Abstract
Objective
ST-segment elevation myocardial infarction is a life-threatening coronary artery disease associated with extensive myocardial injury. Circular RNAs are emerging regulators in cardiovascular disease. This study examined circ-0049271 expression in ST-segment elevation myocardial infarction, its clinical associations, and responsiveness to oxidative stress.
Methods
Fifty-five ST-segment elevation myocardial infarction patients and 35 healthy controls were enrolled. Blood was collected before percutaneous coronary intervention and at 0.5 and 48 h after the intervention. circ-0049271 expression was measured using quantitative reverse transcription polymerase chain reaction, and its correlations with the Gensini score, ischemic duration, cardiac biomarkers, and left ventricular ejection fraction were assessed. Human umbilical vein endothelial cells were exposed to hydrogen peroxide to assess circular RNA regulation under oxidative stress.
Results
circ-0049271 expression was elevated in ST-segment elevation myocardial infarction patients versus controls (p < 0.001) and remained high after percutaneous coronary intervention. It was correlated positively with the Gensini score (r = 0.444), creatine kinase–myocardial band (r = 0.427), and serum potassium (r = 0.322) and negatively with left ventricular ejection fraction (r = −0.281). Receiver operating characteristic analysis yielded an area under the curve of 0.788. In human umbilical vein endothelial cells, circ-0049271 expression increased in a dose-dependent manner with hydrogen peroxide treatment, with elevated oxidative stress markers and reduced superoxide dismutase activity.
Conclusions
circ-0049271 is upregulated in ST-segment elevation myocardial infarction and is correlated with myocardial injury, coronary lesion burden, and oxidative stress, supporting its potential as a diagnostic biomarker and therapeutic target in acute myocardial infarction.
Introduction
ST-segment elevation myocardial infarction (STEMI) is the most acute and life-threatening presentation of coronary heart disease (CHD), contributing significantly to global cardiovascular morbidity and mortality. 1 Timely reperfusion, particularly via percutaneous coronary intervention (PCI), is the cornerstone of STEMI management.2,3 However, despite advances in interventional strategies, many patients experience adverse outcomes such as heart failure, ventricular remodeling, and recurrent ischemic events.4,5
In recent years, noncoding RNAs (ncRNAs) have emerged as pivotal regulators of cardiovascular disease.6,7 Among these, circular RNAs (circRNAs), a class of covalently closed, exonuclease-resistant transcripts, have garnered attention for their stability and diverse regulatory functions.8–10 circRNAs have been implicated in apoptosis, inflammation, oxidative stress, angiogenesis, and cardiac remodeling, making them promising candidates for biomarker discovery and therapeutic intervention.8–10 However, their role in the pathophysiology of STEMI remains largely underexplored.
Preliminary transcriptomic analyses have identified circ-0049271 as one of the most significantly upregulated circRNAs in myocardial infarction (MI).10–12 However, its relationship with STEMI severity and the underlying mechanisms remain unclear. In addition, whether the expression of circ-0049271 is modulated by PCI is yet to be elucidated. Therefore, the present study was designed with the following objectives: (a) characterize the expression profile of circ-0049271 in STEMI patients before and after PCI; (b) evaluate its associations with markers of myocardial injury, ischemic duration, and coronary lesion severity; and (c) investigate its regulation under oxidative stress in vitro. By integrating clinical, biochemical, imaging, and cellular data, this study sought to establish circ-0049271 as a potential biomarker and clarify its role in the pathogenesis of STEMI.
