Abstract
Objectives
This study aimed to investigate the effect of postoperative dexmedetomidine infusion on the recovery of gastrointestinal function in patients undergoing laparoscopic colorectal surgery.
Methods
A total of 128 participants were randomized into dexmedetomidine and control groups. In the dexmedetomidine group, patients received a loading dose of 0.5 μg/kg dexmedetomidine over 15 min before skin incision followed by continuous infusion of 0.05 μg/kg/h starting at the end of surgery and maintained for 48 h. The control group received the same loading dose of dexmedetomidine without the postoperative infusion.
Results
No statistically significant differences were observed between the dexmedetomidine and control groups in time to first flatus, time to first defecation, or time to first oral feeding. The incidences of postoperative gastrointestinal function and delirium were comparable between the two groups. However, patients in the dexmedetomidine group experienced better sleep quality and had lower C-reactive protein levels than those in the control group.
Conclusions
Although postoperative continuous infusion of dexmedetomidine was associated with improved sleep quality in patients undergoing laparoscopic colorectal surgery, it did not accelerate the recovery of gastrointestinal function.
Keywords
Introduction
Postoperative gastrointestinal dysfunction (POGD) is an acute pathophysiological alteration of the gastrointestinal tract, characterized by gastrointestinal mucosal damage, barrier dysfunction, and gastrointestinal motility disorders. 1 Clinically, POGD manifests as nausea and vomiting, abdominal distension, intolerance to solid or semi-liquid foods within 24 h postoperatively, and radiological evidence of postoperative ileus (POI). The incidence of POGD remains high, ranging from 10% to 30%, and it contributes to prolonged hospital stays, increased readmission rates, and elevated healthcare costs. 2
Dexmedetomidine, a highly selective alpha2 (α2)-adrenergic receptor agonist, is characterized by its sedative, analgesic, and antisympathetic effects. 3 It is widely used for perioperative sedation and postoperative analgesia and has recently been approved for reducing the incidence of postoperative delirium 4 and numeric rating scale (NRS) score. 5 However, its effects on gastrointestinal function remain controversial. 6 Several clinical studies have reported that dexmedetomidine promotes bowel recovery after colorectal surgery, 7 cesarean section, 8 and lumbar spinal fusion. 9 In contrast, other studies suggested that it inhibits gastric emptying.10,11 This discrepancy may be partly explained by animal studies demonstrating that dexmedetomidine induces vasoconstriction, thereby impairing microperfusion in the colon, jejunum, and stomach. 12
High-quality studies on the effect of postoperative continuous infusion of dexmedetomidine on gastrointestinal function recovery in patients undergoing laparoscopic colorectal surgery remain limited. Therefore, this study hypothesized that postoperative continuous infusion of dexmedetomidine accelerates the recovery of gastrointestinal function following laparoscopic colorectal surgery.
Materials and methods
Study design and settings
This single-center, randomized controlled study was conducted between 9 January 2024 and 29 December 2024 at the affiliated Suzhou Hospital of Nanjing Medical University, China. The study (Number: J-2023-070-K01) was approved on 30 December 2023 by the Ethics Committee of the affiliated Suzhou Hospital of Nanjing Medical University, located at 358 Shizi Road, Suzhou, Jiangsu, China, and was registered with the Chinese Clinical Trial Registry (Registration Number: ChiCRT2400079610). The study was conducted in accordance with the principles of the Declaration of Helsinki 1975, as revised in 2024. Reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) statement. 13 All participants provided written informed consent.
Participants
Patients aged 18–75 years who were scheduled for elective laparoscopic colorectal surgery with an estimated duration of 1–6 h were screened for eligibility. Exclusion criteria included the following: (a) allergy to dexmedetomidine; (b) refusal to participate; (c) preoperative cognitive impairment (e.g. severe dementia, intellectual disability, or psychiatric disorders); (d) inability to cooperate with assessments (e.g. language barrier, severe hearing or visual impairment, or coma); (e) patients with sick sinus syndrome, severe bradycardia (heart rate <50/min), or second degree or higher atrioventricular block; (f) patients with permanent pacemaker implantation or severe heart failure; (g) patients with Alzheimer’s disease or schizophrenia. Patients should be followed up until the time of first flatus. No patient or member of the general public was involved in the design, conduct, or reporting of the trial.
