Abstract
Objective
The purpose of this study was to investigate the relationship between intraoperative facial nerve monitoring parameters and postoperative facial nerve function during parotid surgeries.
Methods
Registered in PROSPERO, a comprehensive search was conducted in PubMed, Embase, Web of Science, and the Cochrane Library for literature published between 1990 and 2024. Studies reporting the relationship between intraoperative facial nerve monitoring parameters and facial nerve outcomes following parotidectomy were included. Owing to the limited number of studies and heterogeneity, the findings were summarized using a narrative synthesis.
Results
A total of 8 studies, involving 632 participants, were included, among whom 193 developed postoperative facial palsy. Five studies reported that a reduction in amplitude or an increase in threshold was associated with postoperative facial dysfunction.
Conclusion
Current evidence does not provide conclusive support for the predictive value of intraoperative facial nerve monitoring parameters in postoperative facial nerve function. However, given its established protective role and some reported findings (e.g. >50% amplitude drop associated with severe palsy), continued use of intraoperative facial nerve monitoring appears reasonable.
Keywords
Introduction
Facial nerve preservation is a principal concern during parotidectomy because of its anatomical proximity to the parotid gland. Injury to the facial nerve can result in profound functional impairment, including incomplete eye closure, distortion of the commissure, and loss of natural facial expressions, leading to psychosocial morbidity and reduced quality of life. Multiple factors have been associated with an increased risk of nerve injury during parotidectomy, including deep and large tumors, extensive intraoperative hemorrhage, prolonged surgery duration, and the absence of intraoperative facial nerve monitoring (IOFNM).1–4
To minimize the risk of nerve injury, IOFNM, originally introduced in skull base procedures such as acoustic neuroma resection, has become a routine part of parotid surgery for both benign and malignant tumors over the past decades. 5 It provides real-time feedback on the facial nerve by recording electromyography (EMG) signals from facial muscles in response to nerve stimulation, which allows surgeons to locate the nerve and assess its functional integrity.
The use of IOFNM has increased substantially worldwide. According to a study conducted in the United States in 2005, approximately 60% of practicing otolaryngologists utilized IOFNM for parotid surgery. 6 More recent surveys suggest even higher adoption rates in some European countries, with up to 94% of surgeons in Spain reporting routine use. 7 However, utilization of IOFNM remains inconsistent globally, with a Chinese survey demonstrating its application in only about half of otologic procedures. 8
Beyond its role in identifying and protecting the facial nerve in parotid surgery,9,10 IOFNM generates neurophysiological data that may help predict postoperative outcomes. Parameters such as stimulation threshold, EMG amplitude, and response latency provide insights into nerve integrity. For instance, an elevated stimulation threshold or a reduced EMG amplitude may indicate partial nerve injury. 11 In thyroidectomy, IOFNM metrics have been used to predict postoperative recurrent laryngeal nerve dysfunction,12,13 suggesting that similar metrics might help predict facial nerve outcomes in parotidectomy.
Despite the abovementioned potential, there are currently no standardized reference values or cutoff thresholds for interpreting IOFNM parameters in relation to their prognostic value in parotidectomy. Although some studies have reported associations between intraoperative parameters and postoperative facial nerve function, the available evidence remains limited and sometimes inconsistent.
To address this uncertainty, we conducted this systematic review to evaluate the existing literature on the predictive value of IOFNM during parotid surgery. Our aim was to clarify whether parameters such as stimulation thresholds, amplitudes, or latencies can reliably predict postoperative facial function.
Methods
Study design
This systematic review followed the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA) guidelines and was registered in the PROSPERO International Prospective Register of Systematic Reviews (registration number CRD 42022375645). 14 No amendments were made to the original registered protocol. Ethical approval and informed consent were not required because the study is a literature review and does not involve direct patient contact.
Information sources
Two independent reviewers (HC and YZ) searched the PubMed, Web of Science, Embase, and Cochrane Library databases. The literature search was restricted to studies published from 1990 to 2024. This time frame was chosen to minimize the impact of outdated surgical practices on our results, considering the first application of IOFNM in parotid surgery.
