Abstract
Background
Although coronavirus disease 2019 and influenza are recognized risk factors for thrombotic conditions, the incidence of acute ischemic stroke following these infections since 2022 remains unknown.
Methods
Patients aged ≥65 years with no prior history of stroke, coronavirus disease 2019, or influenza who developed acute ischemic stroke from 3 days before to 28 days after a coronavirus disease 2019 or influenza diagnosis were identified using PCORnet and HealthVerity data collected from 1 January to 31 December 2022. Demographic characteristics, underlying conditions, level of care, outcomes, and acute ischemic stroke incidence were described and compared between patients with early acute ischemic stroke occurring from −3 to 7 days from the diagnosis date and late acute ischemic stroke occurring from 8 to 28 days.
Results
Among 245,352 patients in PCORnet and 639,396 patients in HealthVerity aged ≥65 years with COVID-19, the incidence of early acute ischemic stroke following coronavirus disease 2019 (PCORnet: 962 per 100,000; HealthVerity: 447 per 100,000) and influenza (PCORnet: 589 per 100,000; HealthVerity: 387 per 100,000) was significantly higher than that of late acute ischemic stroke (coronavirus disease 2019 PCORnet: 81 per 100,000; HealthVerity: 141 per 100,000; influenza PCORnet: 75 per 100,000; HealthVerity: 15 per 100,000) (all p < 0.01).
Conclusions
Although acute ischemic stroke incidence may have declined since 2020–2021, the persistent risk of acute ischemic stroke associated with acute coronavirus disease 2019 and influenza underscores the importance of continued monitoring and preventive strategies in older adults.
Introduction
Studies have consistently shown that coronavirus disease 2019 (COVID-19) is associated with an increased risk of thrombotic events, including pulmonary embolism and stroke.1,2 Systematic reviews and meta-analyses published in 2020 and 2021 estimated that the risk of stroke following COVID-19 ranged from 0.5% to 8.1%. Furthermore, several studies have reported an increasing risk of acute ischemic stroke (AIS) with greater COVID-19 severity, such as admission to an intensive care unit (ICU).1–4 Subsequent meta-analyses and randomized trials have demonstrated that COVID-19 and stroke are associated with an increased risk of mortality 5 and that therapeutic anticoagulation with heparin improves outcomes among hospitalized patients with COVID-19. 6 Current treatment guidelines recommend anticoagulation with therapeutic doses of unfractionated heparin or low molecular weight heparin for acutely ill hospitalized patients with COVID-19 who are at low risk of bleeding and prophylactic dosing for all other patients.7,8 Although no thromboprophylaxis recommendations exist for influenza, increased mortality during influenza seasons, including deaths from cerebrovascular events, has been recognized since the 19th century.9–12
Hypothesized pathophysiologic mechanisms underlying COVID-19–associated stroke include microvascular thrombosis, epithelial inflammation, and coagulopathy.1–4 These same mechanisms may also be operative in influenza. 12 Risk factors for stroke, including uncontrolled hypertension, diabetes, and cardiovascular disease, are associated with an increased risk of hospitalization for COVID-19 and severe COVID-19 outcomes. 13 Consequently, these factors may jointly contribute to COVID-19 severity and the subsequent risk of stroke in patients with COVID-19.1–4,12
COVID-19 has been shown to increase the risk of stroke across all age groups; however, this risk is higher among adults aged 65 years and older. In addition, the risk of stroke has not been well characterized following the release of thromboprophylaxis recommendations. We examined the incidence of AIS after COVID-19 or influenza diagnosis using two large electronic healthcare data assets: electronic health record (EHR) data from The National Patient-Centered Clinical Research Network (https://pcornet.org/data and https://github.com/PCORnet-DRN-OC/Query-Details/tree/master/therapeutics%20Query, https://pubmed.ncbi.nlm.nih.gov/33002635/), PCORnet (PC) and adjudicated insurance claims data from HealthVerity (HV).