Randomization and group assignment
Eligible participants were randomized into two groups in a 1:1 ratio based on computer-generated random number sequence. Group assignments were concealed in sequentially numbered opaque envelopes 1 h prior to anesthesia induction. Patients were blinded to group assignments throughout the study. Anesthesiologists were aware of patient allocation but did not participate in the follow-up assessments. Follow-up researchers were blinded to the intervention. The blinding was disclosed when a serious adverse reaction was observed.
Anesthesia and analgesia procedures
All participants received a preincisional loading dose of dexmedetomidine (0.5 μg/kg) before skin incision. General anesthesia was induced with propofol (1.0–1.5 mg/kg), sufentanil (0.4 μg/kg), and cisatracurium (0.2 mg/kg). After 2 min of cisatracurium administration, tracheal intubation was performed. End-tidal carbon dioxide (EtCO2) level was maintained at 35–45 mmHg. Anesthesia was maintained using propofol (2–10 mg/kg/h), remifentanil (5–20 μg/kg/h), and sevoflurane (1%–3%) to keep the bispectral index (BIS) in the range of 40–60. Prior to skin incision, 100 mg flurbiprofen axetil and 0.2 μg/kg sufentanil were administered. A heating blanket was used intraoperatively to maintain nasopharyngeal temperature >36°C. Postoperatively, every patient in the dexmedetomidine group received a patient-controlled analgesia (PCA) pump containing dexmedetomidine (0.05 μg/kg/h), sufentanil (0.6 μg/kg/day), and flurbiprofen axetil (200 mg) for 48 h. Every patient in the control group received a PCA pump with sufentanil (0.6 μg/kg) and flurbiprofen axetil (200 mg). Rescue analgesia (100 mg flurbiprofen axetil) was provided when the visual analog scale (VAS; 0 = no pain, 10 = worst pain) score exceeded 4. On postoperative day 1, patients were encouraged to sit at their bedside and begin early ambulation with family assistance.
Outcome measures
The primary outcome was the time to first flatus. Secondary outcomes included the following: postoperative gastrointestinal function assessed using the I-FEED scoring system (intake, feeling nauseated, emesis, physical examination, and duration of symptoms), time to first defecation, time to first oral feeding, incidence of delirium (assessed twice daily for 2 days postoperatively using the 3-min Diagnostic Interview for Confusion Assessment Method (CAM)–defined Delirium (CAM-3D)), sleep quality parameters measured at 24 and 48 h (total sleep time and sleep efficiency, monitored using ActiGraph WGT3X-BT), postoperative pain scores at rest and movement at 24 and 48 h postoperatively, postoperative anxiety level measured using the Self-Rating Anxiety Scale (SAS) at 48 h postoperatively, incidence of postoperative nausea and vomiting (PONV) at 48 h, C-reactive protein (CRP) level at 48 h postoperatively, and length of hospital stay.
The I-FEED scoring system comprises 5 components, each yielding 0 to 2 points. A total score of 0–2 indicates normal gastrointestinal function, 3–5 indicates gastrointestinal intolerance, and 6 indicates POGD. Pain severity was assessed using a 10-point NRS, with 0 representing no pain and 10 representing the worst unbearable pain. Subjective sleep quality was evaluated according to the Pittsburgh Sleep Quality Index (PSQI), whereas objective sleep parameters were measured using a wrist-worn ActiGraph WGT3X-BT monitor. Anxiety was assessed via SAS, which consists of 20 items, each yielding 2 points. Total SAS scores of 50–59, 60–69, and ≥70 were interpreted as indicating mild, moderate, and severe anxiety, respectively.
Postoperative adverse events were systematically recorded at 48 h, including bradycardia (<40 beats per min), cardiac arrest, hypertension (>20% baseline pressure or systolic blood pressure >160 mm Hg), hypotension (>20% baseline pressure or systolic blood pressure <80 mm Hg), and severe allergic reaction.
