Abstract
Background
The World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0) is a widely used instrument for assessing functioning and disability across various health conditions. However, there is limited research on its validation in the context of potentially life-threatening maternal conditions.
Objective
To validate the Tigrigna version of WHODAS 2.0 in women with potentially life-threatening maternal conditions during the immediate postpartum period.
Methods
This cross-sectional study was conducted in hospitals in Tigray, Ethiopia. The Tigrigna version of the WHODAS 2.0 was administered to 121 women with potentially life-threatening maternal conditions. Internal consistency was evaluated using Cronbach’s alpha, convergent validity was preliminarily assessed using Spearman’s correlation, and construct validity was assessed using confirmatory factor analysis.
Result
The 36-item Tigrigna version showed adequate internal consistency for all domain scores, with Cronbach’s alpha values ranging from 0.89 to 0.96 and a summary score Cronbach’s alpha of 0.98. Except for Domains 1 and 2, convergent validity between the 36-item and 12-item versions was demonstrated by strong correlations between similar constructs, with correlation coefficients ranging from 0.86 to 0.96. Confirmatory factor analysis indicated a poor or suboptimal fit for the six-domain model, with a root mean square error of approximation of 0.13, a comparative fit index of 0.84, and a Tucker–Lewis index of 0.82.
Conclusion
This preliminary validation suggests that the Tigrigna version of WHODAS 2.0 demonstrates acceptable reliability and validity for assessing disability in women with a history of potentially life-threatening maternal conditions.
Keywords
Background
Potentially life-threatening maternal conditions (PLTC) are the most common cause of maternal mortality worldwide 1 and have been a leading cause of death in Ethiopia. 2 In Ethiopia, PLTC, which include hypertensive disorders, obstetric hemorrhage, other systemic disorders, and severe management indicators, 3 have the highest reported prevalence globally, at 17.55%. 4 These conditions often result in physical, psychological, and social impairments, making them the third most common contributor to disability-adjusted life years among women aged 15–44 years. 5 Addressing maternal morbidity and related disabilities is a key strategic objective of the Sustainable Development Goals (SDGs) for ending preventable maternal mortality. 6 The importance of information on functioning and disability is gradually increasing in the field of public health. 7
Based on the World Health Organization (WHO) disability report, the global prevalence of disability is 15% 8 ; however, the reported prevalence in Ethiopia was 0.74% in 2022. 9 This discrepancy may stem from Ethiopia’s reliance on physical impairment registries, which often underestimate functional limitations such as mobility, self-care, and social participation. 10 The WHO Disability Assessment Schedule 2 (WHODAS 2.0), which is based on the International Classification of Functioning, Disability and Health (ICF) framework, is well suited for assessing postpartum functional disability in women with PLTC because of its comprehensive domain coverage. Its 36-item version evaluates six domains—cognition, mobility (e.g. walking after cesarean delivery), self-care, getting along, life activities (e.g. caregiving tasks), and participation (e.g. community engagement)—thereby capturing caregiving challenges and recovery-related limitations.11–13
Despite its robust psychometric properties demonstrated in initial studies across 19 countries, the WHODAS 2.0 requires further validation to ensure semantic, conceptual, and measurement equivalence across diverse cultural contexts. 11 Cultural differences in the interpretation of participation and life-activity domains, together with limited invariance testing, underscore the need for context-specific adaptations, particularly in low-resource settings such as Tigray, Ethiopia. Validating the WHODAS 2.0 for PLTC is critical for understanding disability among women facing serious maternal health risks during pregnancy, childbirth, and the postpartum period.
Validation of the WHODAS 2.0 tool for PLTC is an essential step toward improving the understanding of disability among women exposed to serious health risks during pregnancy, childbirth, and the postpartum period. The findings of this study may inform clinical practice, policy development, and interventions aimed at promoting the well-being and outcomes of women experiencing serious maternal health challenges. Therefore, this study aimed to evaluate the reliability and validity of the Tigrigna version of WHODAS 2.0 in assessing disability among women with PLTC during the immediate postpartum period.
