Abstract
Objective
To compare the clinical efficacy and safety of dexmedetomidine and propofol in patients who underwent gastrointestinal endoscopy.
Methods
Relevant studies comparing dexmedetomidine and propofol among patients who underwent gastrointestinal endoscopy were retrieved from databases such as PubMed, Embase, and Cochrane Library.
Results
Seven relevant studies (dexmedetomidine group, n = 238; propofol group, n = 239) met the inclusion criteria. There were no significant differences in the induction time (weighted mean difference [WMD] = 3.46, 95% confidence interval [CI] = −0.95–7.88, I2 = 99%) and recovery time (WMD = 2.74, 95% CI = −2.72–8.19, I2 = 98%). Subgroup analysis revealed no significant differences in the risks of hypotension (risk ratio [RR] = 0.56, 95% CI = 0.25–1.22) and nausea and vomiting (RR = 1.00, 95% CI = 0.46–2.22) between the drugs, whereas dexmedetomidine carried a lower risk of hypoxia (RR = 0.26, 95% CI = 0.11–0.63) and higher risk of bradycardia (RR = 3.01, 95% CI = 1.38–6.54).
Conclusions
Dexmedetomidine had similar efficacy and safety profiles as propofol in patients undergoing gastrointestinal endoscopy.
Keywords
Introduction
Gastrointestinal endoscopy is extremely useful for the diagnosis and treatment of various diseases and conditions, including upper digestive tract bleeding, early gastric cancer, hepatobiliary and pancreatic diseases, and esophageal varices.1,2 Nevertheless, anxiety, pain, fear, and gastrointestinal adverse reactions can lead to poor cooperation during endoscopic procedures, which might result in adverse cardiovascular events.3,4 Therefore, sedatives play an extremely important role in endoscopy, and various sedatives are typically used during the process of endoscopy.
Propofol is a powerful sedative commonly used in gastrointestinal endoscopic surgery with the characteristics of rapid effects, a short action time, and fast recovery. It can cause mild analgesia and adverse reactions, including transient hypotension, dose-dependent respiratory depression, and inadequate ventilation.5,6 Propofol can uncouple the electron transport chain, which can lead to metabolic acidosis, hypotension, bradycardia, arrhythmia, and asystole. 7
Dexmedetomidine was approved for sedation by the US Food and Drug Administration in 1999. Dexmedetomidine is a highly selective α-2 adrenergic agonist with a higher a2/a1 activity ratio than clonidine. It has sedative, analgesic, sympathetic and hemodynamic stability. It has superior characteristics, and accidental overdose does not inhibit breathing, representing another advantage over other sedatives.8,9 However, dexmedetomidine can decrease cardiac output.10,11 Dexmedetomidine has sympatholytic effects that result in reduced blood pressure and heart rate via the activation of α2-adrenal receptors in the brain.12,13 In addition, the postsynaptic activation of endothelial cells induces vasodilatation. 14 Dexmedetomidine only has a mild respiratory depressing effect compared with other analgesics.15,16 Compared with other drugs, dexmedetomidine preserves the hypercapnic response, which limits apnea.15,16 Dexmedetomidine also causes a hypercapnic arousal phenomenon that resembles that of normal sleep. 17
Studies comparing clinical efficacy and safety between dexmedetomidine and propofol have reported inconsistent results. For example, Yan et al. reported that dexmedetomidine is superior to propofol, 8 whereas Tosun et al. stated that propofol is better than dexmedetomidine. 5 Hence, this meta-analysis was conducted to compare the efficacy and safety of propofol and dexmedetomidine in patients undergoing gastrointestinal endoscopy.
Material and methods
Literature search strategy
Published articles comparing dexmedetomidine and propofol among patients undergoing gastrointestinal endoscopy from inception to April 2021 were retrieved from PubMed, Embase, and Cochrane Library using the following keywords: (1) dexmedetomidine or DM; (2) propofol or PF; and (3) gastrointestinal endoscopy. The keywords were assembled with the Boolean operator “and” in the strategy. No restriction was set on the publication language in the literature retrieval. To maximize the search specificity and sensitivity, the reference lists of retrieved studies were also searched to identify any additional relevant studies. The study protocol was registered with INPLASY (INPLASY202160058).
