burden of FI may be underestimated because the condition is frequently underreported due to social stigma and patient embarrassment. 5 The etiology of FI is multifactorial and includes factors such as advanced age, obstetric trauma resulting in anal sphincter injury, gastrointestinal pathologies, and neurological impairments.6–9 Among these factors, aging plays an important role in the development of FI, as it closely affects the function of the enteric neuromuscular system.10,11 Emerging evidence indicates that FI may result from age-related decline and contribute to accelerated deterioration of physical health and functional capacity.12,13
Accelerated aging is a major risk factor for chronic disease development and mortality. 14 In contrast to chronological age, which denotes only the passage of time, biological age represents a superior measure of aging status because it integrates clinical biomarkers that reflect physiological state. 15 Phenotypic age acceleration (PhenoAgeAccel), derived from an algorithm based on an array of clinical biomarkers, quantifies multisystem physiological decline and has demonstrated robust predictive capacity for morbidity and mortality in US populations.16,17 This aging measure provides a valuable tool for identifying high-risk individuals and advancing research on the mechanisms of aging. 18
The potential role of FI in biological aging has been underexplored in the existing literature, and its relationship with mortality risk remains uncertain. Using data from the National Health and Nutrition Examination Survey (NHANES), this investigation examines the associations between FI, accelerated aging, and mortality risk in a nationally representative adult cohort.
Methods
Study design and data source
NHANES, administered by the National Center for Health Statistics (NCHS), is a nationally representative population-based study designed to evaluate the health and nutritional status of the noninstitutionalized civilian population in the US. 19 Employing a complex multistage probability sampling design, NHANES integrates detailed household interviews, standardized physical examinations, and laboratory analyses to collect extensive data on diverse health indicators. 20 The study protocol was approved by the NCHS Research Ethics Review Board, and written informed consent was obtained from all participants. This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. Reporting adhered to the broader recommendations of the Enhancing the Quality and Transparency of Health Research (EQUATOR) Network. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 21
The study population was derived from the 2005–2010 cycles of NHANES, as these were the only cycles that included the Bowel Health Questionnaire (BHQ). Our analysis included individuals aged ≥20 years who had completed the BHQ. Exclusion criteria comprised missing or incomplete data related to FI assessment, PhenoAgeAccel calculation, mortality status, or covariates such as sociodemographic factors, health behaviors, and medical conditions. The detailed process of participant exclusion is illustrated in Figure 1. Finally, 12,581 participants were included in the analysis.

Flowchart of participant selection. FI: fecal incontinence; NHANES: National Health and Nutrition Examination Survey.
FI assessment
FI was assessed using data from the BHQ. The FI survey questions were adapted from the FI Severity Index (FISI), which measures the frequency of involuntary leakage of gas, mucus, liquid, and solid stool. 22 The FISI is a validated instrument whose severity weightings demonstrate a very high correlation between patient and surgeon rankings (r = 0.97), and it has been widely adopted in NHANES research.22–24 In this study, FI was defined as the presence of involuntary loss of mucus, liquid, or solid stool at least once in the past 30 days.
Accelerated aging assessment
PhenoAgeAccel, an established biomarker of biological aging, was originally developed and validated in the US population.16,17 This metric is calculated as the residual derived from a linear regression model in which phenotypic age (PhenoAge) is regressed on chronological age (Supplementary Methods 1). PhenoAge is computed using a previously validated algorithm that integrates chronological age with nine clinical biomarkers: albumin, creatinine, glucose, C-reactive protein, lymphocyte percentage, mean cell volume, red blood cell distribution width, alkaline phosphatase, and white blood cell count. Positive PhenoAgeAccel values reflect a phenotype older than chronological age, whereas negative values correspond to a phenotypic age younger than chronological age. 25
Mortality outcome assessment
Participant mortality status was ascertained through the NHANES Public-Use Linked Mortality Files, which were matched with National Death Index records up to 31 December 2019 (https://www.cdc.gov/nchs/data-linkage/mortality-public.htm). All-cause mortality was documented, and specific causes of death were classified according to the International Statistical Classification of Diseases, Tenth Revision (ICD-10). Cardiovascular mortality included diseases categorized under ICD-10 codes I00-I09, I11, I13, and I20-I51, while cancer mortality was defined by ICD-10 codes C00-C97. The follow-up duration was calculated as the interval from the survey participation date to either the date of death or the end of the study period (31 December 2019) for surviving participants.
