Abstract
This study reported a case of disseminated intravascular coagulation secondary to postpartum hemorrhage complicated by autoimmune hemolytic anemia. A pregnant woman in her early 40s was admitted to The Third Central Hospital of Tianjin with regular contractions in July 2022. The direct antiglobulin test was positive for IgG and C3d. Following the delivery of a baby girl, she developed severe postpartum hemorrhage, and the obstetric disseminated intravascular coagulation score exceeded 8 points. She was diagnosed with disseminated intravascular coagulation secondary to postpartum hemorrhage and warm-antibody type autoimmune hemolytic anemia (IgG + C3). The patient underwent methylprednisone therapy, emergency selective bilateral uterine artery embolization, and transfusion with suspended and washed red blood cells. Subsequently, routine coagulation parameters normalized, and the urine color gradually cleared. Therefore, glucocorticoids, uterine artery embolization, and blood transfusion may be effective for managing disseminated intravascular coagulation secondary to postpartum hemorrhage complicated by autoimmune hemolytic anemia.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) is a complex, potentially life-threatening disorder characterized by widespread activation of coagulation pathways, resulting in microvascular thrombosis, consumption of clotting factors, and subsequent bleeding complications. 1 DIC can occur secondary to various underlying conditions, such as trauma, sepsis, malignancies, and obstetric complications. 2 Autoimmune hemolytic anemia (AIHA) is an acquired condition in which red blood cells (RBCs) are prematurely destroyed by antibodies targeting RBC antigens, with inadequate compensatory production. 3 AIHA is rare in pregnancy, occurring in approximately 1 in 140,000 cases. 4 The co-occurrence of DIC and AIHA has been rarely reported. This study reported a case of DIC secondary to postpartum hemorrhage (PPH) complicated by AIHA. The reporting of this study conforms to the Case Report (CARE) guidelines. 5
Case presentation
History of present illness
A pregnant woman in her early 40s was admitted to The Third Central Hospital of Tianjin with regular contractions at 36 + 6 weeks of gestation in July 2022.
History of past illness
The patient had a history of five pregnancies. She gave birth to a baby boy 16 years ago via cesarean section and had three voluntary noniatrogenic abortions.
Physical examination
Routine indwelling catheterization following admission revealed gross hematuria. She delivered a baby girl vaginally 2 h after admission. Severe PPH occurred, with an estimated blood loss of approximately 1500 mL in 1.5 h, and the patient exhibited signs of shock.
Laboratory examinations
Laboratory examination immediately after admission showed hemoglobin (HB) level of 85 g/L, white blood cell count (WBC) of 14.39 × 109/L, reticulocyte percentage of 6.02%, total bilirubin (TBIL) level of 26.9 µmol/L, indirect bilirubin (IBIL) level of 21.9 µmol/L, platelet count (PLT) of 207 × 109/L, lactate dehydrogenase (LDH) level of 403 U/L, prothrombin time (PT) of 13.5 s, activated partial thromboplastin time (APTT) of 35.0 s, and fibrinogen (FIB) of 3.56 g/L. Postpartum laboratory examination showed PT of 30.4 s, APTT of 46.4 s, and FIB of 0.6 g/L. Laboratory findings on admission suggested hemolysis according to the diagnostic criteria for AIHA in the Guidelines for the Diagnosis and Treatment of Autoimmune Hemolytic Anemia in Chinese Adults (2023 Edition). 6 Direct antiglobulin test (DAT; Coombs test) was positive for IgG and C3d. The erythrocyte monospecific antibody test was positive, and the cold agglutinin test was negative.
Final diagnosis
According to the 2024 Japanese Clinical Practice Guidelines: Management of Diffuse Intravascular Coagulation (http://www.jsognh.jp/dic/, in Japanese), a diagnosis of DIC was established. Decreased levels of HB, bilirubin and LDH; gross hematuria; and later positive RBC autoantibodies all indicated that the patient had hemolysis at the time of admission, resulting in hemolytic anemia. Considering the presence of hemolysis combined with DAT suggestive of AIHA, but also noting that coagulation and fibrinolytic indices were initially normal and that the later abnormal indices were secondary to the PPH (PT, 30.4 s; APTT, 46.4 s; FIB, 0.6 g/L; and D-dimer >10 mg/L), DIC was considered secondary to PPH and AIHA. The patient was finally diagnosed with DIC secondary to PPH and warm-antibody type AIHA (IgG + C3).
