Abstract
Background
Ovarian hyperstimulation syndrome is the most severe and life-threatening iatrogenic complication of controlled ovarian stimulation during assisted reproductive technology procedures. Its diagnosis and severity grading are complex. Despite its debilitating and sometimes fatal consequences, there is a paucity of data on early predictors of ovarian hyperstimulation syndrome.
Objective
This study aimed to identify early predictors of ovarian hyperstimulation syndrome among women undergoing controlled ovarian stimulation for assisted reproductive technology treatment.
Methods
This prospective cohort study included 46 women undergoing assisted reproductive technology treatment from 1 April to 30 November 2023 at Hallmark Medicals, Kumasi, Ghana. Participants were conveniently recruited and received a standard controlled ovarian stimulation protocol. A structured questionnaire was used to collect sociodemographic and clinical data. Approximately 10 mL of venous blood was collected for laboratory analysis of fibrinogen and cytokine markers as well as hepatic and renal function tests. Statistical analyses were performed using R version 4.3.2 and Statistical Package for the Social Sciences version 26.0. A p-value of <0.05 was considered significant.
Results
The incidence of ovarian hyperstimulation syndrome among the study cohort was 10.9%, with a higher prevalence (60%) among the 18–24 year age group (p > 0.05) than among other age groups. Fibrinogen and cytokine levels did not differ significantly between the ovarian hyperstimulation syndrome and non-ovarian hyperstimulation syndrome groups; moreover, there was no significant difference in their levels measured before and after controlled ovarian stimulation (p > 0.05). However, receiver operating characteristic analysis revealed that elevated baseline (pre-controlled ovarian stimulation) levels of fibrinogen (area under the curve = 0.567, p = 0.604), interleukin-8 (area under the curve = 0.628, p = 0.337), tumour necrosis factor-alpha (area under the curve = 0.615, p = 0.337) and interleukin-10 (area under the curve = 0.633, p = 0.334) were associated with an increased risk of ovarian hyperstimulation syndrome. Women with elevated baseline interleukin-10 (≥33.5 ng/L) were at higher risk of ovarian hyperstimulation syndrome, with a sensitivity of 80.0%, specificity of 71.8%, area under the curve of 63.3% and an accuracy of 72.7%.
Conclusion
Interleukin-10 may serve as an early predictor of ovarian hyperstimulation syndrome among women undergoing controlled ovarian stimulation, with high sensitivity, specificity and accuracy. Further large-scale studies are warranted to confirm these findings.
Keywords
Introduction
Infertility affects one in six couples globally, and its prevalence may be as high as 30% in sub-Saharan Africa. 1 In Ghana, the prevalence rate ranges from 11.8% to 15.8%. 2 For most of these couples, assisted reproductive technology (ART) remains the only option to achieve pregnancy. Ovarian hyperstimulation syndrome (OHSS) is an iatrogenic and sometimes life-threatening complication of controlled ovarian stimulation (COS). 3 There is no universal consensus on the definition of OHSS; however, it is generally described as a potentially life-threatening iatrogenic complication that occurs during the early luteal phase and/or early pregnancy due to an exaggerated response to gonadotropin stimulation. It is characterised by ovarian enlargement, increased vascular permeability and a shift of fluid from the intravascular space to the third space. 3 The key features of OHSS include enlarged ovaries, fluid accumulation in the peritoneal cavity with ascites, pleural and pericardial effusions and haemoconcentration. Known risk factors for OHSS include low body mass index (BMI), young age, polycystic ovary syndrome (PCOS), high anti-Müllerian hormone (AMH) levels, history of OHSS, ovulation induction or COS with gonadotropins and beta-human chorionic gonadotropin (β-hCG) administration. 4
The precise pathophysiology of OHSS remains unclear, but the role of β-hCG as a trigger for the cascade of events leading to OHSS is well-established. 5 It stimulates luteinisation and the release or activation of several vasoactive mediators, such as vascular endothelial growth factor (VEGF), components of the renin–angiotensin system and various cytokines, such as interleukin (IL)-8, IL-6 and IL-1β. 6 These vasoactive mediators initiate a cascade of events, including endothelial injury, increased endothelial permeability, plasma leakage into the third space, hypovolaemia, haemoconcentration, thrombosis, electrolyte imbalance and multi-organ failure. 7
The manifestations of the disease range from mild to moderate, severe and critical stages. Mild OHSS presents with abdominal bloating/pain and an increase in ovarian size of up to 8 cc. Moderate disease is characterised by nausea and vomiting, with ultrasound evidence of ascites and increased ovarian volume between 8 and 12 cc. 8 Severe OHSS is characterised by clinical ascites, oliguria, haemoconcentration with haematocrit >45%, hypoproteinaemia and ovarian size >12 cc. The disease is considered critical if there is tense ascites, hydrothorax, oligo/anuria, thromboembolism or acute respiratory distress, with haematocrit >55% and white blood cell count >2500/mL. Severe or critical OHSS can lead to serious sequelae such as liver dysfunction, prerenal failure from hypovolemia, respiratory distress, gastrointestinal symptoms, cerebrovascular complications, adnexal torsion and death. 8
Currently, there is no clear strategy for preventing OHSS. Despite numerous studies on the subject, a recent systematic review of randomised controlled trials (RCTs) concluded that only a few interventions, including COS with gonadotropin-releasing hormone (GnRH) antagonist and agonist trigger, elective embryo cryopreservation, mild stimulation and dopaminergic agonist co-administration, have proven effective in reducing the occurrence and severity of OHSS.9–11 Furthermore, only a few studies have investigated predictors of OHSS.