Methods
This was a cross-sectional methodological study aimed at evaluating the psychometric properties, including reliability and validity, of the Tigrigna version of the WHODAS 2.0 among women with a history of PLTC. A consecutive sampling approach was used, and statistical analyses included assessment of internal consistency, correlation analyses, and confirmatory factor analysis (CFA). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
Study participants, sampling, and data collection procedure
Study participants were consecutively recruited from two comprehensive specialized referral hospitals (Aksum and Ayder) and eight general hospitals (Lemlem Karel, Mekelle, Wukro, Abi-adi, Adigrat, Adwa, St Marry, and Suhul-shire) in Tigray, northern Ethiopia, from 22 December 2023 to 13 January 2024. The inclusion criteria were women aged 18 years or older with a history of PLTC, based on WHO criteria, 3 within the immediate postpartum period. The exclusion criteria included women who were unable to provide informed consent. During the recruitment period, all eligible women were identified across the participating hospitals until. Of these, 121 agreed to participate, yielding a participation rate of 97.58%, while 3 women were excluded because of refusal.
The short 12-item form was not administered independently because its items are a direct subset of the 36-item version, allowing the extraction of 12-item scores from the full administration. This approach minimized participant response burden, avoided redundancy, and reduced potential fatigue in a vulnerable postpartum population. Data were collected using the Kobo mobile application at the time of discharge.
The study protocol was reviewed and approved by the Institutional Review Board of the College of Health Sciences, Mekelle University (approval no. MU-IRB 2129/2023). Informed verbal consent was obtained from all participants. All patient data were deidentified to protect privacy. The study was conducted in accordance with the Declaration of Helsinki of 1975, as revised in 2024.
Measurements
The WHODAS 2.0 is a generic, multidimensional disability assessment questionnaire. The full version (36 items) comprises six domains: Domain 1 (D1), “Cognition” (six items); Domain 2 (D2), “Mobility” (five items); Domain 3 (D3), “Self-care” (four items); Domain 4 (D4), “Getting along” (five items); Domain 5 (D5), “Life activities” (eight items: four related to household activities and four related to work/school activities); and Domain 6 (D6), “Participation” (eight items). The short-form version (12 items) provides a brief assessment of overall functioning and disability in clinical and research settings when time is limited. The 12 items of WHODAS 2.0 are labeled S1 to S12. The 12-item WHODAS 2.0 is derived from the 36-item version, with two questions selected from each domain: D1, “Cognition” (D1.1 = S6, D1.4 = S3); D2, “Mobility” (D2.1 = S1, D2.5 = S7); D3, “Self-care” (D3.1 = S8, D3.2 = S9); D4, “Getting along” (D4.1 = S10, D4.2 = S11); D5, “Life activities” (D5.1 = S2, D5.5 = S12); and D6, “Participation” (D6.1 =S4, D6.5 = S5). Responses for each item are rated on a five-point scale reflecting the difficulty experienced in the previous 30 days: none = 0, mild = 1, moderate = 2, severe = 3, and extreme/cannot do = 4. For participants who are not employed or enrolled in school, the 36- and 12-item versions are reduced to 32 and 11 items, respectively. The overall WHODAS 2.0 score ranges from 0 to 100, with higher scores indicating greater disability in daily functioning. 11
Sociodemographic information was collected using structured interviewer-administered questions. The number of morbidities was obtained from medical records and verified for PLTC-related conditions, including hypertensive disorders, obstetric hemorrhage, and other systemic disorders. Morbidities were categorized as one, two, or three or more based on the count of distinct conditions experienced. All responses were recorded electronically using the Kobo Toolbox mobile application.
Translation and adaptation
The WHODAS 2.0 items were translated into Tigrigna following permission obtained from the WHO and the WHO guidelines for tool adaptation and translation (WHO 2015). The forward translation was performed by the first author, a native Tigrigna speaker with expertise in maternal and reproductive health and fluency in English. The back translation was conducted by an independent bilingual (English–Tigrigna) English language teacher, who was also a native Tigrigna speaker, to ensure linguistic accuracy and cultural relevance. The translated draft was reviewed by a panel of experts in public health, gynecology, nursing, and midwifery. Linguistic adaptations addressed cultural and contextual nuances; for example, “Getting out of your home” was translated as “Move out of the yard/corridor” to reflect local housing structures in Tigray, where homes often include enclosed yards. Similarly, “Maintaining friendships” was adjusted to “Continuing friendships” to capture ongoing social relationships in the Tigrigna context.
A pilot study was conducted by administering the third draft of the Tigrigna WHODAS 2.0 to 15 women with PLTC. Face-to-face interviews were conducted by trained data collectors, followed by debriefing sessions to assess comprehension, clarity, and cultural appropriateness. Minor wording adjustments were made based on participant feedback, including simplification of response options for women with low literacy.