Methods
Study design and participants
In this case–control study, a total of 55 patients with STEMI and 35 age- and sex-matched healthy controls were enrolled. STEMI was diagnosed according to the standard criteria: (a) typical ischemic chest pain lasting >20 min; (b) new ST-segment elevation in ≥2 contiguous electrocardiographic leads (≥1 mm in limb leads or ≥2 mm in precordial leads); and (c) elevated cardiac biomarkers, such as troponin I or T above the 99th percentile upper reference limit. All patients underwent PCI as part of routine care. The control group consisted of apparently healthy volunteers recruited from individuals undergoing routine health examinations at our institution during the same study period. To ensure appropriate comparability, controls were screened using a structured questionnaire, clinical examination, and laboratory and imaging evaluations. Exclusion criteria for controls included the following: (a) history of angina, MI, coronary revascularization, or any form of ischemic heart disease; (b) structural heart disease or left ventricular systolic dysfunction (left ventricular ejection fraction (LVEF) <50% on echocardiography); (c) arrhythmia, heart failure, or valvular disease; (d) systemic inflammatory or autoimmune disorders; (e) renal or hepatic dysfunction; and (f) acute or chronic infection within the past 4 weeks. All control participants had normal cardiac structure and function, as confirmed on transthoracic echocardiography without any evidence of ischemic symptoms or electrocardiographic abnormalities. Written informed consent was obtained from all participants prior to enrollment, and patient details were de-identified. The study protocol was approved by the institutional ethics committee, and the study was conducted in accordance with the Declaration of Helsinki of 1975, as revised in 2024. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Study variables
Peripheral venous blood samples were collected from STEMI patients at three predefined time points to capture dynamic changes in biomarker expression: (a) prior to PCI, (b) 30 min post-PCI, and (c) 48 h post-PCI. For healthy controls, a single blood sample was obtained during a routine health examination under fasting conditions. All blood samples were processed immediately and stored at −80°C for subsequent RNA extraction and analysis. Baseline data, including demographic information, blood pressure, body mass index (BMI), smoking status, diabetes mellitus, hyperlipidemia, and use of medications (e.g. antihypertensives, statins, and antiplatelets), were collected at enrollment. Laboratory data were collected for the following parameters: lipid profile, cardiac biomarkers (creatine kinase–myocardial band (CK-MB), cardiac troponin I (cTnI), and N-terminal pro-B-type natriuretic peptide (NT-proBNP)), renal function (serum creatinine and estimated glomerular filtration rate (eGFR)), and serum electrolytes. LVEF was assessed using transthoracic echocardiography, which was performed within 72 h of admission, according to the guidelines of the American Society of Echocardiography. 14
Coronary angiography and Gensini scoring
All STEMI patients underwent coronary angiography as part of their standard clinical evaluation and management. The severity and extent of CHD were assessed using the Gensini scoring system, a widely used method for quantifying coronary atherosclerotic burden. 15 Specifically, the Gensini score is calculated by assigning a severity score to each coronary stenosis based on the degree of luminal narrowing: 25% stenosis = 1 point, 50% = 2 points, 75% = 4 points, 90% = 8 points, 99% = 16 points, and complete occlusion = 32 points. This score is then multiplied by a weighting factor that reflects the functional importance of the lesion’s location in the coronary arterial tree (e.g. left main, proximal left anterior descending (LAD), and distal right coronary artery (RCA)). These base scores are then multiplied by weighting factors, depending on the location of the lesions: left main artery ×5; proximal LAD ×2.5, mid-LAD ×1.5, and distal LAD ×1; first diagonal branch ×1 and second diagonal branch ×0.5; proximal left circumflex artery (LCX) ×2.5, distal LCX and posterior descending branch ×1, and obtuse marginal branch ×0.5; proximal, mid, and distal RCA, and posterior descending branch ×1. The total Gensini score is the sum of the weighted scores across all affected segments, with higher scores indicating more severe and extensive coronary obstruction. Coronary angiograms were independently reviewed by two experienced interventional cardiologists blinded to the study outcomes to ensure consistency and accuracy in scoring.
Circ-0049271 assessment using quantitative reverse transcription polymerase chain reaction (qRT–PCR)
Total RNA was extracted from peripheral blood samples using TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. The quality and concentration of RNA were assessed using a NanoDrop spectrophotometer. Complementary DNA (cDNA) was synthesized from 1 µg of total RNA using a reverse transcription kit (PrimeScript RT Reagent Kit, Takara) following the recommended protocol. qRT–PCR was performed using a SYBR Green master mix (e.g. SYBR® Premix Ex Taq™, Takara) on a real-time PCR detection system (Applied Biosystems 7500 or equivalent). Specific primers targeting circ-0049271 were designed to span the back-splice junction to ensure circRNA specificity. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal control for normalization. Each reaction was run in triplicate, and the relative expression levels of circ-0049271 were calculated using the 2−ΔΔCt method. Melt curve analysis was performed to confirm amplification specificity.
