Abstract
Objective:
The spread of Severe Acute Respiratory Syndrome Corona Virus-2 (SARS-CoV-2) in Ethiopia is below par understood and to date has been poorly characterized by a lower number of confirmed cases and deaths as compared with other regions of the Sub-Saharan African (SSA) countries. We aimed to investigate the seroprevalence of SARS-CoV-2 specific IgG antibodies, using the Abbott anti-nucleocapsid IgG chemiluminescent microparticle immunoassay, in two COVID-19 diagnosed and treatment centers of quarantined population during the first wave of the COVID-19 pandemic (since 30 April–30 May 2020).
Methods:
We analyzed data of 446 quarantined individuals during the first wave of COVID-19 pandemic. The data were collected using both interviewed and blood sample collection. Participants asked about demographic characteristics, COVID-19 infection symptoms, and its practice of preventive measures. Seroprevalence was determined using the severe acute respiratory syndrome coronavirus 2 IgG test.
Results:
The mean (± standard deviation) age of the respondent was 37.5 (±18.5) years. The estimated SARS-CoV-2 infection seroprevalence was found 4.7% (95% confidence interval: 3.1–6.2) with no significant difference on age and gender of participants. Severe acute respiratory syndrome coronavirus 2 antibody seroprevalence was significantly associated with individuals who have been worked by moving from home to work area (adjusted odds ratio = 7.8, 95% confidence interval: 4.2–14.3,
Conclusion:
Our study concluded that lower coronavirus disease 2019 seroprevalence, yet the large population in the community to be infected and insignificant proportion of seroprevalence, was observed between age and sex of respondents. Protective measures like contact tracing, face covering, and social distancing are therefore vital to demote the risk of community—strengthening factors should be continued as effect modification of anticipation for severe course of coronavirus disease 2019.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),1,2 poses huge health and societal burden globally. SARS-CoV-2 is easily transmitted from person to person 3 via droplets from the respiratory tract of infected people, including asymptomatic individuals. 4 The spectrum of the COVID-19 severity varies broadly, from asymptomatic infection to severe complication like organ failure to death. 5 Globally, more than 60 million confirmed cases of COVID-19, including 1,416,292 deaths, have been reported by World Health Organization (WHO) as of 26 November 2020 during the first wave of pandemic.6,7 There has been substantial evidence that a large proportion of the people who were infected with SARS-CoV-2 are asymptomatic, but they can infect others. 8 It has been reported based on an analysis of published reports that asymptomatic cases could account for 5% to 80%. The main symptom of the COVID-19 disease ranges from mild (asymptomatic) to severe illness characterized by fever, dry cough, dyspnea, headache, sore throat, and rhinorrhea rarely associated with hemoptysis and respiratory collapse.9,10
Epidemiologically, asymptomatic silent spreaders have been one of the major concerns as suggested by a systematic review—could be as low as 1.6% confirmed cases in China or as large as 51.7% in Diamond Princess cruise. 11 The seroprevalence of SARS-CoV-2 antibodies varies substantially between settings, 12 reflecting the variation in countries’ experience of the pandemic. Countries implementing successful public health measures to reduce transmission—including physical distancing, effective quarantining of high-risk individuals, and strict border controls—have reported a low prevalence of SARS-CoV-2 antibodies (seroprevalence) in the population. 13 There were also inter-regional variations of seroprevalence among population levels; for instance, 12.7% in North America, 8.5% in Europe, 8.2% in Africa, 11 and 1% in Southern United States. 14 Existing evidence from a growing body of research on the risk factors for early SARS-CoV-2 prevalent revealed that old age is a strong predictor to develop COVID-19; in addition, cardiovascular disease (CVD), kidney disease, cancer, and dementia are risks for hospitalization.2,15