Both data assets include information on underlying medical conditions that increase the risk of stroke and severe outcomes from COVID-19 or influenza. The study objective was to describe the incidence, timing, characteristics, and clinical outcomes of AIS among patients aged 65 years and older with COVID-19 or influenza who were diagnosed with AIS from 1 January to 31 December 2022.
Methods
PC
PC provided access to outpatient and inpatient EHR data from 40 sites, each representing one or more health care systems. Participating sites generated aggregate, site-level data using a single statistical program package run locally at each PC site. The results were returned to the Harvard Pilgrim Health Care Institute, the Coordinating Center for the Centers for Disease Control and Prevention (CDC)–funded surveillance project, and combined into a single aggregate report without individual-level data from all responding sites.
Inclusion criteria–PC
The aggregated report included patients aged 65 years and older who received care in PC-affiliated health care systems from 1 January to 31 December 2022. Patients with COVID-19 had EHR documentation of a positive severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) viral test result; an International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) diagnostic code for COVID-19 (U07.1 or U07.2); or a prescription or administration of a COVID-19 treatment, including nirmatrelvir/ritonavir or molnupiravir prescription or remdesivir or monoclonal antibody administration, and had no prior COVID-19 diagnosis within 6 months before the index date. Patients with influenza had EHR documentation of a positive influenza test result; an ICD-10-CM diagnostic code for influenza (J09*, J10*, or J11*); or a prescription or administration of influenza medication, including oseltamivir or baloxivir. For both conditions, the earliest date of a positive test, diagnosis, or therapy was defined as the index date. Patients were excluded if they had any prior ICD-10-CM code for ischemic stroke or transient ischemic attack in any PC records. Demographic and clinical information was examined for all patients with COVID-19 and influenza from 1 January to 31 December 2022.
HV
HV performs privacy-preserving record linkage of patient-level data from more than 75 data partners. We used CDC-licensed, HV adjudicated payor insurance claims data linked to SARS-CoV-2 testing data from commercial laboratories (August 2023 data release). The study period spanned from 1 January to 31 December 2022, with a 60-day follow-up period extending to 1 March 2023 after acute illness.
Inclusion criteria–HV
The study population included patients aged 65 years and older who were continuously enrolled in a closed payer system from 1 January 2019 to 31 December 2022, including the 60 days following the index date. Patients with COVID-19 had a documented COVID-19 ICD-10-CM code (U07.1 or U07.2) or a positive test for SARS-CoV-2 between 1 January and 31 December 2022, with no prior COVID-19 diagnosis within 6 months before the index date. Patients with influenza were defined by a documented ICD-10-CM code for influenza (J09*, J10*, or J11*) and no prior influenza diagnosis within 6 months. Patients were excluded if they had any prior ICD-10-CM code for ischemic stroke or transient ischemic attack since 1 January 2019.
Incident AIS was defined as one or more documented ICD-10-CM codes (I63) for ischemic stroke occurring from 3 days before to 28 days after the index date. The 3-day pre-index window was included because disease onset may occur before testing, diagnosis, or treatment for either infection, whereas the 28-day post-index window was included because complications may occur after acute illness. Early AIS was defined as occurring from 3 days before to 7 days after the index date, whereas late AIS was defined as occurring between 8 and 28 days post-index. Supplemental tables provide the number and timing of other thrombotic and inflammatory conditions that occurred after COVID-19 and influenza diagnoses in patients with AIS.
