Abstract
Objective
To describe patients with severe asthma treated with or eligible for monoclonal antibodies, assessing the health and economic burden using the Italian National Healthcare Service (SSN) administrative data.
Methods
From 4.6 million inhabitants, among patients with asthma from 1 January to 31 December 2022, those with severe asthma were identified by monoclonal antibody dispensation (cohort A) and by eligibility for monoclonal antibodies, defined as continuous treatment with medium- or high-dose inhaled corticosteroids and long-acting beta agonists and the occurrence of exacerbations (cohort B-narrow and cohort B-broad according to “narrow” and” broad” definitions, respectively). One-year exacerbations, healthcare utilization, and direct costs were assessed.
Results
Of the 128,621 patients with asthma (51.8% women; mean age, 54 years), patients with severe asthma were identified as follows: cohort A (n = 3046; 2.4%), cohort B-narrow (n = 3517; 2.7%), and cohort B-broad (n = 7621; 5.6%). Compared with cohort A, patients in cohorts B-narrow and B-broad were older, had more comorbidities, experienced more moderate/severe exacerbations (70.9%–57.3% vs. 46.7%), and had higher hospitalization rates and greater drug use but fewer specialist visits. The annual SSN costs averaged €7512 for cohort A versus €2911–€2351 for cohorts B-narrow and B-broad. Cohort A incurred higher costs for asthma drugs, whereas cohorts B-narrow and B-broad incurred higher costs for concomitant drugs, hospitalizations, and specialist care.
Conclusions
A significant disease burden exists in patients with uncontrolled severe asthma who are potentially eligible for monoclonal antibodies in Italy.
Introduction
Asthma is one of the most common chronic respiratory diseases and a major noncommunicable disease, affecting approximately 20% of children and 5%–10% adults worldwide. 1 Symptoms include wheezing, breathlessness, chest tightness, and cough that vary over time and in intensity, together with expiratory airflow limitation that may later become persistent. 2 An estimated 5%–10% of patients with asthma have the severe form of the disease, the definition of which remains heterogeneous depending on the guideline and classification used.1,3 More comprehensively, severe asthma (SA) is defined by poor symptom control and/or frequent moderate or severe exacerbations (i.e. uncontrolled asthma) despite adherence to maximal optimized treatment with medium- or high-dose inhaled corticosteroids (ICS) plus a second controller (i.e. long-acting beta agonist (LABA) and/or oral corticosteroids (OCS)) and management of contributory factors (e.g. inhaler technique and adherence), or asthma that worsens when high-dose treatment is reduced.1,3,4 Overall, SA is associated with frequent symptoms, an increased risk of exacerbations, greater healthcare utilization, and reduced health-related quality of life.4–6
Patients with SA frequently experience multimorbidity, which reduces quality of life. Indeed, some comorbidities can complicate asthma treatment, increase the risk of poor asthma-related outcomes, and cause significant productivity losses and healthcare costs up to fivefold higher than those attributable to asthma alone. 7 Patients with SA often require urgent healthcare services, such as OCS prescriptions, specialist consultations and diagnostic investigations, emergency department (ED) visits, and hospitalizations, leading to a substantial burden on healthcare systems, patients, and caregivers. 6 Recommendations for the management of SA are therefore aimed at improving health status and quality of life, particularly by reducing the frequency of exacerbations and eliminating the need for maintenance OCS. 2 These recommendations include patient education on asthma and inhaler technique; nonpharmacological interventions (e.g. smoking cessation and weight loss); personalized management of modifiable risk factors, including comorbidities, throughout the asthma care pathway, possibly through a multidisciplinary approach; and add-on nonbiologic and, when appropriate, biologic therapies according to steps 4 or 5 of the Global Initiative for Asthma (GINA) guidelines.1–4,7
Type 2 inflammation is very common in patients with SA; it is characterized by the presence of cytokines such as interleukin (IL)-4, IL-5, and IL-13 and is often associated with elevated blood or sputum eosinophils (EOS) or increased fractional exhaled nitric oxide, together with atopy and elevated immunoglobulin (Ig) E levels. 4 In the presence of type 2 airway inflammation, patients with SA may be relatively refractory to high-dose ICS; therefore, the addition of a biologic therapy for a trial period of 4 months is recommended if the patient continues to experience exacerbations or poor symptom control despite 3–6 months of adherence to maximally optimized high-dose ICS treatment.3,4 Currently, type 2-targeted biologic therapies reimbursed by the Italian National Healthcare Service (Servizio Sanitario Nazionale (SSN)) for SA in Italy include anti-IgE agents such as omalizumab, 8 anti-IL-5 receptor alpha (IL-5Rα) agents such as benralizumab, 9 anti-IL-5 agents such as mepolizumab, 10 anti-IL-4Rα agents such as dupilumab, 11 and the anti-thymic stromal lymphopoietin (TSLP) monoclonal antibody (mAb) tezepelumab, which has recently been approved for reimbursement. 12
As studies based on real-world data addressing SA are few and heterogeneous, large population databases are necessary to describe the management of this condition and generate real-world evidence.