Data quality control
To ensure data quality, the following procedures were implemented:
Supervisors and data collectors received 2 days of training. The data collection process was closely monitored. The Kobo Toolbox mobile application was used for data collection and real-time monitoring. Its form logic and automated validation checks minimized skipped questions, response errors, and missing data, improving accuracy and completeness. The WHODAS 2.0 was administered by interviewers in a face-to-face format to accommodate varying literacy levels, as self-administration could introduce response bias.
Data analysis
Data were analyzed using Statistical Package for the Social Sciences (SPSS) version 24 and Stata version 17. We used descriptive statistics to summarize the sociodemographic characteristics of the participants. The Shapiro–Wilk test (p < 0.01) indicated non-normal distribution, leading to the use of Spearman’s correlation to assess convergent validity between the 36-item and 12-item WHODAS 2.0 scores.
Reliability
Internal consistency was assessed using Cronbach’s alpha coefficient, interpreted as follows: >0.9 = excellent, >0.8 =good, >0.7 = acceptable, >0.6 =questionable, >0.5 = poor, and <0.5 = unacceptable. 15 Item-deleted Cronbach’s alpha coefficients were calculated to determine whether individual items were aligned with the same construct. Internal consistency was further examined using item–total correlations (the correlation between each item and the overall scale score). For scales with only a couple of items, Spearman–Brown coefficients were also calculated to provide a more accurate estimate of internal consistency. 16
Face and content validity
Face and content validity were assessed using input from experts in maternal health, including one gynecologist, three midwives, four public health professionals, and one nurse. Efforts were made to ensure that experts clearly understood the purpose and scope of the tool. Experts participated in a panel discussion to provide feedback for tool improvement, conducted face-to-face to facilitate clear communication. In the first step, experts reviewed the items for face validity and confirmed that the instrument was appropriate for measuring functioning and disability in the study area (Tigray region).
Content validity was evaluated to determine whether the WHODAS 2.0 tool adequately captures disability in the local Tigray context. The content validity index (CVI) was used, including both item-level CVI (I-CVI) and scale-level CVI (S-CVI). Nine experts in public health, midwifery, nursing, and obstetrics rated each item on a four-point scale: 1 = not relevant, 2 = somewhat relevant, 3 = quite relevant, and 4 = highly relevant. Scores of 1 or 2 indicated disagreement (irrelevant), while scores of 3 or 4 indicated agreement (relevant). According to the literature, with at least nine experts, a CVI of ≥0.78 is considered acceptable. 17
Construct validity
Convergent validity was assessed using Spearman’s correlations due to non-normal data distribution. Preliminarily evaluation involved correlations between the 36-item and 12-item WHODAS 2.0 versions, which primarily examines equivalence rather than convergence with external variables. Correlation strength was interpreted as follows: 0–0.19 = very weak, 0.2–0.39 = weak, 0.4–0.59 = moderate, 0.6–0.79 = strong, and 0.8–1.0 = very strong. 18 It was hypothesized that higher disability scores on the 36-item version would correspond to higher scores on the 12-item version. 11
CFA
Construct validity was further evaluated using CFA to test the hypothesized six-domain structure of the 36-item Tigrigna WHODAS 2.0 (cognition, mobility, self-care, getting along, life activities, and participation. 11 Given the ordinal nature of the Likert-scale responses, CFA was performed using robust weighted least squares estimation. Model fit was assessed using the following indices: χ2/df (<3 = acceptable fit), comparative fit index (CFI > 0.90), Tucker–Lewis index (TLI > 0.90), root mean square error of approximation (RMSEA < 0.08), and standardized root mean residual (SRMR < 0.08). 19 Standardized factor loadings were examined to confirm item–domain relationships, with loadings ≥0.4 considered acceptable. The sample size (n = 121) was adequate for basic psychometric analyses, as indicated by a high Kaiser–Meyer–Olkin value (KMO = 0.93), but was limited for CFA, which may reduce statistical power and compromise model stability.
We calculated the composite reliability (CR) and average variance extracted (AVE) to further evaluate internal consistency and convergent validity, respectively. CR values ≥ 0.7 indicate acceptable internal reliability, AVE values > 0.5 indicate convergent validity, and CR > AVE indicates construct robustness. 20
Results
Characteristics of study participants
A total of 121 women who experienced PLTC during the immediate postpartum period participated in the study. The mean age was 29.36 years (SD: ±5.98). Most participants were married (n = 116, 95.9%), housewives (n = 78, 64.46%), and residents of urban areas (n = 91, 75.20%) (Table 1).
Sociodemographic characteristics and number of maternal complications among study participants.