Search strategy
The two reviewers independently conducted cross-searches using predefined keywords in each database. A concise string for the PubMed search strategy is as follows: (“intraoperative neurophysiological monitoring”[Mesh] OR “facial nerve monitoring” OR “electromyography” OR “motor evoked potentials” OR “electrical stimulation”) AND (“parotidectomy” OR “parotid gland”[Mesh] OR “parotid neoplasms”[Mesh] OR “salivary gland surgery”). The detailed search strategy (omitted here for brevity) and other database strategies are provided in the supplementary materials (Supplementary 1).
Eligibility criteria
Studies were included if they met the following criteria:
Studies reporting definitive values of IOFNM parameters, including amplitude, latency, or stimulation threshold; Studies reporting outcomes in patients with or without facial palsy; Studies investigating the relationship between IOFNM parameters and postoperative facial palsy. Studies including surgeries such as revision parotidectomy or bilateral parotidectomy (bilateral cases were excluded because compensatory mechanisms may affect facial nerve function, making direct comparisons challenging); Studies including populations under 18 years of age; Case reports or abstracts with insufficient data for inclusion; Studies without available English translation.
Studies were excluded if they met any of the following criteria:
Study selection
The articles identified in the initial search underwent a two-stage screening process. In the first stage, titles and abstracts were reviewed, duplicates were removed, and studies not meeting the inclusion criteria were excluded. In the second stage, full-text articles were obtained and assessed. Eligibility was independently evaluated by the two reviewers, and any disagreements were resolved through discussion.
Data collection
Data were extracted into purpose-built data sheets by the first author (HC) and checked by the second author (YZ). Additional information from the study authors was not required. Data collected included first author, year of publication, country, inclusion and exclusion criteria, type of parotid surgery, sample size, method of facial nerve function evaluation, involved IOFNM parameters and their relative cutoff values, and study design and conclusions.
Quality appraisal
The quality of the selected studies was independently assessed by two reviewers using an adapted version of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 15 All included studies were scored according to 11 specific criteria derived from items 5, 6, 7, 8, 10, 12, 13, and 15 of the original checklist. For each criterion, a positive score (√) was assigned when the requirement was met and a negative score (×) was assigned when it was not met (Supplementary 2). Based on the total score, studies were classified as having a low risk of bias (8–11 points), moderate risk (4–7 points), or high risk (≤3 points).
Bias assessment
Statistical assessments for reporting bias, such as funnel plot asymmetry analysis, were not performed because fewer than 10 studies were included and a meta-analysis was not conducted. Nevertheless, potential publication bias cannot be excluded, and the results should be interpreted with caution.
Synthesis of results
The primary outcome was facial nerve function, evaluated using the House–Brackmann (HB) facial paralysis grading scale or other facial grading systems. Differences were evaluated pre- and post-parotidectomy in patients presenting with facial palsy. A narrative analysis was used to summarize the characteristics and outcomes of the included studies.
Certainty assessment
The certainty of evidence for each outcome was assessed using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach, adapted for narrative synthesis without pooled effect estimates. For each outcome, study limitations (risk of bias), inconsistency, indirectness, imprecision, and potential publication bias were considered.