This retrospective observational cohort study of Italian administrative healthcare data aimed to describe patients with SA, comparing those receiving treatment with mAbs with those eligible for such treatment, in terms of 1-year healthcare utilization and related direct costs borne by the SSN.
Methods
Data source
The Fondazione Ricerca e Salute (ReS) database collects and integrates administrative healthcare data that Italian local and regional healthcare authorities are required to provide annually to the Italian Ministry of Health for reimbursement purposes, as the SSN provides universal coverage. The ReS database includes information from approximately 5 million inhabitants per year (2014–2023), representing approximately 9% of the Italian population. Its age-group distribution is consistent with that reported by the Italian Institute of Statistics (Figure S1). 13 Additional information on Fondazione ReS and its database is provided in the Supplementary File (Box S1).
Data were anonymized at the source and provided in aggregated form in accordance with European Regulations 2014/536 14 and 2016/679, 15 as agreed by the regional and local authorities that own the data. The need for informed consent was waived in accordance with the specific provisions of the Italian Privacy Authority. 16 Data cannot be traced to individual patients. The need for ethical approval was waived in line with European Regulation 2014/536; 14 moreover, national legislation on observational studies requires a positive opinion from the Ethics Committee only for prospective observational studies, not for retrospective studies. 17 The study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 18
Study cohort identification
From the total population available in the ReS database between 1 January and 31 December 2022 (accrual period; 4,602,017 inhabitants), patients with analyzable data from at least 3 previous years (i.e. lookback period; Figure S2) were screened. Patients with asthma were identified using three different proxies: (a) disease waiver claim code for asthma; (b) dispensation of mAbs; and/or (c) dispensation of other drugs for obstructive airway diseases. All criteria and corresponding codes are listed in Table S1.
Patients with SA were analyzed from 2014 to 2022 in accordance with steps 4 and 5 of the GINA’s treatment recommendations. Step 4 corresponds to patients experiencing asthma exacerbations despite continuous treatment with medium- or high-dose ICS plus LABA (i.e. potentially eligible for mAbs), and step 5 corresponds to patients receiving mAbs.
Patients with SA were classified as follows:
Cohort A. Patients receiving reimbursed mAbs for asthma during 2014–2022 (i.e. omalizumab, mepolizumab, benralizumab, or dupilumab);
Cohort B (eligibility for mAbs).
“Narrow” definition. Patients who experienced at least two moderate or one severe exacerbation despite treatment with medium- or high-dose ICS in combination with LABA (cohort B-narrow); “Broad” definition. Patients who experienced at least one moderate or severe exacerbation despite treatment with medium- or high-dose ICS in combination with LABA (cohort B-broad).
According to GINA guidelines, at least two dispensations of OCS or one hospitalization due to asthma were considered as proxies for moderate or severe exacerbations, respectively (Table S2). 4 Although GINA defines moderate exacerbations as at least two events, patients with SA often have OCS available at home for use during moderate exacerbations. Because such home provisions are not captured in administrative data, cohort B-broad was also included in the analysis to avoid underestimating moderate exacerbations.
The algorithm and study design were validated following established scientific methodology, including a comprehensive review of the literature and consultation with a leading pneumologist specializing in asthma, immunology, and allergology who participated in the study.19–21
The index date was defined as the earliest date of dispensing asthma medication and/or waiver if the first record of asthma occurred during the accrual year (i.e. 2022) or as 1 January 2022 if the first administrative record of asthma occurred during the lookback period.