Face and content validity
Expert input was used to assess face and content validity. Experts reviewed the items for face validity and confirmed that the instrument was appropriate for measuring functioning and disability in the Tigray context. During translation, two items were adapted for cultural relevance: “Getting out of your home” was translated to “Move out of the yard/corridor,” and “Maintaining friendships” was adapted to “Continuing friendships.”
Content validity was assessed to determine whether the WHODAS 2.0 adequately addressed disability in the Tigray context. The CVI, including I-CVI and S-CVI, was calculated based on ratings from nine experts. The results indicated I-CVI values ranging from 0.78 to 1.0 and an S-CVI of 0.96, demonstrating acceptable content validity.
WHODAS 2.0 domain scores
The overall mean (SD) scores for the 36-item and 32-item WHODAS 2.0 were 20.55 (20.99) and 18.63 (21.48), respectively. The minimum and maximum mean domain scores were observed in the “Getting along with others” domain (14.91) and “Life activities” domain (23.56), respectively. D5 (work or school activities, items 5.5–8) was assessed in only 37 participants. For the 12-item WHODAS 2.0, mean scores ranged from 13.84 to 27.70. The median (interquartile range (IQR)) for the 36-item and 32-item versions were 14.48 (38.40) and 9.17 (33.87), respectively (Table 2).
Descriptive analysis of 36-item and 12-item WHODAS 2.0 scores among study participants.
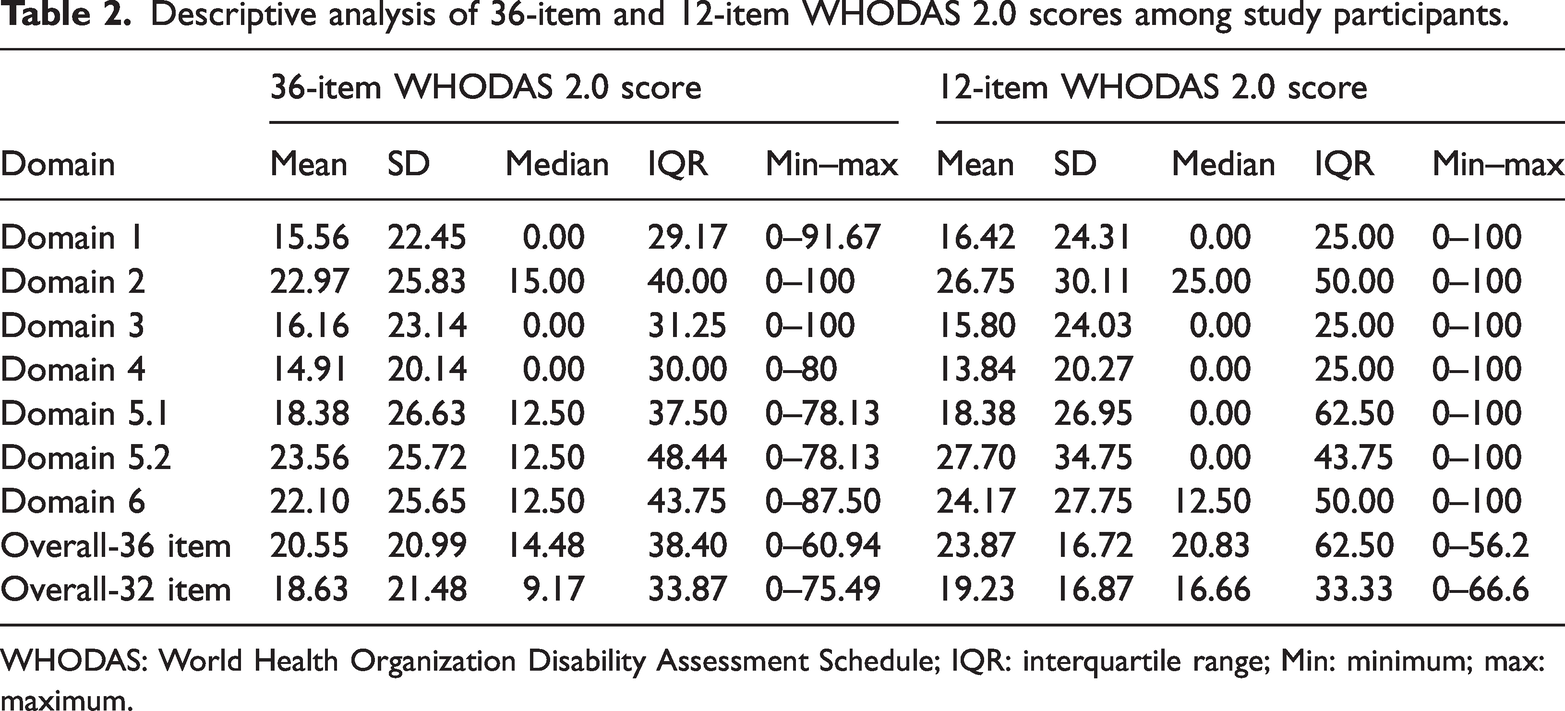
WHODAS: World Health Organization Disability Assessment Schedule; IQR: interquartile range; Min: minimum; max: maximum.