Results
Search and study selection
Four databases, namely, PubMed, Embase, Web of Science, and the Cochrane Library, were searched, yielding 1738 results. After removing duplicates, 1057 studies remained. Screening of titles and abstracts led to the exclusion of 983 studies that did not meet the inclusion criteria, leaving 74 studies for full-text review. Of these, 66 were further excluded for various reasons, as outlined in Figure 1 using the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) flowchart. 16 Overall, eight studies were included.5,17–23
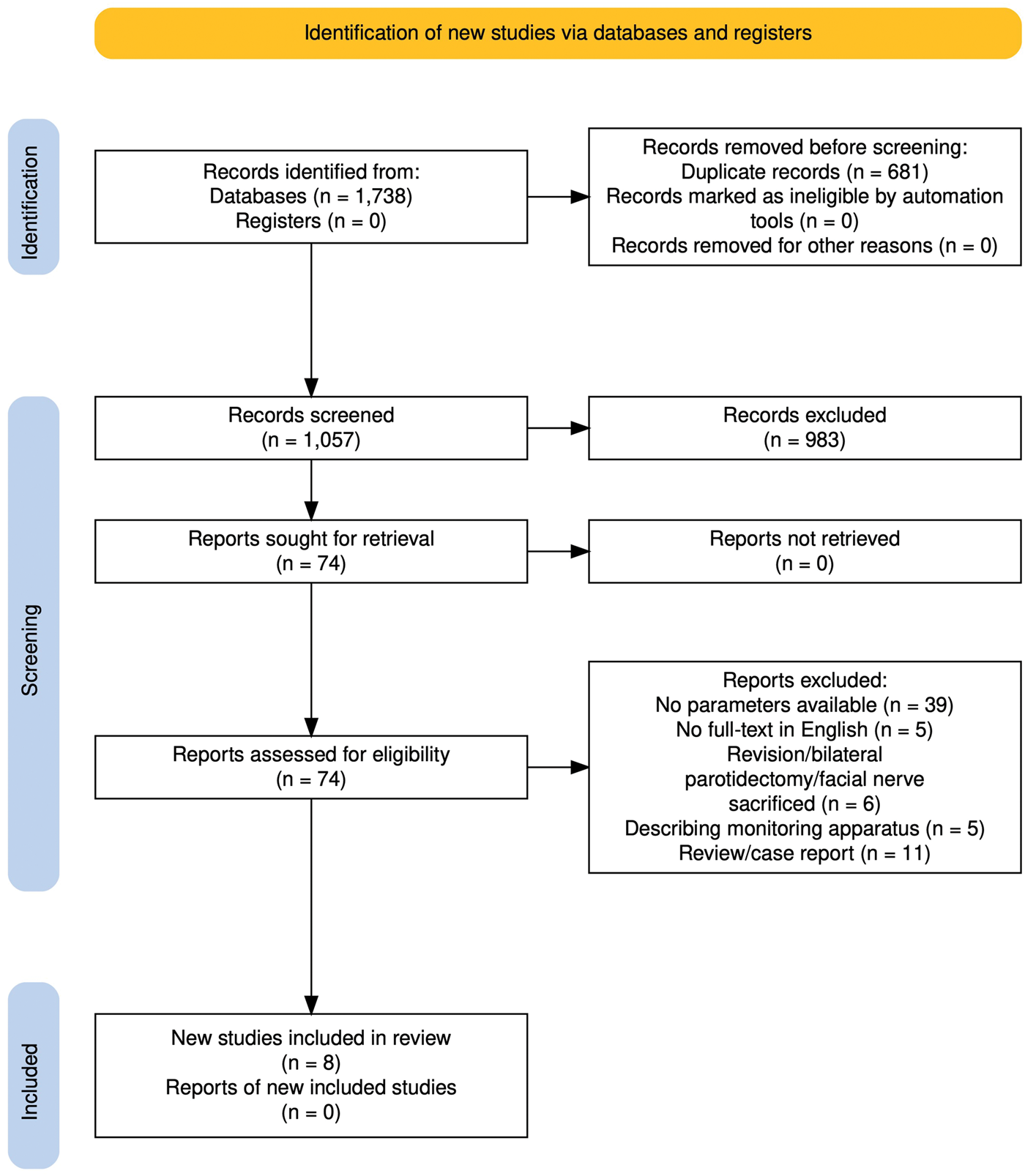
Flow chart of workflow.
Study characteristics
All the included studies were observational in nature. Of the eight studies, two were retrospective, and six were prospective. Study characteristics are summarized in Table 1. A total of 632 participants were included, with sample sizes ranging from 25 to 222 per study. Facial nerve weakness was observed in 193 of the 632 patients.
Characteristics of included studies.

HB: House–Brackmann; ARR: amplitude response ratio; mA: milliampere.
Quality analysis
All studies were assessed using the adapted STROBE guidelines for observational studies (Supplementary 2). 15 None of the included studies were classified as having a high risk of bias. Six studies were rated as having a low risk of bias, and two as having a moderate risk. The studies identified as having a low risk of bias18–23 received a score of 9, 9, 9, 9, 9, and 8, respectively. The study with the highest risk of bias 5 scored 5 (moderate risk). Regarding the evaluation of variables (i.e. distinct IOFNM parameters), evaluation of facial nerve function, and reporting of results, all studies described their methodology. However, none of the studies explained how the sample size was determined, reported the number of participants with missing data, or described how missing data were addressed. The presence of significant clinical heterogeneity among the included studies rendered meta-analysis unsuitable.
