Abstract
Objectives
To investigate the effects of day 5 embryo transfer (D5ET) compared with day 3 embryo transfer (D3ET) in patients at high risk of developing ovarian hyperstimulation syndrome (OHSS); to analyse factors affecting blastocyst formation.
Methods
Patients at high risk of developing OHSS underwent either D3ET or D5ET.
Results
A total of 253 patients received D3ET; 263 received D5ET. The number of embryos transferred was lower in the D5ET group than in the D3ET group. There were no between-group differences in pregnancy or live birth rates. Implantation rate was higher, and multifetation rate lower, in the D5ET group compared with the D3ET group. In addition, the incidence of moderate or severe OHSS was lower in the D5ET group than in the D3ET group. The woman’s age, gonadotrophin dosage and insemination method were associated with the quality of blastocyst formation.
Conclusions
In patients with a high risk of developing OHSS, compared with D3ET, D5ET decreased the multifetation rate and the incidence of moderate or severe OHSS, but did not affect the pregnancy or live birth rate. Women of a younger age, who have had an appropriate gonadotrophin dose and insemination by in vitro fertilization, are suitable candidates for blastocyst transfer.
Introduction
Ovarian hyperstimulation syndrome (OHSS) is a common complication of assisted reproductive technology. The incidence of clinically significant OHSS is 2–3%, but severe OHSS can be life-threatening. 1 Cancellation of fresh embryo transfer and cryopreservation of all embryos is an effective strategy to avoid the development of severe OHSS in women at high risk of the complication; high risk is indicated by increased E2, follicular size and number. 2 However, the pregnancy rate is lower in freeze–thaw cycles than in fresh cycles. 2
If additional time is available to monitor and relieve symptoms in patients at high risk of developing OHSS, it may be possible to determine those who can undergo fresh embryo transfer, which would increase both the opportunity for (and the security of) fresh-cycle transfers. Extending the culture time and performing blastocyst transfer may provide more time to assess and treat the patient. It is therefore worth investigating whether day 5 embryo transfer (D5ET) can lead to better outcomes than day 3 embryo transfer (D3ET), in patients at high risk of developing OHSS. The extended culture time need for blastocyst transfer is thought to result in embryos with better implantation potential and a higher live birth rate. 3 However, blastocyst culture is not successful in all patients. Patients face the risk that there is a poor or absent blastocyst formation on day 5. Some studies have investigated the factors affecting blastocyst formation.4,5 However, little has been published on the use of blastocyst transfer in patients at high risk of OHSS.
In the present study, the effects of D5ET or D3ET on in vitro fertilization–embryo transfer (IVF–ET) outcomes were observed in patients at high risk of developing OHSS. In addition, factors affecting the formation of high-quality blastocysts were analysed, to provide a more reliable basis for blastocyst culture in these patients.
Patients and methods
Patients
Patients who had a high risk of developing OHSS (following controlled ovarian hyperstimulation during their first IVF–ET cycle) at the Reproductive Medical Centre, First Affiliated Hospital of Zhengzhou University, Zhengzhou, China, between January 2009 and January 2012, were enrolled in the present study. A high risk of developing OHSS was indicated by a serum oestradiol level > 5000 pg/ml on the day of human chorionic gonadotrophin (HCG) administration and the presence of >18 ovarian follicles with a diameter >11 mm. 6 The presence of more than four high-quality eight-cell embryos after in vitro culture for 3 days was a further inclusion criterion for the study. Embryos used for preimplantation genetic diagnosis or cryopreserved IVF cycles were excluded from the study.
Information concerning age, body mass index, duration and cause of infertility, basal follicle stimulating hormone (FSH) level, antral follicle count, duration of gonadotrophin treatment, gonadotrophin dose, oestradiol level on the day of HCG administration, number of retrieved oocytes, in vitro insemination method, semen source and number of embryos transferred were recorded for each patient.
On the basis of information provided about the risks and benefits of D5ET and D3ET, each patient selected which procedure to follow. Written informed consent was obtained from all study participants. The study protocol was approved by the Institutional Review Board and Ethics Committee of the First Affiliated Hospital of Zhengzhou University, Zhengzhou, China.
Controlled ovarian hyperstimulation
Controlled ovarian hyperstimulation was performed according to the standard protocol used at our centre. 7 After the initiation of treatment, the gonadotrophin dose was adjusted according to the ovarian response. Once the recruited follicles were >18 mm in diameter, independent of the serum oestradiol level, oocyte maturation was obtained by administration of 5000 IU HCG. Oocytes were collected by transvaginal ultrasound-guided puncture, 34–36 h later. After ovum pick-up, type-B ultrasound examination, routine blood tests and clotting function tests were performed. In patients at high risk of developing OHSS, hydroxyethyl starch 500 ml/day was injected from the day of oocyte pickup to embryo transfer. Fixed doses of aspirin (100 mg/day) and methylprednisolone (4 mg/day) were administered from the day of oocyte pickup to 10 days after embryo transfer.