Inclusion and exclusion criteria
The inclusion criteria were as follows: randomized controlled trials; comparison of dexmedetomidine and propofol; and inclusion of patients undergoing gastrointestinal endoscopy. The exclusion criteria were as follows: case studies, meta-analyses, letters to editors, or otherwise unsuitable; lack of a comparison between dexmedetomidine and propofol; patients did not undergo gastrointestinal endoscopy; data were insufficient or limited; and duplicate studies. If two studies were published by the same authors, the latest data were included.
Data extraction and quality assessment
Two reviewers independently extracted the data. For each study, the data collected included the date of publication, first author, study design, country, number of patients enrolled and randomized in each study, age (years), sex, American Society of Anesthesiologists physical status I to II, and concentration of dexmedetomidine. We extracted data for the induction time, the recovery time, and complications. Any disagreements were resolved by a third reviewer.
The Cochrane risk of bias assessment tool for randomized studies of interventions (ROB 2.0) was individually applied to all selected studies.18,19 The risk of bias of each study was rated as “high risk,” “low risk,” or “unclear” according to the match level between the extracted information and evaluation criteria. Despite every effort to maintain fairness during the quality assessment, minor grading errors were possible. This manuscript adheres to the applicable EQUATOR guidelines. 20 The quality assessment was also performed using the GRADE methodology. 19
Statistical analysis