Study design
Despite variations in research design, the eight included studies investigated the correlation between IOFNM results and postoperative facial nerve function using two primary approaches. The first approach compared intraoperative parameters between patients who developed facial palsy postoperatively and those with normal facial nerve function. The second approach assessed changes in parameters before and after parotidectomy in patients with facial palsy to determine cutoff values.
Certainty of evidence
The certainty of evidence for the association between an amplitude reduction >50% and postoperative facial palsy was rated as low due to concerns about risk of bias and imprecision. For stimulation threshold and latency as predictors, the certainty was rated as very low, owing to serious inconsistency and imprecision across studies. Evidence supporting the amplitude response ratio (ARR) as a predictor was also rated as low certainty, as it was based on a single observational study with a small sample size.
Parameters in IOFNM
IOFNM often records parameters such as stimulation threshold, latency, and amplitude at a given current intensity. Specifically, a parameter known as the ARR was used to predict postoperative facial nerve function. ARR is defined as the ratio of electrical potential in response to trunk stimulation and branch stimulation after tumor resection (trunk/periphery). Several studies further calculated cutoff values and their accuracy, which served as indicators of poor postoperative facial nerve function.
Postoperative facial nerve weakness
The included studies used four distinct evaluation methods, with the HB grading system being the most frequently applied (studies 1 and 5–8), which is also commonly used in clinical practice. The remaining three studies employed alternative approaches: study 4 classified facial nerve function as weak or not weak; study 2 assessed each nerve branch individually; and study 3 graded function into four levels (normal, mild, moderate, and severe) based on dynamic movement of muscle groups across four facial regions (Table 1).
The relationship between parameters and facial nerve weakness
A narrative summary of the evidence regarding the prognostic value of IOFNM is provided in Table 2. Six studies evaluated amplitude. Stanković et al. 21 investigated the effect of continuous IOFNM and found a significant correlation between a drop in amplitude (<50% of the baseline amplitude) and postoperative facial paralysis. Chiang et al. 17 observed an 81.25% incidence of facial palsy (13/16) in patients with postoperative amplitude decreases >50%, compared with no facial palsy in those with no decrease or a decrease of <50%. Mamelle et al. 20 reported higher maximal response amplitudes at 2 mA in patients with normal facial function than in those with poor outcomes. Higashino et al. 22 found that ARR (trunk/periphery) was less than 0.5 in damaged branches and approximately 1 in normal branches. Eren et al. 19 and Ozturk et al. 23 compared the amplitude before and after parotidectomy and facial nerve dissection, respectively, in patients with facial palsy, revealing no significant difference.
GRADE narrative summary table of the evidence regarding the prognostic value of IOFNM in parotid surgery.

IOFNM: intraoperative facial nerve monitoring; ARR: amplitude response ratio; mA: milliampere.
Five studies evaluated the IOFNM threshold. Haring et al. reported that the mean post-dissection threshold was higher in patients with immediate postoperative nerve weakness (0.29 mA) than in those with normal postoperative nerve function (0.2 mA), recommending a cutoff value of 0.25 mA, derived from the receiver operating characteristic (ROC) curve, to predict nerve injury. No significant difference was observed between the facial weakness and normal groups in studies by Mamelle et al. 20 and Brennan et al., 5 who concluded that minimal stimulation level did not predict isolated nerve branch weakness. Eren et al. 19 and Ozturk et al. 23 found no threshold difference before and after parotidectomy in patients with facial weakness.
Two studies comparing latency before and after surgery reported no significant differences.
Three studies further evaluated the predictive value of IOFNM parameters. Higashino et al. 22 established a cutoff value of 0.55 for ARR, yielding sensitivity, specificity, and accuracy rates of 96.5%, 93.1%, and 96.8%, respectively, for the diagnosis of postoperative facial nerve palsy. Stanković et al. 21 reported that a 50% amplitude drop had a sensitivity of 87.5% (area under the curve (AUC): 0.75) and a negative predictive value of 83.3%. Haring identified an optimal post-dissection threshold cutoff of 0.25 mA, defined as the point on the ROC curve that maximized sensitivity and specificity for facial nerve outcome, with a sensitivity of 47%, specificity of 82%, positive likelihood ratio of 2.70, and negative likelihood ratio of 0.64.