Oxidative stress model in human umbilical vein endothelial cells (HUVECs)
HUVECs were cultured under standard conditions and treated with different concentrations of hydrogen peroxide (H2O2; 0, 100, and 400 µmol/L) for 12 h. Following treatment, cell viability was assessed using the cell counting kit-8 (CCK-8) assay. Levels of reactive oxygen species (ROS), malondialdehyde (MDA), lactate dehydrogenase (LDH), superoxide dismutase (SOD), and endothelin-1 (ET-1) were measured using enzyme-linked immunosorbent assay (ELISA) kits. All assays were performed in accordance with the manufacturers’ protocols, and measurements were obtained using a microplate reader. Each experiment was conducted in triplicate to ensure reproducibility.
Statistical analyses
Continuous variables were expressed as mean ± SD for normally distributed data and as median and interquartile range (IQR) values for non-normally distributed data. Categorical variables were presented as frequencies (percentages). For comparisons between these two groups, Student’s t-test was used for normally distributed variables, and the Mann–Whitney U test was applied for skewed data. For comparisons involving more than two groups, one-way analysis of variance (ANOVA) or Kruskal–Wallis test was used, depending on data distribution. Correlation analyses were conducted using the Pearson correlation coefficient for parametric data and Spearman rank correlation coefficient for nonparametric data. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the diagnostic performance of circ-0049271 and calculate the area under the curve (AUC), sensitivity, and specificity. A two-sided p value <0.05 was considered statistically significant. All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) software (version 25.0, IBM Corp., Armonk, NY, USA) and R software.
Results
Baseline characteristics
Although participants were recruited to achieve age- and sex-comparable groups, the STEMI cohort had a slightly higher mean age, reflecting the real-world demographic pattern of MI patients. As shown in Table 1, patients in the STEMI group were significantly older than those in the control group (58.9 ± 13.7 vs. 51.4 ± 9.4 years, p = 0.001). The prevalence rates of hypertension (54.5% vs. 11.4%, p = 0.001), diabetes mellitus (30.9% vs. 8.6%, p = 0.013), and smoking (47.3% vs. 20.0%, p = 0.009) were higher in the STEMI group. No significant differences were noted between the two groups in terms of BMI, hyperlipidemia, renal dysfunction, alcohol use, and family history of STEMI.
Comparison of baseline characteristics between the STEMI and control groups.

STEMI: ST-segment elevation myocardial infarction; BMI: body mass index; CHD: coronary heart disease.
Data are presented as mean ± SD or n (%).
Comparison of circ-0049271 expression between the STEMI and control groups
Before PCI, the expression of circ-0049271 in the STEMI group was 1.45 ± 0.39, which was significantly greater than that in the control group (1.05 ± 0.26; p = 0.001). At 0.5 and 48 h post-PCI, the STEMI group had significantly higher circ-0049271 expression (1.47 ± 0.44 and 1.57 ± 0.54, respectively) than the control group (1.05 ± 0.26, p = 0.001 for both comparisons).
Correlation between circ-0049271 expression and ischemic duration in STEMI patients
Based on ischemic duration, patients were categorized into three groups: 0–2 h (n = 19), 2–6 h (n = 20), and >6 h (n = 16) groups. The expression of circ-0049271 differed significantly between the three groups (p = 0.001; Table 2). Post-hoc multiple comparisons revealed that the 2–6 h group had significantly higher circ-0049271 expression than the 0–2 h group (p = 0.007); the >6 h group had significantly higher expression than the 0–2 h group (p = 0.001). Although the >6 h group had higher expression than the 2–6 h group, the difference was not statistically significant (p = 0.246).
The relative expression of circ-0049271 according to ischemic duration in STEMI patients.

STEMI: ST-segment elevation myocardial infarction.
p value indicates statistical significance among these three groups.
p1: Comparison between the 0–2 h and 2–6 h groups.
p2: Comparison between the 0–2 h and >6 h groups.
p3: Comparison between the 2–6 h and >6 h groups.
Correlation between circ-0049271 expression and ischemic location, culprit vessel, and number of diseased vessels in STEMI patients
The expression of circ-0049271 was positively correlated with the Gensini score, with a correlation coefficient of r = 0.444 (p = 0.001). Patients in the STEMI group were grouped by ischemic location (anterior, extensive anterior, lateral, inferior, inferoposterior, or posterior wall), culprit vessel (LAD artery, circumflex artery, diagonal branch, and RCA), and number of diseased vessels (single-, double-, or triple-vessel disease). As summarized in Table 3, there were no statistically significant differences in circ-0049271 expression based on ischemic location (p = 0.434), culprit vessel (p = 0.162), or number of diseased vessels (p = 0.933).