Demographics and comorbidities
The overall cohort of patients with asthma as well as cohorts A and B were characterized by sex, age, and prevalence of asthma on the index date. In addition, the frequencies of analyzable comorbidities—those relevant in the overall population and in patients with SA—were described (Table S3). These comorbidities included arterial hypertension, dyslipidemia, thyroid disease, neoplasia, depression, diabetes, osteoporosis, arrhythmias, glaucoma, nasal polyposis, coronary artery disease, psychosis, chronic liver disease, cerebrovascular disease, heart failure, and chronic kidney disease.
The rate of moderate or severe exacerbations in each cohort during the first year following the index date was also assessed (Table S2).
Healthcare resource utilization and direct cost analyses
Cohorts A, B-narrow, and B-broad were observed throughout the first year following the index date (follow-up; Figure S2) to assess the following:
Consumption of mAbs reimbursed by the SSN for asthma (i.e. omalizumab, benralizumab, mepolizumab, and dupilumab) as well as drugs for obstructive airway diseases according to the active substance (5th-level Anatomical Therapeutic Chemical (ATC) code; Table S1), reported as the distribution of patients treated and defined daily dose (DDD); Concomitant drugs according to the chemical subgroup (4th level ATC code), with a focus on systemic antibacterials (1st level ATC code: J01), reported as the distribution of patients treated and DDD; All-cause and asthma-related overnight hospitalizations, reported as the distribution of admitted patients and mean length of in-hospital stay (days); ED visits, reported as the distribution of admitted patients and mean number of visits per patient; Local outpatient specialist services (i.e. diagnostic tests and invasive/noninvasive procedures), reported as the distribution of patients and mean number of services performed per patient; Per-patient mean annual direct healthcare costs reimbursed by the SSN, provided both as individual cost items (i.e. pharmaceuticals, hospitalizations, and local outpatient specialist services) and as the integration of all administrative healthcare databases (Box S1).
Descriptive statistical analyses