Discussion
IOFNM has become a vital component of modern parotid surgery. Most existing research on IOFNM has emphasized its protective role during parotidectomy, particularly compared with traditional methods without IOFNM.9,24 These studies found that the absence of IOFNM increases the risk of facial nerve injury. 25 Moreover, intraoperative amplitude changes can occur due to surgical manipulation itself, without actual impairment of facial nerve function. 26 However, limited attention has been given to the predictive value of intraoperative neurophysiological data for facial nerve outcomes. 3 Accurate prediction not only helps provide patients with informed expectations but can also aid in refining surgical techniques and optimizing perioperative care. 3
In this review, we systematically analyzed the literature regarding the prognostic use of IOFNM in parotid surgery. Eight studies, encompassing a total of 632 patients, were included for detailed evaluation of the relationship between IOFNM parameters and postoperative facial nerve function. Of these, three studies found no association, whereas five reported a potential predictive relationship. However, because of limited data and considerable heterogeneity in assessed parameters, study designs, and outcome reporting, meta-analysis was not feasible. Therefore, current evidence does not support a definite conclusion regarding the prognostic value of IOFNM in parotidectomy.
All included studies assessed at least one IOFNM parameter, such as amplitude, threshold, or latency, and evaluated its correlation with postoperative facial nerve outcomes. Amplitude was analyzed in six studies, threshold in five, and latency in two. Overall, amplitude and threshold appear more promising for prediction.
Amplitude changes were the most commonly investigated parameter and are directly correlated with the number of activated axons.27,28 Theoretically, the amplitude of the facial nerve response is closely related to the degree of nerve damage. A reduction in IOFNM amplitude after facial nerve dissection suggests a decreased number of motor units contributing to polarization. Among the included studies, Stanković et al. 21 and Chiang et al. 17 reported that an intraoperative amplitude reduction of >50% was associated with significant postoperative facial dysfunction. In contrast, Eren et al. 19 and Ozturk et al. 23 found no clear correlation between amplitude and postoperative outcomes. These discrepancies may be explained by differences in stimulation parameters and patient cohorts. For example, Ozturk et al. 23 observed only 4 mild facial paresis cases (HB Grade 2) among 25 patients, with amplitude measured using a fixed 0.5 mA current, whereas Eren et al. 19 reported 5 mild (Grade 2), transient cases among 34 patients using threshold stimulation. Both studies focused on mildly affected patients, limiting the sensitivity for detecting meaningful amplitude–function relationships. In contrast, Stanković et al. 21 included patients with HB Grade 3 dysfunction and stratified them by amplitude drop >50% or <50%, while Chiang et al. 17 and Mamelle et al. 20 also included patients with moderate dysfunction using supramaximal stimulation.
The stimulation threshold of a specific nerve region reflects that region’s response to an incoming impulse. 29 However, the included studies showed inconsistent results. Haring et al. 18 identified a post-dissection stimulation threshold >0.25 mA as predictive of facial nerve injury. In contrast, Mamelle et al. 20 and Brennan et al. 5 did not find significant associations between stimulation thresholds and postoperative outcomes. These discrepancies likely arise from differences in threshold definitions. Haring et al. defined threshold as the minimum current required to evoke a compound muscle action potential (CMAP) in each peripheral facial nerve branch after tumor resection, with a sensitivity window of 50–500 µV. Eren et al. 19 used the lowest current that produced visible muscle contraction or fasciculation, while Mamelle et al. 20 used a fixed threshold of 100 µV. The definition of threshold directly affected its sensitivity and specificity. Furthermore, the absolute post-dissection threshold appears to have greater clinical relevance than the magnitude of intraoperative change. Haring et al. analyzed absolute post-dissection threshold values between patients with normal and impaired postoperative facial nerve function. In contrast, Eren et al. 19 and Mamelle et al. 20 evaluated changes or ratios in stimulation thresholds (e.g. pre- vs. post-dissection or pre- vs. post-resection) and found these measures to be of limited value in predicting postoperative outcomes.