Embryo culture and transfer
In vitro culture was performed using G-Series™ media (Vitrolife, Gothenburg, Sweden). Oocytes and embryos were incubated at 37℃ with 5% oxygen, 6% carbon dioxide and 89% nitrogen. Short-term insemination or intracytoplasmic sperm injection (ICSI) was performed 39–40 h after HCG administration (day 0). Pronuclei were observed 16–20 h after insemination. On day 3, embryos were evaluated in terms of fragmentation rate and the number and size of blastomeres. 4 Day 3 embryos with eight blastomeres, <20% fragmentation, of uniform size and with no multinucleated blastomeres were regarded as being of high quality. 4
Four high-quality embryos were selected on day 3 to continue in culture to day 5; all other available day 3 embryos were frozen. The day 5 blastocysts were graded as described by Gardner et al. 8 The inner cell mass and outer trophoblast were graded (based on the cell number, cohesion and regularity) as A, B or C; blastocyst quality was categorized as excellent (AA), good (AB, BA or BB), fair (BC or CB) or poor (CC). 5 In the present study, blastocysts categorized as excellent (AA) were regarded as being of high quality.
High-quality embryos with eight cells were used in all patients receiving D3ET. In patients receiving D5ET, high-quality blastocysts were used, wherever possible. The number of embryos transferred was in line with standards set by the Ministry of Public Health of China. 9
Assessment of OHSS and pregnancy confirmation
The occurrence of moderate OHSS was defined as the presence of ascites on ultrasound examination, moderate haemoconcentration and elevated leucocyte levels. Symptoms of OHSS include rapid weight gain, abdominal distension, nausea and vomiting. Severe OHSS was defined as the presence of increased fluid shifts into the peritoneal (and possibly the pleural and pericardial) cavity, leading to hypovolaemia and severe haemoconcentration; hepatorenal failure, acute respiratory distress syndrome, haemorrhage from ovarian rupture and thromboembolism may also occur.
Clinical pregnancy was confirmed by visualization of fetal cardiac activity on ultrasound on day 35 following embryo transfer. The numbers of fetuses and live births were recorded.
Statistical analyses
Data analyses were performed using Student’s t-test for numerical variables and χ2-test for categorical variables. To determine the effects of blastocyst transfer on IVF-ET outcomes, the relative risk and corresponding 95% confidence interval were calculated. Factors affecting the formation of high-quality blastocyts were analysed using multivariate logistic regression. A P-value of <0.05 was considered to be statistically significant. All statistical analyses were performed using SAS software, version 9.1 (SAS Institute, Cary, NC, USA).
Results
Characteristics and clinical features in patients at high risk of developing ovarian hyperstimulation syndrome receiving either day 3 embryo transfer (D3ET) or day 5 embryo transfer (D5ET).
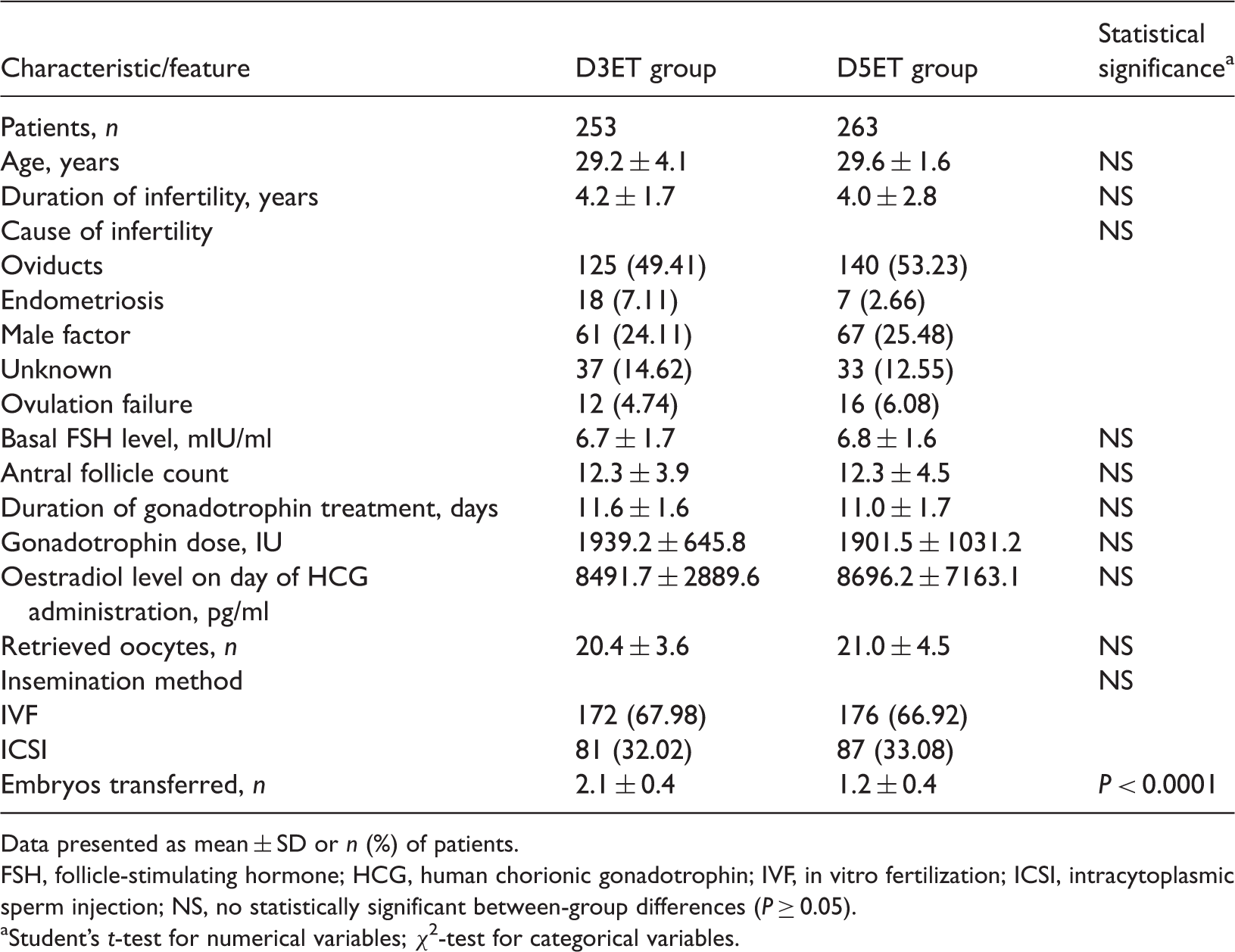
Data presented as mean ± SD or n (%) of patients.
FSH, follicle-stimulating hormone; HCG, human chorionic gonadotrophin; IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection; NS, no statistically significant between-group differences (P ≥ 0.05).
Student’s t-test for numerical variables; χ2-test for categorical variables.