Differential diagnosis
The case reported here differed from cholestasis, as the total bile acid level at admission was 5 µmol/L (within the normal range), and the patient reported no pruritus. It also differed from HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome, which typically presents with hemolysis, elevated liver enzymes, and low platelets, often accompanied with elevated blood pressure. The patient’s blood pressure at admission was 119/60 mmHg, with no increase observed during pregnancy or hospitalization. Proteinuria was negative throughout pregnancy. Alanine aminotransferase (ALT) was 10 U/L, aspartate aminotransferase (AST) was 28 U/L, and PLT was 207 × 109/L. PLT, ALT, and AST remained within normal ranges throughout pregnancy, whereas LDH and bilirubin were elevated at admission, indicating hemolysis. Furthermore, the case differed from acute fatty liver. According to the Swansea diagnostic criteria for acute fatty liver, the patient met only three criteria: elevated bilirubin, elevated WBC, and mild prolongation of APTT. Blood glucose, ALT, AST, creatinine, uric acid, and liver ultrasound findings were all normal.
Treatment
Written informed consent for all treatments related to delivery and its complications was obtained from the patient at admission. During the PPH rescue process, 40 mg methylprednisolone once daily was urgently administered for 3 days to reduce hemolysis. Diuretic therapy and kidney-protective treatment were administered based on volume expansion. For elevated bilirubin, 0.25 g ursodeoxycholic acid was given three times daily. Elective bilateral uterine artery interventional embolization was urgently performed, effectively controlling the bleeding. The patient was administered the following sequentially: 600 mL of fresh frozen plasma at 4 h after admission, 2 g of FIB at 6 h, 600 U of prothrombin complex at 8 h, 600 mL of fresh frozen plasma at 11 h, 20 U of cryoprecipitate at 14 h, 2 U of suspended RBCs at 16 h (HB had dropped to 44 g/L), 2 U of suspended RBCs at 22 h, and 4 U of washed RBCs at 39 h. At 34 h postpartum, the patient was administered 40 mg/d enoxaparin for anticoagulation therapy, which was continued until recovery 5 days after delivery. After discharge, enoxaparin was continued until 7 days postpartum. HB levels stabilized, and IBIL, PT, APTT, FIB, and LDH returned to normal (Figure 1). Hemolytic remission was achieved, and the urine color gradually cleared.
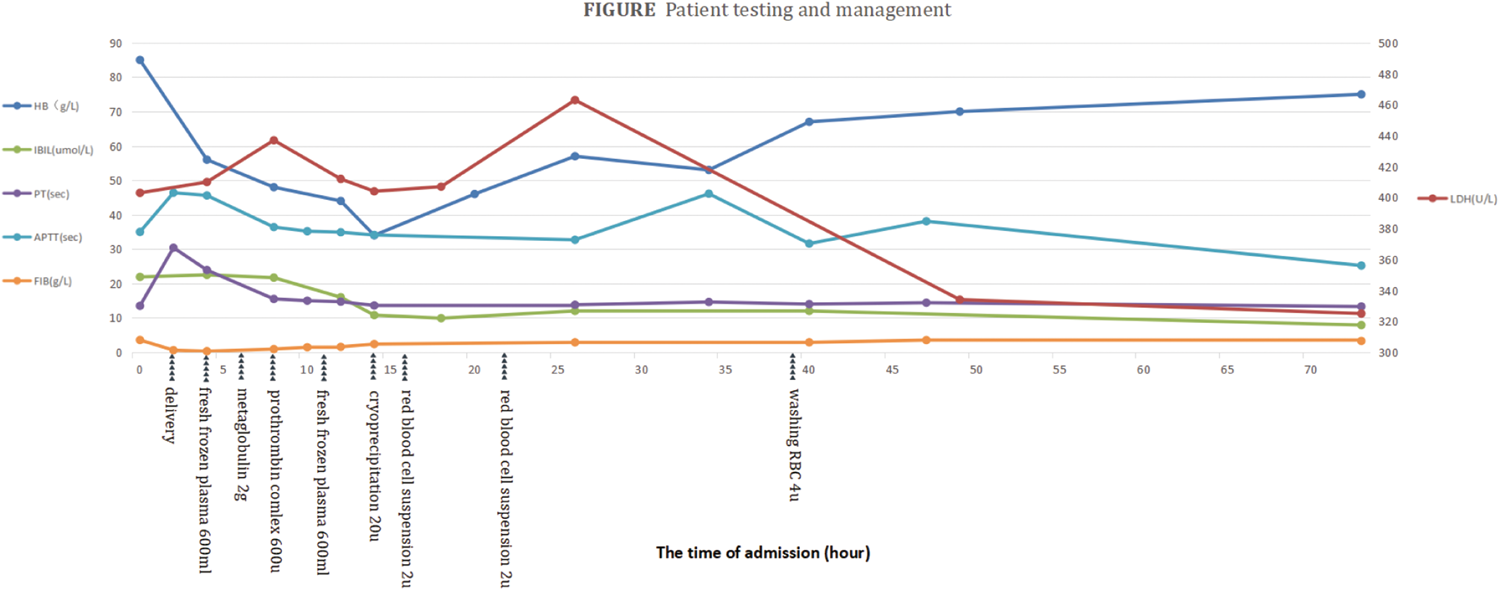
Changes in hemoglobin (HB), indirect bilirubin (IBIL), prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen (FIB), and lactate dehydrogenase (LDH) over time.
