Abstract
Fabry disease is a rare X-linked lysosomal storage disorder caused by a defect in glycosphingolipid metabolism, which leads to the accumulation of globotriaosylceramide (Gb3) in various cell types. It has diverse clinical manifestations, often making diagnosis challenging. This report presents the case of a male patient in his mid-20s with Fabry disease who presented with recurrent lower limb edema and proteinuria. He was ultimately diagnosed and treated with enzyme replacement therapy in combination with a nonsteroidal mineralocorticoid receptor antagonist. Since 2018, the patient’s serum creatinine level and 24-h urinary protein excretion had progressively increased, while his estimated glomerular filtration rate and hemoglobin levels had decreased. Following the addition of the nonsteroidal mineralocorticoid receptor antagonist to the treatment regimen, these parameters gradually improved and stabilized. Long-term follow-up of the patient is ongoing. This case suggests that combining enzyme replacement therapy with a nonsteroidal mineralocorticoid receptor antagonist aids in arresting disease progression and improving prognosis in patients with Fabry disease.
Keywords
Introduction
Fabry disease (FD) is an X-linked genetic disorder caused by mutations in GLA, which encodes α-galactosidase A. This leads to the accumulation of globotriaosylceramide (Gb3) in various cell types. The abnormal deposition of glycosphingolipids results in a wide range of clinical signs and symptoms, contributing to significant morbidity and mortality. 1 FD involves multiple systems and is characterized by angiokeratoma, acroparesthesia, hypohidrosis, corneal verticillata, and chronic or episodic pain as well as cardiovascular, cerebrovascular, and renal manifestations such as cardiomyopathy, arrhythmias, stroke, and proteinuria. 2 This disease is relatively rare in both male and female populations. The estimated prevalence of FD is approximately 1 in 3000 individuals, with the prevalence in female carriers ranging from 1:6000 to 1:40,000.3,4 The prevalence of classic FD in men ranges between 1/117,000 and 1/8454. The disease occurs across all ethnicities and races. 5 Nonspecific clinical manifestations often lead to initial misdiagnosis. Moreover, late-onset, cardiac, and renal variants may present with delayed or atypical symptoms, further complicating diagnosis. 6 FD is associated with significantly increased mortality and morbidity; even with treatment, life expectancy is reduced by approximately 20 years in men and 10 years in women, primarily due to end-stage renal disease and life-threatening cardiovascular and cerebrovascular events. 7 Treatment for FD includes specific and supportive approaches. Specific therapies consist of recombinant α-galactosidase A (enzyme replacement therapy (ERT)) and migalastat, an oral pharmacological chaperone that stabilizes specific mutant forms of α-galactosidase A, thereby promoting proper lysosomal trafficking and enhancing enzymatic activity. Supportive management focuses on treating complications such as chronic kidney disease (CKD) along with cardiac, neurological, and gastrointestinal manifestations. 8
This report details the case of a patient with an atypical presentation who was eventually diagnosed with FD and treated with ERT combined with a nonsteroidal mineralocorticoid receptor antagonist (MRA).
Case presentation
The patient was a man in his mid-20s who presented with recurrent bilateral lower limb edema, frothy urine, and lower back pain. He was readmitted to our department in April 2022 for further evaluation and management. His medical history spans >10 years when he first developed a burning pain in his extremities and anhidrosis but did not seek medical attention. He subsequently experienced tinnitus, hearing loss, and a red macular rash. Approximately 8 years ago, he noticed recurrent bilateral lower limb edema accompanied with lower back pain and frothy urine, although his urine output remained normal. He was first admitted to our department and received initial treatment in 2015. Urinalysis at that time showed 2+ proteinuria; 24-h urinary protein excretion was 1.55 g/24 h. He was initially diagnosed with “chronic glomerulonephritis” and received renal protective and antiproteinuric treatments, including a renin–angiotensin system blocker (valsartan, 80 mg daily). His condition worsened following corticosteroid therapy. In December 2015, a left renal biopsy was performed, which confirmed FD (Figure 1). His family history indicated that his mother and two maternal uncles had also been diagnosed with FD (Figure 2).