Statistical analyses
The incidence of AIS among patients with COVID-19 or influenza was calculated by dividing the total number of patients with AIS by the total number of patients with COVID-19 or influenza, respectively. Incidence rates per 100,000 people are presented in the text, and percentages are presented in the Tables and Figure. The 95% confidence intervals (CIs) were calculated based on a standard t-distribution. In addition to overall AIS incidence, stratified incidence was calculated because individual-level data for adjustment were not available. Stratification was performed by sex (male and female), age group for HV only (65–74 years, 75–84 years, and ≥85 years), race and ethnicity (non-Hispanic Asian, non-Hispanic Black, non-Hispanic multiracial, non-Hispanic Other, non-Hispanic White, Hispanic, and unknown), and illness severity within 3 days before to 28 days after the index date, as indicated by an outpatient visit, emergency department visit, hospitalization, or ICU admission (nonexclusive categories). Estimates were stratified by seven underlying conditions documented at least twice in the previous three years: hypertension, type 1 or 2 diabetes mellitus, hyperlipidemia, history of coronary artery disease, obesity, and smoking status, with ICD-10-CM codes provided in the supplemental appendix. Outcomes included in the PC data included discharge to hospice care, a nursing facility, discharge with home oxygen, or death within 90 days (non-exclusive categories). The proportion of patients with COVID-19 or influenza experiencing AIS in the early period versus the late period was compared using Pearson’s chi-squared tests. p-values <0.05 were considered statistically significant.
This activity was reviewed by the CDC, deemed not research, and conducted in accordance with applicable federal law and CDC policy, US Department of Health and Human Services. This study was conducted in accordance with the Declaration of Helsinki 1975, as revised in 2024, and followed standard guidelines for the reporting of observational cross-sectional studies. 14
Results
COVID-19 and ischemic stroke
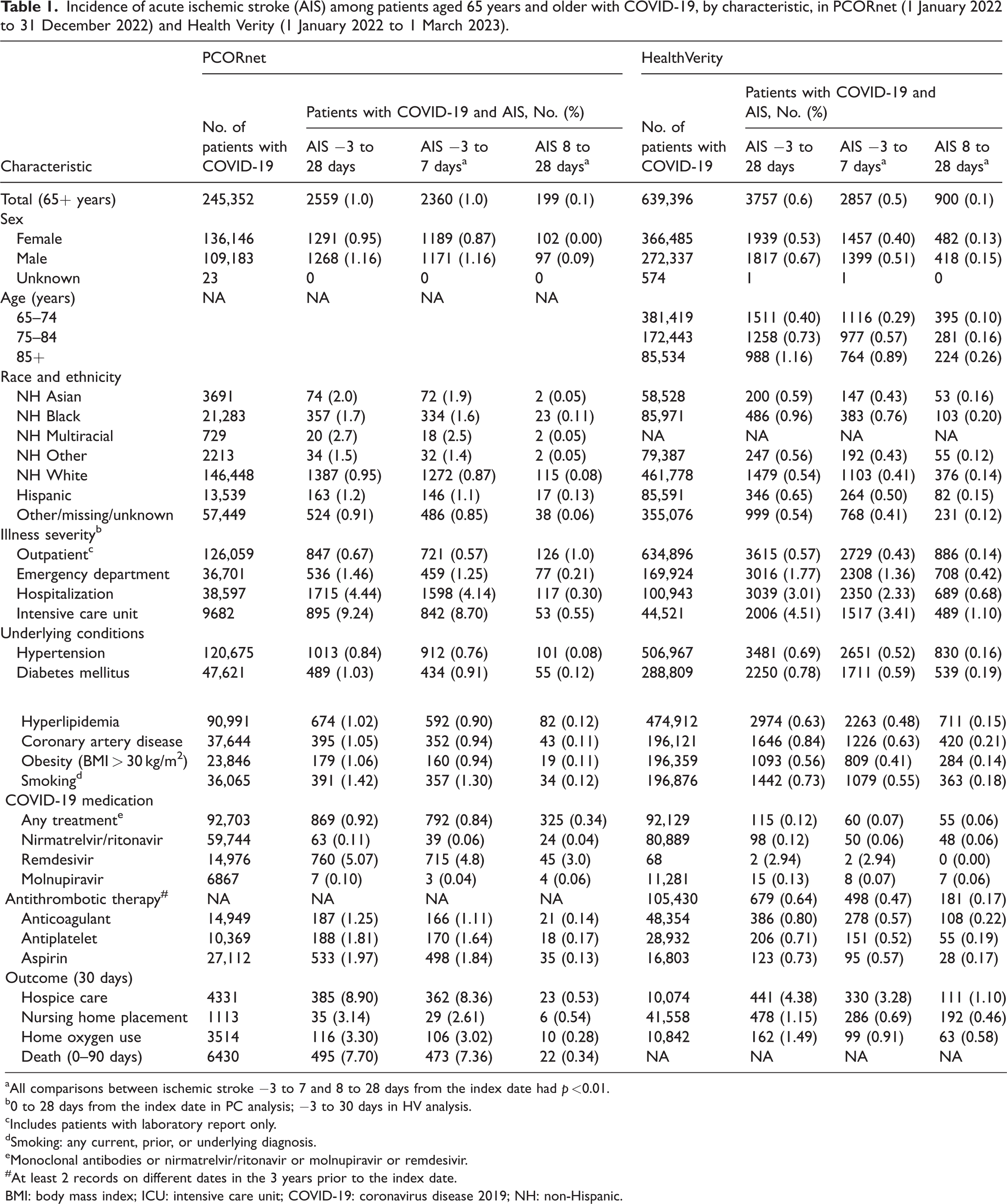
Overall, 245,352 patients in PC and 639,396 patients in HV met the COVID-19 inclusion criteria (Table 1). Among patients with COVID-19, 2559 in PC and 3757 in HV had an ICD-10-CM code for AIS from 3 days before to 28 days after the COVID-19 index date. The overall incidence of AIS was 1043 per 100,000 patients (95% CI: 1003–1083) in PC and 588 per 100,000 patients (95% CI: 569–618) in HV. Characteristics of patients with COVID-19 and AIS from PC and HV are shown in Table 1. Overall, 