Reliability
The Tigrigna version of WHODAS 2.0 demonstrated excellent internal consistency, as indicated by Cronbach’s alpha values across domains: cognition (D1 = 0.97), mobility (D2 = 0.93), self-care (D3 = 0.95), getting along (D4 = 0.89), housework (D5.1–4 = 0.97), work/school activities (D5.5–8 = 0.96), and participation (D6 =0.96). The overall internal consistency of the 36-item and 32-item WHODAS 2.0 (after exclusion of work/school activities) was 0.98 and 0.97, respectively. Correlations between each item and the overall score ranged from 0.73 to 0.95 and were statistically significant (p < 0.001) (Table 3).
Descriptive statistics and reliability of the Tigrigna version of WHODAS 2.0.

WHODAS: World Health Organization Disability Assessment Schedule; D: domain.
Domain 5.5–8 (work/school activities) was tested in 37 participants.
For the 12-item WHODAS 2.0 and the 11-item version (after omitting work/school activities), Cronbach’s alpha coefficients were 0.94 and 0.93, respectively. We used the Spearman–Brown coefficient to assess internal consistency for domains with a couple of items within the domain. The Spearman–Brown score coefficient yielded coefficients of 0.92 for cognition, 0.86 for mobility, 0.95 for self-care, 0.87 for life activities, 0.71 for participation, and 0.85 for getting along.
Validity
Convergent validity was preliminarily assessed using correlations between the 36-item and 12-item WHODAS 2.0 versions; this approach primarily evaluates equivalence rather than convergence with external variables. All 36-item domains were positively correlated with the corresponding 12-item domains, indicating that higher scores on the full version corresponded to higher scores on the short form. Correlations between the 12-item composite score and subscale scores supported the convergent validity of the 36-item version (r = 0.61–0.78, p < 0.01). Discriminant validity was not fully satisfied for several items, as these items demonstrated similar or higher correlations with other domains than with their intended domain, indicating a violation of discriminant validity criteria (Table 4).
Correlations between WHODAS 2.0 full-form (36-item) domains and short-form (12-item) items.

Correlation is significant at the 0.01 level (two-tailed).
Correlation is significant at the 0.05 level (two-tailed).
WHODAS: World Health Organization Disability Assessment Schedule.
A correlation matrix of the six WHODAS 2.0 domains and the overall score was developed to assess the validity of domain selection. Strong, statistically significant correlations were observed in all cases (p < 0.001). Notably, strong correlations were found between the cognition and self-care domains (r = 0.92), between self-care and getting along (r = 0.92) and life activities (r = 0.90), and between household and work/school life activities (r = 0.92). Correlations between each domain and the overall assessment of functioning, disability, and health ranged from 0.87 to 0.90 (Table 5).
Correlation between WHODAS 2.0 domains and the overall summary score.

WHODAS: World Health Organization Disability Assessment Schedule.
We evaluated the theoretical validity by analyzing the internal structure, including correlations between individual items and their respective domains, as well as between items and the overall score. Strong correlations were observed between item scores and the Domain score (r = 0.79–0.97) and between individual items and the overall score (r = 0.75–0.88), all statistically significant at p < 0.001 (Table 6).
Correlation between WHODAS 2.0 items, their respective domain scores, and the overall (total) score.

WHODAS: World Health Organization Disability Assessment Schedule.
CFA
After excluding work/school activity items in D5, CFA was conducted on the 32-item WHODAS 2.0 to examine the dimensionality of the Tigrigna version. Data suitability was supported by a KMO measure of 0.93 and a significant Bartlett’s test of sphericity (p < 0.01). Model fit indices were as follows: CFI = 0.84, TLI = 0.82, RMSEA = 0.13, and SRMR = 0.06, indicating a poor or suboptimal model fit, likely due to the limited sample size (N = 121). Standardized factor loadings ranged from 0.64 to 0.97, demonstrating acceptable to strong item–domain relationships. The six-domain CFA model is presented in the path diagram (Figure 1).