Renal biopsy findings (light microscopy, electron microscopy, and immunofluorescence). (a) and (b) show the results of light microscopy: HE staining showing an increase in the number of cells in the glomerulus, and PAS showing slight proliferation of mesangial cells and matrix. (c,d) PASM staining showing that the capillary loops were open, and the basement membrane was not obviously thickened. Masson staining showing no obvious deposition of erythrophil in the glomerulus. (e) and (f) show the electron microscopic images of the glomerulus: one glomerulus was detected microscopically. Capillary endothelial cells are markedly vacuolated and degenerated, and erythrocytes are visible in individual lumens; there is no obvious endothelial cell proliferation, and the capillary loops are open. There is no obvious thickening and delamination of the wall layer of the renal capsule, vacuolar degeneration of the cells of the wall layer, or obvious proliferation. Basement membrane: no obvious thickening, with thickness of approximately 200–400 nm. Epithelial cells of the visceral layer are swollen and show vacuolar degeneration; foamy, secondary lysosomes are increased, and a large number of medullary vesicles and zebra bodies are seen. The peduncles are diffusely fused. Mesangial cells and matrix show no significant hyperplasia. No electron-dense deposits are observed. Vacuolar degeneration of renal tubular epithelial cells is visible. No specific lesions are present in the renal interstitium. Within the renal interstitial vasculature, erythrocytes are seen in the lumen of individual capillaries. Immunofluorescence: IgG1, negative; IgG4, negative; PLA2R, negative; HE: hematoxylin and eosin; PAS: periodic acid–Schiff; PASM: periodic acid–silver methenamine stain; IgG1: immunoglobulin G1; IgG4: immunoglobulin G4; PLA2R: phospholipase A2 receptor.
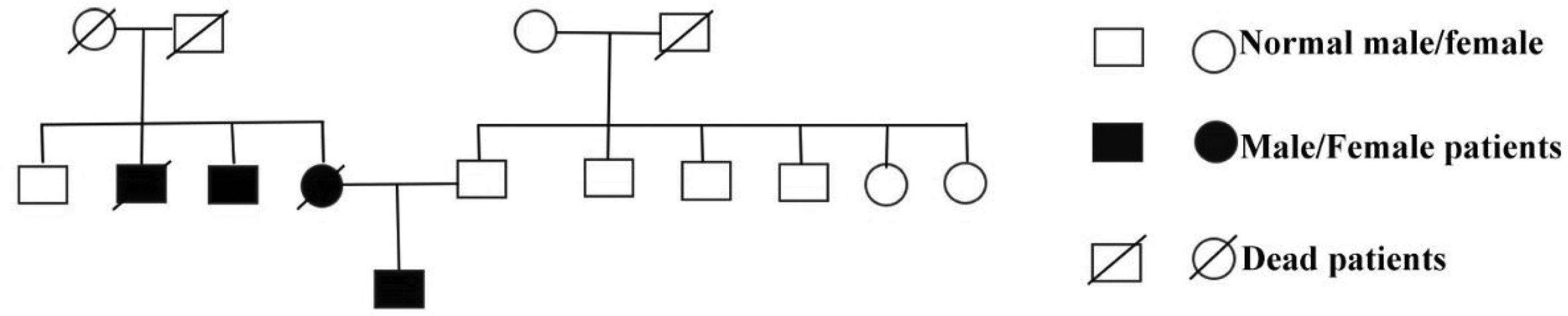
Family pedigree of the patient.
Following the diagnosis, corticosteroids were gradually tapered and discontinued, while renal protective therapy with valsartan (80 mg daily) was continued. Long-term outpatient follow-up until 2018 showed that his 24-h urinary protein excretion fluctuated between 0.6 g/24 h and >2.0 g/24 h. The patient was subsequently lost to follow-up after 2018 due to a fracture, and he discontinued all treatment. In August 2020, genetic testing confirmed FD; his α-galactosidase A level was 0.26 μmol/L, and globotriaosylsphingosine (lyso-GL-3) level was 92.61 ng/mL (Table 1). The patient received no disease-specific treatment until April 2022 when he began receiving intravenous agalsidase alfa infusions (10.5 mg every 2 weeks) at our department. Valsartan was continued throughout this period. Subsequently, in April 2023, oral finerenone (10 mg once daily) was added to his regimen. The patient has since remained on this combination therapy of agalsidase alfa, valsartan, and finerenone. Follow-up laboratory results are summarized in Table 2 and Figure 3.