Ethiopia reported the first case in March 2020 and subsequently identified the epicenter at the national capital Addis Ababa; meanwhile, a cluster was also detected in Asosa, the capital city of Benishangul-Gumuz Region, North West Ethiopia. 12 During the first COVID-19 pandemic as of 8 June 2020, there were over 87,000 cases and more than 2000 deaths.5,16 After an influx or entry of COVID-19 disease in Ethiopia, quarantining has been imposed citywide and in areas where potential exposure to the contagious disease has likely happened in a large population, to ascertain if they become unwell. 1 This will help to reduce the risk of them infecting others by the implementation of separation and restriction of movement,7,17 besides curb on the social gathering of the population, 5 and enforcement of quarantining for individuals having contact and travel history during the first wave of COVID-19 pandemic. 1
Different studies have also assessed the seroprevalence among close contacts and quarantined population, 18 as part of the national sample, and findings have documented a higher infection rate reported than the general population.5,19 Moreover, evidence on the risk factors for acquiring COVID-19 disease among isolated populations specifically at high-risk individuals remains elusive and poorly defined. 18 As a response to this pandemic, Ethiopia enforced strict quarantine, contact tracing, and physical distancing policies resulting in one of the lowest numbers of individuals infected with SARS-CoV-2 globally. These serosurvey findings are highly important for the enhancement of preventive measures to mitigate COVID-19 risks among identified breaks accordingly. This study investigated the seroprevalence of SARS-CoV-2-specific IgG antibodies, using the Abbott anti-nucleocapsid IgG chemiluminescent microparticle immunoassay (CMIA), in two COVID-19 patient diagnosed and treatment centers among quarantined population during the first wave of the COVID-19 (since 30 April to 30 May 2020).
Methods
Study area and study design
The study was conducted in Benishangul-Gumuz Region in the North-West part of Ethiopia; which is located at longitude 34°10′N and 37°40′E; and at latitude of 09°17′N 12°06′N. 20 The capital city of the region is Asosa, located at a distance of 659 km in the west, and Pawe is located at 565 km, North West direction of Addis Ababa the capital city of Ethiopia.21,22 According to the 2019, national population projection estimations; the regions has an estimated 1.21 million population were there. 23 Nationally, starting from the COVID-19 outbreak, the regional health bureau has establishedmianly two COVID-19 diagnosed and treatment centers for suspected SARS-CoV-2 as samples collection and diagnosing centres.
Study periods
We analyzed the COVID-19 serosurvey data of 446 quarantined individuals during the first wave of the COVID-19 pandemic from 30 April to 30 May 2020.
Sample size determination
The sample size of 450 was calculated using single population proportion formula,
Participant, quarantining, and study procedure
Population assumed to be more likely within high risk for COVID-19 acquiring with individuals having overseas traveling history, contact history, hotel hostess, health workers, street children, prisoners, and car station workers of whom were suspected for COVID-19 disease were quarantined during the first wave of COVID-19 pandemic since 30 April to 30 May 2020 in two centers (Asosa and Pawe). Totally, 446 individuals were isolated out of 450 participants and interviewed with written informed consent of each participating case after the purpose, risk, benefit, confidentiality, and degree of involvement were explained. Finally, participants were asked about the history of symptoms within the last 2 months compatible with COVID-19 (fever, chills, fatigue, myalgia, sore throat, cough, shortness of breath, chest pain, headache, nausea, vomiting, and anosmia). They were also asked about contact with suspected or confirmed cases and their practice of COVID-19 prevention measures (i.e. wearing face masks, social distancing). Individuals who had a history of diagnosed as suspected cases of COVID-19 infection were excluded during the interview whatever serologic tests were done. In this, for instance, population assumed to be more likely within high risk for COVID-19 acquiring with individuals having traveling history, hotel hostess, health workers, street children, prisoners, and station workers of whom were suspected for COVID-19 disease and quarantined within two centers were included for interview.