Statistical analyses
The sample size was determined based on the incidence of POGD. According to the pilot study, the incidence of POGD was equivalent to 30% in the control group. Based on preliminary observations, the incidence of POGD was approximately 7% in patients receiving dexmedetomidine. Assuming a 1:1 ratio of patients in the two groups, a two-sided test with a significance level (α) of 0.05, and a power (1−β) of 0.90, the required sample size was 60 in each group. Considering an 8% withdrawal and loss to follow-up, 65 patients were included in this study.
Statistical analyses were performed using Graphpad Prism 9.0 software. The Kolmogorov–Smirnov test was used to identify the normality of continuous variables. For continuous variables, mean ± SD or median ± interquartile range (IQR) values were reported according to the normality of the distribution. Categorical variables were presented as percentages. Differences between the dexmedetomidine and control groups were analyzed using the unpaired t-test for normally distributed continuous variables and the Mann–Whitney U test for non-normally distribution continuous variables. Fisher’s exact test was used for categorical datasets. A two-tailed P values <0.05 was considered statistically significant.
Results
Of the 130 patients screened for eligibility, 2 were excluded. The remaining 128 patients were randomly assigned to the dexmedetomidine (n = 64) and control (n = 64) groups (Figure 1). During the study, 4 patients in dexmedetomidine group were excluded (1 due to unplanned intensive care unit (ICU) admission, 2 due to changes in surgical plan, and 1 due to postoperative withdrawal). Similarly, 4 patients in the control group were excluded (3 due to changes in the surgical plan and 1 due to postoperative withdrawal). Consequently, 120 patients (60 per group) were included in the per-protocol analysis.
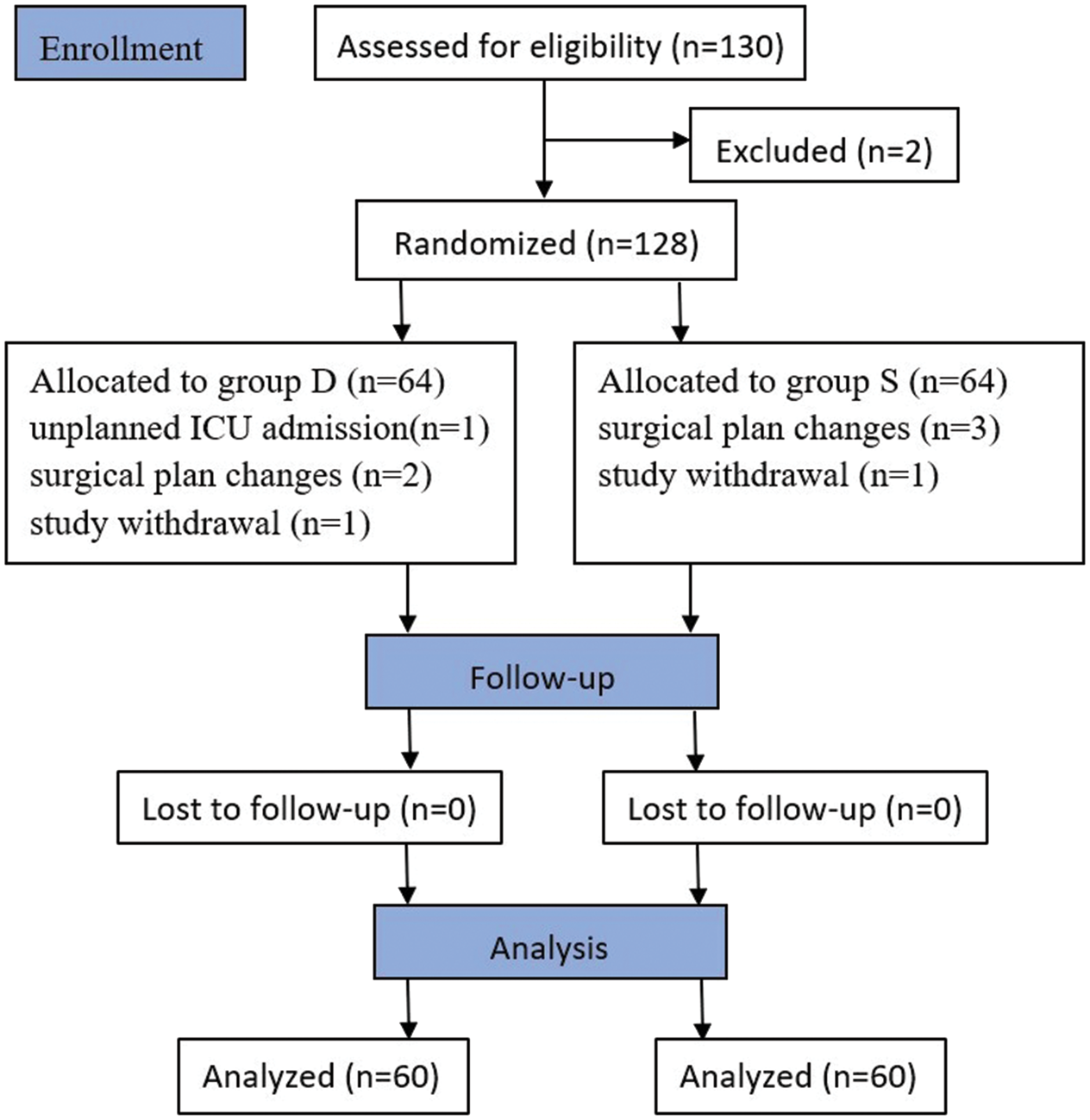
CONSORT flow diagram. CONSORT: Consolidated Standards of Reporting Trials.
In total, 83 patients (69.2%) were male and 37 (30.8%) were female. Baseline characteristics (age, sex, height, and weight) of the dexmedetomidine and control groups were comparable, with no statistically significant differences in American Society of Anesthesiologists status, type of surgery, surgical time, anesthesia duration, and albumin levels. There was no difference in the output of urine between the dexmedetomidine and control groups (Table 1).