Results of genetic testing.

This variant is a nonsense mutation, which may lead to premature termination of protein translation or nonsense-mediated mRNA decay, thereby resulting in the loss of the protein’s normal function.
AD: autosomal dominant; AR: autosomal recessive; XL: X-linked.
Reference genome version: GRCh37/hg19.
Patient follow-up results.
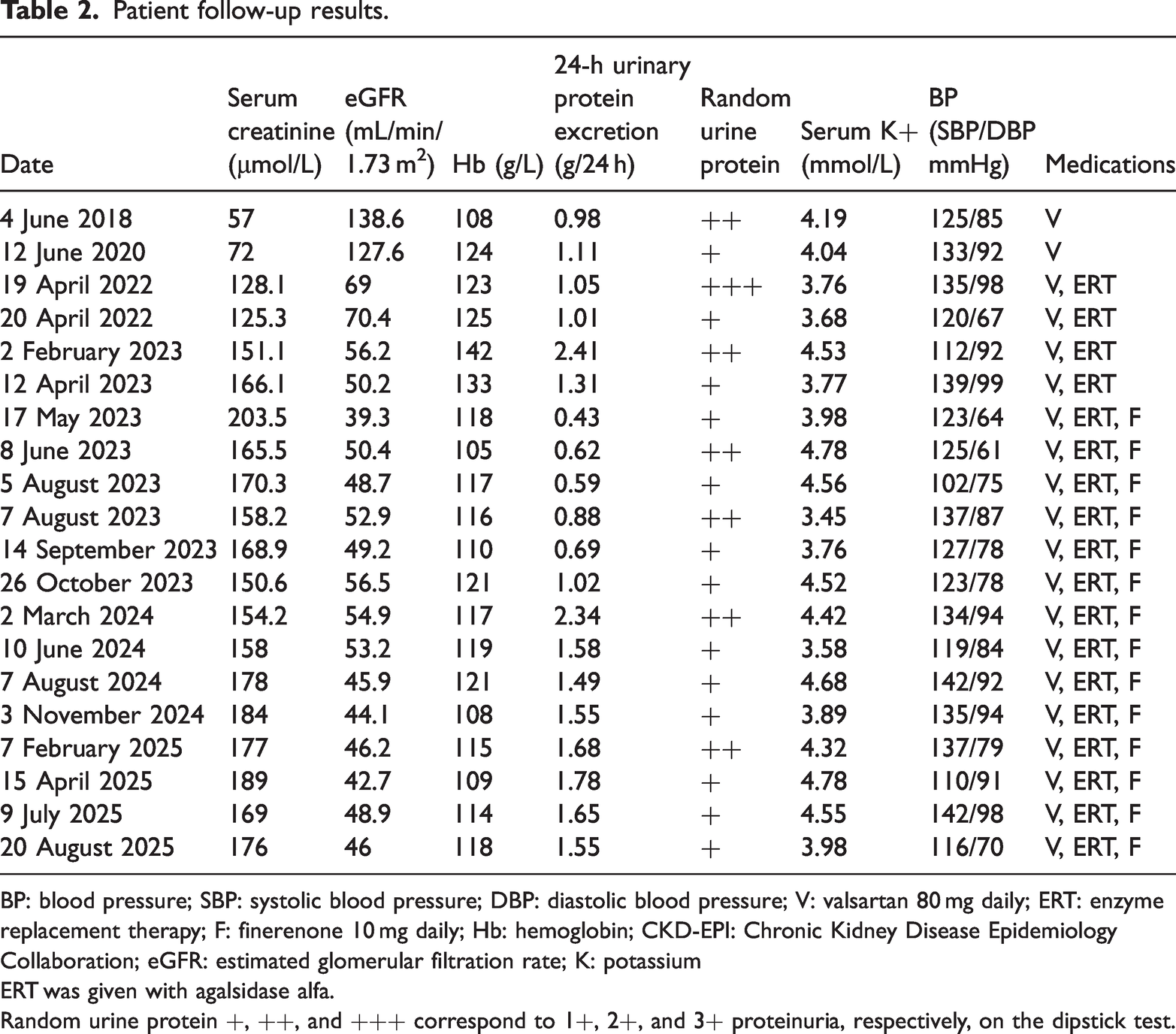
BP: blood pressure; SBP: systolic blood pressure; DBP: diastolic blood pressure; V: valsartan 80 mg daily; ERT: enzyme replacement therapy; F: finerenone 10 mg daily; Hb: hemoglobin; CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration; eGFR: estimated glomerular filtration rate; K: potassium
ERT was given with agalsidase alfa.