Fibrinogen and cytokines are possible biomarkers of OHSS. Fibrinogen levels increase in response to systemic inflammation and hypercoagulation, reflecting haemoconcentration and the risk of thrombosis—key features of OHSS. 12 Luteinised granulosa cells and other ovarian immune cells also secrete VEGF and other cytokines that cause endothelial damage and increased vascular permeability. 13 A rise in cytokine levels reflects endothelial activation and inflammation or OHSS severity. Fibrinogen and cytokines are therefore potential biomarkers of OHSS, as they directly mirror its key pathological features, including haemoconcentration/hypercoagulability and endothelial injury/vascular permeability. 14
However, there is limited literature on fibrinogen and cytokine profiles in women undergoing ART treatment. This study aimed to examine the pre- and post-COS levels of fibrinogen and cytokines, including IL-8, tumour necrosis factor-alpha (TNF-α) and IL-10, as biomarkers of OHSS in women undergoing ART treatment.
If validated in larger, multicentre studies, fibrinogen or cytokine testing could serve as an early and objective diagnostic tool that could be incorporated into pre- or post-treatment screening protocols for ART candidates. Additionally, the study’s focus on biochemical markers detectable in peripheral blood enhances its feasibility and applicability in clinical settings, particularly in resource-limited environments where access to advanced diagnostic techniques may be unavailable. This approach would allow clinicians to stratify risk in women undergoing COS and tailor stimulation regimens, apply preventive measures (e.g. GnRH antagonist COS and agonist trigger, elective embryo cryopreservation, mild stimulation and dopaminergic agonist co-administration) or intensify post-procedural monitoring in high-risk individuals. Consequently, the incidence of severe OHSS may be reduced, patient safety enhanced and ART treatment outcomes optimised in both high- and low-resource settings.
Materials and methods
Study design, setting, population and sampling
This prospective analytical cohort study was conducted from 1 April 2023 to 30 November 2023 among women undergoing ART treatment at Hallmark Medicals, Ahensan Estate, Kumasi, Ghana. Hallmark Medicals is a general hospital that provides ART treatment as its flagship service. Sixty women aged 18–42 years who were undergoing self-cycle in vitro fertilisation (IVF)/intracytoplasmic sperm injection (ICSI) or donating oocytes for IVF/ICSI treatment of women with infertility were conveniently recruited and followed up to assess changes in their coagulation and cytokine profiles for predicting OHSS.
Using the Yamane formula (1967) and assuming the number of IVF cases during the study period to be 50 with a precision level of 0.05, the calculated sample size was 42. However, 60 women were screened; of these, 14 were excluded (8 with PCOS, 5 with a history of OHSS and 1 on antithrombotic treatment), resulting in a final sample size of 46 participants. Although this was a prospective study, funding constraints prevented recruitment beyond the minimum sample size. Only patients with early or primary OHSS were included. As a standard precaution against secondary OHSS, self-cycle ART patients who developed primary OHSS did not undergo embryo transfer; instead, their embryos were electively frozen for frozen–thawed embryo transfer at a later date. Women with PCOS (because of the known high risk of OHSS), a history of OHSS (because of the risk of recurrence) or coagulopathy and those who received treatment with antithrombotic agents were excluded from the study. Written informed consent was obtained from all participants.
COS and egg retrieval
All participants received one of the two standard treatment protocols for COS: the luteal-phase long GnRH agonist (GnRHa) protocol or the short GnRHa protocol. In both protocols, a GnRHa was administered to downregulate the pituitary production of follicle-stimulating hormone (FSH) and luteinising hormone (LH), thereby suppressing spontaneous follicular growth and LH surge/ovulation in the IVF/ICSI cycle. In the luteal-phase long agonist protocol, patients received one intramuscular (IM) Depo injection of 3.6 mg of goserelin on menstrual cycle Day 21 before the IVF/ICSI cycle. A daily injection of 0.25 mg of buserelin was administered from Day 1 or 2 of the IVF/ICSI cycle in the short GnRHa protocol.
COS was performed by administering highly purified FSH, with or without human menopausal gonadotropin (HMG), using standard doses depending on the patient’s age, BMI and antral follicle count (AFC). Gonadotropin administration was started on Day 2 or 3 of the menstrual cycle. Ovarian response to COS was monitored using serial transvaginal scans on stimulation Days 6, 9 and 11, and the FSH dose was adjusted according to the observed response. The targeted follicular number per patient was 8–16. An ovulation trigger was administered as a 5000–10,000 IU of β-hCG injection (depending on the response to COS) on stimulation Day 11 or 12, when the follicles reached a diameter of 18 mm.
Standard operative procedures were conducted to prevent OHSS among participants. These included the exclusion of patients with known risk factors (e.g. PCOS) from receiving a GnRHa protocol and recruitment of such patients for antagonist protocols; administration of tailored doses of FSH based on ovarian reserve assessment and prediction of ovarian response using age, AMH and AFC; transvaginal ultrasound follicular tracking to detect hyper-response; coasting of COS; and withholding of the β-hCG trigger from patients considered at significant risk of OHSS during COS.
Transvaginal ultrasound-guided oocyte retrieval was performed under general anaesthesia 34 h after the β-hCG injection. This was followed by oocyte fertilisation and embryo culture using standard laboratory procedures for IVF/ICSI. All embryos formed from self-cycle patients who developed OHSS were electively frozen for transfer in a subsequent frozen–thawed cycle. All participants were admitted for the first 24 h for postoperative recovery and monitoring for OHSS and were followed up until all reported symptoms resolved.
For this study, OHSS was defined as the presence of significant abdominal pain, ultrasound or clinical ascites, oliguria and haemoconcentration with haematocrit (>45%).
All patients who developed OHSS received standard treatment, including oral dopamine agonists (e.g. cabergoline), parenteral infusion of colloids and human albumin and thromboprophylaxis.