50% were female; 14% in PC and 13% in HV were non-Hispanic Black, 6.4% in PC and 9.2% in HV were Hispanic, and 54% in PC and 39% in HV were non-Hispanic White. Underlying conditions were common, particularly hypertension, which was present in 40% of PC patients and 93% of HV patients. Among patients with AIS, at least two-thirds were hospitalized, and 10% or more were discharged to hospice care. Other inflammatory conditions recorded among these patients, by days after the index date, are provided in Supplemental Table 1. Among the 245,352 patients in PC with COVID-19, information on coinfection was available. Overall, 779 patients (0.3%) were also diagnosed with influenza within 14 days after COVID-19 diagnosis; 3 experienced early AIS and 2 experienced late AIS.
Incidence of acute ischemic stroke (AIS) among patients aged 65 years and older with COVID-19, by characteristic, in PCORnet (1 January 2022 to 31 December 2022) and Health Verity (1 January 2022 to 1 March 2023).
All comparisons between ischemic stroke −3 to 7 and 8 to 28 days from the index date had p <0.01.
0 to 28 days from the index date in PC analysis; −3 to 30 days in HV analysis.
Includes patients with laboratory report only.
Smoking: any current, prior, or underlying diagnosis.
Monoclonal antibodies or nirmatrelvir/ritonavir or molnupiravir or remdesivir.
At least 2 records on different dates in the 3 years prior to the index date.
BMI: body mass index; ICU: intensive care unit; COVID-19: coronavirus disease 2019; NH: non-Hispanic.
The distribution of age, race and ethnicity, illness severity, underlying conditions, and most outcomes was similar between patients with early and late AIS (data not shown). Among patients with COVID-19 and AIS, 92% in PC and 76% in HV experienced early AIS (Figure 1). The incidence of early AIS was significantly higher than that of late AIS in PC (early AIS incidence, 962 per 100,000 patients; 95% CI: 923–1001 vs. late AIS incidence, 81 per 100,000 patients; 95% CI: 70–92) and HV (early AIS incidence, 447 per 100,000 patients; 95% CI: 430–473 vs. late AIS incidence, 141 per 100,000 patients; 95% CI: 132–156). Differences between early and late AIS were also observed over time. In PC, there were 87.4 early AIS events per 100,000 person-days and 2.8 late AIS events per 100,000 person-days, with an incidence rate ratio (IRR) of 30.8 (95% CI: 26.8–35.8). In HV, there were 40.6 early AIS events per 100,000 person-days and 6.7 late AIS events per 100,000 person-days, corresponding to an IRR of 6.0 (95% CI: 5.6–6.5). The incidence of early AIS was significantly higher than that of late AIS across all subgroups examined (Table 1).