Sample processing and laboratory investigation of SARS-CoV-2 IgG test
After the completion of the interview, our phlebotomist collected 5 mL of blood from each participant via venipuncture and then the blood specimen was kept in an extraction tube filled with extraction buffer (300 µL) after withdrawing from each participant at room temperature (15°C–30°C) for up to 2 h before testing. 5 In addition, the refrigerator temperature requirement for the SARS-CoV-2 IgG assay was monitored on samples of each day of use based on the manufacturer’s instruction. The serum was separated and used to test for antibodies using the Elecsys Anti-SARS-CoV-2 assay, an electrochemiluminescence immunoassay using a recombinant protein representing the nucleon-capsid (N) antigen for the determination of high-affinity antibodies (including IgG) against SARS-CoV-2.7,17 This assay employs a cutoff index (COI) that is automatically calculated from two calibration standards—a COI of 1.0 or more is considered reactive/positive, and a COI less than or equal to 1.0 is reported as non-reactive/negative. The assay sensitivity and specificity were reported to be 97.2% (95.4–98.4) and 99.4% (99.1–99.8), respectively.12,24,25 A unique identification number was used to link the interview information and laboratory results. The blood specimen was kept in an extraction tube filled with extraction buffer (300 µL) at room temperature (15°C–30°C) for up to 2 h before testing. In addition, the refrigerator temperature requirement for the SARS-CoV-2 IgG assay was monitored on samples of each day of use based on the manufacturer’s instructions. Serum was collected for anti-SARS-CoV-2 nucleocapsid IgG using the Abbott ARCHITECT SARS-CoV-2 IgG CMIA qualitative assay, as per the manufacturer’s specifications. Specimen shipment, package, and label were conducted in compliance with applicable and available national and international regulations covering the transport of clinical specimens and infectious substances.
Outcome ascertainment
The outcome variable for this research was tested serum result of positive/negative, whereas the socio-demographic factors, including individual background information (sex, age, resident, marital status, levels of education, religions, economic status, comorbidity status) and COVID-19 prevention information sources (TV, radio, social media usage), were incorporated as independent variables for determining the seroprevalence of quarantined individuals.
Operational definitions
Proper hand hygiene practice
Proper hand hygiene practice was defined as a person who washes hands—the front, back, fingertips, rub thumb, and palms with adequate water and detergent at least for 20 to 30 s or using sanitizer/hand rub to the level of compliance before getting in the facility or taking the services.
Proper physical distance
Proper physical distance was defined as a person keeping 1 m away from another person during getting services, during greetings, during shopping, during the discussion, or during praying.
Proper mask wearing
Mask-wearing practice while leaving home was defined as a person covering the mouth and nose with a mask or any type of cloth or handkerchief tact.1,5
Quarantine
Quarantine is for individuals who have been in close contact (within 6 ft of someone for a cumulative total of 15 min or over 24 h) with someone who has COVID-19, unless they have been fully vaccinated. However, fully vaccinated people should get tested 5 to 7 days after their exposure, even if they do not have symptoms, and wear a mask indoors in public for 14 days following exposure or until their test result is negative. 1
Data collection procedure and quality control
Data regarding the socio-demographic source of information for exposure risk and prevention practicing for COVID-19 were collected through face-to-face interviews using a structured questionnaire adapted from the different previous research literature,5,12 and WHO Survey tools for COVID-19 prevention measures, and validated by WHO.26,27 Three BSc nurses and two laboratory technicians recruited with collected data process were strictly supervised by two medical doctors (MD), who managed the overall data collection process. A day of data collection training was given; regarding on data collection tools; techniques, and ethical issue of the study participants inside the quarantine rooms.