Correlation between circ-0049271 expression and ischemic location, culprit vessel, and number of diseased vessels in STEMI patients.

STEMI: ST-segment elevation myocardial infarction.
Correlation between circ-0049271 expression and traditional risk factors
To assess whether circ-0049271 expression is associated with traditional risk factors for CHD, STEMI patients were stratified based on several variables, including age (≥75 vs. <75 years), BMI (≥24 vs. <24 kg/m2), presence of hypertension, diabetes, hyperlipidemia, smoking, and family history of CHD; the analysis revealed no statistically significant differences in circ-0049271 expression among the groups stratified by these risk factors (all p > 0.05; Table 4).
Correlation between circ-0049271 expression and risk factors for CHD.
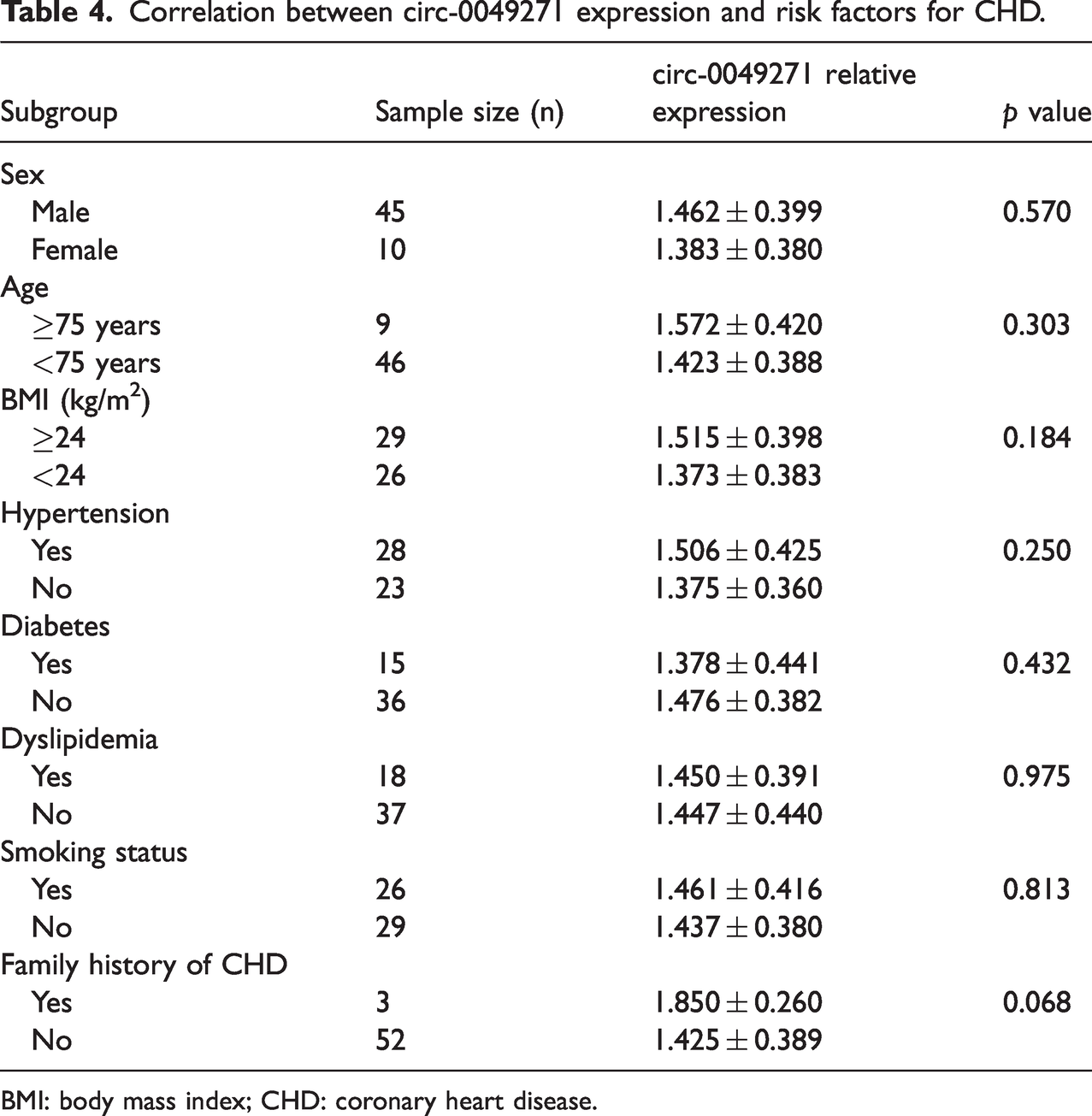
BMI: body mass index; CHD: coronary heart disease.