The study participants were divided into two groups based on outcomes: women who developed OHSS and those who did not. At the end of the study period, 46 participants were included in the statistical analyses.
Sociodemographic data and sample collection
A structured questionnaire was used to collect sociodemographic information, including age, anthropometric measurements (such as BMI) and clinical data from participants’ medical records. Approximately 10 mL of venous blood was drawn from the antecubital fossa of each participant, not more than 1 h before the first gonadotropin injection for COS and less than 1 h after egg collection. Pre-stimulation blood samples were collected to determine baseline fibrinogen and cytokine levels. These samples were obtained on Day 1 of COS when all patients presented to begin stimulation. A few participants received their gonadotropin and β-hCG supplies at facilities closer to their homes. For this group, β-hCG injections—often administered at night—were given at the local facilities. Therefore, post-egg collection samples were more feasible to obtain than the preferred post-β-hCG samples, as all participants necessarily returned to the main facility for egg collection.
Three millilitres of blood were dispensed into a gel separator tube, 3 mL into an ethylenediaminetetraacetic acid (EDTA) tube and 4 mL into a citrate tube. Samples in EDTA tubes were used for complete blood count analysis. Haematological parameters, including haematocrit, were measured using an automated five-part differential haematology analyser (XN-350 Sysmex, Germany). Citrated samples were used to estimate fibrinogen levels, while serum samples in gel tubes were used to measure cytokines as well as liver and renal function.
Laboratory estimation of fibrinogen and cytokines (IL-8, TNF-α and IL-10)
All test samples were analysed using enzyme-linked immunosorbent assay (ELISA) kits. The microtitre plate wells for each test were coated with purified human antibodies (solid-phase antibodies). After the addition of samples and horseradish peroxidase (HRP) enzyme, an antibody–antigen–enzyme complex was formed. Following thorough washing and the addition of chromogens, colour development occurred, and the reaction was terminated by adding a stop solution. The resulting colour intensity was measured spectrophotometrically at a wavelength of 450 nm, and the analyser was used to calculate and display the concentration of each sample.
Quality control materials simulating the characteristics of patient samples were used to monitor the performance of immunochemical assays. Quality control materials were included in each batch of samples analysed in accordance with good laboratory practice and laboratory accreditation requirements.
Ethical considerations
Ethical approval for this study (CHRPE/683/23; dated 2 August 2023) was obtained from the Committee for Human Research and Publication Ethics, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana. Written informed consent was obtained from all participants before enrolment. Participants were assured of confidentiality and informed of their right to withdraw from the study at any time without consequence. All ethical principles outlined in the Declaration of Helsinki (1975), as revised in 2024, were strictly followed.
Data management and statistical analysis
The data obtained were entered into Microsoft Excel 2021. R Language for Statistical Computing (version 4.3.2) and Statistical Package for the Social Sciences (SPSS, version 26.0; IBM Corp., Armonk, NY, USA) were used for data analysis. A bar chart was used to illustrate the prevalence of OHSS. Categorical variables were presented as frequencies and percentages. Parametric continuous variables were expressed as mean ± standard deviation, while non-parametric continuous variables were expressed as median and interquartile range after assessing normality using the Shapiro–Wilk test. The association between sociodemographic and clinical variables and OHSS status was determined using the chi-squared test. The outcomes for fibrinogen, cytokine profiles, haematological parameters and liver and renal function test parameters were compared using the independent-samples t-test for parametric variables and the Mann–Whitney U test for non-parametric variables. Pre- and post-COS levels were compared using the Wilcoxon signed-rank test. The receiver operating characteristic (ROC) curve analysis was used to evaluate the diagnostic accuracy of fibrinogen, IL-8, TNF-α and IL-10 for OHSS. A p-value of <0.05 was considered statistically significant. All participant details were de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 15
Results
Sociodemographic and clinical characteristics of study participants
The study included equal numbers of egg donors (50.0%) and self-cycle participants (50.0%) undergoing COS. Among the participants, 60.9% had secondary infertility, whereas 39.1% had primary infertility, with approximately two-thirds (65.2%) experiencing infertility for 7–12 years. The mean age of the participants was 27.89 years, with the majority falling within the 18–24 years age range (45.7%), followed by 25–34 years (30.4%) and 36–41 years (23.9%). Most participants had attained a senior high school education (41.3%), and approximately one-third had attained a tertiary education (32.6%). Approximately 39.1% and 32.6% of the participants had achieved at least one and two pregnancies before, respectively. More than half of the participants were self-employed (54.3%), and over 90% were Christians (91.3%). Almost all participants were Ghanaians (97.8%), and the majority were Asantes (69.6%). The participants’ mean weight, height and BMI were 70.20 kg, 1.64 m and 26.09 kg/m2, respectively (Table 1).
Sociodemographic and clinical characteristics of study participants.