Continuous variables were presented as means ± standard deviation and median (interquartile range). Categorical variables were reported as frequencies and percentages within each category. Data extraction was performed using Oracle SQL Developer, Italian version 18.1.0.095 (Redwood City, CA), and subsequent analyses were conducted using Microsoft Excel Office 365 (Microsoft Corporation, Redmond, WA). A chi-squared test was performed to compare frequencies of patient demographics, comorbidities, dispensations of concomitant drugs, hospital and ED admissions, and use of local outpatient specialist services between cohort A (reference) and cohorts B-narrow and B-broad. Differences were considered statistically significant at p < 0.05.
Results
Demographics and clinical characteristics
From the ReS population in 2022 who had analyzable data from at least 3 previous years, 128,621 patients with asthma were identified (prevalence 29.3 per 1000 inhabitants). Among them, 3046 (2.4%) patients were treated with mAbs (cohort A) at baseline (2014–2022; Figure 1), whereas 125,575 (97.6%) patients were not treated with mAbs. Of these, 3517 (2.7%) patients met the narrow definition of eligibility for mAbs, and 7621 (5.6%) met the broad definition (Figure 1).
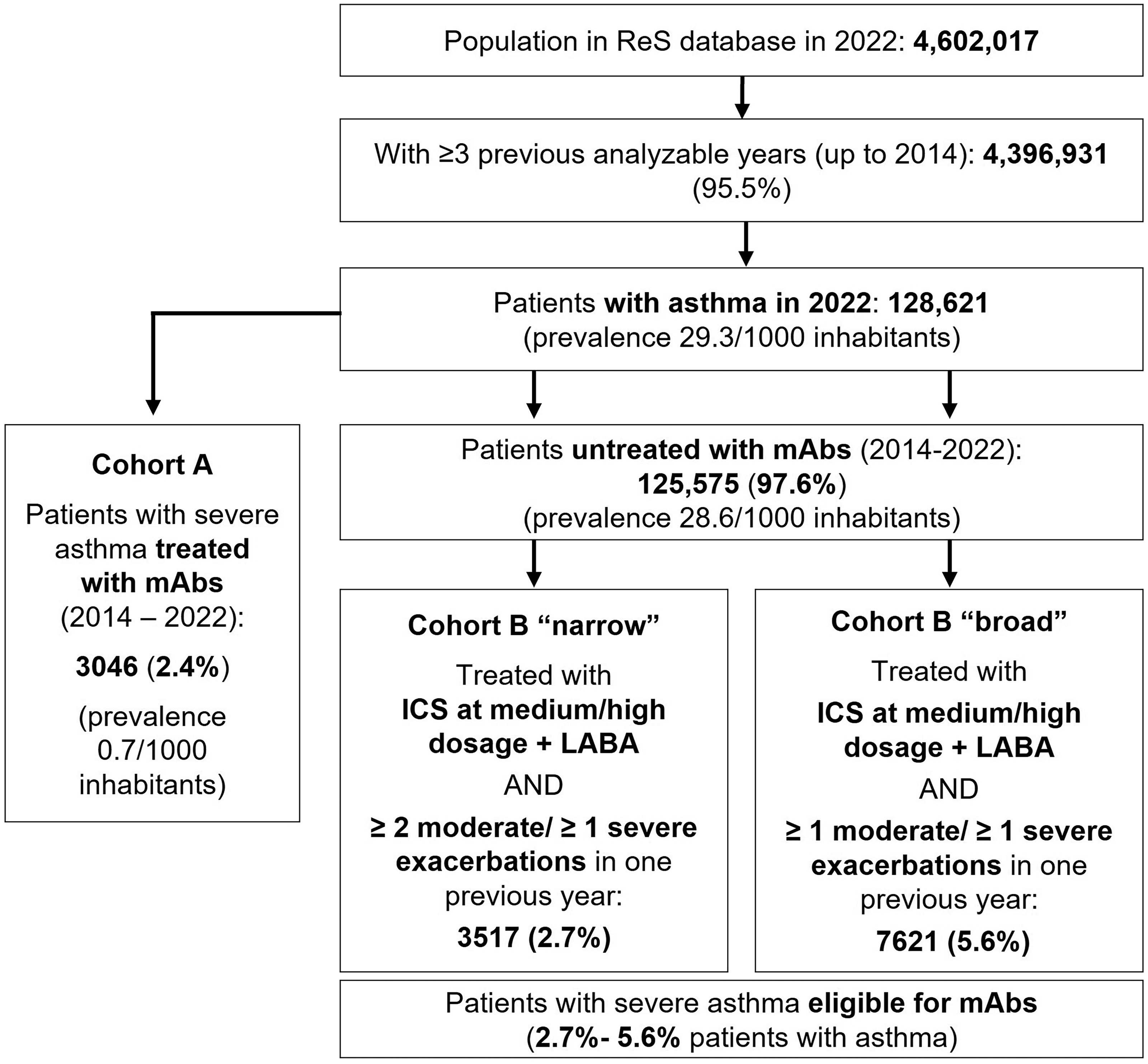
Identification of patients with severe asthma in 2022 from the ReS database, treated with or eligible for mAbs. ReS: Ricerca e Salute; mAbs: monoclonal antibodies; ICS: inhaled corticosteroid; LABA: long-acting beta agonist.
The overall age distribution showed that 10.2% of patients were aged 13–25 years and 20.4% were aged 46–59 years (Table 1). mAb use was highest among patients aged 46–59 years. Use of mAbs increased progressively until the age of 60–69 years and then declined sharply, whereas continuous use of medium- or high-dose ICS/LABA combinations increased until a peak at 70–79 years (Table 1, Figure S3). Female patients predominated among all patients with asthma and within each cohort, with a significantly higher prevalence observed in cohort A.
Demographics and clinical characteristics of patients with asthma in 2022 and severe asthma, by cohort.

Differences between cohorts A and B-narrow and between cohorts A and B-broad were statistically significant if the p-value was <0.05 using a chi-squared test between frequencies.
Compared with patients eligible for mAbs, those receiving treatment with mAbs were younger, with at least 8 years lower mean age (Table 1). Overall, patients in cohorts B-narrow and B-broad had a higher burden of comorbidities than those in cohort A, except for nasal polyposis, which was more prevalent in cohort A than in cohorts B-narrow and B-broad (8.3% vs. 2.7%–3.5%; p < 0.05) (Table 1).
During follow-up, patients in cohorts B-narrow and B-broad experienced significantly higher rates of moderate asthma or SA exacerbations than those in cohort A (57.3%–70.9% vs. 46.7%) (Table 1).
