Abstract
Objective
Postoperative pain is one of the most common complications after anorectal surgery and can delay recovery, lengthen hospital stay, and reduce patient comfort. Non-steroidal anti-inflammatory drugs are widely used due to their strong analgesic and opioid-sparing effects but are associated with gastrointestinal, renal, and bleeding risks. Magnesium may provide analgesic benefits with fewer adverse effects. However, evidence regarding its efficacy in the oral form and direct comparison with non-steroidal anti-inflammatory drugs remains limited. In this study, we aimed to compare the analgesic effectiveness of oral magnesium with that of oral ketorolac to identify the more suitable analgesic drug in these patients.
Methods
In this double-blind, randomized controlled trial, 104 patients undergoing anorectal surgery were randomly divided into 2 groups. Group 1 received oral magnesium (250 mg daily), and Group 2 received oral ketorolac (10 mg daily). The medicine was given to the patients 2 h after the operation and then every 12 h for 10 days. Pain levels were measured at 24-hour intervals after the surgery using the visual analog scale.
Results
Both treatments significantly reduced postoperative pain over time (p < 0.001). The ketorolac group showed lower mean pain scores on days 1, 3, and 5 (p < 0.001), whereas no significant differences were observed on postoperative days 7 and 10 (p = 0.089 and 0.092, respectively). Narcotic consumption was higher in the magnesium group than in the ketorolac group (p < 0.001).
Conclusions
Oral magnesium demonstrated a clinically meaningful analgesic effect comparable to that of oral ketorolac from postoperative day 5 onward, suggesting that it is a safe non-opioid alternative for postoperative pain management in anorectal surgery. Further multicenter trials with larger samples are warranted to confirm these findings.
Introduction
Benign anorectal diseases, including anal pain, rectal bleeding, fissures, fistulas, and hemorrhoids, are common complaints in patients referred to clinics. In the United States, on the basis of screening colonoscopy, a prevalence of 4%–8% has been reported for hemorrhoids. It has been reported that cryptoglandular infections cause 80% of anorectal fistulas, and the main treatment for these benign anorectal diseases is surgery.1,2 Nonsurgical treatments include bowel management programs, medications, and perineal exercises that aim to normalize the patient’s bowel pattern. Patients with more severe diseases may require surgeries, such as hemorrhoidectomy, fistulotomy, or sphincteroplasty, to correct abnormalities.2,3
Postoperative pain is one of the most common complications following anorectal surgery and can significantly delay recovery and prolong hospital stay. 2 Non-steroidal anti-inflammatory drugs (NSAIDs) are frequently used because they offer effective pain control and an opioid-sparing effect; however, their gastrointestinal (GI), renal, and bleeding risks limit their routine use in some patients. Ketorolac is an NSAID with analgesic properties that effectively reduces postoperative pain. 4 Ketorolac provides some beneficial effects for patients; it can reduce hospital stay and does not increase the risk of bleeding. 5 However, some NSAIDs are associated with an increased leakage rate in GI anastomotic surgeries, and ketorolac can also increase the risk of pancreatic fistula. 6 Preoperative prescription of ketorolac significantly increases the risk of postoperative pancreatic fistula (POPF) in a dose-dependent manner, whereas a 25-mg increase in the preoperative dose of ketorolac increases the risk of fistula by 10%. 6 Previous research on ketorolac has demonstrated its efficacy in reducing postoperative pain; however, safety concerns remain, especially in GI surgeries.7,8 Conversely, magnesium has emerged as a potential analgesic adjunct through its antagonism of the N-methyl-D-aspartate (NMDA) receptor; however, data on its oral administration and comparative efficacy remain scarce. The NMDA receptor may play a role in chronic neuropathic pain. Moreover, NMDA receptor antagonism can offer an alternative analgesic option for patients who are unresponsive to conventional analgesic drugs.9,10 Magnesium is also a glutamate receptor antagonist that alters the perception and duration of pain. 11 It has been reported that magnesium contributes substantially to reducing central sensitization and attenuating established pain hypersensitivity. 12 Recent clinical studies have investigated magnesium as an adjunct analgesic, primarily via the intravenous (IV) route, showing consistent reductions in postoperative pain scores and opioid requirement with minimal adverse effects. 13 Oral magnesium has also been explored in smaller-scale trials for neuropathic and postoperative pain, suggesting comparable analgesic benefits and excellent tolerability when administered within physiological dose ranges.14,15 Unlike NSAIDs, oral magnesium is not associated with GI bleeding, renal dysfunction, or delayed wound healing, making it a promising non-opioid alternative for pain management. However, evidence on its efficacy in surgical populations remains limited, and data from randomized controlled trials using the oral form are still scarce. 16
The current standard analgesic regimens typically include NSAIDs and opioids, either alone or in combination. Among the routes of administration, the systemic route seems to offer better therapeutic efficacy in pain management. 16 Opioids, such as morphine and pethidine, provide potent analgesia but are associated with well-known adverse effects including nausea, constipation, urinary retention, and risk of dependence.17,18 Additionally, some anticonvulsants and antidepressant drugs aid in pain management.19,20 The pattern of analgesic medications used varies among surgeons, and opioids are the treatment of choice for many surgeons. 21 However, the currently used analgesic drugs do not provide consistent outcomes in all patients; in some patients, they are associated with inappropriate responses. Therefore, there is a need to develop more effective treatment options to fulfill the unmet therapeutic needs in challenging cases.