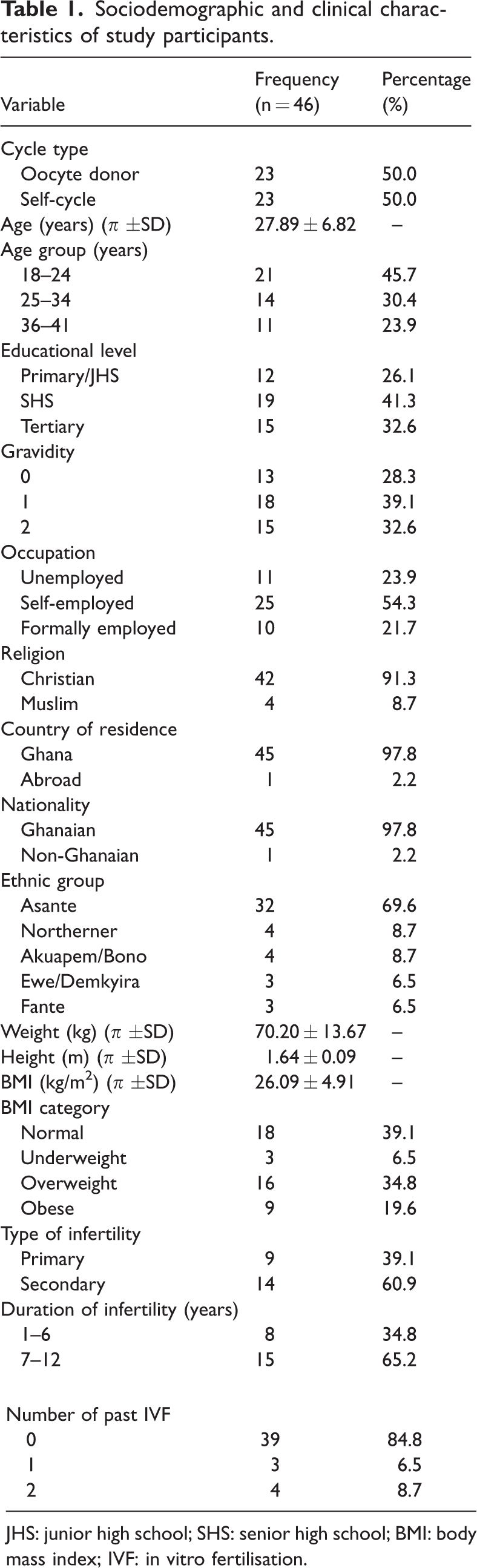
JHS: junior high school; SHS: senior high school; BMI: body mass index; IVF: in vitro fertilisation.
Prevalence of OHSS among women who underwent COS
A prevalence of 10.9% for OHSS was observed among women who underwent COS, comprising 60.0% donors and 40.0% self-cycle participants (Figure 1).
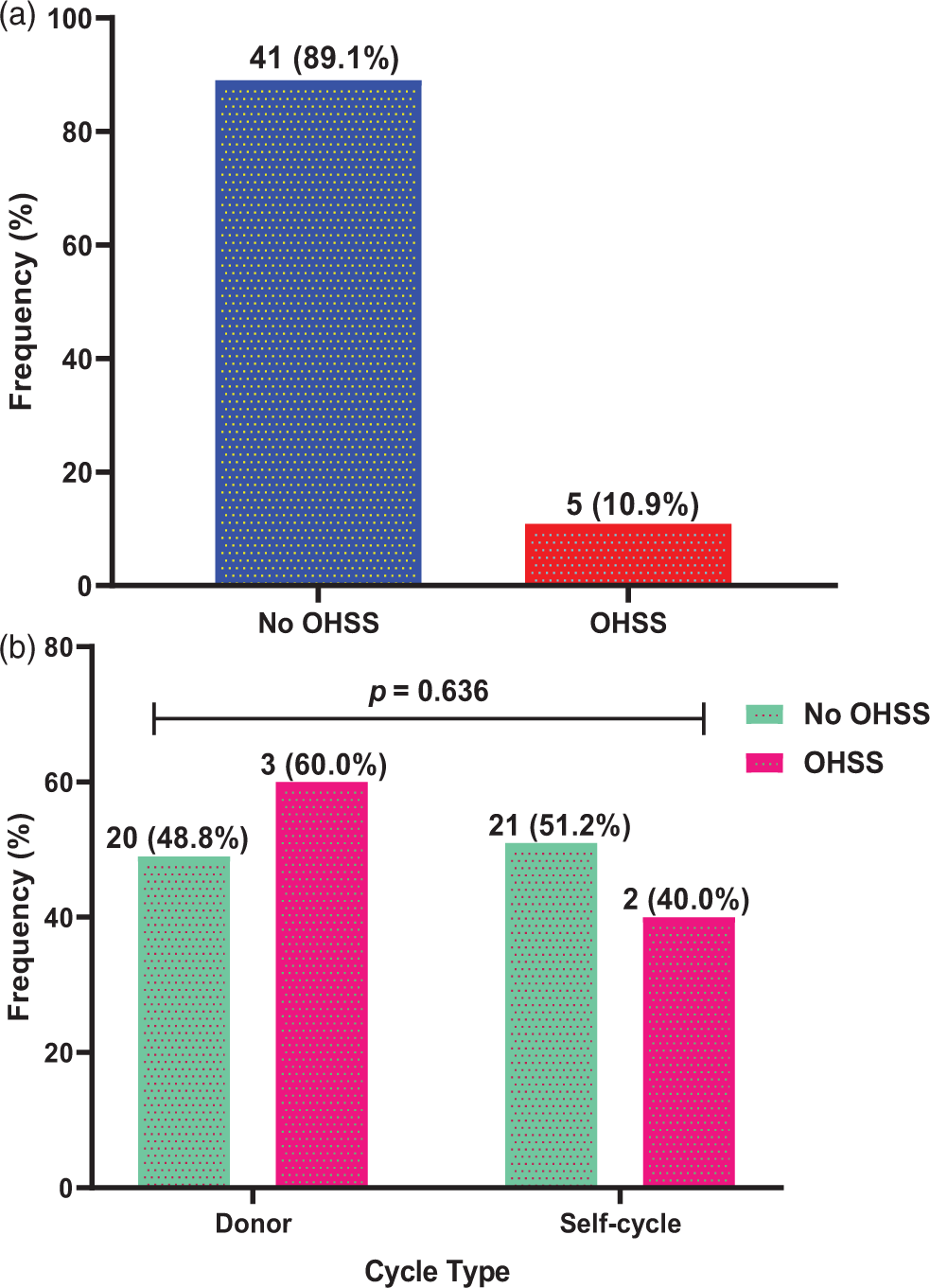
Incidence of OHSS among women who underwent COS. OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation.