Correlation between circ-0049271 expression and clinical laboratory and imaging parameters
circ-0049271 was positively correlated with CK-MB (r = 0.427) and serum potassium (r = 0.322) and negatively correlated with LVEF (r = −0.281), with all correlations reaching statistical significance (p < 0.05; Figure 1). However, there were no significant correlations between circ-0049271 expression and cTnI, NT-proBNP, serum sodium, serum calcium, or low-density lipoprotein (LDL) cholesterol (all p > 0.05).

Correlation between circ-0049271 expression and clinical parameters in STEMI patients.
ROC curve analysis of circ-0049271 expression for assessing its diagnostic performance in STEMI patients
The AUC for circ-0049271 was 0.7875. Using a cutoff value of 1.475, the sensitivity was 56.4%, and the specificity was 91.4%, indicating a moderately good discriminatory ability (Figure 2).

Diagnostic performance of circ-0049271 for STEMI. Receiver operating characteristic (ROC) curve showing the ability of circ-0049271 to distinguish STEMI patients from healthy controls. The area under the curve (AUC) was 0.7875. STEMI: ST-segment elevation myocardial infarction.
circ-0049271 expression under oxidative stress in HUVECs
To evaluate the relationship between circ-0049271 expression and oxidative stress in vascular endothelial cells, HUVECs were treated with varying concentrations of H2O2 (0, 100, and 400 μmol/L) for 12 h to induce oxidative stress. The results demonstrated a dose-dependent decline in cell viability (Figure 3) and a corresponding increase in oxidative stress, as evidenced by elevated levels of ROS, MDA, and LDH, along with reduced SOD activity (Figure 4). Additionally, ET-1 levels rose with increasing H2O2 exposure, indicating impaired endothelial function. Notably, circ-0049271 expression increased progressively with higher H2O2 concentrations (Figure 5), suggesting a potential link between its upregulation and the severity of oxidative stress.

Effect of oxidative stress on HUVEC viability. The viability of human umbilical vein endothelial cells (HUVECs) after treatment with hydrogen peroxide (H2O2) at concentrations of 0, 100, and 400 μmol/L for 12 h was assessed using the CCK-8 assay. A dose-dependent decline in cell viability was observed. HUVEC: human umbilical vein endothelial cell; CCK-8: cell counting kit-8.

Changes in the levels of oxidative stress markers in HUVECs exposed to hydrogen peroxide. Levels of reactive oxygen species (ROS), malondialdehyde (MDA), lactate dehydrogenase (LDH), superoxide dismutase (SOD), and endothelin-1 (ET-1) after treatment with 0, 100, and 400 μmol/L H2O2 for 12 h. ROS, MDA, LDH, and ET-1 levels increased, while SOD activity decreased in a dose-dependent manner. HUVECs: human umbilical vein endothelial cells. *p < 0.05.

circ-0049271 expression in HUVECs under oxidative stress. Relative expression levels of circ-0049271 in HUVECs treated with 0, 100, and 400 μmol/L H2O2 for 12 h. A significant dose-dependent increase in circ-0049271 expression was observed with rising H2O2 concentrations. HUVECs: human umbilical vein endothelial cells. *p < 0.05, **p < 0.01
Discussion
This study is the first to comprehensively characterize the expression and biological relevance of circ-0049271 in patients with STEMI. Our findings demonstrate that circ-0049271 is significantly upregulated in STEMI patients and remains elevated in the early post-PCI period. Its expression is correlated with markers of myocardial injury, left ventricular dysfunction, ischemic duration, and coronary lesion burden and is responsive to oxidative stress in vitro.