Covariate assessment
Several covariates were considered in the analysis, including sociodemographic characteristics, health-related behaviors, and medical conditions. Sociodemographic covariates included age, sex, race, educational level, marital status, and the poverty–income ratio (PIR). Smoking status was categorized as never, former, or current smoker. Physical activity level was determined from self-reported data and classified as inactive, moderately active, or active. Dietary quality was assessed using the Healthy Eating Index-2015 (HEI-2015), a measure of adherence to the Dietary Guidelines for Americans. 26 Details on the components and scoring criteria of HEI-2015 are provided in Supplementary Methods 2. Body mass index (BMI) was calculated from measured weight and height and classified as normal (<25), overweight (≥25 and <30), or obese (≥30). 27 A history of hypertension was defined by self-reported clinician diagnosis, use of antihypertensive medications, or measured blood pressure values consistent with the American College of Cardiology/American Heart Association guidelines (systolic ≥130 mmHg or diastolic ≥80 mmHg). 28 A history of diabetes was determined based on self-reported diagnosis, use of insulin or oral hypoglycemic agents, or satisfaction of any of the following biochemical criteria: fasting plasma glucose ≥126 mg/dL, 2-hour oral glucose tolerance test result ≥200 mg/dL, or hemoglobin A1c ≥6.5%. 29 History of cardiovascular disease or cancer was established through self-report of a physician’s diagnosis.
Statistical analysis
The analysis accounted for the complex survey design of NHANES, including sample weights, clustering effects, and stratification. Baseline characteristics were presented as weighted means with corresponding 95% confidence intervals (CIs) for continuous variables and as weighted proportions with 95% CIs for categorical variables. Comparisons between participants with and without FI were conducted using weighted linear regression models for continuous variables and weighted chi-square tests for categorical variables. The association between FI and PhenoAgeAccel was evaluated using multivariable linear regression models, with results reported as coefficients (β) and 95% CIs (Supplementary Methods 3). The log-rank test and Kaplan–Meier curves were used to compare survival between the two groups. Mortality risk was assessed using Cox proportional hazards models, and hazard ratios (HRs) with 95% CIs were reported (Supplementary Methods 3). Sensitivity analysis was conducted by excluding participants who died within the first year of follow-up to evaluate the robustness of the findings. Subgroup analyses and interaction tests were performed to assess potential differences in associations across groups stratified by age, sex, BMI, and comorbidities. Mediation analysis using the product of coefficients approach was performed to quantify the potential mediating role of PhenoAgeAccel in the relationship between FI and mortality risk. Significance was assessed using nonparametric bootstrapping with 1000 replicates. A significant mediating effect was established when all the following criteria were met: a significant total effect, a significant indirect effect, and a significant proportion mediated. All statistical analyses were performed using R (version 4.4.0, http://www.R-project.org). Two-sided
Results
Baseline characteristics
A total of 12,581 US adults were included in the analysis, with a weighted mean age of 46.68 years (95% CI: 45.97, 47.39). Of the participants, 48.84% were male, and 8.38% reported FI. The characteristics of the participants are summarized in Table 1. Participants with FI were more likely to be older, female, non-Hispanic White, widowed, divorced, or separated and had lower educational level and PIR. The FI group had higher rates of current smoking, physical inactivity, and obesity. Furthermore, this population exhibited a higher prevalence of comorbidities, including hypertension, diabetes, cardiovascular disease, and cancer. Notably, although negative PhenoAgeAccel values in both groups indicate a phenotypic age younger than the chronological age, the FI group had a significantly less negative value, suggesting an accelerated pace of biological aging relative to the non-FI group. Consistently, participants with FI experienced substantially higher all-cause mortality rates.