Path diagram of the six-domain confirmatory factor analysis model for the 36-item WHODAS 2.0 among women with potentially life-threatening maternal conditions. WHODAS: World Health Organization Disability Assessment Schedule.
To address limitations of the 32-item CFA due to the sample size (N = 121), a secondary CFA was conducted on the 12-item WHODAS 2.0, which provides a more appropriate participant-to-item ratio (approximately 10 participants per item). The 12-item CFA did not improve model fit, with indices remaining suboptimal: RMSEA = 0.17 (threshold < 0.08), CFI =0.86, TLI = 0.83 (threshold > 0.90), and SRMR = 0.07 (acceptable). Standardized factor loadings remained strong (0.55–0.92), indicating that the 12-item version retains robust item representation.
Construct validity was further evaluated using AVE, calculated as the sum of squared factor loadings divided by the number of items within each domain. AVE values for the Tigrigna WHODAS 2.0 ranged from 0.5 to 1.25, proving evidence of convergent validity (Table 7).
AVE and CR of WHODAS 2.0 domains among women with a history of PLTC.
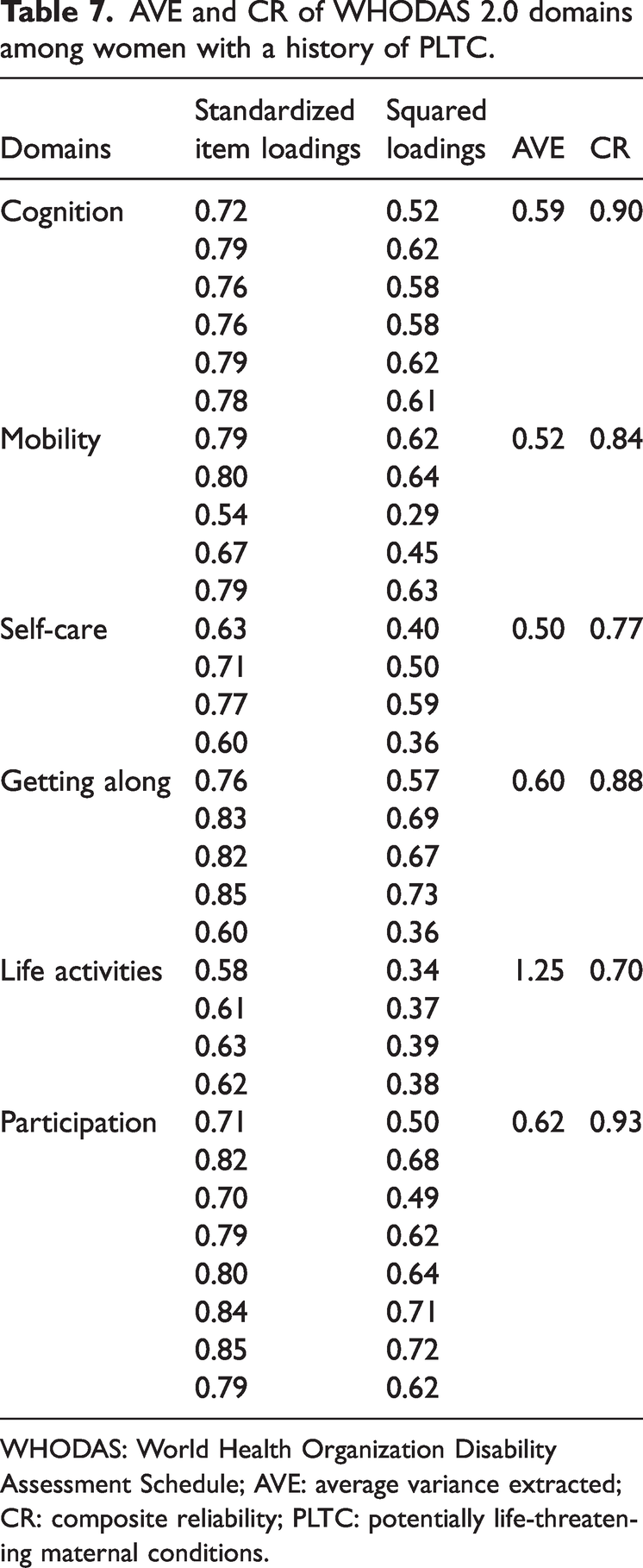
WHODAS: World Health Organization Disability Assessment Schedule; AVE: average variance extracted; CR: composite reliability; PLTC: potentially life-threatening maternal conditions.