In this study, we aimed to compare the analgesic effectiveness of oral magnesium with that of oral ketorolac to choose the appropriate analgesic drug for perioperative pain management in terms of patients’ pain, comfort, and normal defecation during anorectal surgery. Ketorolac was selected as the comparator because it represents the current standard non-opioid analgesic used for postoperative pain management following anorectal and other soft-tissue surgeries. It provides potent analgesic effects and reduces opioid requirement without causing sedation, making it a reliable benchmark for evaluating novel agents. However, its use may be limited by GI irritation, bleeding risk, and potential renal dysfunction, particularly with prolonged administration. In contrast, magnesium offers analgesic properties through NMDA receptor antagonism and modulation of central sensitization and has a more favorable safety profile. Although IV magnesium has been investigated in several perioperative trials, data on the efficacy of oral magnesium remain scarce. Therefore, the present randomized double-blind trial was designed to fill this evidence gap by directly comparing the analgesic effectiveness and safety of oral magnesium versus oral ketorolac in patients undergoing anorectal surgery.
Methods
This study was a double-blind, randomized controlled trial investigating the effectiveness of oral magnesium with that of oral ketorolac on pain during anorectal surgery. The study was approved by the Ethics Committee of Qom University of Medical Sciences (ethics code: IR.MUQ.REC.1399.184). The date of first registration for this study was 18 November 2020, with registration number IRCT20120118008769N3. The study was conducted from 6 October 2020 with follow-up completed on 6 January 2021. This study has been reported in line with the Consolidated Standards of Reporting Trials (CONSORT) criteria and has been conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. 22 The research population included all patients who were anorectal surgery candidates and were referred to our tertiary center. After assigning the ethics code, patients who provided informed consent to participate in the study were enrolled and randomly divided into two groups; both patients and the person evaluating the outcomes were unaware of the type of treatment allocation. The research sample included 104 candidates for anorectal surgery who were admitted to the surgical department of Shahid Beheshti Hospital, Qom City. After the researcher explained the purpose and implementation of the project, the consenting patients signed a consent form and were enrolled in the study. Written informed consent was obtained from the patients included in this study.
Eligibility
The inclusion criteria were as follows: patients who were candidates for surgery involving fissures, fistulas, or hemorrhoids and were aged ≥11 years. The exclusion criteria were as follows: age <11 years, malignancy, inflammatory bowel disease, complex fistulas, chronic renal dysfunction or history of peptic ulcers, preanalytical sample collection errors, use of other painkillers in addition to the given drugs, history of drug sensitivity, and manifestations of any drug adverse effects, such as renal dysfunction or other symptoms. In total, 110 patients were initially screened for eligibility. Of these, six were excluded before randomization, including four who did not meet the inclusion criteria (two with chronic renal disease, one with active peptic ulcer, and one who had recently taken analgesics) and two who declined study participation. The remaining 104 eligible patients were randomized equally into two groups (52 each in the magnesium and ketorolac groups) (Figure 1).