Basic characteristics of the study participants.
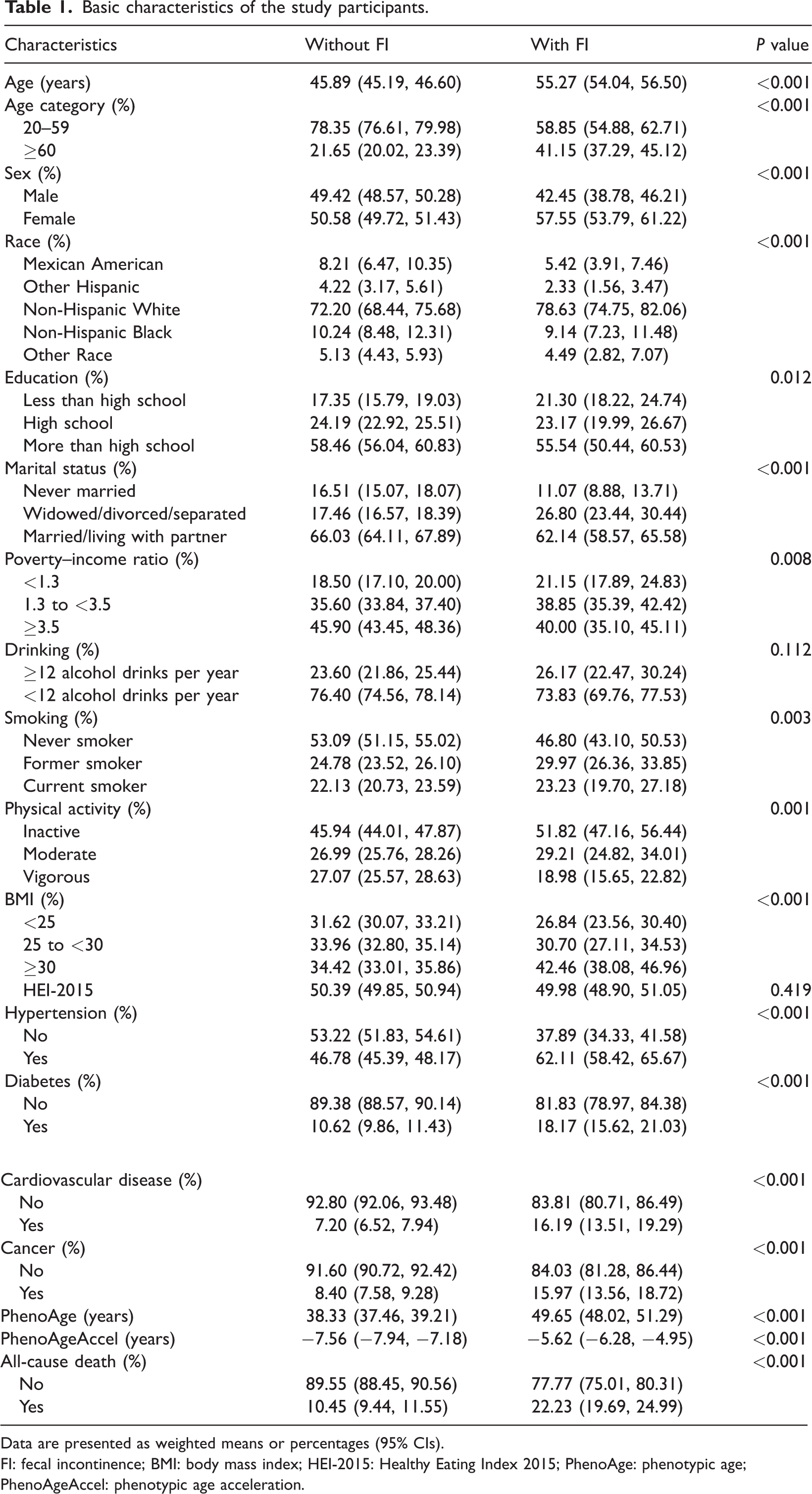
Data are presented as weighted means or percentages (95% CIs).