Patient characteristics and intraoperative data.

ASA: American Society of Anesthesiologists; BMI: body mass index; IQR: interquartile range.
Class I indicates normal health and class II indicates mild systemic disease.
Values are rounded to the nearest 100 mL.
Primary outcome
The median time of first flatus did not differ between the dexmedetomidine and control groups, with values reported as 59 h (50.5–66.9) and 63.5 h (49.25–81.6), respectively (P = 0 .15; Table 2).
Postoperative clinical outcomes.

Values are rounded to the nearest hour.
χ2 value.
Assessed twice daily using the 3-min Diagnostic Interview for CAM–defined Delirium, which evaluates acute onset and fluctuating course, inattention, disorganized thinking, and altered level of consciousness.
IQR: interquartile range; VAS: visual analog scale; CRP: C-reactive protein; SAS: Self-Rating Anxiety Scale; CAM: Confusion Assessment Method.
Secondary outcomes
The I-FEED scores of the dexmedetomidine and control groups were comparable (Figure 2). There were no statistically significant differences between the groups in the time to first oral feeding and time to first defecation. The incidence of delirium in the dexmedetomidine and control groups was 10 (16.7%) and 8 (13.3%), respectively, at 24 h postoperatively and 3 (5%) and 2 (3.3%), respectively, at 48 h postoperatively.

I-FEED score between the dexmedetomidine and control groups. I-FEED indicates intake, feeling nauseated, emesis, physical examination, and duration of symptoms. POGI: postoperative gastrointestinal injury; POGD: postoperative gastrointestinal dysfunction.
Patients in the dexmedetomidine group had longer total sleep time and higher sleep efficiency at 24 h (P < 0.0001) and 48 h (P = 0.0031) postoperatively.
Although there was no statistically significant difference between the two groups in the resting VAS scores at 24 h postoperatively, patients in the dexmedetomidine group reported lower VAS scores upon movement. However, VAS scores were comparable between the two groups at 48 h postoperatively.
Compared with the control group, patients in the dexmedetomidine group exhibited significantly lower CRP levels (P = 0.0087). In the dexmedetomidine group, fewer patients exhibited anxiety at 48 h postoperatively. There were no differences between the two groups in the incidence of PONV, hypertension, and bradycardia at 48 h postoperatively (Table 2).