Flowchart of the study participants.
Procedure and follow-up
After the patients were selected, they were randomly divided into two groups: group 1 (n = 52) received oral magnesium (125 mg every 12 h), and group 2 (n = 52) received oral ketorolac (10 mg daily). Both medications were prepared by the hospital pharmacy under the supervision of the research team. To maintain blinding, magnesium and ketorolac tablets were encapsulated in identical gelatin capsules and packaged in identical unlabeled containers, coded as “A” or “B.” The capsules were identical in size, color, and taste, ensuring that neither the patients nor the clinical staff could distinguish between treatments. Each patient received medications and were blinded to group allocations. The randomization codes were kept by a third party not involved in the study who alone had access to the allocation list. The medicine was given to the patients 2 h after the operation and then every 12 h for 10 days. The selected doses were based on prior pharmacokinetic and clinical evidence: 250 mg/day oral magnesium has been shown to maintain normal serum magnesium levels while providing NMDA receptor antagonism and analgesic benefit without toxicity. 23 Moreover, 10 mg/day oral ketorolac represents the lowest effective dose for short-term postoperative pain control with reduced GI and renal risk. 7 All procedures were performed according to a standardized anesthesia protocol consisting of induction with propofol (2 mg/kg) and fentanyl (2 µg/kg), followed by maintenance with isoflurane in 50% oxygen/nitrous oxide. No intraoperative magnesium or ketorolac was administered in addition to the assigned study medication. Postoperatively, all patients received routine care, including IV fluids and local wound management. Breakthrough pain was managed with 25 mg intramuscular pethidine as the rescue analgesic, which was given upon patient request if the visual analog scale (VAS) pain score exceeded 4. The same criteria and dosing were applied to both groups to ensure comparability. Two patients in the ketorolac group left the study because they were unable to continue participation. Finally, 52 and 50 patients were analyzed in the magnesium and ketorolac groups, respectively. A flowchart of the study participants is shown in Figure 1.
Variables and measurements
In the present study, age, sex, treatment group, narcotic use, and pain level were the variables. Pain levels were measured at 24-hour intervals postoperatively on days 3, 5, 7, and 10. Postoperative pain intensity was evaluated using the VAS, 24 with scores ranging from 0 (no pain) to 10 (worst pain). The VAS was designated as the primary assessment tool to verify the consistency of patient-reported pain. VAS scores were recorded on postoperative days 1, 3, 5, 7, and 10 by a single trained nurse who was blinded to treatment allocation. In this study, we also investigated the effects of age and sex as independent variables on the frequency of pain in patients. Additionally, the number of narcotics received was recorded in the patient file and measured separately in both groups. Furthermore, the effects of oral magnesium with oral ketorolac on pain after anorectal surgery in patients were determined and compared. Moreover, for patients who used other painkillers, the names of the consumed drugs were recorded.
Randomization
Participants were randomly assigned to two equal groups using permuted block randomization to ensure balanced group sizes throughout the trial. Four blocks containing codes “A” and “B” were initially prepared, and the sequence of each block was randomly determined using dice. This process was repeated 26 times for the assignment of all 104 participants. Final allocation of the treatment codes to the magnesium and ketorolac groups was also performed randomly using the coin toss method. 25 The randomization procedure was performed by a research assistant who was not involved in patient care or outcome assessment. The surgeons and investigators were blinded to group allocation and avoided any actions or discussions with patients or nurses that could reveal the type of medications used. All possible blocks were considered and assigned a corresponding number during the random allocation process.
At each stage, a sequence of 1s or 2s was generated using R software, and the corresponding block was selected and added sequentially. Participants were allocated to each group. The design analyzer generated a randomized sequence, and nontransparent envelopes were used for the random allocation process, which was overseen by an individual not involved in the research. The participants remained in the same group throughout the study based on their randomized allocation.
Sample size
The sample size was initially calculated using previously reported mean and SD values for pain scores (intervention mean =1.28, SD = 1.43; control mean = 2.06, SD =1.48), with α = 0.05 and target power =0.90. 26 Based on these parameters, the calculated sample was 52 per group (total n = 104). We did not apply any additional formal inflation to manage dropouts in the original calculation. Two participants in the ketorolac group subsequently refused to receive the allocated intervention, resulting in 52 and 50 participants in the magnesium and ketorolac groups, respectively (total n = 102). The minimum required sample was calculated as 52 for each group, and a total of 104 patients were included in the study.