Sociodemographic and clinical factors associated with OHSS among women who underwent COS
As shown in Table 2, educational level was significantly associated with OHSS, with 80.0% of participants with OHSS having tertiary education compared with 20% with basic education (junior high school) (p = 0.044). However, OHSS was more prevalent among younger age groups, particularly those aged 18–24 years (60.0%; mean age: 27.40 years), than among the non-OHSS group, which included participants with a mean age of 27.95 years (p > 0.05).
Sociodemographic and clinical factors associated with OHSS among women who underwent COS.
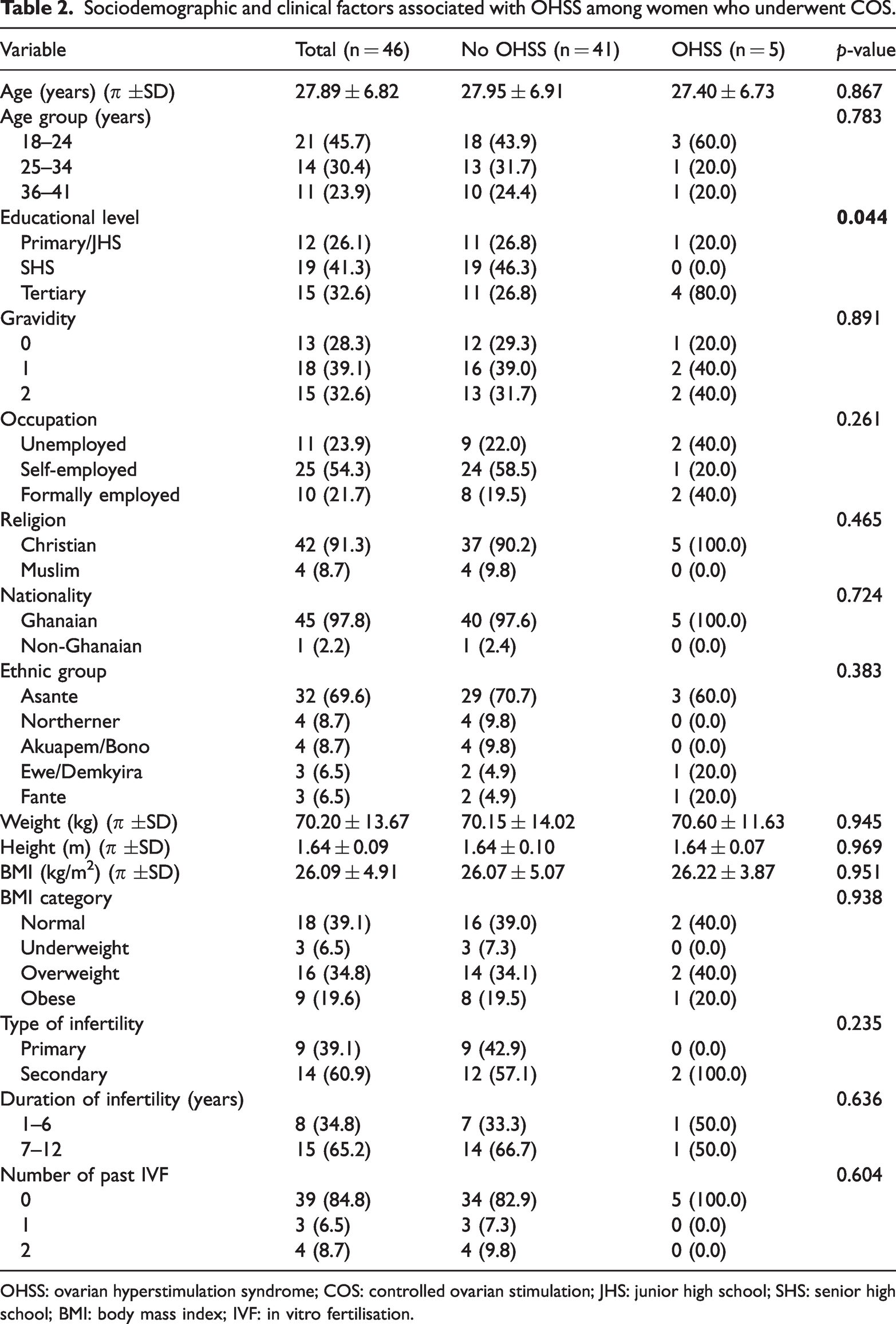
OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation; JHS: junior high school; SHS: senior high school; BMI: body mass index; IVF: in vitro fertilisation.
Gravidity (p = 0.891), occupation (p = 0.261) and religion (p = 0.465) did not predict OHSS. Similarly, ethnicity (p = 0.383) and duration of infertility (p = 0.636) were not significantly associated with OHSS among study participants (Table 2).
Comparison of pre- and post-COS levels of fibrinogen, IL-8, TNF-α and IL-10 between OHSS and non-OHSS participants
Comparison of fibrinogen and cytokine biomarkers among study participants showed no significant differences between OHSS and non-OHSS participants in pre- and post-COS levels of fibrinogen, TNF-α, IL-8 and IL-10 (p > 0.05).
However, post-COS levels of TNF-α (243.00 ng/L) were significantly higher than pre-COS levels (212.25 ng/L, p = 0.004) among the study participants (Table 3).
Comparison of pre- and post-COS levels of fibrinogen and cytokines between OHSS and non-OHSS participants.

p-values for between‐subject effects (No OHSS vs OHSS) were computed via Mann–Whitney U test, while p-values for within‐subject effects (pre-ovarian stimulation vs post-ovarian stimulation) were computed via Wilcoxon signed-rank test. p < 0.05 indicated statistically significant differences.
OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation; TNF-α: tumour necrosis factor-alpha; IL: interleukin.
Comparison of pre- and post-COS levels of liver function parameters between OHSS and non-OHSS participants
No significant differences were observed between OHSS and non-OHSS participants in pre- and post-COS levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT) (p > 0.05). Similarly, there were no significant differences between the two groups in pre- and post-COS levels of total protein, albumin, total bilirubin and direct bilirubin (p > 0.05).
However, post-COS levels of ALT (21.31 IU/L) were significantly higher than pre-COS levels (16.70 IU/L, p = 0.029) among the study participants (Table 4).
Comparison of pre- and post-COS levels of liver function parameters between OHSS and non-OHSS participants.
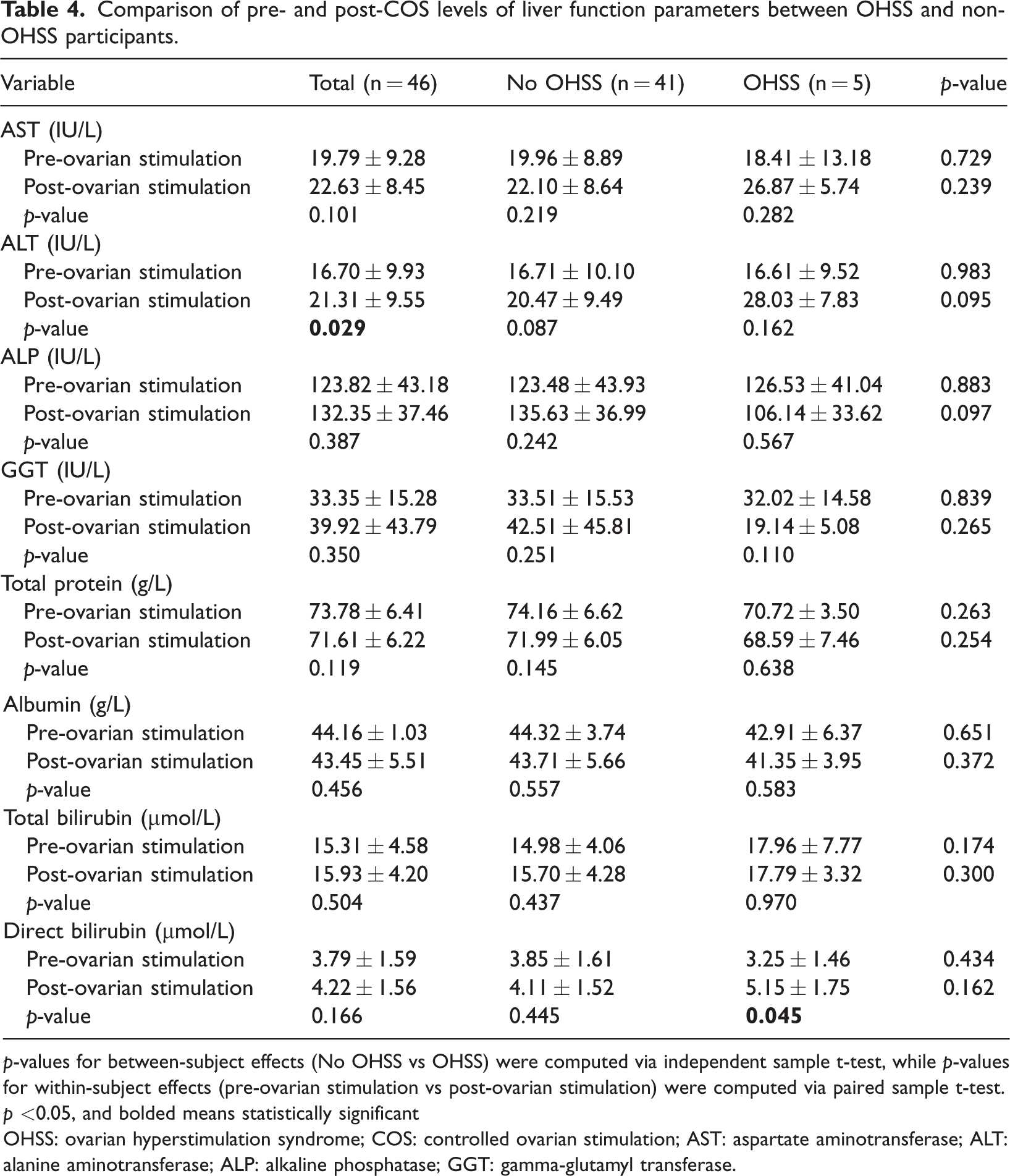
p-values for between‐subject effects (No OHSS vs OHSS) were computed via independent sample t-test, while p-values for within‐subject effects (pre-ovarian stimulation vs post-ovarian stimulation) were computed via paired sample t-test. p <0.05, and bolded means statistically significant
OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; GGT: gamma-glutamyl transferase.
Comparison of pre- and post-COS levels of renal function parameters between OHSS and non-OHSS participants
There were no significant differences between OHSS and non-OHSS participants in pre- and post-COS levels of urea, creatinine, K+, Na+ and Cl− (p > 0.05). Similarly, pre- and post-COS levels of these parameters were not significantly different among the study participants (p > 0.05) (Table 5).
Comparison of pre- and post-COS levels of renal function parameters between OHSS and non-OHSS participants.

p-values for between‐subject effects (No OHSS vs OHSS) were computed via Mann–Whitney U test, while p-values for within‐subject effects (pre-ovarian stimulation vs post-ovarian stimulation) were computed via Wilcoxon signed-rank test. p < 0.05 indicated statistically significant differences.
OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation; K+: potassium; Na+: sodium; Cl−: chloride.