Review Manager (version 5.2, The Cochrane Collaboration, 2011) was adopted to estimate the effects of the outcomes among the selected studies. Continuous variables were reported as the weighted mean difference (WMD) and 95% confidence interval (CI). Complications were reported as the relative risk (RR) and 95% CI. The number needed to treat (NNT) was also calculated for each complication. Heterogeneity was assessed in this study using the I2 statistic as follows: <50%, low; 50% to 75%, moderate, and >75%, high. If I2 >50%, the potential sources of heterogeneity were analyzed via sensitivity analysis. In addition, a random-effects model was used when heterogeneity was observed, whereas a fixed-effects model was adopted when no heterogeneity was observed. A funnel plot was not used to test potential publication bias because the number of studies was fewer than 10. 19 Sensitivity analyses were performed to examine the robustness of the results.
Results
Search process
The electronic search retrieved 151 articles. After a thorough review, 39 studies met the preliminary criteria. During further screening, 32 articles were excluded because of issues regarding the study design (n = 32), intervention (n = 3), comparator (n = 6), and outcomes (n = 5). Finally, seven studies were selected for the meta-analysis. Figure 1 presents a flowchart of the identification, inclusion, and exclusion of studies, reflecting the search process and the reasons for exclusion.
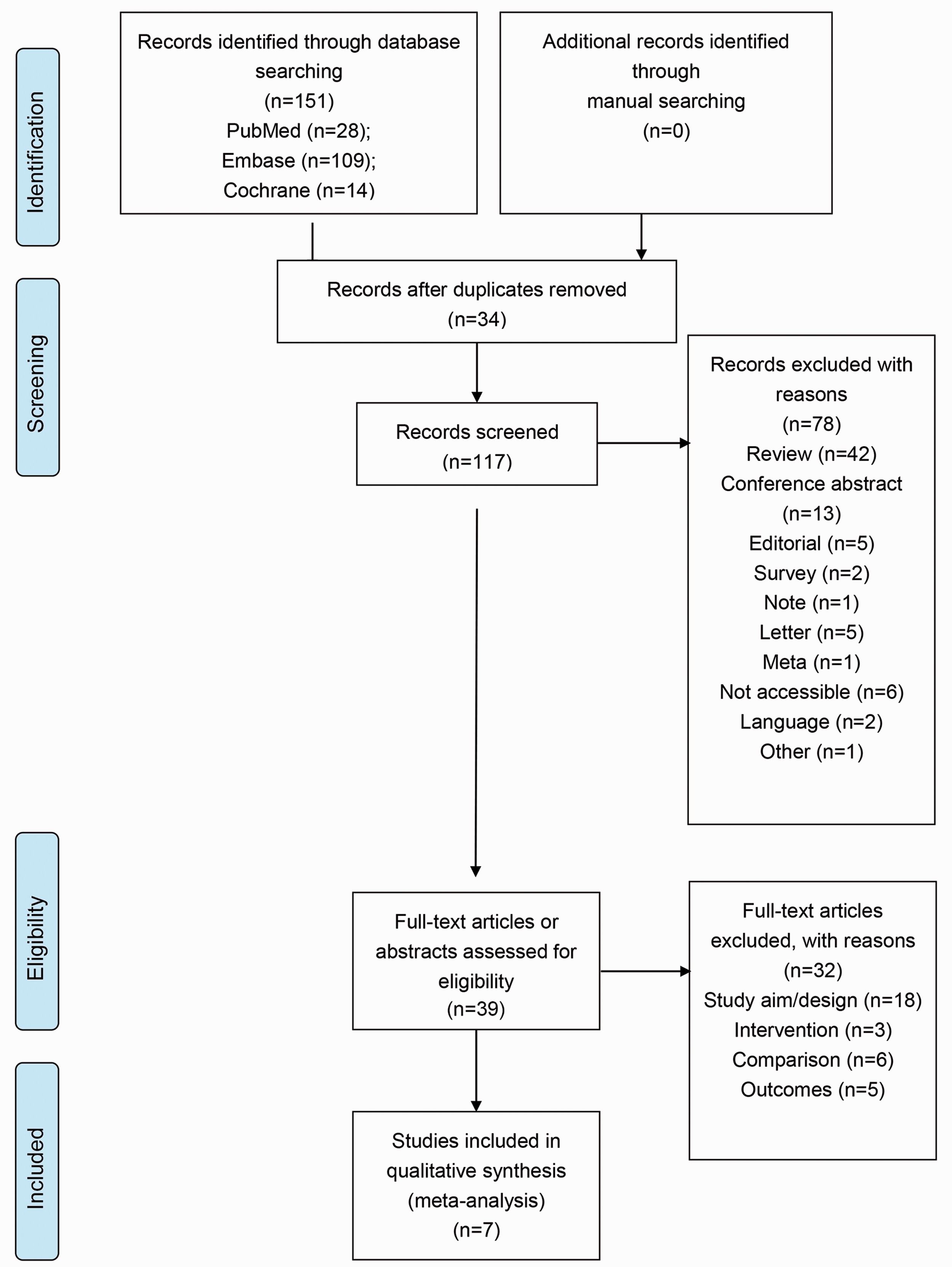
Flow diagram of the study selection process.
Characteristics of the included studies
Table 1 summarizes the type of study and the total number of patients associated with each group. The characteristics include first author, publication year, country, age, gender, group, sample size, and recruitment period.
Characteristics of the studies included in the meta-analysis.
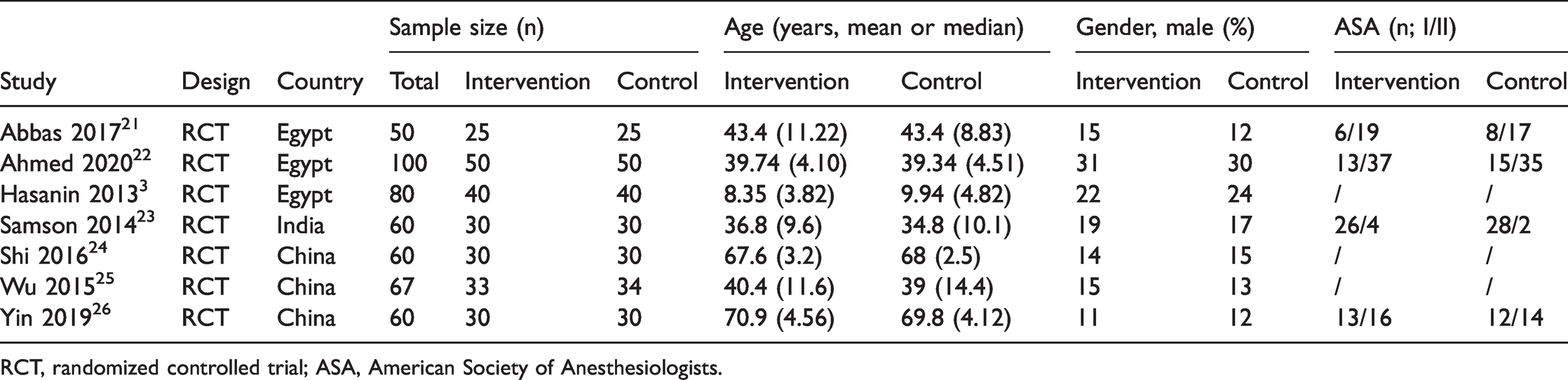
RCT, randomized controlled trial; ASA, American Society of Anesthesiologists.