Random urine protein +, ++, and +++ correspond to 1+, 2+, and 3+ proteinuria, respectively, on the dipstick test.
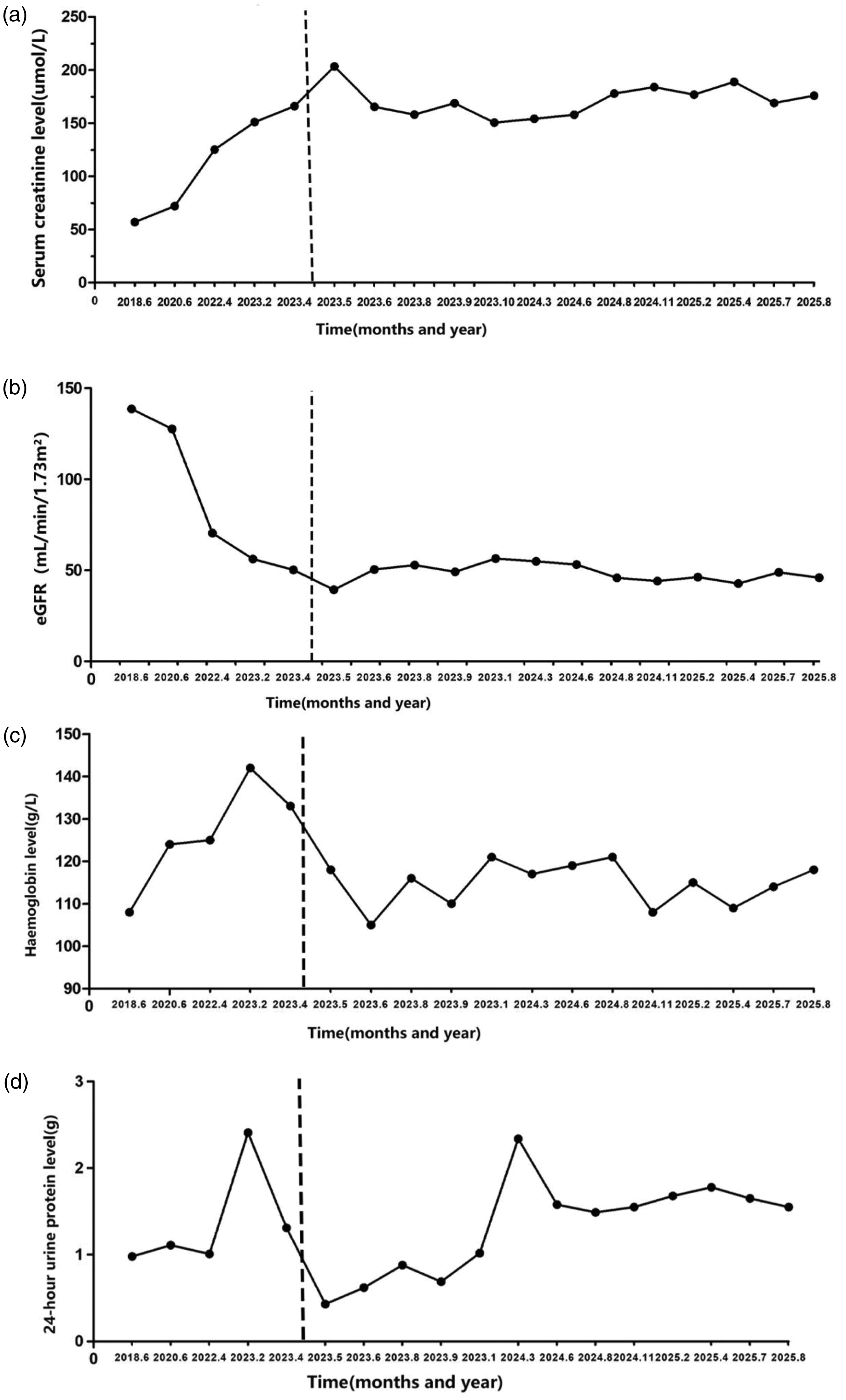
Patient follow-up results. The chart shows changes in key laboratory indicators. The vertical dashed line indicates the initiation of finerenone therapy on 18 April 2023. (a) Serum creatinine levels over time. (b) Estimated glomerular filtration rate (eGFR) over time. (c) Hemoglobin levels over time. (d) 24-h urinary protein excretion over time. The graphs show that the levels of serum creatinine, hemoglobin, and 24-h urinary protein excretion as well as estimated glomerular filtration rate of the patient after the use of finerenone tablets on 18 April 2023 stabilized over time.
Follow-up data showed that the patient’s serum creatinine level began to rise in 2018, peaking at 203.5 μmol/L in May 2023. Following the initiation of finerenone, it decreased to 165.5 μmol/L within approximately 1 month (by June 2023) and subsequently stabilized. Similarly, the estimated glomerular filtration rate (eGFR; calculated using the 2021 chronic kidney disease epidemiology collaboration (CKD-EPI) creatinine equation) 9 declined from 2018 to a nadir of 39.3 mL/min/1.73 m2 in May 2023 but has since improved and remains approximately 50 mL/min/1.73 m2. His hemoglobin level dropped to 105 g/L in June 2023 but later showed gradual improvement. His 24-h urinary protein excretion peaked at 2.41 g/24 h in February 2023 and decreased after starting finerenone, eventually reaching a stable range. Notably, serum potassium levels remained normal (range: 3.4–5.0 mmol/L) throughout the follow-up period after finerenone initiation, with no recorded episodes of hyperkalemia or other adverse events. Key follow-up parameters, including blood pressure, are presented in the chart (Table 2 and Figure 3). The most recent follow-up in August 2025 confirmed that the patient’s parameters have remained stable with the continued combination therapy.
This case report conforms to the Case Report (CARE) guidelines, 10 and written informed consent for treatment and publication was obtained from the patient. All patient details have been deidentified.
Discussion
This case illustrates key aspects of FD diagnosis and highlights a novel therapeutic combination for Fabry nephropathy. First, clinical presentations can be atypical. Although nonobstructive concentric left ventricular hypertrophy (LVH) is a common finding in FD, a high index of clinical suspicion is essential for timely diagnosis. 11 The initial laboratory test for suspected FD involves the measurement of α-galactosidase A activity in the plasma or leukocytes, with reduced activity indicating FD. 12