Pre-COS levels of fibrinogen, IL-8, TNF-α and IL-10 as diagnostic biomarkers of OHSS among women who underwent COS
ROC analysis was used to evaluate the diagnostic performance of pre-COS levels of fibrinogen, IL-8, TNF-α and IL-10 as diagnostic biomarkers of OHSS among women who underwent COS. Although there were no significant differences in fibrinogen and cytokine levels before and after COS, women with elevated baseline (pre-COS) levels of fibrinogen (area under the curve (AUC) = 0.567, p = 0.604), IL-8 (AUC = 0.628, p = 0.337), TNF-α (AUC = 0.615, p = 0.337) and IL-10 (AUC = 0.633, p = 0.334) were more likely to develop OHSS (Figure 2).
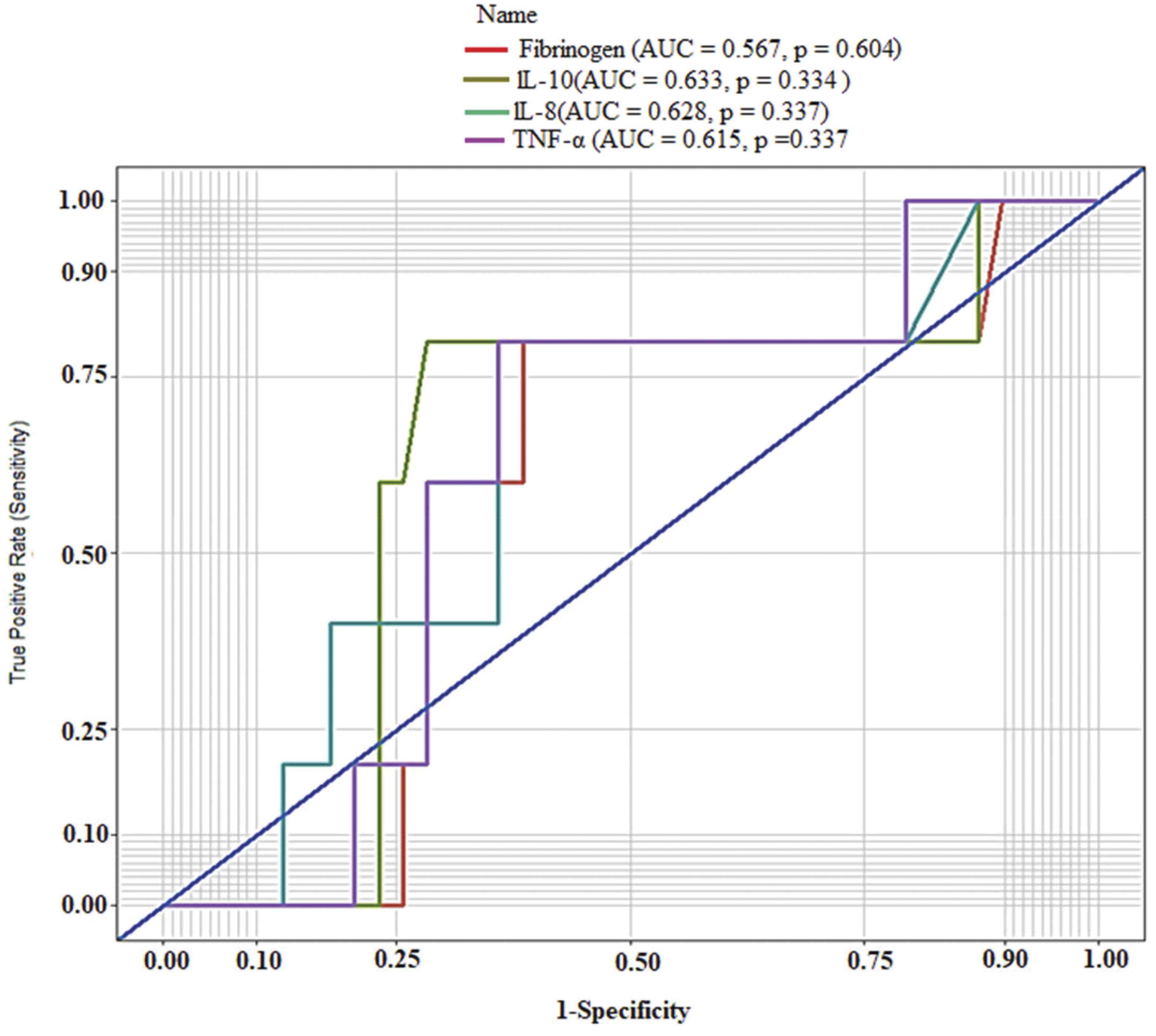
The ROC curves of pre-COS levels of fibrinogen, IL-8, TNF-α and IL-10 as diagnostic biomarkers of OHSS among women who underwent COS. ROC: receiver operating characteristics; COS: controlled ovarian stimulation; OHSS: ovarian hyperstimulation syndrome; IL: interleukin; TNF-α: tumour necrosis factor-alpha.
At a cut-off value of ≥33.5 ng/L, IL-10 was the optimal cytokine for detecting OHSS among women who underwent COS, with a sensitivity of 80.0%, specificity of 71.8%, positive predictive value of 26.7%, negative predictive value of 96.6%, AUC of 63.3% and accuracy of 72.7% (Table 6).
Diagnostics performance of pre-COS levels of fibrinogen, IL-8, TNF-α and IL-10 as diagnostics biomarkers of OHSS among women who underwent COS.

At a cut‐off of ≥33.5 ng/L, IL-10 was the optimal biomarker for detecting OHSS among women who underwent COS, with a sensitivity of 80.0%, specificity of 71.8%, PPV of 26.7% and NPV of 96.6% as well as area under the curve and accuracy of 63.3% and 72.7%, respectively.