FI: fecal incontinence; BMI: body mass index; HEI-2015: Healthy Eating Index 2015; PhenoAge: phenotypic age; PhenoAgeAccel: phenotypic age acceleration.
Association between FI and PhenoAgeAccel
Weighted multivariable linear regression models were used to evaluate the association between FI and PhenoAgeAccel. As shown in Table 2, FI was significantly positively associated with PhenoAgeAccel in the unadjusted model (β: 1.94, 95% CI: 1.35, 2.53), partially adjusted model (β: 1.80, 95% CI: 1.22, 2.38), and fully adjusted model (β: 0.98, 95% CI: 0.43, 1.58).
Association between FI and PhenoAgeAccel.

FI: fecal incontinence; PhenoAgeAccel: phenotypic age acceleration; HEI-2015: Healthy Eating Index 2015; BMI: body mass index.
Model 1: not adjusted for any covariates.
Model 2: adjusted for age category, sex, and race.
Model 3: adjusted for age category, sex, race, educational level, marital status, poverty–income ratio, BMI, drinking, smoking, physical activity, HEI-2015, and history of hypertension, diabetes, cardiovascular disease, and cancer.
Subgroup analyses by age, sex, BMI, hypertension, diabetes, cardiovascular disease, and cancer showed no significant interactions (all
Relationship of FI with all-cause and cause-specific mortality
Within the cohort of 12,581 participants, a total of 1,949 deaths were recorded during a median follow-up period of 11.33 years (139,757.08 person-years), including 482 from cardiovascular disease and 467 from cancer. Kaplan-Meier survival curves (Supplementary Figure 2) showed significantly higher all-cause, cardiovascular, and cancer mortality rates in participants with FI compared to those without (log-rank
Relationship of FI with all-cause and cause-specific mortality.