Healthcare resource utilization and direct cost analyses
During follow-up, more than 90% of patients in each cohort received at least one drug for obstructive airway disease (ATC R03) (Table S4). Fixed-dose combination (FDC) ICS/LABA therapy was the most frequently administered treatment across all cohorts. More than half of the patients in cohort A continued to receive mAbs, with an average consumption of up to 230.1 DDD for omalizumab, benralizumab, and mepolizumab (in descending order of dispensation) and 187.7 DDD of dupilumab. Less than 1% of patients eligible for mAbs initiated such treatment during follow-up, including dupilumab and benralizumab. Across all cohorts, approximately one quarter of patients received ICS, and fewer than one fifth received LABA and/or long-acting muscarinic antagonists (LAMA), administered as either open or fixed combinations (Table S4).
At least one concomitant drug was dispensed to more than 90% of patients in each cohort (Table 2). Consistent with the comorbidity findings, significantly higher proportions of patients receiving treatment were observed among those eligible for mAbs across almost all of the first 10 chemical subgroups (Table 2). OCS, proton pump inhibitors (PPIs), and combinations of penicillins were the most frequently dispensed drug classes in each cohort. During follow-up, 69.7% of cohort A and 72.6%–79.2% of cohorts B-narrow and B-broad received at least one systemic antibacterial drug; the most dispensed drugs were, in descending order of use, combinations of penicillins, macrolides, third-generation cephalosporins, and fluoroquinolones.
Free filled prescriptions of concomitant drugs (first 10 chemical subgroups) during the follow-up year, by cohort.
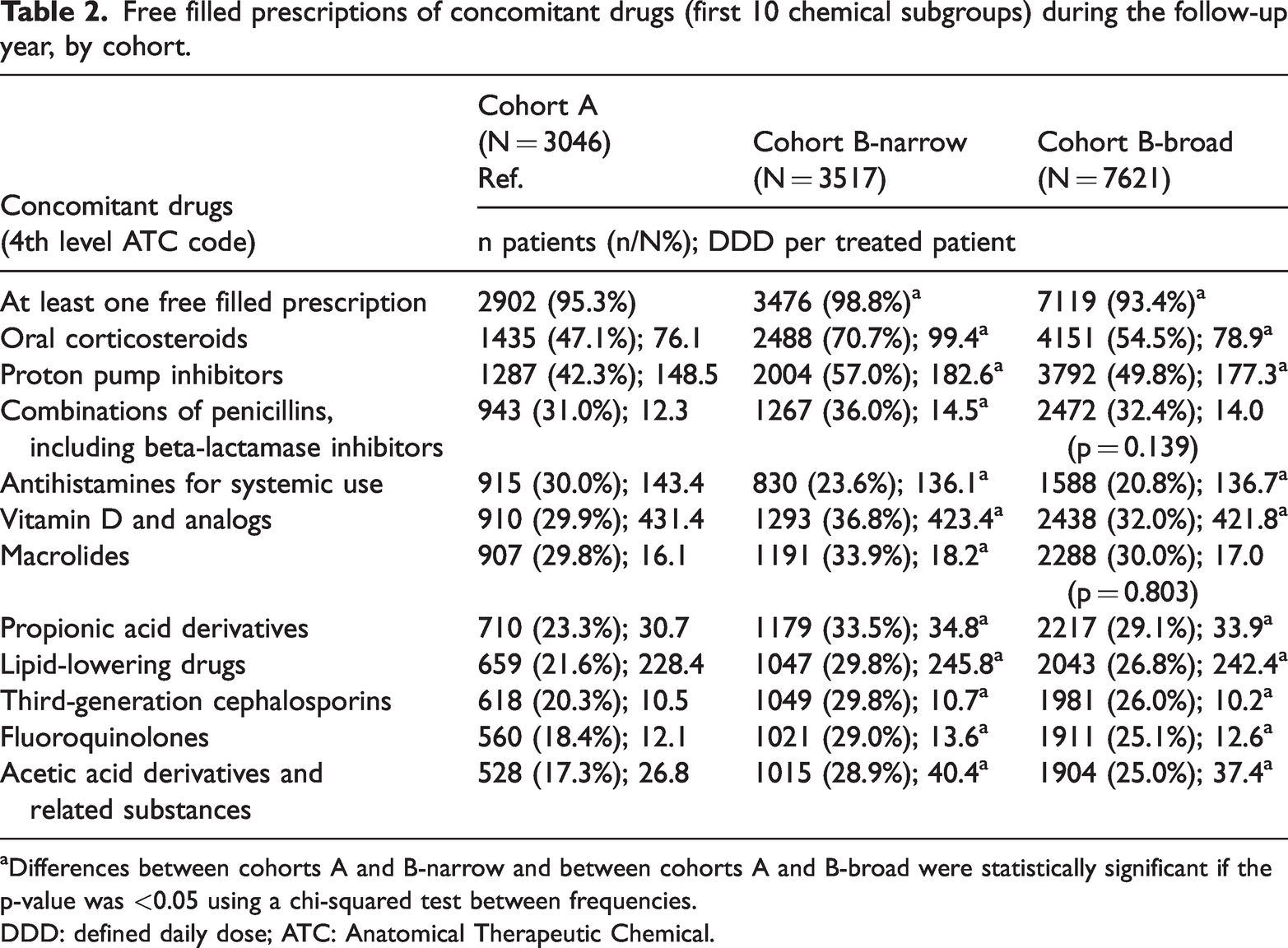
Differences between cohorts A and B-narrow and between cohorts A and B-broad were statistically significant if the p-value was <0.05 using a chi-squared test between frequencies.