Outcome and follow-up
The patient was followed up for 2 months, during which no hemolysis was observed.
Discussion
This study reported a rare case of DIC secondary to PPH combined with AIHA. Treatment with glucocorticoids, uterine artery embolization (UAE), and blood transfusion may be effective in managing DIC secondary to PPH combined with AIHA.
Several studies have reported the co-occurrence of DIC and AIHA. For example, Klapper et al. reported a fatal case of AIHA presenting with massive hemolysis and DIC; 7 Damani et al. reported a fatal case of COVID-19 in a patient who developed AIHA and DIC; 8 and Orvain et al. described a patient with chronic lymphocytic leukemia complicated by AIHA who developed acute hepatitis and DIC. 9 However, to the best of our knowledge, this is the first reported case of concomitant PPH, DIC, and AIHA. During pregnancy and delivery, the immune system can become unbalanced. 10 It is reported that only 0.1% of pregnant women produce anti-RBC antibodies. 11
The literature contains no reports of PPH with autoimmune anemia complicated by DIC. Therefore, no standard treatment exists for such patients. The first-line therapy for AIHA is glucocorticoids. 12 According to the Chinese Guidelines for the Treatment of Adult Autoimmune Hemolytic Anemia (2023) Edition, 6 symptomatic supportive treatment of AIHA involves RBC component transfusion, diuresis, and choleretic therapy. The treatment of PPH complicated by DIC focuses on coagulation dysfunction, with transfusion of blood components, including RBCs, platelets, fresh frozen plasma, and cryoprecipitate, to address anemia and coagulation factor depletion. UAE or transarterial embolization (TAE) is considered the first-line treatment option for PPH combined with DIC. The antifibrinolytic drug tranexamic acid can be used to reduce hyperfibrinolysis. In this patient, hemolysis resolved after treatment with glucocorticoids. Blood transfusion is an effective treatment for AIHA; however, plasma antibodies can lead to agglutination of both the patient’s and donor’s erythrocytes, affecting blood typing, crossmatching, and overall transfusion safety. According to the Chinese Guidelines for the Treatment of Autoimmune Hemolytic Anemia in Adults (2023) Edition, 6 symptomatic supportive treatment of AIHA may include RBC component transfusion, although the guidelines do not emphasize on the use of washed RBCs during rescue. Nevertheless, patients with AIHA often display autoantibodies, resulting in a positive DAT. In such cases, washed RBCs are used for patients with severe anemia, as they reduce antibodies and complement components in plasma, thereby lowering the risk of hemolytic transfusion reactions.