This meta-analysis included 477 patients. All seven articles3,21–26 were published between 2013 and 2020. The sample size of the studies ranged 60 to 100 patients. The analysis included 238 patients in the dexmedetomidine group and 239 in the propofol group.
Results of quality assessment
The quality of the studies was assessed using the risk of bias table in the Review Manager 5.2 tutorial. Figure 2 presents the quality evaluation in this meta-analysis. There was limited bias among the included studies. Table 2 presents the GRADE analysis of the variables examined in this meta-analysis. The certainty was moderate for induction time; low for recovery time, hypoxia, hypotension, and bradycardia; and very low for nausea and vomiting.

Risk of bias graph. (a) Overview of the judgment of each risk of bias item presented as a percentage across all included studies. (b) Overview of the judgment about each risk of bias item for each included study.
GRADE approach for assessing quality of evidence. Author(s): Question: Dexmedetomidine compared with propofol for gastrointestinal endoscopy Setting: Bibliography:

CI, Confidence interval; MD, Mean difference; RR, Risk ratio.
Explanations
a. No description of the blinding method.
b. I2 = 98.1%, P = 0.000.
c. Unclear bias arising from the selection of the reported results.
d. Large confidence interval is large.
Results of the meta-analysis
Meta-analysis of the induction time (minutes)
Three included studies reported the induction time. The forest plot for the induction time between the dexmedetomidine and propofol groups is presented in Figure 3. The combined result suggested no statistically significant difference in the induction time (WMD = 3.46, 95% CI = −0.95–7.88; I2 = 99%, Pheterogeneity < 0.001).

Forest plot of the induction time between dexmedetomidine and propofol.
Meta-analysis of the recovery time (minutes)
Five included studies compared the recovery time between the dexmedetomidine and propofol groups. As illustrated in the forest plot (Figure 4), the meta-analysis revealed no statistically significant difference between the two groups (WMD = 2.74, 95% CI = −2.72; I2 = 98%, Pheterogeneity < 0.001).

Forest plots of the recovery time between dexmedetomidine and propofol.
Meta-analysis of complications
The subgroup analysis of complications between the groups identified no statistically significant in the risks of hypotension (RR = 0.56, 95% CI = 0.25–1.22) and nausea and vomiting (RR = 1.00, 95% CI = 0.46–2.21). However, dexmedetomidine carried a lower risk of hypoxia (RR = 0.26, 95% CI = 0.11–0.63) and a higher risk of bradycardia (RR = 3.01, 95% CI = 1.38–6.54 than propofol (Figure 5). Table 3 reveals that the NNT required to observe one instance of nausea and vomiting was 29.7, whereas those to prevent one event were 36.8 for hypotension, 8.3 for hypoxia, and 10.9 for bradycardia.

Forest plots of subgroup analyses of complications between dexmedetomidine and propofol.
Number needed to treat analysis for complications.

*number needed to treat to harm.
**number needed to treat to benefit
RR, risk ratio; CI, confidence interval.