DDD: defined daily dose; ATC: Anatomical Therapeutic Chemical.
Patients eligible for mAbs were more frequently admitted for overnight hospitalizations (9.5%–12.0%) and ED visits (18.9%–20.9%) compared with cohort A (10.5% and 19.0%, respectively), although these differences were not statistically significant. Overnight hospitalizations were mainly due to pulmonary and cardiovascular (CV) diseases, while general and respiratory-related symptoms were the most common causes of ED visits. Patients in cohort A accessed local outpatient specialist services significantly more often than those in cohorts B-narrow and B-broad (88.4% vs. 76.8%–81.9%; p < 0.05), with the highest rates of access observed for pulmonary examinations, unspecified specialist examinations, and in-depth pulmonary evaluations (Table 3).
Use of healthcare services during the follow-up year, by cohort.

Differences between cohorts A and B-narrow and between cohorts A and B-broad were statistically significant if the p-value was <0.05 using a chi-squared test between frequencies.
Analysis of annual direct costs showed that, on average, the SSN spent €7512 per patient in cohort A and €2911–€2351 per patient in cohorts B-narrow and B-broad. Pharmaceuticals were the main cost component across all cohorts, accounting for up to 77.7% of total expenditure per patient in cohort A, primarily due to the use of mAbs (Table 4). In contrast, for patients eligible for mAbs, the mean per-patient costs for concomitant drugs, overnight hospitalizations, and outpatient specialist services were approximately twice those for patients treated with mAbs (37.1%–37.9% vs. 15.9%, 27.2%–29.4% vs. 17.3%, and 13.1%–13.7% vs. 5.0%, respectively).
Per-patient mean direct healthcare costs borne by the Italian National Healthcare Service during the follow-up year, by cohort.

Discussion
This retrospective observational study provides a comprehensive description of patients with SA, comparing those receiving treatment with mAbs with those eligible for such treatment but not receiving it. Using Italian administrative healthcare data, the study examined patient characteristics, the burden of SA and comorbidities, treatment patterns, healthcare resource utilization, and associated costs.
Administrative data have previously been validated as a reliable source for identifying and analyzing patients with asthma.19–21 The overall prevalence of 29.3 asthma patients per 1000 inhabitants of any age in Italy in 2022, as observed in this study, falls within the range of crude prevalence rates reported in previous Italian studies (2.7%–11%) 19 and in worldwide literature (1%–20%). By adopting a comprehensive definition of SA, this study identified affected patients using proxies based on therapeutic strategies and exacerbations. However, reliance on these proxies may have led to underestimation or overestimation of the number of patients with SA and may not have captured the heterogeneity of its different phenotypes.
The study identified notable differences between patients with SA treated with mAbs (cohort A) and those potentially eligible for such treatment (cohorts B-narrow and B-broad). Patients eligible for mAbs exhibited poorer clinical characteristics: they were older and had higher prevalence of most analyzed comorbidities, with statistically significant differences. Moreover, compared with patients receiving mAbs, eligible patients experienced more exacerbations, were more frequently prescribed concomitant medications—particularly OCS, PPIs, and systemic antibacterials—and accessed local outpatient specialist services more often. These differences were statistically significant.
Although this study was not designed to evaluate the association between biologic use and comorbidities, significant differences in comorbidity rates were observed between patients treated with mAbs and those eligible for treatment. It is plausible that patients receiving mAbs were less affected by comorbidities that increase the risk of asthma exacerbations, healthcare utilization, and costs, thereby contributing to improved clinical outcomes and quality of life. Further details on this aspect are provided in Box S1.