Monitoring for adverse events
All participants were monitored daily for potential adverse events throughout the 7-day intervention period. Patients were specifically asked about GI symptoms (nausea, vomiting, abdominal discomfort, diarrhea, and constipation), dizziness, and headache during each follow-up. Any reported event was recorded; graded as mild, moderate, or severe; and assessed for its possible relationship to the study medication. Serious adverse events were defined as any event requiring hospitalization or treatment discontinuation.
Statistical analyses
The data were entered into the Statistical Package for Social Sciences (SPSS) software for Windows, version 26 (IBM Corp., Armonk, NY, USA), and descriptive indices and parametric tests such as independent t tests were used to evaluate differences in the mean scores between the two groups. Sensitivity analysis was also performed after excluding patients with a history of opioid use to evaluate the robustness of the primary outcomes. Additionally, the repeated measures (RM) analysis of variance (ANOVA) test was used to perform linear regression analysis. Given the observed effect size used for the sample size calculation (Cohen’s d ≈ 0.54), a post-hoc power calculation indicated that the analyzed sample (n1 = 52, n2 = 50) yielded an approximate power of 76.5% to detect the prespecified difference with a two-sided α of 0.05. p < 0.05 was considered to indicate statistical significance.
Results
Demographic characteristics
The study included a total of 104 patients, 52 of whom were given magnesium and 52 of whom were given ketorolac. Two patients from the ketorolac group withdrew their consent for study participation and therefore did not receive the assigned intervention (Figure 1). No serious adverse effects were documented during treatment with ketorolac or magnesium. Mild GI symptoms were the most commonly reported adverse effects and included nausea (magnesium group: 3.8% and ketorolac group: 4.9%), abdominal discomfort (magnesium group: 2.9% and ketorolac group: 2.9%), and constipation (magnesium group: 1.9% and ketorolac group: 3.9%). Dizziness was reported by one patient from each group. All symptoms were self-limiting and required no additional treatment. There were no statistically significant differences in the incidence of any adverse event between the two groups (p > 0.05). The study revealed that the magnesium group had a mean age of 50.10 ± 8.74 years (range: 37–67 years), whereas the ketorolac group had a mean age of 46.88 ± 6.82 years (range: 35–66 years). However, the difference in the ages between the two groups was statistically insignificant (p = 0.510). Sex distribution was almost equal in both groups, with 50% women and 50% men in the magnesium group and 48% women and 52% men in the ketorolac group. This difference was not statistically significant (p = 0.840). However, the proportion of patients receiving narcotics was significantly higher in the magnesium group than in the ketorolac group (34.6%, n = 18 vs. 2%, n = 1; p < 0.001). The demographic data for the study groups are presented in Table 1.
Baseline demographic and clinical characteristics of participants in the magnesium and ketorolac groups.