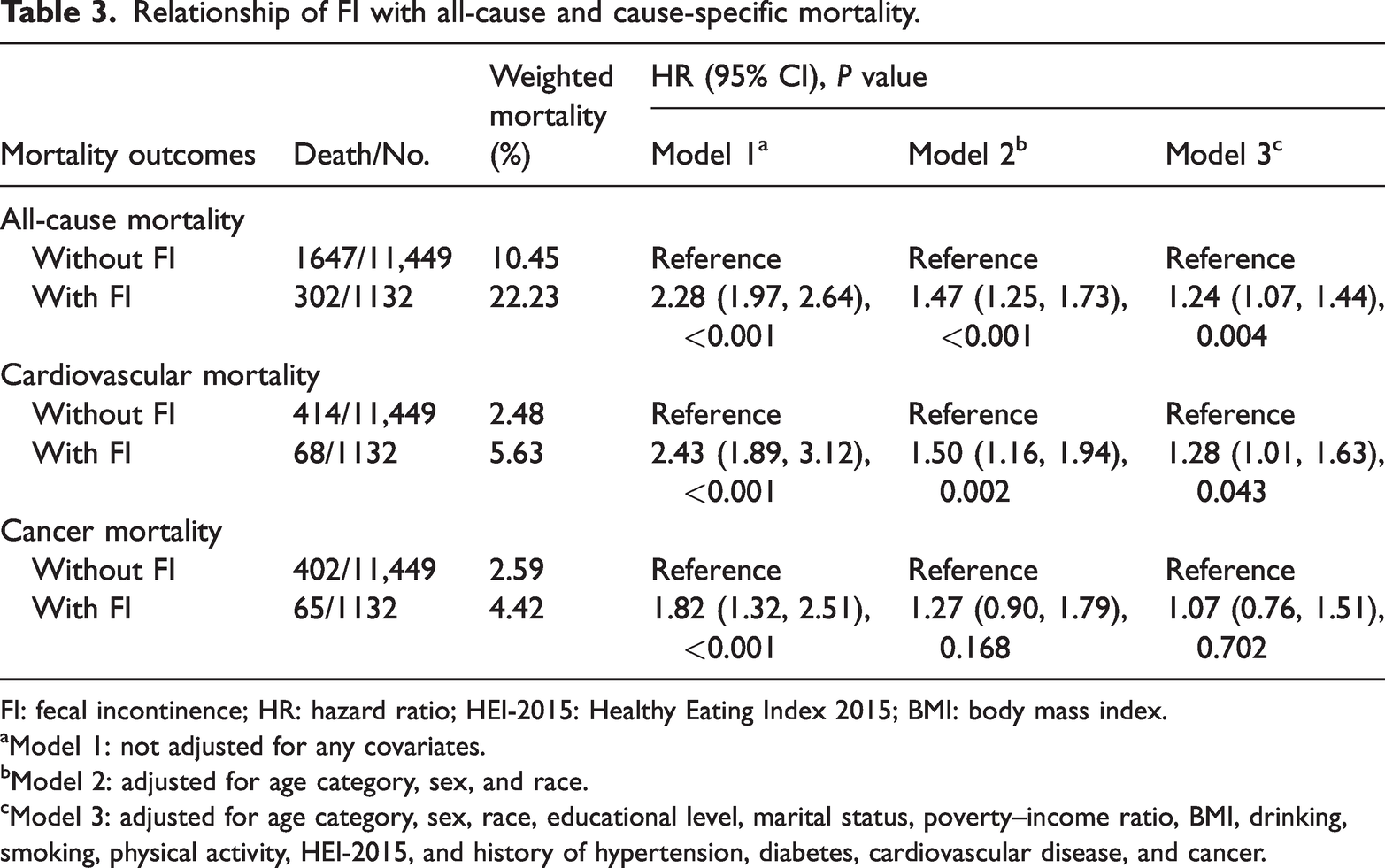
FI: fecal incontinence; HR: hazard ratio; HEI-2015: Healthy Eating Index 2015; BMI: body mass index.
Model 1: not adjusted for any covariates.
Model 2: adjusted for age category, sex, and race.
Model 3: adjusted for age category, sex, race, educational level, marital status, poverty–income ratio, BMI, drinking, smoking, physical activity, HEI-2015, and history of hypertension, diabetes, cardiovascular disease, and cancer.
Subgroup analyses revealed that the association between FI and mortality outcomes was generally consistent across different subgroups (
In the sensitivity analysis excluding deaths within the first year of follow-up, the associations of FI with all-cause mortality (HR: 1.24, 95% CI: 1.07, 1.45) and cardiovascular mortality (HR: 1.28, 95% CI: 1.00, 1.67) remained significant, and no association was observed with cancer mortality (HR: 1.09, 95% CI: 0.83, 1.43) (Supplementary Table 3).
Mediating effect of PhenoAgeAccel in the associations of FI with all-cause and cause-specific mortality
Mediation analyses were conducted to evaluate the potential mediating role of PhenoAgeAccel in the associations of FI with mortality. The analyses demonstrated that PhenoAgeAccel served as a significant partial mediator, accounting for 9.56% of the total effect of FI on all-cause mortality (Figure 2). For cardiovascular mortality, a significant indirect effect was observed, but the direct effect was nonsignificant. No significant mediation effect was observed for cancer mortality (Supplementary Table 4).

Mediating effect of PhenoAgeAccel on the association between FI and (a) all-cause mortality and (b) cardiovascular mortality. PhenoAgeAccel: phenotypic age acceleration.