Incidence (%) of acute ischemic stroke after COVID-19 and influenza among adults aged 65 years and older, by days since diagnosis date (−3 to 28 days, −3 to 7 days, and 8 to 28 days), PCORnet (1 January 2022 to 31 December 2022), and HealthVerity (1 January 2022 to 1 March 2023). COVID-19: coronavirus disease 2019.
Influenza and ischemic stroke
A total of 14,609 patients in PC and 59,736 patients in HV met the influenza inclusion criteria (Table 2). Among patients with influenza, 97 in PC and 318 in HV had an ICD-10-CM code for AIS from 3 days before to 28 days after the influenza index date. The overall incidence of AIS was 664 per 100,000 patients (95% CI: 532–796) in PC and 532 per 100,000 patients (95% CI: 474–591) in HV. Characteristics of patients with influenza and AIS from PC and HV are shown in Table 2. Overall, 44% of PC patients and 52% of HV patients were female; 22% in PC and 13% in HV were Black, 4% in PC and 11% in HV were Hispanic, and 52% in PC and 40% in HV were White. Underlying conditions were common, particularly hypertension, which was present in 40% of PC patients and 94% of HV patients. Among patients with AIS, at least two-thirds were hospitalized, and 10% or more were discharged to hospice care. Other inflammatory conditions recorded among these patients, by days after the index date, are provided in Supplemental Table 1. Among the 14,609 patients in HV with influenza, information on coinfection was available. Overall, 790 patients (5.4%) were also diagnosed with COVID-19 within 14 days after influenza diagnosis. Among these, three experienced early AIS and two experienced late AIS.
Incidence of acute ischemic stroke (AIS) among patients aged 65 years and older with influenza, by characteristic, in PCORnet (1 January to 31 December 2022) and Health Verity (1 January to 31 December 2022, with follow-up to 1 March 2023).

All comparisons between ischemic stroke −3 to 7 and 8 to 28 days from the index date had p <0.01.
0 to 28 days for PC analysis; −3 to 30 days for HV analysis.
Includes patients with laboratory report only.
Smoking: any current, prior, or underlying diagnosis.
At least 2 records on different dates in the 3 years prior to the index date.
BMI: body mass index; ICU: intensive care unit; NH: non-Hispanic.
The distribution of age, race and ethnicity, illness severity, underlying conditions, and most outcomes was similar between patients with early and late AIS (data not shown). Among patients with influenza and AIS, 89% in PC and 73% in HV experienced early AIS (Figure 1). The incidence of early AIS was significantly higher than that of late AIS in PC (early AIS incidence, 589 per 100,000 patients; 95% CI: 465–713 vs. late AIS incidence, 75 per 100,000 patients; 95% CI: 31–120) and HV (early AIS incidence, 387 per 100,000 patients; 95% CI: 337–436 vs. late AIS incidence, 15 per 100,000 patients; 95% CI: 12–18). Differences between early and late AIS were also observed over time. In PC, there were 53.5 early AIS events per 100,000 person-days and 3.6 late AIS events per 100,000 person-days, with an IRR of 14.6 (95% CI: 8.2–29.2). In HV, there were 35.2 early AIS events per 100,000 person-days and 7.0 late AIS events per 100,000 person-days, corresponding to an IRR of 5.0 (95% CI: 4.0–6.5). The incidence of early AIS was significantly higher than that of late AIS across all subgroups examined (Table 1).