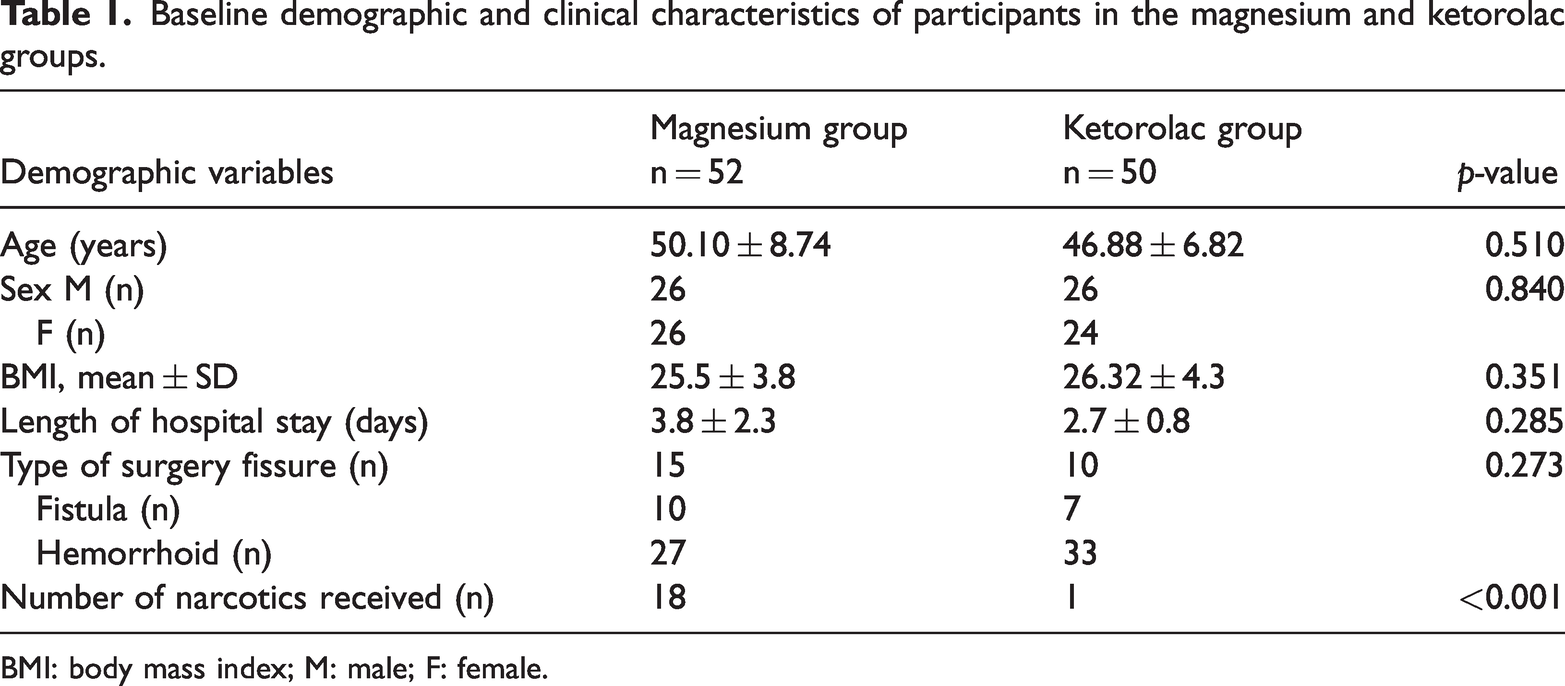
BMI: body mass index; M: male; F: female.
Pain severity on postoperative day 1
On postoperative day 1, approximately 52% (n = 27) of the patients in the magnesium group experienced severe pain, approximately 31% (n = 16) experienced moderate pain, and the remaining patients experienced mild or no pain. Among those in the ketorolac group, 14% (n = 7) experienced severe pain, 66% (n = 33) experienced moderate pain, and the remaining patients experienced mild or no pain, representing a statistically significant difference (p < 0.001).
Pain severity on postoperative day 3
On postoperative day 3, approximately 36% (n = 19) of the patients in the magnesium group experienced severe pain, approximately 46% (n = 24) experienced moderate pain, and the remaining patients experienced mild or no pain. In the ketorolac group, no patient experienced severe pain, approximately 32% (n = 16) experienced moderate pain, and the remaining patients experienced mild or no pain, representing a statistically significant difference (p < 0.001).
Pain severity on postoperative day 5
On postoperative day 5, approximately 13% (n = 7) of the patients in the magnesium group experienced severe pain, 48% (n = 25) experienced moderate pain, and the remaining patients experienced mild or no pain. In the ketorolac group, no patient experienced severe pain, 24% (n = 12) experienced moderate pain, 68% (n = 34) experienced mild pain, and the remaining patients experienced no pain, representing a statistically significant difference (p < 0.001).
Pain severity on postoperative day 7
On postoperative day 7, approximately 4% (n = 2) of the patients in the magnesium group reported severe pain, approximately 36% (n = 19) reported moderate pain, and approximately 56% (n = 29) reported mild pain. In the ketorolac group, no patient reported severe or moderate pain, 70% (n = 35) reported mild pain, and 30% (n = 15) reported no pain. The difference was not statistically significant (p = 0.089).