The observed positive correlation between circ-0049271 and CK-MB, along with its inverse relationship with LVEF, suggests that circ-0049271 is involved in the acute phase of myocardial necrosis and subsequent cardiac dysfunction. 16 These associations underscore its potential role in myocardial injury and adverse remodeling, although the exact underlying mechanisms remain unclear. Notably, circ-0049271 expression was not correlated with cTnI or NT-proBNP; moreover, it was not associated with traditional risk factors such as age, hypertension, and hyperlipidemia. This indicates that its elevation may reflect a distinct molecular axis of STEMI pathophysiology, independent of conventional clinical risk markers. 17
Importantly, circ-0049271 expression was significantly associated with the Gensini score, a validated metric of coronary atherosclerotic burden. Thus, circ-0049271 may not only reflect acute myocardial injury but also serve as an indicator of chronic vascular disease severity. 18 Furthermore, its stepwise increase with longer ischemic duration supports its potential utility as a time-sensitive biomarker. This is particularly relevant in cases where the onset of chest pain is unclear or undocumented, such as in older individuals, those who are unconscious, and those with atypical presentations.
Our in vitro experiments further support the link between circ-0049271 expression and ischemic injury, demonstrating dose-dependent upregulation of its expression in endothelial cells exposed to H2O2. This experimental model mimics the oxidative microenvironment characteristic of ischemic myocardium. The concurrent increases in ROS, MDA, and ET-1 levels, along with decreased SOD activity, suggest that circ-0049271 is involved in the amplification of oxidative stress and endothelial dysfunction, the two key contributors to infarct expansion, microvascular injury, and post-MI complications. 19
The role of circRNAs in oxidative stress regulation has gained increasing attention. For example, circNCX1 promotes cardiomyocyte apoptosis via miR-133a-3p/CDIP1 signaling under oxidative conditions, 20 and circ-0000285 has been implicated in endothelial apoptosis through the miR-625 pathway. 21 In line with these findings, our bioinformatics analysis identified miR-29a-3p and miR-17-3p as potential downstream targets of circ-0049271; furthermore, both of these play recognized roles in oxidative balance, inflammation, and cardiac repair.22,23 Although functional validation is needed, these results suggest that circ-0049271 participates in a broader regulatory network affecting redox homeostasis and vascular integrity.
To further explore clinical correlates, STEMI patients were divided into high and low circ-0049271 expression groups according to the median expression level. Compared with the low expression group, patients in the high expression group exhibited significantly elevated CK-MB levels and Gensini scores as well as reduced LVEF (all p < 0.05). No significant differences were observed in demographic variables or traditional cardiovascular risk factors between the two groups. The subgroup comparison between high and low circ-0049271 expression further supports its close association with myocardial injury and coronary lesion severity, independent of baseline clinical risk factors.
Several important questions remain. First, the specific molecular mechanisms by which circ-0049271 influences myocardial and endothelial responses to injury remain unknown. Further investigation is needed to determine whether it modulates apoptosis, inflammation, fibrosis, or angiogenesis, either directly or via microRNA (miRNA) sponging. Second, standardization of detection methods remains a technical hurdle due to the unique circular structure and potential amplification artifacts of circRNAs. Third, although the current findings establish correlation, prospective studies are needed to determine whether circ-0049271 has prognostic value or therapeutic implications in STEMI.
Emerging therapeutic strategies targeting circRNAs, including circRNA mimics, antisense oligonucleotides, and clustered regularly interspaced short palindromic repeats (CRISPR)-mediated circularization modulation, are under development in the fields of oncology and neuroscience. Cardiovascular applications are still in their early stages; however, the robust association of circ-0049271 expression with myocardial injury, ischemic burden, and oxidative stress provides a compelling rationale for further mechanistic and translational research in this area. Taken together, circ-0049271 is a novel, clinically relevant circRNA that integrates multiple dimensions of STEMI pathology, including ischemic duration, coronary lesion burden, myocardial necrosis, and oxidative stress. Its potential as a biomarker and therapeutic target merits further exploration.