Discussion
Among patients aged 65 years and older, the incidence of new AIS was highest from 3 days before to 7 days after the index date for COVID-19 and influenza. Although AIS incidence was significantly higher during the early period (−3 to 7 days) than during the later period (8 to 28 days) for both illnesses, the magnitude was highest for COVID-19 in PC. This may be because the PC sample, unlike the HV sample, included patients who received COVID-19–specific medication. As a result, more patients with COVID-19 and consequently more patients with AIS may have been captured in PC, resulting in a higher estimate than HV.
The finding that acute stroke occurs in the early part of COVID-19 illness aligns with population-based studies from Israel, Denmark, Sweden, and Scotland conducted in 2020.15–18 In these studies, between 66% and 75% of patients with stroke were hospitalized during the acute phase of COVID-19 illness. The overall proportion of patients with AIS following influenza in our study (0.6% to 1.0%) was consistent with findings from pre–COVID-19 pandemic evaluations of nonrespiratory diagnoses associated with influenza (0.9%) 17 and from other studies examining the risk of stroke associated with influenza.9–12,19 These studies also reported a higher incidence of stroke during the first 7–10 days of illness, corroborating our finding of increased stroke risk in the early post-infection period.
This study covered calendar year 2022, following the release of recommendations for the use of antithrombotic prophylaxis among hospitalized patients with COVID-19 in August 2021. 6 Unfortunately, we were unable to assess the use of prophylaxis use among hospitalized patients to determine its role in the development of AIS. Other studies examining COVID-19–related stroke incidence were conducted predominantly in 2020 and 2021, before the release of thromboprophylaxis recommendations. For example, in a study of hospitalized patients with COVID-19 conducted in 2020, the risk of stroke was 4% among 165 patients and 5%–6% in those with severe COVID-19. 3 A meta-analysis of 2020 data reported a pooled incidence of 1.4% for ischemic and hemorrhagic stroke following COVID-19, with a range of 0.5%–8.1% across studies. 4 Our findings fall within the lower range of these estimates, which may reflect changes in COVID-19 severity due to viral variants or population immunity, the effects of thromboprophylaxis, or other factors. Data on stroke incidence during the thromboprophylaxis era or the Omicron era remain limited. Nevertheless, an increased risk of mortality associated with COVID-19 persists among patients who also experience stroke, even in the setting of prophylaxis. 5
This study had several limitations. First, these observations were derived from patients who accessed health care, and the findings may not be generalizable to other populations. Second, this study was designed to report surveillance estimates of AIS at the population level. Because only aggregated data were available from PC, we were unable to adjust for age, other risk factors, or acute therapies, including receipt of thromboprophylaxis, when estimating the incidence of AIS. Third, because the diagnosis of AIS was not confirmed with imaging studies, some misclassification may have occurred. As a result, some patients with AIS may not have been captured, whereas others may have been included despite not having AIS. This limitation may have obscured potential associations between specific stroke subtypes and COVID-19 or influenza. Finally, these data may not have captured all occurrences of COVID-19, influenza, and AIS, particularly if patients received care at institutions not represented in the data sources.