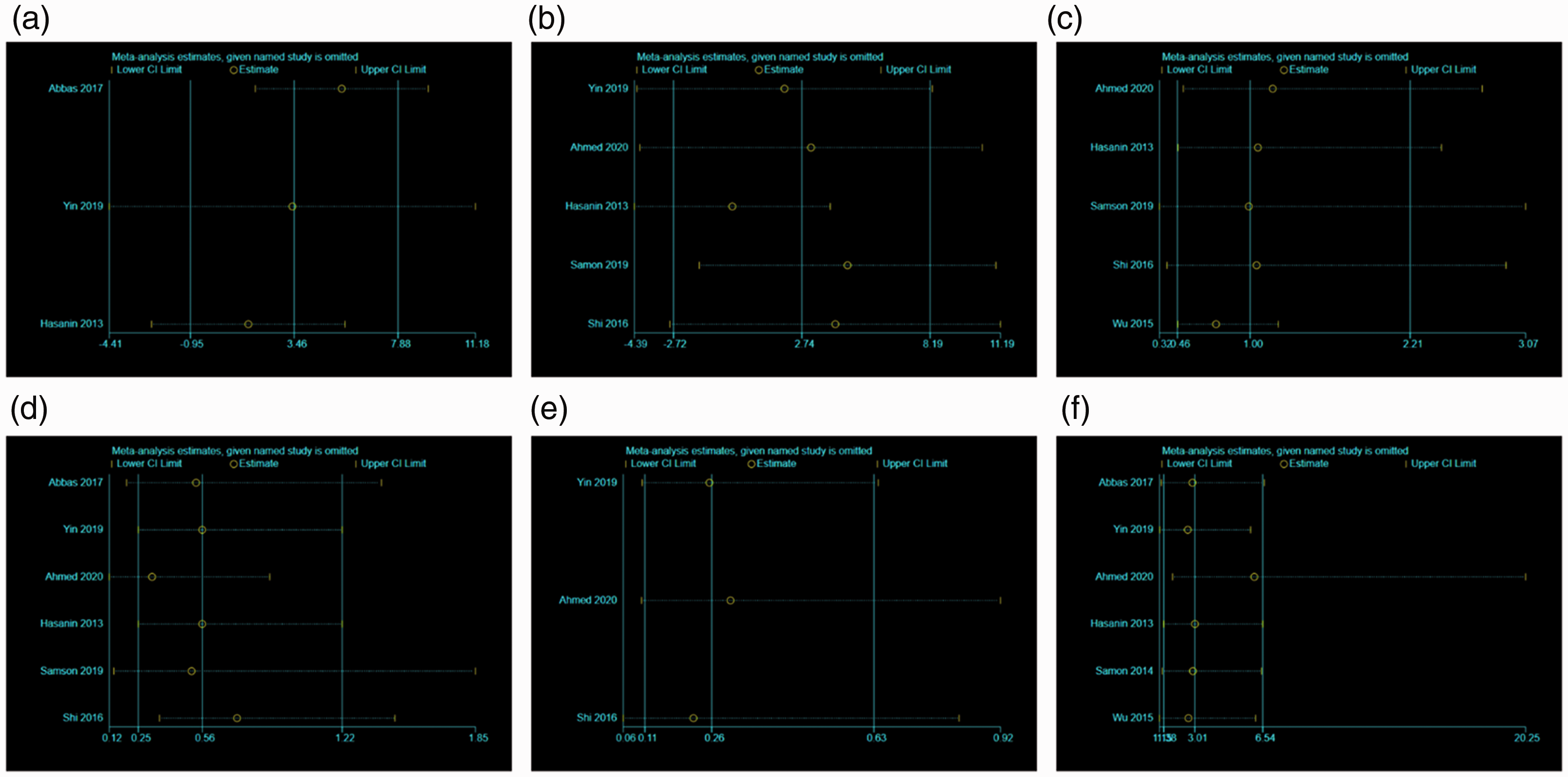
Results of sensitivity analysis and publication bias
As presented in Appendix file 1, the sensitivity analyses illustrated that the results were robust.