It remains to be determined whether the observed upregulation of circ-0049271 is specific to STEMI or represents a broader phenomenon across the acute coronary syndrome (ACS) spectrum. STEMI is characterized by complete coronary occlusion and extensive transmural necrosis, whereas non-ST-elevation MI (NSTEMI) and unstable angina typically involve partial occlusion or transient ischemia with lesser degrees of myocardial injury. Although our study focused exclusively on STEMI, emerging transcriptomic evidence suggests that certain circRNAs demonstrate progressively higher expression levels from stable angina to NSTEMI and STEMI, reflecting increasing ischemic severity and oxidative stress burden. Given that circ-0049271 is responsive to oxidative stress and is correlated with myocardial injury markers, it is plausible that its elevation is not restricted to STEMI but rather scales with the extent of ischemia and necrosis. Future comparative studies involving patients with NSTEMI and unstable angina are warranted to delineate whether circ-0049271 serves as a STEMI-specific biomarker or as a general indicator of acute myocardial ischemia within the ACS continuum.
Strengths and limitations
This study offers several notable strengths. To the best of our knowledge, this is the first study to systematically evaluate the expression of circ-0049271 in STEMI patients, integrating clinical, imaging, biochemical, and cellular data. The study design included dynamic time-point sampling (pre-PCI and post-PCI), and the observed associations with established clinical markers, such as CK-MB, LVEF, and Gensini score, support the biological relevance of circ-0049271. The incorporation of an oxidative stress model using HUVECs provides complementary mechanistic insights, demonstrating a dose-dependent response of circ-0049271 under simulated ischemic conditions. However, certain limitations should be acknowledged. First, this was a single-center study with a relatively small sample size, which may limit the generalizability of the findings. Second, the cross-sectional and observational nature of the analysis precludes causal inference. Third, although the in vitro oxidative stress model provided preliminary mechanistic evidence, we did not conduct functional validation experiments such as gene knockdown or overexpression to confirm the specific role of circ-0049271. Fourth, the lack of longitudinal follow-up data restricts our ability to assess the prognostic value of circ-0049271 for adverse cardiovascular outcomes. Fifth, we did not compare circ-0049271 and high-sensitivity (hs) cardiac troponin, which is currently the gold standard for diagnosing myocardial injury. Although we included conventional cardiac biomarkers such as cTnI and CK-MB, we did not evaluate the prognostic ability of hs-troponin in our cohort. Future studies should include hs-troponin to validate the diagnostic and prognostic potential of circ-0049271 relative to this widely accepted biomarker. Sixth, the sample size of our study, which consisted of 55 STEMI patients, is relatively small. Although the observed trends are promising, larger sample sizes would provide more robust statistical power and enhance the generalizability of our findings. We recognize that a larger cohort would also allow a more detailed exploration of subgroup differences and strengthen the evidence for the use of circ-0049271 as a potential biomarker in STEMI. Finally, another limitation is the relatively limited assessment of infarct size and location in our analysis. Although we correlated circ-0049271 expression with the Gensini score, a measure of coronary atherosclerotic burden, and ischemic duration, we did not fully evaluate the infarct size or specific infarction locations in our study cohort. More detailed imaging techniques such as cardiac magnetic resonance imaging (MRI) or serial echocardiographic assessments could provide additional insights into how circ-0049271 levels are correlated with infarct size, location, and myocardial injury severity. These aspects would help refine our understanding of circ-0049271’s role in STEMI pathophysiology.
Conclusion
circ-0049271 is significantly upregulated in patients with STEMI and is associated with myocardial injury severity, coronary lesion burden, ischemic duration, and oxidative stress. Its persistent elevation post-reperfusion and correlation with key clinical indicators support its potential role as a diagnostic and mechanistic biomarker. In vitro evidence suggests that circ-0049271 contributes to endothelial dysfunction under oxidative stress, a hallmark of myocardial ischemia–reperfusion injury. These findings highlight circ-0049271 as a promising target for future research. Further mechanistic studies and prospective cohort investigations are warranted to validate its clinical utility and explore therapeutic interventions aimed at modulating its expression or downstream pathways.
Footnotes
Acknowledgments
We gratefully acknowledge the contributions of all participants and the dedicated support of our colleagues.
Author contributions
HCL and DCX designed this study; HCL, CX, and WWL curated the data; WWL analyzed the data; HCL wrote this manuscript; CX, WWL, and DXC revised this manuscript.
Data availability statement
Data are available upon reasonable request to the corresponding author.
Declaration of conflicting interests
None.
Funding
None.