OHSS: ovarian hyperstimulation syndrome; COS: controlled ovarian stimulation; CI: confidence interval; PPV: positive predictive value; NPV: negative predictive value; LR+: positive likelihood ratio; LR−: negative likelihood ratio; TNF-α: tumour necrosis factor-alpha; IL: interleukin.
Discussion
One in 10 participants in this study developed OHSS, the most severe and life-threatening iatrogenic complication of COS during ART procedures. This prevalence is higher than the 1.2% reported in Europe but lower than the 24.8% reported among a select population of Chinese women with PCOS undergoing IVF/ICSI.16,17 The disparity in prevalence rates may be attributed to methodological differences in sample size, study design, study population and sampling techniques. The higher prevalence of OHSS among the younger age group aligns with existing evidence identifying young age as a critical risk factor for OHSS.18,19 A previous study comparing the peritoneal fluid of OHSS and non-OHSS participants reported higher levels of IL-8 and IL-10 in the OHSS group. 20 In contrast, the present study found no significant differences in fibrinogen and cytokine levels between the OHSS and non-OHSS groups; moreover, no significant differences were observed between pre- and post-COS levels within groups. However, ROC analysis identified IL-10 as a potential biomarker of OHSS with relatively high sensitivity and specificity. This novel finding could represent an important advancement in the prediction of OHSS, although larger, more robust studies are needed to validate it.
There is currently no definitive conclusion regarding the pathophysiology of OHSS. However, the roles of β-hCG and several pro-inflammatory mediators, such as VEGF and the renin–angiotensin system, as well as their associations with various cytokines, including TNF-α, IL-1β and IL-10, are well-established. Recent discoveries have revealed that increased immunoactivity in receptors such as the cholinergic receptor muscarinic 1 (CHRM1), transient receptor potential canonical (TRPC1) ion channel and transient receptor potential melastatin 2 (TRPM2) channel may play significant roles in OHSS, suggesting multiple avenues for OHSS prevention.21,22 Additionally, evidence indicates that prophylactic administration of dopamine agonists, such as cabergoline or clarithromycin, can prevent or improve OHSS outcomes when administered before the β-hCG trigger.11,23 Furthermore, nifedipine has been shown to inhibit VEGF more effectively through TRPC1 than cabergoline. 22 The identification of a reliable OHSS predictor, such as IL-10, could therefore enable early identification of individuals at risk and facilitate timely prophylactic interventions, ultimately improving patient safety and treatment outcomes in ART practice.
The absence of significant differences in renal and liver function parameters between OHSS and non-OHSS participants in this study is consistent with previous reports. This finding suggests that despite the cardinal features of haemoconcentration and increased thrombosis risk in OHSS, the livers and kidneys remain unaffected. This may be because OHSS primarily results from increased vascular permeability rather than direct hepatic or renal cellular damage, particularly in mild cases. Moreover, hepatic and renal impairment that occurs in severe cases is typically transient and reversible, and standard liver and kidney function tests may not detect subclinical abnormalities. 24 This explains the remarkable recovery often observed following prompt fluid replacement and thromboprophylaxis.
Limitations
The relatively small sample size, short duration of follow-up and single-site setting in Ghana may limit the generalizability of this study to other populations or settings. The observed OHSS incidence of 10.9%, compared with the international incidence of 1%–2%, was relatively high. This may reflect the small sample size and the inclusion of a large proportion of oocyte donors, who are known to be at higher risk of OHSS.
The short follow-up period allowed for the assessment of biomarkers only immediately after egg collection, which may have led to missed cases of late-onset (secondary) OHSS because some participants were unwilling to return for follow-up. Moreover, the findings may have been influenced by unaccounted confounding factors, such as BMI, smoking status or egg collection- and anaesthesia-related effects, which could have affected biomarker levels.
Conclusion
IL-10 may serve as a useful diagnostic biomarker for the early detection of OHSS among women undergoing ovarian stimulation, demonstrating high sensitivity, specificity and accuracy. However, the small sample size and single-centre design limit the strength and generalisability of these findings. Larger, multicentre studies are warranted to validate the diagnostic role of IL-10 in OHSS.
Footnotes
Acknowledgements
The authors express their gratitude to all study participants and to the management of Hallmark Medicals, Ahinsan estate, Kumasi, Ghana, for their support in the successful implementation of this study.
Authors’ contributions
Conception of the research proposal: Herbert Ekoe Dankluvi, Millicent Osaah, Ellis Fleischer Djoleto, Charles Mawunyo Senaya, Francis Jojo Moses Kwadzo Damalie, Edward Daasah, Enoch Odame Anto, Benedict Sackey, Ebenezer Senu
Data collection and analysis: Herbert Ekoe Dankluvi, Enoch Odame Anto, Millicent Osaah, Elikplim Adzo Damalie, Benedict Sackey, Ebenezer Senu, Ellis Fleischer Djoleto, Edward Dassah, Francis Jojo Moses Kodzo Damalie
Manuscript writing: Herbert Ekoe Dankluvi, Enoch Odame Anto, Elikplim Adzo Damalie, Benedict Sackey, Ebenezer Senu, Edward Dassah, Charles Mawunyo Senaya, Francis Jojo Moses Kodzo Damalie
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Ethical clearance (CHRPE/683/23; dated 2 August 2023) was obtained from the Committee for Human Research and Publication Ethics, Kwame Nkrumah University of Science and Technology, prior to the commencement of the study, in accordance with the Declaration of Helsinki (1975, as revised in 2024). The study protocol was explained to all participants, and written informed consent was obtained.
Funding
This study did not receive funding from any private, government or not-for-profit organisation. It was self-funded.