Latency is the time from the onset of the stimulus artifact to the onset of the evoked potential, reflecting the sum of processes from nerve conduction and neuromuscular transmission to the generation of an action potential on muscle fiber membranes. Various technical differences in nerve conduction measurements—such as stimulation site, intensity, and recording electrode placement—can affect the accuracy and comparability of results. 30 The two included studies compared latency changes before and after surgery and found an approximate 1-ms difference.19,23 Therefore, it is not surprising that no direct association between latency and postoperative outcomes was observed, as latency is less sensitive than amplitude and cannot be reliably quantified or used as a standalone prognostic indicator.
IOFNM is routinely employed in acoustic neuroma and skull base surgeries, where its predictive value has been well established.31–33 A meta-analysis summarizing direct electrical stimulation (DES) in vestibular schwannoma surgery showed that a CMAP amplitude below 500 μV is sensitive for predicting poor short-term facial function, whereas minimal stimulation threshold (MST) values of 0.05 mA and 0.10 mA are both sensitive and specific for long-term outcomes. 11 However, because the cerebellopontine segment lacks an epineurium and the distance between the stimulation site and facial muscles is greater, these findings from acoustic neuroma resection cannot be directly applied to parotidectomy.
We attempted a meta-analysis but were unable to proceed due to several limitations. The eight included studies demonstrated significant heterogeneity, as described above. Most lacked control groups, sample size calculation, missing data management, or detailed information regarding the type of parotidectomy, tumor size, location, extent of resection, and facial nerve dissection. More importantly, only four studies provided sufficient data to calculate a cutoff value, making quantitative synthesis unfeasible.
Despite these limitations, this study provides valuable insights into the role of IOFNM in parotid surgery. Current evidence does not conclusively link intraoperative parameters with postoperative facial nerve function, and the predictive value of IOFNM remains uncertain. However, given its protective and potential predictive benefits, routine IOFNM is recommended, with careful attention to the parameters obtained. Despite the heterogeneity of existing studies, a significant intraoperative amplitude drop (e.g. >50%) appears to be more predictive of moderate to severe postoperative facial palsy. This review highlights the need for further investigation into predictive IOFNM parameters.
Conclusion
Current evidence does not conclusively link IOFNM parameters with postoperative facial nerve function. Despite heterogeneity among studies, a significant intraoperative amplitude drop (e.g. >50%) may predict moderate to severe facial palsy. Given its protective and potential predictive benefits, routine IOFNM is recommended, with careful attention to recorded parameters.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251403833 - Supplemental material for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value
Supplemental material, sj-pdf-1-imr-10.1177_03000605251403833 for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value by Yang Zhao, Hanlin Chen, Songbo Xue and Guodong Feng in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251403833 - Supplemental material for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value
Supplemental material, sj-pdf-2-imr-10.1177_03000605251403833 for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value by Yang Zhao, Hanlin Chen, Songbo Xue and Guodong Feng in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251403833 - Supplemental material for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value
Supplemental material, sj-pdf-3-imr-10.1177_03000605251403833 for Intraoperative facial nerve monitoring in parotidectomy: A systematic review of its predictive value by Yang Zhao, Hanlin Chen, Songbo Xue and Guodong Feng in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank Dr Nan Zhao for her valuable guidance on the systematic review.
Author contributions
Y. Zhao and H. Chen designed the study, initiated the collaborative project, and revised the manuscript. H. Chen and G. Feng monitored data collection and drafted the manuscript. H. Chen and S. Xue cleaned and analyzed the data. Y. Zhao and G. Feng provided administrative and technical support.
Availability of data and materials
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
AI tool application
DeepSeek was partially used to assist with language refinement during manuscript preparation; all content was verified by the authors.
Declaration of conflicting interests
The authors declare no conflicts of interest related to this work.
Ethics approval and consent to participate
The need for informed patient consent was waived due to the retrospective nature of the study.
Clinical trial registration number: Not applicable.
PROSPERO registration number: CRD42022375645.
Human Ethics and Consent to Participate declarations: Not required.
Informed Consent: Not required.
Funding
This work was supported by the Natural Science Foundation of Beijing Municipality (No. 7242107).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