Pain severity on postoperative day 10
On postoperative day 10, approximately 2% (n = 1) of the patients in the magnesium group reported severe pain, approximately 7% (n = 4) reported moderate pain, and 83% (n = 43) reported mild pain. In contrast, among those in the ketorolac group, no patient reported severe or moderate pain, 44% (n = 22) reported mild pain, and 56% (n = 28) reported no pain; this difference was not statistically significant (p = 0.092).
Pain frequency
The pain frequency was recorded for patients in both treatment groups who underwent anorectal surgery and received oral magnesium or ketorolac. When patients with prior opioid use (n = 18 in the magnesium group and n = 1 in the ketorolac group) were excluded, the differences in the postoperative pain scores between the two groups remained statistically significant (p < 0.05). The mean VAS scores in the magnesium group were 6.08 ± 2.57, 5.54 ± 2.18, 4.33 ± 2.05, 2.94 ± 1.84, and 1.69 ± 1.44 on postoperative days 1, 3, 5, 7, and 10, respectively. In the ketorolac group, the corresponding mean VAS scores were 4.74 ± 1.63, 4.10 ± 1.48, 2.46 ± 1.38, 1.20 ± 1.03, and 0.44 ± 0.50. The VAS scores (mean ± SD) of each group are summarized in Table 2. The average pain score of the magnesium group on postoperative day 1 was 1.3 times higher than that of the ketorolac group, representing a statistically significant difference (p < 0.001). In this study, age and sex were two independent variables that affected the frequency of pain in patients. Pain frequency was lower in men than in women, and in both groups, it decreased with increasing age. On postoperative day 1, the pain score decreased by an average of 0.07 units for each 1-year increase in age, indicating that pain levels decrease with age as an independent factor. The average pain score on postoperative day 3 in the magnesium group was approximately 1.4 times higher than that in the ketorolac group, representing a statistically significant difference (p < 0.001). On postoperative day 3, the pain score decreased by an average of 0.05 units for each 1-year increase in age. The average pain score on postoperative day 5 in the magnesium group was approximately 2 times higher than that in the ketorolac group, and this difference was statistically significant (p < 0.001). On postoperative day 5, the pain score decreased by an average of 0.05 units for every 1-year increase in age. On postoperative day 7, the average pain score in the magnesium group was 2.5 times higher than that in the ketorolac group, although this difference did not reach statistical significance (p = 0.093). On postoperative day 7, the pain score decreased by an average of 0.02 units for every 1-year increase in age. The average pain score of the magnesium group on postoperative day 10 was 3.8 times higher than that in the ketorolac group; however, this difference was not statistically significant (p = 0.088). On postoperative day 10, the pain score decreased by an average of 0.01 units for every 1-year increase in age.
The average VAS scores for patients in both treatment groups who underwent anorectal surgery.

VAS: visual analog scale.
Discussion
Various medications have been used to relieve suffering, facilitate function, enhance recovery, and satisfy patients who undergo anorectal surgery. 27 Some studies have used non-opioid analgesic drugs to control postoperative pain and minimize opioid-related adverse effects. In a clinical trial, De Oliveiraet al. 4 aimed to optimize the dose and route of administration for systemic single-dose ketorolac for postoperative pain prevention. The findings revealed that the use of single-dose systemic ketorolac can reduce not only postoperative pain but also postoperative nausea and vomiting. The 60-mg dose achieved significant reduction in postoperative pain, while no such effect was observed with the 30-mg dose. The use of ketorolac in the early postoperative period can lead to the development of biochemical (grade A) fistulae after pancreaticoduodenectomy (PD).
In a retrospective study, Kowalsky et al. 6 reported an association between ketorolac use and POPF after PD in a dose-dependent manner. Sueda et al. 28 reported a significant association between clinically relevant POPF and ketorolac use in 464 patients who underwent pancreatectomy. In addition, ketorolac has been associated with renal dysfunction when used for postoperative pain prevention across multiple clinical settings.29–31 Torabi et al. 32 did not find an increase in the incidence of acute kidney diseases in trauma patients who underwent ketorolac for acute pain management. De Oliveira et al. 16 investigated the effect of the systemic administration of perioperative magnesium to mitigate postoperative pain in surgical patients. They enrolled 1257 participants in the study and demonstrated that magnesium can control excessive pain during states of rest and activity. These data from more than 1200 patients receiving magnesium proved that the continuous use of prescribed magnesium reduces postoperative pain, and the use of narcotics reduces pain. Postoperative defecation pain is a distinctive complication in patients who undergo hemorrhoidectomy. 33 Yano 34 proposed that harder stools would exert additional physical impact on the wound, thereby causing more pain. As one of the practices recommended by surgeons is the administration of laxatives,33,35 the laxative effects of magnesium may aid postoperative pain prevention in patients who undergo anorectal surgery.