Discussion
In public health research and clinical practice, health assessment scales have become increasingly prevalent. 21 Comprehensive evaluation of health requires tools that capture multiple aspects of health conditions; thus, disability assessment instruments must demonstrate validity and reliability. 11 When instruments developed in one cultural context are applied in another, careful adaptation is necessary, as a measure that is reliable in one setting may not be meaningful or interpretable in another. 22
Cultural adaptation of items, such as translating “Getting out of your home” to “Move out of the yard/corridor” and “Maintaining friendships” to “Continuing friendships,” ensured linguistic equivalence in Tigray context. However, these adjustments, which emphasize communal aspects of life, may influence construct equivalence, enhancing local relevance while necessitating caution in cross-cultural comparisons. 23
The reliability analysis of the Tigrigna WHODAS 2.0 demonstrated very high Cronbach’s alpha values for cognition, life activity, participation, and the overall scale (>0.95), indicating excellent internal consistency. However, alpha values above 0.95 may suggest item redundancy, where items are highly similar, potentially inflating reliability without contributing unique information. 24 This phenomenon is consistent with the WHODAS 2.0, which includes deliberate item overlap according to the ICF framework. 11 High item–total correlations (0.738–0.955) support internal consistency, and similar patterns observed in other studies 22 indicate that psychometric performance is not compromised. Future research could apply item response theory (IRT) to address potential redundancy while preserving cross-cultural validity. Nevertheless, these findings affirm the reliability of the Tigrigna WHODAS 2.0 for assessing disability in women with PLTC, with the high alpha values likely influenced by the relatively homogeneous experience of maternal morbidity in the study sample.
In women with PLTC, functional disability primarily presents as mobility limitations (highest median score in D2), restrictions in life activities, and barriers to participation, likely resulting from physical sequelae such as hemorrhage-related weakness or hypertensive complications. These findings are consistent with prior studies on severe maternal morbidity.25,26 The validated Tigrigna WHODAS 2.0 is therefore relevant for follow-up care in Tigray, supporting routine postpartum screening to identify rehabilitative needs (e.g. physical therapy for mobility) and monitor recovery trajectories.
The 30-day recall period of WHODAS 2.0, used in this study during the immediate postpartum period, primarily captures acute disabilities, such as mobility limitations following cesarean delivery, but may not reflect long-term functioning. This temporal limitation restricts generalizability beyond the early postpartum period. Previous research indicates that a 30-day recall may overlook fluctuating postpartum health states. 12 Delayed or repeated assessments are recommended to capture persistent or evolving disabilities. 27 Similar temporal considerations have been observed in psychiatric remission studies. 28 Longitudinal research is warranted to distinguish acute from chronic disability trajectories.
The correlation coefficients between the 36-item and 12-item WHODAS 2.0 domains were generally consistent, except for the mobility domain. The 36-item domains of self-care, getting along, life activities, and participation were highly correlated with their corresponding 12-item domains, supporting convergent validity. Although there was a statistically significant difference between D1 (cognition) of the 12-item and 36-item versions, the correlation coefficient was weak (<0.40), indicating a limited relationship. This suggests that the 12-item Tigrigna WHODAS 2.0 is reliable and demonstrates preliminary validity for clinical use, but it may not comprehensively capture all components assessed by the 36-item version. 22 Moreover, no correlation was observed in the mobility domain between the two forms, potentially due to differences in measurement depth; the 36-item version captures a broader range of functional variation. 22 In the analysis of divergent (external) validity, the cognition and mobility domains showed violations, likely attributable to overlapping items between domains. 29
The validity of domain selection ranged from strong to very strong. Correlations between individual domains and the overall disability score ranged from 0.87 to 0.90, while interdomain correlations ranged from 0.66 to 0.92. These values are slightly higher than those reported in validations of the Polish and traditional Chinese WHODAS 2.0 versions.30,31 The high interdomain correlations may raise concerns regarding discriminant validity, suggesting that the six WHODAS 2.0 domains may not be fully distinct, potentially reflecting near-unidimensionality. Discriminant validity is generally supported when interdomain correlations are moderate (e.g. <0.70), allowing domains to measure unique functional aspects. 32 Correlations exceeding 0.70, such as the 0.92 observed between cognition and self-care, indicate considerable overlap, where changes in one domain mirror changes in others. 33 This pattern is common in WHODAS 2.0 validations, particularly in homogeneous clinical populations in which disability domains are interconnected—for example, mobility impairments (D2) can influence life activities (D5) due to the holistic nature of the ICF framework underpinning the tool.34–36 High correlations (>0.70) have also been reported in studies of chronic disease and general populations, indicating that WHODAS 2.0, although theoretically multidimensional, may function as a unidimensional measure in certain contexts, likely reflecting the shared impact of maternal morbidity. 37