Discussion
This randomized trial investigated the effects of postoperative continuous infusion of dexmedetomidine on the recovery of gastrointestinal function. The results indicated that continuous postoperative dexmedetomidine infusion did not reduce the time to first flatus, first defecation, and first oral intake. Additionally, it had no statistically significant effect on the incidence of delirium up to 2 days postoperatively. However, compared with the control group, the dexmedetomidine group showed lower postoperative VAS scores during movement at 24 h postoperatively, better sleep quality in the first 2 postoperative days, lower SAS scores, and reduced CRP levels within the first 48 h after surgery.
Our results differ from those of previous studies. In laparoscopic colorectal cancer resection, patients who received a loading dose of dexmedetomidine (1 μg/kg) before anesthesia induction and continuous infusion (0.3 μg/kg/h) during the surgery experienced a shorter time to first flatus and first defecation. 14 Another clinical trial reported that single-dose administration of 0.5 μg/kg dexmedetomidine for 15 min followed by continuous infusion of 0.2 μg/kg/h dexmedetomidine until 30 min before the end of surgery not only shortened the time of first flatus but also reduced the NRS scores 6 h postoperatively and decreased the incidence of PONV. 15 In contrast, no acceleration in gastric emptying was observed in critically ill patients who received intraoperative dexmedetomidine. 16 Similarly, our results did not demonstrate any beneficial effect of dexmedetomidine on postoperative bowel function, although some patients exhibited an earlier recovery of first flatus. Although dexmedetomidine clearly attenuated the stress response and improved the VAS scores, consistent with other trials, 17 its role in promoting gastrointestinal recovery remains unconfirmed in our study cohort.
The following explanations may account for the inconsistencies observed in the data. First, as an α2-adrenergic receptor agonist, dexmedetomidine at low doses reduces sympathetic tone and may promote gastrointestinal transit via central α2-adrenergic receptors. In contrast, higher doses can inhibit transit through excitatory cholinergic pathways via peripheral α2-adrenergic receptors inhibition.18,19 Second, although patients experienced better sleep quality and demonstrated lower CRP levels through the antisympathetic and antistress effect of dexmedetomidine, the recovery of intestinal function depends on a complex interplay of pathophysiological mechanisms. These include interactions between pharmacological factors, ischemic–reperfusion injury, inflammation, ambulation, and early diet initiation after the surgery. Moreover, the inhibitory effects of opioids on gastric motility and bowel propulsion cannot be overlooked. 20 Recently, new drugs such as alvimopan and methylnaltrexone have demonstrated efficacy in blocking the peripheral effects of μ-opioid receptors in the gastrointestinal tract while preserving opioid-mediated central analgesia. 21 Dexmedetomidine may promote postoperative recovery of gastrointestinal function by reducing the need for intraoperative and postoperative administration of sufentanil and remifentanil; 22 however, this effect was not observed in our study. Furthermore, patients in the dexmedetomidine group exhibited lower CRP levels than those who did not receive dexmedetomidine via the analgesia pump. This effect is likely due to the ability of dexmedetomidine to attenuate ischemic–reperfusion injury and suppress inflammatory responses. 23 In addition, gastrointestinal injury is an overlooked factor that must be considered.
Our study has three limitations. First, the precise effects of dexmedetomidine on gastrointestinal function remain unclear. Therefore, multicenter controlled trials are needed to clarify its role. Second, subgroup analysis based on tumor location (e.g. rectum, left colon, and right colon) was not performed. Third, potential influence of age on postoperative intestinal function recovery was not explored, which warrants future investigation.
Conclusions
Although dexmedetomidine improved postoperative sleep quality, it did not accelerate gastrointestinal recovery after laparoscopic colorectal surgery in our trial. Therefore, larger-scale studies are warranted to draw definitive conclusions.
Footnotes
Acknowledgments
We thank the doctors of the Gastrointestinal Surgical Department of The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou, Jiangsu province, China, for their kind help for sample collection. We also thank all the participants for their cooperation.
Author contributions
Fei Wang wrote the original draft and analyzed the data. Xiaoqian Liu and Jiaqi Gu were responsible for materials preparation, data collection, and project administration. Xian Chen interpreted the data and revised the manuscript. Yumin Zhu designed the study and revised the manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors report no conflicts of interest in this work.
Funding
Not applicable.