During follow-up, patients in cohorts B-narrow and B-broad experienced significantly more frequent moderate or severe exacerbations than those in cohort A. This finding aligns with evidence suggesting that patients with asthma who have recently experienced an exacerbation are six to eight times more likely to experience subsequent exacerbations. 22 It also indicates that these patients may have had uncontrolled disease for which biologic therapy was warranted. Consistently, a significantly higher proportion of patients in cohorts B-narrow and B-broad received OCS during follow-up, while fewer than 1% were treated with mAbs. The elevated rate of exacerbations requiring OCS highlights an important unmet medical need, underscoring the necessity for improved patient management.22,23 According to GINA guidelines, if asthma remains uncontrolled after 3–6 months of optimized management, specialists should consider the use of biologic therapies based on eligibility, with the goal of achieving long-term clinical remission.2,4 The selection of a specific mAb is guided by SA criteria and phenotype-specific eligibility. During the study period, the following mAbs were reimbursed in Italy: anti-IgE omalizumab for severe allergic asthma in patients aged over 6 years, anti-IL-5Rα mepolizumab and anti-IL-5 benralizumab for severe EOS asthma in patients aged over 18 years, and anti-IL-4Rα dupilumab severe EOS/type 2 asthma or asthma requiring maintenance OCS in patients aged over 6 years.4,8–10,24 In February 2024, tezepelumab was approved and reimbursed in Italy for uncontrolled SA in patients aged over 12 years. 12
To date, the low utilization of biological therapies may be attributed to several factors, including their high cost and limited evidence regarding effectiveness in specific populations, such as adolescents and children. 3
As expected, most patients in all cohorts received ICS/LABA therapy during follow-up for maintenance purposes.4,6,25 However, substantial proportions of patients (approximately 50%–70%) were also treated with OCS. Analysis of DDD indicated that patients eligible for mAbs were significantly more likely to receive OCS than those already receiving biologic therapy. Consequently, patients not treated with mAbs were potentially at higher risk of serious adverse events and development of OCS-related comorbidities.4,7,26 Evidence indicates that each OCS prescription contributes to a cumulative health burden, regardless of dose or duration, highlighting the importance of implementing OCS-sparing strategies to improve patient outcomes. 27 Recent expert consensus and asthma guidelines emphasize minimizing OCS use, the utility of biologic therapies as steroid-sparing agents, and the importance of systematical evaluating eligibility for biologic treatment.3,4,28,29
Analysis of concomitant drug use revealed a high utilization of systemic antibacterials, particularly penicillins and macrolides, which were dispensed to a significantly higher proportion of patients eligible for mAbs than to those already receiving treatment.3,4,26 Overall, the benefits of antibacterial therapy should be carefully weighed against potential risks, considering the local antimicrobial resistance context. 30
Patients with SA contribute substantially to healthcare utilization and direct costs, as well as to indirect costs related to loss of productivity at work and school, particularly in cases of severe uncontrolled disease.22,31 In line with this, the study showed direct costs incurred by the SSN, comparing patients receiving mAbs with those eligible for such treatment. As expected, total costs were higher for patients treated with mAbs, primarily due the high cost of these drugs. Conversely, patients eligible for mAbs incurred higher costs for concomitant medications, hospitalizations, and local outpatient specialist services. These findings are consistent with previously published literature.5,32
Strengths and limitations
A key strength of this study is the coverage provided by the ReS database, including approximately 9% of the Italian population, its alignment with the Italian National Statistics by age group, and the universal coverage of the SSN, which reduces regional disparities. 33 Thus, the ReS database can be considered representative of the Italian population and generalizable to patients with SA treated with or eligible for mAbs. Specific strengths include the use of a tailored algorithm to identify patients with SA, incorporating a minimum 2-year lookback period, medications aligned with the step-care approach to asthma therapy (i.e. GINA steps 4–5), and hospitalizations due to asthma as a proxy for severe exacerbations, along with the exclusion of patients with COPD.2,19–21 This approach aligns with the necessary differential diagnosis between asthma and other conditions that may present with asthma-like symptoms.1,4,19,21
Nevertheless, a direct linkage with clinical, primary care, or anatomic–pathological databases was not possible. Consequently, confirmation of asthma diagnosis or severity and assessment of contributing factors to symptoms and exacerbations could not be performed due to the absence of clinical and laboratory data documenting lung function (e.g. forced expiratory volume, bronchial provocation tests, and peak expiratory flow) and other relevant information such as inhaler technique, therapy adherence, comorbidities not captured by administrative data (e.g. chronic sinusitis, obesity, and anxiety), socioeconomic status, smoking habits, and environmental exposures.1,4,19,27 In addition to clinical variables such as serum EOS and IgE levels, administrative healthcare databases do not capture socioeconomic status or race/ethnicity, limiting interpretation of the results. Future integration of multiple data sources may help overcome these limitations. The higher utilization observed among patients eligible for mAbs, compared with cohort A, may partly be due to the management of their significantly higher frequency of moderate exacerbations, which were identified via OCS dispensations. Another limitation of this study is its purely descriptive design; establishing causal or correlational relationships would require more detailed statistical analyses.