Path coefficients and 95% CIs were derived from mediation analyses using nonparametric bootstrapping with 1000 replicates. Path a represents the effect of FI on PhenoAgeAccel; Path b represents the effect of PhenoAgeAccel on mortality, adjusted for FI; and Path c’ represents the direct effect of FI on mortality, adjusted for PhenoAgeAccel. This analysis quantified the total effect (the overall association between FI and mortality outcomes), direct effect (the total effect of FI on mortality outcomes independent of PhenoAgeAccel), and indirect effect (the effect of FI on mortality outcomes mediated by PhenoAgeAccel). The proportion of mediation refers to the percentage of the total effect explained by PhenoAgeAccel as the mediating variable in the relationship between FI and mortality outcomes.
Discussion
In this nationally representative cohort of US adults, FI was significantly associated with accelerated aging. FI was also linked to increased risks of all-cause and cardiovascular mortality, whereas no significant association was observed with cancer mortality. Notably, subgroup analyses revealed a significantly higher risk of cancer mortality among participants aged <60 years. Mediation analysis further demonstrated that accelerated aging partially mediated 9.56% of the association between FI and all-cause mortality.
FI is a prevalent condition in older adults and encompasses a spectrum of physiological alterations linked to aging. 30 Our study advances prior research by establishing an association between FI and PhenoAgeAccel, providing a quantifiable measure of its impact on accelerated aging. This connection may be explained through specific biological pathways involving key components of the PhenoAgeAccel algorithm. FI appears to accelerate biological aging by triggering chronic low-grade inflammation, reflected in elevated C-reactive protein levels, and potentially through systemic nutrient malabsorption or dysregulation, indicated by decreased albumin concentrations. This mechanistic pathway aligns with evidence demonstrating that FI is correlated with fundamental aging processes, including age-related degenerative changes of the enteric neuromuscular apparatus (particularly the internal anal sphincter), 31 increased inflammatory signaling, 32 oxidative stress, 33 and accumulation of senescent cells. 11 The consistency between these molecular features of aging and the specific biomarkers affected in FI provides compelling evidence that FI actively contributes to biological aging processes, extending beyond its recognized association with functional declines such as reduced gait speed and impaired balance. 13
Even after accounting for comorbid chronic conditions, older adults with FI exhibit greater declines in physical function compared with those without FI. 13 This association remained statistically significant after comprehensive adjustment for a wide range of sociodemographic, lifestyle, and clinical factors, highlighting the stability of the relationship.
Abnormalities in gastrointestinal health are associated with various adverse health outcomes.34,35 A large-scale New Zealand cohort study demonstrated a significant relationship between FI and mortality among community-dwelling older adults. 36 However, a Canadian cohort study reported that although FI was initially associated with elevated mortality, this relationship became nonsignificant after adjustment for confounders, 37 suggesting that other variables may explain much of the observed association. The association observed in our study, in contrast to some previous reports, may be attributable to our comprehensive adjustment for lifestyle confounders, such as dietary quality and physical activity, which were not fully accounted for in earlier studies and may have obscured this relationship. Previous studies have also identified a link between gastrointestinal dysfunction and cardiovascular events, suggesting that impaired gastrointestinal health may elevate the risk of cardiovascular death.38–40 In line with this evidence, our results indicate an increased risk of cardiovascular mortality in individuals with FI. Although no significant association was found between FI and cancer mortality in the overall population, subgroup analyses revealed a significant relationship between FI and cancer mortality among participants aged <60 years. This age-dependent association likely reflects distinct etiological patterns. In older adults, FI typically arises from age-related degenerative changes, while in younger individuals, it indicates occult gastrointestinal malignancies. 41 Younger patients often present with more aggressive tumor characteristics, including larger size, poorer differentiation, and higher rates of peritoneal recurrence.42–44 Moreover, younger patients exhibit fewer alarm symptoms than older individuals, a presentation that often delays diagnosis and may ultimately compromise survival. 45
Our study is the first to investigate accelerated aging as a mediator in the relationship between FI and mortality. Although prior research has documented an association between FI and elevated mortality, it remains unclear whether this risk is directly driven by FI or reflects the influence of frequently accompanying comorbidities.46,47 Our findings suggest that accelerated aging may represent a partial pathway through which FI affects long-term health outcomes. Consequently, interventions targeting biological aging mechanisms could represent a novel therapeutic approach to reduce the burden of FI-related health deterioration. The partial nature of the observed mediation also indicates that additional mechanisms likely contribute to the observed associations, warranting further research. Given the cross-sectional assessment of FI and PhenoAgeAccel, the mediation results should be interpreted as identifying a biologically plausible pathway rather than confirming causal direction or temporal sequence.