Discussion
Sedation in invasive surgery can provide appropriate care and help to complete the operation. Although usually safe and effective, adverse reactions may occur, especially in patients with complications.27–29 Appropriate sedation during surgery can help to reduce anxiety/pressure and the incidence of complications as well as promote patient cooperation, which can improve the success rate of endoscopy and patient satisfaction. 30 In the present meta-analysis of randomized controlled trials of patients undergoing gastrointestinal endoscopy, there were no differences between propofol and dexmedetomidine regarding the induction time, recovery time, and risks of hypotension and nausea and vomiting, but dexmedetomidine had an advantage in terms of hypoxia and a disadvantage regarding bradycardia.
Propofol, a phenolic derivative, has sedative and hypnotic effects mediated by the γ-aminobutyric acid receptor but no analgesic effect. However, propofol carries a risk of rapid onset of deep sedation, which may cause respiratory and cardiovascular depression.31,32 Compared with traditional sedatives, propofol, when used as a sedative during gastrointestinal endoscopy, has a shorter recovery time and better sedative effect, and it does not increase the incidence of cardiopulmonary complications.33,34
Dexmedetomidine is a new type of α2-adrenergic receptor agonist with high selectivity, and the drug is characterized by its ability to cause sedation, memory elimination, and sympathetic and analgesic effects. In a phase III study, dexmedetomidine (0.2–0.7 μg/kg/hour) produced clinically effective sedation and significantly reduced the analgesic requirements of ventilating patients in the intensive care unit.35–37
In the present meta-analysis, there were no differences in the induction and recovery times between the two drugs. These results are in agreement with a trial concluding that dexmedetomidine was not inferior to propofol or midazolam for maintaining sedation. 38 Wanat et al. reported that dexmedetomidine provided more rapid recovery than propofol after cardiovascular interventions, 39 and this was also observed after colonoscopy. 37 However, differences in study populations could explain these discrepancies. Physical capacity can vary by age, which can influence patient response to sedation. In addition, patients often have a variety of comorbidities that require various drugs and treatments, which could not be controlled in the present study.
In this study, there were no differences in the risk of hypotension between the two groups. This contradicts the results of a study reporting that dexmedetomidine reduced the risk of hypotension during colonoscopy compared with propofol. 40 Another study observed severe hypotension in patients treated with dexmedetomidine that necessitated premature treatment termination. 41 On the contrary, a meta-analysis by Nishizawa et al. 42 revealed no differences between dexmedetomidine and propofol regarding the risk of all complications, supporting the present analysis. The present study included some studies of older people, who often experience and require treatment with various drugs that could influence the results.
Regarding other complications, there were no differences in the risk of nausea and vomiting, in line with the findings of a meta-analysis by Nishizawa et al., 42 but dexmedetomidine was linked to lower rates of hypoxia and higher rates of bradycardia. Higher rates of bradycardia were observed in patients treated with dexmedetomidine in previous studies,38,40,41,43 as were lower rates of hypoxia.43,44 Again, differences in populations, comorbidities, procedures, and diseases could explain the difference findings among the studies. A meta-analysis by Pereira et al. 45 reported that dexmedetomidine reduced the risk of delirium versus propofol, but this adverse reaction could not be examined in the present meta-analysis.
This study had some limitations. First, more indicators evaluating other aspects between propofol and dexmedetomidine should have been included, although these indicators be analyzed in the future. Second, some comparisons of certain subgroups were not conducted, but such comparisons could also be analyzed in the future. Third, race was not included as a factor for subgroup analysis.
Conclusions
Dexmedetomidine was not associated with differences in the induction time and recovery time compared with propofol among patients undergoing gastrointestinal endoscopy. Although the drugs did not differ concerning the risks of nausea/vomiting and hypotension, dexmedetomidine carried a lower risk of hypoxia and a higher risk of bradycardia.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211032786 - Supplemental material for Comparison of clinical efficacy and safety between dexmedetomidine and propofol among patients undergoing gastrointestinal endoscopy: a meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605211032786 for Comparison of clinical efficacy and safety between dexmedetomidine and propofol among patients undergoing gastrointestinal endoscopy: a meta-analysis by Weihua Liu, Wenli Yu, Hongli Yu and Mingwei Sheng in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