In the early stages of FD, glomerular hyperfiltration may mask underlying renal dysfunction. However, as the disease progresses, fibrosis, sclerosis, and tubular atrophy develop, often culminating in renal failure by the third to fifth decade of life. 13 Renal involvement is a major cause of morbidity and mortality in FD. The 5-year survival rate for patients with FD-related nephropathy is 41% compared with 68% for those with other nephropathies, highlighting its poor prognosis.14,15
Cardiac involvement is a major cause of mortality in FD, most commonly manifesting as LVH. Although rare in children, LVH typically presents in men at approximately 40 years of age and appears later in women. 16 The hypertrophy is usually concentric; however, asymmetric septal hypertrophy can occur, and obstructive forms are uncommon. Multimodal cardiac imaging—including echocardiography, cardiac magnetic resonance (CMR), nuclear imaging, and positron emission tomography—has significantly enhanced both diagnostic accuracy and prognostic assessment. 17
Renal involvement in FD is characterized by proteinuria and progressive renal failure, primarily resulting from Gb3 deposition, particularly in podocytes. Proteinuria often serves as the earliest indicator of renal impairment. Factors associated with accelerated renal disease progression in male FD patients include baseline proteinuria, reduced glomerular filtration rate, and hypertension, 18 consistent with findings in this case. Studies from Germany have further linked severe proteinuria to rapid decline in renal function, indicating that a urine protein-to-creatinine ratio >1.5 signifies an increased risk. 19
Given the role of mineralocorticoid receptor overexpression in promoting oxidative stress, inflammation, and fibrosis—key drivers of cardiovascular disease and CKD—early intervention is crucial. Finerenone, a novel nonsteroidal, highly selective MRA, can achieve significant reductions in cardiac and renal endpoints in clinical trials, positioning it as a promising therapeutic tool for CKD.20,21 As a third-generation MRA, finerenone exhibits higher receptor selectivity than spironolactone and greater receptor affinity than eplerenone. Its distinct mechanism involves altering the MR conformation via its side chain, which impedes the receptor’s activation. This unique action likely underpins its potent anti-inflammatory and antifibrotic properties. Furthermore, finerenone achieves a nearly balanced distribution between cardiac and renal tissues and can deliver comparable efficacy at lower doses while avoiding the sex hormone–related adverse reactions associated with nonselective blockers. 22 Furthermore, studies have indicated that finerenone delays kidney disease progression and reduces cardiovascular events in CKD patients. 23 Consequently, continuous finerenone treatment in this case showed no adverse reactions, with improvements in both proteinuria and renal function. We observed a significant improvement in serum creatinine levels and proteinuria within 1 month of initiating finerenone, with subsequent sustained stability. The patient’s serum potassium level remained normal, underscoring the safety of this agent in this context.