Finally, it should be noted that the results pertain to the Italian healthcare system, characterized by a single-payer model, and therefore may not be generalizable to other healthcare systems.
Conclusions
The results of this study indicate that patients with uncontrolled SA who are potentially eligible for mAbs in Italy experience a substantial disease burden. A complex interplay exists between asthma, multimorbidity, and polypharmacy, which requires management by specialists, preferably within a multidisciplinary team. Real-world claims data, such as administrative healthcare databases, represent a valuable source for studying routine clinical practice and patient burden, complementing findings from clinical trials and registries. This study provides a comprehensive overview of the real-world management of patients with SA who are treated with mAbs or remain uncontrolled and potentially eligible for such therapies. These insights can support payers, healthcare providers, and policymakers in identifying unmet medical needs and guiding future research to address gaps and improve patient outcomes.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251409996 - Supplemental material for Burden of severe asthma in Italy: Patients treated with versus eligible for monoclonal antibodies in a large real-world study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251409996 for Burden of severe asthma in Italy: Patients treated with versus eligible for monoclonal antibodies in a large real-world study by Letizia Dondi, Giulia Ronconi, Silvia Calabria, Irene Dell’Anno, Leonardo Dondi, Alice Addesi, Immacolata Esposito, Giorgio Walter Canonica, Nello Martini and Carlo Piccinni in Journal of International Medical Research
Footnotes
Acknowledgments
The authors acknowledge the use of the Deep L AI tool to improve the English language of the manuscript.
Authors’ contributions
Conceptualization, LD, GR, SC, GWC; Data curation, LD, LeonardoD; Formal analysis, LD, LeonardoD; Funding acquisition, NM, CP, IE; Investigation, LD, LeonardoD, GR, ID; Methodology, LD, GR; Project administration, LD, GR, SC, AA, IE; Software, LD, LeonardoD; Supervision, NM; Validation, CP, SC, ID, GWC; Writing—original draft, SC, CP, ID; Writing—review and editing, SC, LD, GR, ID, CP, GWC. All authors have read and approved the published version of the manuscript.
Availability of data and materials
The datasets analyzed in this study are not publicly available and cannot be provided by the corresponding author upon request, as they are owned by the Italian regional/local health authorities and cannot be shared by Fondazione ReS.
Consent for publication
Not applicable.
Declaration of conflicting interests
LD, GR, SC, ID, LeonardoD, CP, and NM are employees of Fondazione ReS and report no competing interests with any financial organization regarding the material discussed in the manuscript.
AA and IE are employees of Drugs and Health Srl and report no competing interests with any financial organization regarding the material discussed in the manuscript.
GWC has received research grants, lecture fees, or advisory board fees from Menarini, Allergy Therapeutics, AstraZeneca, Chiesi Farmaceutici, Faes, Firma, Genentech, Guidotti-Malesci, GlaxoSmithKline, Hal Allergy, Innovacaremd, Novartis, OmPharma, RedMaple, Sanofi-Aventis/Sanofi-Genzyme, Stallergenes-Greer, and Uriach Pharma outside the submitted work.
Ethics approval and consent to participate
This retrospective observational study used anonymized Italian administrative data provided in an aggregated form according to agreements with the regional/local health authorities, owners of the data, and the European privacy regulations. Ethical approval and consent to participate were therefore not applicable.
Funding
This report received unconditional funding from AstraZeneca S.p.A. The funding agreement ensured maintenance of author independence in study design, data interpretation, writing, and publication decisions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