The results of this study demonstrate that despite the analgesic properties of magnesium and the common adverse effects of ketorolac, magnesium can be useful in surgical patients. The analgesic effects of magnesium are primarily attributed to its antagonism of the NMDA receptor and modulation of voltage-gated calcium channels. By reducing calcium influx into neuronal cells, magnesium decreases central sensitization and neuronal excitability within the dorsal horn, thereby attenuating the amplification of nociceptive transmission. This mechanism differs from that of ketorolac, which act peripherally through cyclooxygenase inhibition and prostaglandin suppression. The gradual onset and sustained nature of analgesia observed with magnesium in this study may reflect its central, modulatory mechanism rather than a rapid anti-inflammatory effect. Additionally, magnesium exerts a mild relaxant effect on intestinal smooth muscle and may enhance bowel motility, thus benefiting patients after anorectal surgery by reducing postoperative discomfort related to sphincter spasm or delayed defecation. These pharmacologic properties could explain the prolonged yet well-tolerated analgesic profile observed over the postoperative course.15,36–38
In this study, we aimed to compare the effects of oral magnesium with those of oral ketorolac on pain reduction after anorectal surgery. In total, 104 candidates for anorectal surgery who met the inclusion criteria were enrolled in the study via randomization and double blinding. Ketorolac and magnesium tablets were used, and postoperative pain was evaluated in both groups according to the VAS criteria. Findings revealed reduced postoperative pain in patients receiving magnesium. Pain reduction was also observed in the ketorolac group, and this reduction was more than that in the magnesium group. Moreover, the pain intensity in the ketorolac group was lower than that in the magnesium group on all treatment days. The number of narcotics received by patients in both magnesium and ketorolac groups was monitored, and the value was higher in the magnesium group. Notably, the difference in the dose of narcotics received between the two groups was statistically significant. The difference in pain intensity between the magnesium and ketorolac groups was statistically significant on postoperative days 1, 3, and 5 but not on postoperative days 7 and 10. The mean differences in the VAS scores (≈1.2–1.5) were comparable to the minimal clinically important difference reported for postoperative pain (≥1.5), indicating potential clinical relevance in addition to statistical significance. The analgesic effect of oral magnesium tablets in patients undergoing anorectal surgery may be similar to that of oral ketorolac tablets and can be used from postoperative day 5 onward. Additionally, magnesium tablets can be administered from postoperative days 1 to 5, accompanied with another narcotic instead of ketorolac, to prevent adverse effects, such as leakage in the GI anastomosis or increased risk of pancreatic fistula. Our results add to a growing body of evidence stating that magnesium, traditionally studied via IV administration, has clinically relevant analgesic properties in the perioperative setting. Systematic reviews of perioperative systemic magnesium report reductions in postoperative pain and opioid consumption across a range of surgical procedures. Smaller randomized trials of oral magnesium in dental and minor surgical models have also suggested analgesic benefit and favorable tolerability.13,14 Our trial extends these data by providing randomized, double-blind evidence in an anorectal surgery population using a simple, once-daily oral regimen. Together, these findings suggest that oral magnesium can be considered a feasible adjunct for postoperative analgesia when parenteral routes are impractical or when clinicians prefer an oral, low-risk agent.13,14,16 Based on the balance of efficacy and safety observed here and in previous reports, oral magnesium may be particularly useful for the following patients: (a) those who are undergoing minor outpatient procedures such as anorectal surgery where prolonged IV access is not intended; (b) those who have relative contraindications to NSAIDs (history of peptic ulcer disease, increased bleeding risk, or marginal renal function); or (c) those who would benefit from opioid-sparing strategies but for whom routine opioids are undesirable. Importantly, because our data show a stronger early analgesic effect for ketorolac but comparable pain control during the later postoperative period, a practical clinical strategy might be to use short-term NSAIDs for immediate postoperative control when safe or use oral magnesium as an alternative or adjunct in patients at higher risk of NSAIDs or in cases where clinicians prefer to avoid NSAIDs altogether.