When DIC secondary to PPH coexists with AIHA, the pathophysiological interactions and management are highly complex. Hemolytic activity in AIHA can synergistically worsen DIC by increasing the burden of free HB and inflammatory mediators, further activating coagulation cascades. In the postpartum period, hormonal therapy, particularly corticosteroids used for AIHA, may have nuanced effects on maternal recovery, influencing infection risk, glucose regulation, wound healing, and potentially thromboembolic risk. DIC and AIHA independently trigger systemic inflammation and endothelial activation, which amplifies the risk of microvascular thrombosis and bleeding. 13 Hemolysis due to AIHA exacerbates DIC by releasing free HB and cellular debris, which activate the coagulation cascade, worsen endothelial dysfunction, and contribute to further consumption of clotting factors.7,14,15 The literature documents cases in which the coexistence of AIHA and DIC resulted in a synergistically negative prognosis, often associated with poor response to standard therapy or greater organ damage.7,14 High-dose corticosteroids, the first-line treatment for AIHA, is a mainstay during pregnancy and postpartum due to their established efficacy and relative safety. Most patients demonstrate improvement, although the time to response may be delayed (typically 7–26 days for AIHA responders). 16 Although steroid therapy is necessary for AIHA, it increases infection risk and may impair wound healing or glycemic control, complicating postpartum recovery. 16 For DIC, the underlying trigger, PPH, should be urgently addressed with volume resuscitation, replacement of depleted clotting factors (e.g. fresh frozen plasma and platelets), and control of the primary hemorrhage. 13 Immunosuppressive therapy for AIHA (steroids or second-line agents) may exacerbate sepsis or infections if present, further worsening DIC. In refractory AIHA, IVIG or rituximab is often used; however, these therapies are considered on a case-by-case basis due to safety concerns during postpartum and potential effects on lactation. 16 Hemolysis is a well documented exacerbator of DIC due to the release of procoagulant materials and direct endothelial damage. Clinical reports have shown that outcomes are often worse when both conditions coexist.7,14,15 Complement-mediated hemolysis observed in some AIHA cases is particularly prothrombotic, further increasing the risk of DIC.7,15 High-dose corticosteroids (e.g. prednisone and methylprednisolone) are the mainstay treatment for AIHA even during pregnancy and postpartum. Most patients demonstrate good hematologic responses; however, infection, hyperglycemia, and impaired tissue healing may be more pronounced during the postpartum period. 16 There is no strong evidence that hormonal therapy for AIHA (primarily steroids) independently worsens routine postpartum recovery. The primary complications instead relate to immunosuppression and metabolic side effects. When AIHA, DIC, and PPH coexist, early multidisciplinary management is essential, including hemostatic support, immunosuppressive therapy, infection control, and careful monitoring for complications such as thromboembolism or sepsis.13,16 Blood product support is often necessary but may be complicated by crossmatching issues in AIHA and increased consumption or bleeding risk in DIC. 16 These complexities underscore the need for tailored and closely monitored postpartum care, a balance between rapid immunosuppression for AIHA, and an aggressive supportive therapy for DIC, with attention to potential mutually exacerbating effects.7,13–16
Regarding potential mechanisms, they remain hypothetical due to the rarity of this case. Nevertheless, emerging evidence suggests that acute hemolysis, as seen in AIHA, may actively contribute to coagulation derangements through the release of phosphatidylserine-expressing erythrocyte microparticles, free heme, and ADP, all of which are potent inducers of thrombin generation and platelet activation. These hemolysis-related factors create a procoagulant and proinflammatory state, amplifying DIC-driven consumption and FIB depletion initiated by PPH. This pathophysiological interplay between complement activation and microparticle release following hemolysis may explain the more precipitous drop in FIB observed in our case, bridging the gap between PPH as a trigger and the unexpectedly severe coagulation abnormalities. Additional literature on AIHA and DIC supports this amplification of consumptive coagulopathy by hemolytic processes.15,17,18
In this case, emergency selective bilateral UAE effectively controlled PPH, and administration of methylprednisolone suppressed the immune response underlying AIHA. Transfusion of suspended and washed RBCs helped stabilize the patient’s HB levels and contributed to the observed hemolytic remission.
Conclusion
This study reported a case of DIC secondary to PPH combined with AIHA, which may offer valuable insights for clinicians managing similarly complex clinical scenarios.
Footnotes
Acknowledgments
None.
Authors’ contributions
Chunlei Ma and Qian Liang carried out the studies, participated in collecting data, and drafted the manuscript. Chunlei Ma, Qian Liang, Shurong Song, Zhenlin Wu and Ke Li participated in acquisition, analysis, interpretation of data, and designing and drafting of the manuscript. All authors read and approved the final manuscript.
Consent for publication
Not applicable.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This work has been carried out in accordance with the Declaration of Helsinki (2000) of the World Medical Association. This study was approved by the Tianjin Third Central Hospital Medical Ethics Committee, and all participants provided written informed consent.
Funding
None.