This study has several notable strengths. First, the analysis leverages a large, nationally representative, population-based sample of US adults with extended follow-up, enhancing the generalizability and reliability of the findings. Second, the incorporation of PhenoAgeAccel as an objective biomarker of biological aging provides a quantifiable measure linking FI to accelerated aging. Third, rigorous adjustment for potential confounders and comprehensive subgroup analyses further strengthen the validity and robustness of the results.
Several limitations should also be acknowledged. First, the cross-sectional assessment of FI and PhenoAgeAccel precludes determination of temporal sequence. Future prospective studies are needed to dynamically monitor the occurrence of FI and changes in PhenoAgeAccel to verify causal direction. Second, the NHANES dataset lacks detailed FI characteristics, including severity, frequency, and etiology, which may introduce residual confounding. Third, as NHANES is not specifically designed for oncological studies, it lacks detailed clinical information, such as cancer stage, histological subtype, and treatment history, which may confound analyses of cancer-specific mortality. Fourth, cause-specific mortality was defined using ICD-10 codes; although standard, misclassification or incomplete information may affect the precision of cardiovascular and cancer mortality estimates. Finally, PhenoAgeAccel may not capture the full spectrum of biological aging processes. Future research should include a broader range of aging biomarkers to provide a more comprehensive assessment of the relationship between FI, accelerated aging, and health outcomes.
Conclusion
This study demonstrated that accelerated aging partially mediates the associations of FI with all-cause and cardiovascular mortality in the US population. These findings offer valuable insights for developing strategies to mitigate the broader health impacts of FI and promote long-term health.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261425322 - Supplemental material for Accelerated aging mediates the association between fecal incontinence and mortality: Evidence from the National Health and Nutrition Examination Survey
Supplemental material, sj-pdf-1-imr-10.1177_03000605261425322 for Accelerated aging mediates the association between fecal incontinence and mortality: Evidence from the National Health and Nutrition Examination Survey by Yizhou Lu, Zixin Yu, Hongxin Chen and Hong Shen in Journal of International Medical Research
Footnotes
Acknowledgments
We thank all NHANES staff and participants for their contributions.
Consent for publication
Not applicable.
CRediT authorship contribution statement
Yizhou Lu: Conceptualization, Methodology, Formal Analysis, Investigation, Software, Data Curation, Visualization, Writing–Original Draft.
Zixin Yu: Conceptualization, Methodology, Formal Analysis, Investigation, Software, Data Curation, Visualization, Writing–Original Draft.
Hongxin Chen: Supervision, Funding Acquisition, Writing–Review & Editing.
Hong Shen: Supervision, Funding Acquisition, Writing–Review & Editing.
All authors contributed to the article and approved the submitted version.
Data availability statement
Declaration of conflicting interests
The authors report no conflicts of interest.
Ethics approval and consent to participate
The National Center for Health Statistics Research Ethics Review Board provided ethics approval (Protocol #2005-06) for all NHANES study protocols. Written informed consent was obtained from all participants. Therefore, no additional external ethical approval or consent was required.
Funding
This work was supported by the Internal Research Fund Project of the Second Affiliated Hospital of Nanjing University of Chinese Medicine (Jiangsu Second Chinese Medicine Hospital) (SEZRC202504, SEZJY202505).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