The absence of a control group is a major limitation of this study, as comparing the effectiveness of magnesium and ketorolac without a placebo or no-treatment control group makes it difficult to attribute the observed effects solely to the treatments. However, owing to the possibility of postoperative pain, the allocation of patients to the control group is not allowed in line with medical ethics. The reduction from the planned 104 to 102 participants reduced the power by <1%. Thus, although two participants did not complete the intervention, this minor loss had a negligible effect on the statistical power and did not alter the overall study conclusions. In relevant studies, magnesium absorption depends on the type of magnesium salt and other food elements that may augment or abate it. An imbalance in baseline opioid exposure was noted, with a higher proportion of prior opioid users in the magnesium group. This may have theoretically influenced pain perception or analgesic response. However, a sensitivity analysis excluding these participants did not substantially alter the results, suggesting that this imbalance had minimal impact on the overall findings. Future trials should consider randomization stratified by prior opioid use to avoid this confounding factor. This study also has a relatively small sample size and single-center design, which may restrict the generalizability of the findings and reduce the statistical power, limiting its ability to detect smaller differences. Future multicenter trials with larger sample sizes are warranted to validate these results and enhance their validity. Moreover, future studies with larger and more heterogeneous samples should investigate potential demographic moderators (e.g. sex or age) of the analgesic outcomes to oral magnesium and oral ketorolac.
Conclusion
It is crucial for practitioners to possess current knowledge of pain management and its associated adverse effects in patients who have undergone anorectal surgery. In this randomized, double-blind clinical trial, both oral magnesium and oral ketorolac effectively reduced postoperative pain following anorectal surgery. Although the early analgesic response was more pronounced with ketorolac, pain intensity in the magnesium group declined steadily and reached comparable levels by the end of the first postoperative week. Magnesium was well tolerated, with no serious adverse events observed throughout the study. These findings suggest that oral magnesium is a safe and practical non-opioid adjunct for postoperative analgesia, particularly in patients for whom NSAIDs are contraindicated or poorly tolerated. Its distinct central mechanism of action and favorable GI safety profile further support its use as part of a multimodal pain management strategy in anorectal procedures.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251409937 - Supplemental material for Comparison of oral ketorolac and oral magnesium for postoperative pain management in anorectal surgery: A randomized double-blind clinical trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605251409937 for Comparison of oral ketorolac and oral magnesium for postoperative pain management in anorectal surgery: A randomized double-blind clinical trial by Seyed Jalal Eshagh Hoseini, Farzaneh Sadat Ghazi, Mohsen Eshraghi, Mostafa Vahedian, Mohammad Reza Pashaei, Mojdeh Bahadorzadeh and Sajjad Ahmadpour: For The Alzheimer’s Disease Neuroimaging Initiative in Journal of International Medical Research
Footnotes
Acknowledgment
The authors would like to thank Urmia University of Medical Sciences for their support.
Authors’ contributions
SE, FG, and ME wrote the manuscript; MV and MP performed the procedures and managed the patients; MH reviewed and revised the final manuscript; MB and SA are the corresponding authors. All the authors have read and approved the final manuscript.
Availability of data and materials
The datasets used in the current study are available from the corresponding author upon reasonable request. All the material is owned by the authors, and/or no permissions are needed.
Consent for publication
The patients provided consent for the publication of this study.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Qom University of Medical Sciences (ethics code: IR.MUQ.REC.1399.184) and registered at the Center for Clinical Trials (No. IRCT20120118008769N3). We adhered to the Helsinki ethical statement in all stages of the study (Declaration of Helsinki 1975, as revised in 2024). The clinical trial registration is available at ![]() . Written informed consent was obtained from the patients for the publication of this article.
. Written informed consent was obtained from the patients for the publication of this article.
Funding
The authors have not received any funding for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