Statistical analyses
The data entry was performed using statistical program EpiData, version 3.1, and then exported into STATA/R version 14 (SE) for analysis. The descriptive statistics were presented by narration and tabulations. Binary logistic regression was performed to identify candidate transferee variables for multivariable logistic regression using a cutoff
Results
Between 30 April and 30 May 2020, we did a round of serosurvey among 446 quarantined individuals who were participated in two COVID-19 treatment centers. The majority, 342 (76.5%), of respondents were diagnosed from Asosa centers versus 107 (23.4%) were from Pawe. Almost similar proportions of the male participant were reported in both Asosa and Pawe centers, 211/339 (61.6%) and 73/107 (68.2%), respectively. The mean (± standard deviation (SD)) age of the respondent was 44.1 (±18.5) years, and nearly half 215 (48.10%) of them were ⩾65 age groups. Moreover, the majority, 223 (50.1%), of participants had completed primary education versus 69 (15.47%) had diploma and above certificate (Table 1).
Baseline socio-demographic characteristics of quarantined individuals in two centers, North West Ethiopia, 2020.

COVID-19: coronavirus disease 2019.
Behavioral and clinical characteristics
When asked about practicing physical distancing recommendations, more than half, 238 (53.3%), of the respondents did report practicing physical distancing recommendations accordingly. Similarly, from the overall respondents, 287 (64.9%) did not use face masks while leaving home, 335 (75.1%) avoid religious or other social gatherings involving more than four individuals outside of their households/residence, and 412 (92.38%) washing hands with soap and water during leaving home. Majority; 262 (58.5%) of the respondents had television and radio messages about COVID-19 prevention, but nearly one in five, 78 (17.5%), respondents had chronic comorbidity follow-up (Table 2).
Selected clinical and behavioral characteristics of study participants for SARS-CoV-2 seroprevalence, North West Ethiopia, 2020.

SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; COVID-19: coronavirus disease 2019.
Seroprevalence of the quarantined population
Overall, 21 of 446 (4.69%; 95% CI: 3.1–7.2) individuals who participated were seropositive for SARS-CoV-2 IgG antibody during recruitment into the study (Table 2). Nevertheless, this seroprevalence estimation did not vary by age and gender difference. The prevalence of SARS-CoV-2 among participants who lived in urban was 5.5 times higher prevalence to rural inhabitants. The corresponding estimates were 7.8% (95% CI: 5.5–8.2) and 2.1% (95% CI: 1.75–2.9) with statistically significant difference (