In summary, COVID-19 and influenza were temporally associated with AIS among individuals aged 65 years and older, with the highest incidence occurring from 3 days before to 7 days after diagnosis. The higher risk of AIS observed with COVID-19 may be attributable to multiple factors that warrant further investigation, including underlying risk factors, protective factors, and disease-specific complications such as inflammation. These findings underscore the importance of preventive measures and close monitoring of older adults with COVID-19 or influenza, particularly during the first week of illness, when stroke risk is highest. Vaccination against COVID-19 and influenza, together with effective management of chronic conditions such as diabetes and cardiovascular disease, which are key contributors to stroke and severe COVID-19, remains essential for reducing thrombotic events. Improved understanding of barriers to thrombotic event prevention among these patients may inform effective interventions to further reduce risk. In addition, increasing awareness of early signs of stroke among patients and health care providers is critical for prompt recognition and timely intervention, which can substantially improve outcomes.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261416675 - Supplemental material for Acute ischemic stroke incidence after influenza or coronavirus disease 2019 among older US adults: A cross-sectional study using electronic health records
Supplemental material, sj-pdf-1-imr-10.1177_03000605261416675 for Acute ischemic stroke incidence after influenza or coronavirus disease 2019 among older US adults: A cross-sectional study using electronic health records by Emilia H Koumans, Julia Raykin, Fátima Coronado, Sharon Saydah, Tegan K Boehmer, Jennifer Wiltz, Shikha Garg, Carol E Desantis, Thomas W Carton, Lindsay G Cowell, Deepika Thacker, Jonathan Arnold, Sonja A Rasmussen, Samantha J Smith, Kimberly Barrett, Christine Draper, Elizabeth A Lundeen, Rebecca C Woodruff and Jason P Block in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605261416675 - Supplemental material for Acute ischemic stroke incidence after influenza or coronavirus disease 2019 among older US adults: A cross-sectional study using electronic health records
Supplemental material, sj-pdf-2-imr-10.1177_03000605261416675 for Acute ischemic stroke incidence after influenza or coronavirus disease 2019 among older US adults: A cross-sectional study using electronic health records by Emilia H Koumans, Julia Raykin, Fátima Coronado, Sharon Saydah, Tegan K Boehmer, Jennifer Wiltz, Shikha Garg, Carol E Desantis, Thomas W Carton, Lindsay G Cowell, Deepika Thacker, Jonathan Arnold, Sonja A Rasmussen, Samantha J Smith, Kimberly Barrett, Christine Draper, Elizabeth A Lundeen, Rebecca C Woodruff and Jason P Block in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank Evelyn Twentyman for initiating this project and Sarah Hamid for statistical consultation.
Author contributions
Emilia H. Koumans initiated the project; managed and analyzed data; and drafted, reviewed, and approved the final manuscript. Julia Raykin managed and analyzed data and reviewed and approved the final manuscript. Fátima Coronado provided technical input, reviewed and approved the final manuscript, and assisted with revisions. Sharon Saydah managed and analyzed data, reviewed and approved the final manuscript, and assisted with revisions. Tegan K. Boehmer initiated the project, managed data, and reviewed and approved the final manuscript. Jennifer Wiltz initiated the project and reviewed and approved the final manuscript. Shikha Garg provided technical input and reviewed and approved the final manuscript. Carol E. DeSantis managed data and reviewed and approved the final manuscript. Thomas W. Carton coordinated data collection and reviewed and approved the final manuscript. Lindsay G. Cowell coordinated data collection and reviewed and approved the final manuscript. Deepika Thacker coordinated data collection and reviewed and approved the final manuscript. Jonathan Arnold coordinated data collection and reviewed and approved the final manuscript. Sonja A. Rasmussen coordinated data collection and reviewed and approved the final manuscript. Samantha J. Smith managed data and reviewed and approved the final manuscript. Kimberly Barrett managed data and reviewed and approved the final manuscript. Christine Draper, MPH, managed data and reviewed and approved the final manuscript. Elizabeth A. Lundeen provided technical input and reviewed and approved the final manuscript. Rebecca C. Woodruff provided technical input and reviewed and approved the final manuscript. Jason P. Block initiated the project, coordinated data collection, and reviewed and approved the final manuscript.
Data availability statement
Because PCORnet, not CDC, maintains the data, the data are not publicly available.
Disclaimer
The opinions expressed by the authors contributing to this report do not necessarily reflect the official position of the Centers for Disease Control and Prevention or the institutions with which the authors are affiliated.
Funding
No funding was received to assist with the preparation of this manuscript.
Financial interests
The authors declare that they have no financial interests. The following authors declare they have no financial interests to disclose: EHK, JR, TKB, SS, JW, SG, CD, TWC, LGC, DT, JA, SAR, SJS, KB, CB, FC, EL, RW, and JPB.
Non-financial interests
SAR serves on advisory committees for pregnancy registries for medications manufactured by Axsome Therapeutics, Harmony Biosciences, Myovant Sciences, Novo Nordisk, and Pfizer, and receives royalties for an article on influenza and pregnancy from UpToDate.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