Regarding the use of domain scores in isolation versus the total score, the high interdomain correlations suggest caution when interpreting individual domains independently, as they may not provide unique information beyond the overall construct. 34 The WHO manual recommends using the total score as the primary metric for general disability assessment, particularly in contexts where the measure functions unidimensionally, while domain scores may serve a supplementary role in guiding targeted interventions, such as prioritizing mobility or participation. The total score is more robust for evaluating overall disability, reflecting maternal morbidity’s broad impact across physical, psychological, and social domains, whereas domain scores require careful interpretation considering interdomain correlations. 11 Methodologically, the homogeneous PLTC experience, small sample size, and cultural factors in Tigray (e.g. communal living arrangements) may increase shared variance, amplifying interdomain overlaps. 38
In CFA, the strong factor loadings indicate that the observed variables are well-represented by the latent factors. However, the poor model fit suggests that the overall model does not fully capture the data structure. This poor fit may be attributable to the small sample size, which can produce unstable estimates, inflated factor loadings, and negatively affect fit indices. 39
The sample size of 121 participants, although adequate for a KMO value of 0.93 and a significant Bartlett’s test (p < 0.01), is relatively small for CFA of a 32–36-item instrument, as guidelines recommend 5–10 participants per item. 40 This may partly explain the poor model fit and the potential for sample-dependent results. While the Tigrigna WHODAS 2.0 demonstrates preliminary reliability and validity in women with PLTC, the small sample limits the stability of factor structure and the generalizability of findings. Future studies with larger samples and test–retest reliability assessments are recommended to enhance CFA robustness and confirm the instrument’s dimensionality.
Clinically, the WHODAS 2.0 can inform rehabilitative interventions (e.g. mobility training), mental health support (e.g. addressing participation barriers), and social-support programs (e.g. community reintegration following PLTC). As an outcome measure, it enables the evaluation of quality-of-care improvements by tracking disability reductions in maternal health programs aligned with SDG targets. The validated Tigrigna version also facilitates international comparisons of disability outcomes in PLTC, supporting benchmarking against global studies. 41 This approach promotes equity in maternal health research by highlighting region-specific burdens (e.g. higher mobility-related disabilities in low-resource settings) and informing cross-cultural appropriate interventions.
Convergent validity was not fully assessed because correlations with related constructs (e.g. quality of life or clinical severity scores) were not included; future research should incorporate such measures. The relatively small sample size may reduce the reliability of parameter estimates, affecting the robustness and stability of the CFA results. Future studies should aim for at least 200 participants to ensure adequate statistical power and more stable model estimates. Additionally, the absence of test–retest assessments limits evaluation of temporal stability, while the lack of concurrent validity, such as correlations with clinical severity or external functional measures, constrains the assessment of discriminant and construct validity. Addressing these gaps should be prioritized in subsequent research to strengthen the psychometric evidence for the Tigrigna WHODAS 2.0.
Conclusion
The Tigrigna version of WHODAS 2.0 demonstrated preliminary evidence of acceptable reliability and validity for assessing disability in women with PLTC in Tigray, Ethiopia. However, the small sample size and suboptimal CFA fit indices limit the robustness of the factor structure, indicating that this validation is preliminary. Large-scale studies, including test–retest assessments, are warranted to confirm these findings and refine the instrument for wider clinical and research use.
Footnotes
Acknowledgments
We would like to thank the World Health Organization, the original developer of the WHODAS 2.0 tool, for granting permission to adapt and use it. We also extend our gratitude to the study participants, data collectors, and experts who participated in the translation process, evaluated the semantic equivalence, and assessed the face and content validity of the tool.
Authors’ contributions
FT designed the study, analyzed the data, and drafted the manuscript. FT, GF, YZ, and AB contributed to the study design and critically reviewed the manuscript. All the authors have read and approved the final version of the manuscript.
Availability of data
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare no conflicts of interest.
Ethics approval and consent to participate
The study protocol was reviewed and approved by the Institutional Review Board of Mekelle University College of Health Sciences (approval no. MU-IRB 2129/2023). Informed consent was obtained from all participants. All patient details were deidentified to protect privacy. The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024.
Funding
This research did not receive specific funding from any public, private, or nonprofit organization.