SARS-CoV-2 seroprevalence by selected socio-demographic and other factors for suspected and quarantined individuals in North West Ethiopia, 2020.
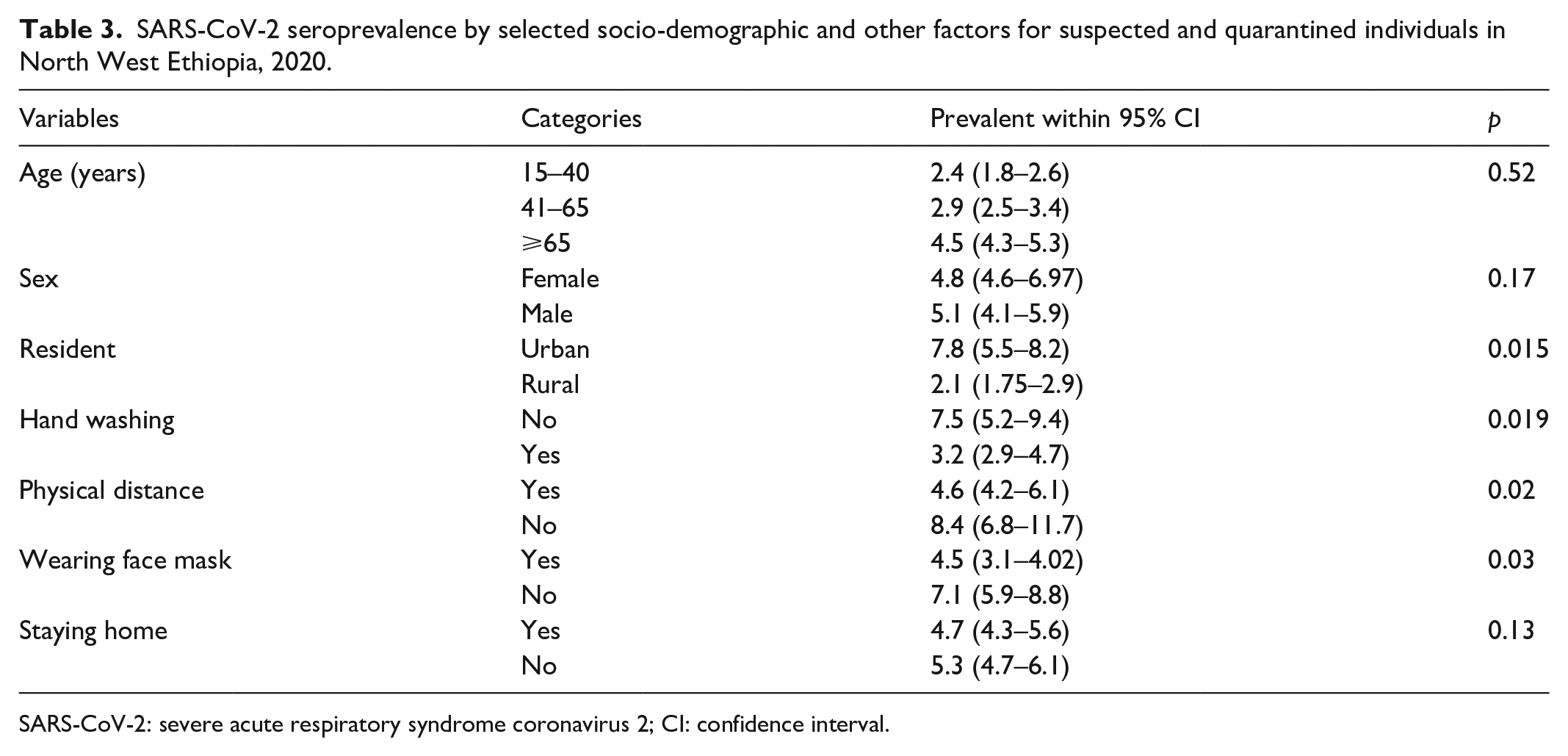
SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; CI: confidence interval.
Predictors of SAR-CoV-2 antibody seroprevalence
Table 4 shows the multiple logistic regression models using the results of serological test (positive or negative) of the quarantined population, and retaining those variables with a
Multivariable logistic regression analysis for predictors of SARS-CoV-2 risk of airing for quarantined among individuals in North West Ethiopia, 2020.

SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; AOR: adjusted odds ratio.
Discussion
The spread of SARS-CoV-2 infection in Ethiopia has poorly documented and; characterized by a lower number of confirmed cases and deaths to as compared with other regions of the Sub-Saharan Africa (SSA) countires.
1
In our serosurvey reported, the over all estimated seroprevalence of SARS-CoV-2 infections was determined as 4.7% (95% CI: 3.1–7.2) among quarantined populations. Of the largest 95.3%, proportions of isolated population were not infected at the times of survey, meaning that the prevention and control measures would be vital to interrupt transmission of COVID-19.
1
The estimated prevalence of SARS-CoV-2 serum antibodies during the first wave of the epidemic in our study is higher than previously reported in Addis Ababa (1.9%),
17
Dire Dawa (3.2%),
5
Central Ethiopia (3.5%),
12
Turkey (2.7%),
29
Israel (1.1%),
28
California (2.5%),
4
and Germany (0.12%).30,31 During this study population levels preventive measures that included school closing, restriction in social gathering, and physical distancing rules were in place in Ethiopia. In addition, the serological assays were used in the study, and the circulation of SARS-CoV-2 in the community is related with individuals’ adherence to stringent infection control measures, and high availability and utilization of personal protective equipment. Despite the substantial decrease in the prevalence of SARS-CoV-2 antibodies, in our study, previous findings in Saudi Arabia (6%),
7
Brazil (12%),
30
India (39.6% and 57.9%),25,32 and the Philippines (94.0%)
33
are inconsistent. The observed differences might be due to differences in the stages of the pandemic at the time of the surveys, with surveys conducted at the earlier stage of the pandemic more likely to report lower prevalence compared to those conducted at the later stage.