Recently, sodium–glucose cotransporter 2 (SGLT2) inhibitors have emerged as pivotal supportive therapies in CKD, including potential applications in rare diseases. 24 Their renoprotective effects, independent of glycemic control, suggest a potential synergistic role with finerenone in future multi-mechanistic approaches to manage Fabry nephropathy. Furthermore, no clinically significant drug interactions were observed between finerenone, valsartan, and α-galactosidase.
Given the diagnostic challenges, diverse manifestations, and therapeutic complexity of FD, management requires a multidisciplinary approach tailored to disease severity and individual symptoms. Over the past two decades, two ERTs (agalsidase alfa and agalsidase beta) and the chaperone therapy using migalastat have been approved; these therapies have aided in stabilizing symptoms and reducing the disease burden. Additional therapeutic agents remain under investigation. 25
This case report has certain limitations. It is a single case report, and the findings require validation in larger, controlled studies. The observed stabilization could be influenced by the synergistic effect of finerenone with ERT; the efficacy of finerenone alone in Fabry nephropathy remains unknown. Furthermore, long-term outcomes beyond the current follow-up period need to be monitored.
Conclusion
This single case suggests that ERT combined with a nonsteroidal MRA aids in preventing the progression of renal dysfunction in patients with FD. This combination was well-tolerated and associated with improvement in biochemical parameters. Further studies are needed to confirm these preliminary findings and define the role of this therapeutic strategy in Fabry nephropathy.
Footnotes
Acknowledgments
We would like to thank our colleagues for their review of our article and for their invaluable suggestions. The manuscript was professionally edited for English language by a professional editing service.
Authors’ contributions
Guan-qiang Tian and Ping Yang contributed to conception, methodology, data curation, analysis, and writing. Zu-chun Liao and Wen Xiao contributed to data collection. Jun Li and Bin Song provided guidance and supervision. All authors read and approved the final manuscript.
Consent to participate
Informed consent to participate in this study was obtained from the patient.
Consent for publication
Informed consent to publish the relevant data was obtained from the patient.
Data availability statement
The data supporting this study’s findings are available within the article and its supplementary materials.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Ethical approval for this case report was waived by the Ethics Committee of People’s Hospital of Deyang City as it is a retrospective analysis of a single case and does not constitute a research study per institutional policy.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