1
The difference might be that slow spread of the virus during the first COVID-19 pandemic was an artifact of limited testing capacity.
19
The possible explanation might be a constrained transmission due to environmental factors. A significant body of evidence supports a relationship between climatic and environmental conditions, namely, increased ultraviolet (UV) light, temperature, and humidity which reduced the spread of SARS-CoV-2.17,29 A previous study finding18,28,34 of a multivariable model showed age had statistical association with likelihood of SARS-CoV-2 seroprevalence with increased trend. In our finding, a positive but non-statistically significant association was found between age during a quarantined of SARS-CoV-2 antibody sero-positivity test (
Limitations of the study
There are factors which may have contributed to the low seroprevalence of SARS-CoV-2 IgG in our reported, at first initial, we did not use power approaches for sample size estimation, coupled with the performance of the test depends on time since infection, with limited sensitivity for recent infections were listed restrain. Secondly we only tested seroprevalance rate for quarantined population under estimated the magnitudes of the infection.
Conclusion
We affirmed that our study concluded lower COVID-19 seroprevalence, yet a large population in the community to be susceptible for infection, and an insignificant proportion of COVID-19 seroprevalence was observed between the age and sex of respondents. We also found that the impact of comorbidities, not using face mask, was more substantial among individual’s isolated respondents, and our study suggests essential preventive measure to cut asymptomatic transmission power in the community. In the face of the ongoing COVID-19 pandemic wave, it is important to define the epidemiology, and magnitude of this infection at population levels with wide screening for antibodies to SARS-CoV-2 can profile transmission dynamics on future practice and response to protect vulnerable groups.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221076931 – Supplemental material for Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2 ) IgG-antibody seroprevalence among quarantined population, during the first wave of COVID-19 pandemic, In North West Ethiopia (from 30 April to 30 May 2020)
Supplemental material, sj-docx-1-smo-10.1177_20503121221076931 for Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2 ) IgG-antibody seroprevalence among quarantined population, during the first wave of COVID-19 pandemic, In North West Ethiopia (from 30 April to 30 May 2020) by Fassikaw Kebede, Tsehay Kebede and Birhanu Kebede in SAGE Open Medicine
Footnotes
Acknowledgements
We would like thanks of all Pawe Woreda Health Bureau staffes, and the two hospitals administrative members for their unreserved supporting during the data collection of this research. Finally we also thanks and appreciated of all data collectors and the two supervisors for their commitment and timely finishing of the data collection process.
Author contributions
F.K. and T.K. conceived the study. F.K., T.K., and B.K. supervised the data collection. T.K. and B.K. did the analysis and wrote the manuscript. The authors commented, edited the draft, and approved the final version of this manuscript.
Availability of data and materials
All data that support the findings of this study is available from the corresponding author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
We obtained ethical clearance from our institutions committee by looking at the research aims and methodologies of researchers to make sure that the research will be conducted in a way that protects the dignity, rights, and safety of the admitted coronavirus disease 2019 (COVID-19) participants, and that the research design is ethically sound and is likely to render the anticipated result. The ethical and research board of Pawe Woreda Health Bureau approved this proposal for data collection within the protocol Refill No. PWOH/84/13/21 for written informed consent was permission from each patient is enough declaration. Data collection permission was also sent for two centers with a meeting number 012/2013.
Informed consent
Patients’ written informed consent of every participating case was obtained after the purpose, risk, benefit, confidentiality, and degree of involvement were fully explained to caregivers by their local language before starting the data collection. For participants less than 18 years of age, written and signed assent was obtained from legally authorized representatives. Data security and participants’ confidentiality were maintained at all levels of data management.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