IVF–ET outcomes
In vitro fertilization–embryo transfer outcomes in patients at high risk of developing ovarian hyperstimulation syndrome (OHSS) receiving either day 3 embryo transfer (D3ET) or day 5 embryo transfer (D5ET).

χ2-test.
Total pregnancies listed in this table excludes cases of ectopic pregnancy (n = 15).
NS, no statistically significant between-group differences (P ≥ 0.05).
Day 5 blastocyst formation
A total of 1003 high-quality day 3 embryos were used for blastocyst culture. The overall blastocyst formation rate was 69.0% (692/1003); of these, 194 were of excellent quality, giving a high-quality blastocyst formation rate of 19.3% (194/1003). Of the 263 patients receiving D5ET, 178 received high-quality blastocysts.
Multivariate logistic regression analysis of factors affecting high-quality blastocyst formation in patients at high risk of developing ovarian hyperstimulation syndrome.

IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection.
Discussion
Blastocyst transfer is thought to result in the selection of embryos with better developmental potential, thus enabling the transfer of fewer embryos and a reduction in multiple pregnancies. However, the major disadvantage of extended culture is the risk that no blastocysts will be available for transfer on day 5. Therefore, blastocyst transfer is not widely used in clinical practice. In the present study, only four of the available high-quality day 3 embryos underwent extended culture, with the other available embryos being frozen. This enabled patients to more easily accept the use of high-quality day 3 embryos for blastocyst culture and transfer.
Blake et al. 10 and Guerif et al. 11 reported that blastocyst transfer was associated with a higher clinical pregnancy rate and live birth rate than D3ET, whereas in the present study there were no significant differences in the clinical pregnancy or live birth rates between the D5ET and D3ET groups. Thomas et al. 5 reported clinical pregnancy and live birth rates with D5ET of 64.7% and 58%, respectively, which are higher than those in the present study. These differences may be due to different patient populations. The present study population was confined to patients at high risk of developing OHSS; a high level of serum oestradiol (>5000 pg/ml) on the day of HCG administration may not be conducive to embryo implantation, leading to a decreased pregnancy rate. 12 However, in the present study D5ET was associated with a lower rate of multiple pregnancies, compared with D3ET. The most effective method to decrease multifetation is to reduce the number of embryos transferred. 13 In the present study, the number of embryos transferred was lower in the D5ET group than in the D3ET group, but the rate of embryo implantation was higher in the D5ET group than in the D3ET group. These results indicate that extending the culture of embryos to the blastocyst stage can select embryos with a high implantation potential, enabling the transfer of fewer embryos and reducing the number of multiple pregnancies.
Another important finding in the present study is that D5ET significantly decreased the incidence of moderate or severe OHSS. This may be partly due to the prolonged in vitro culture time, which gave clinicians more time to monitor and relieve the patient’s symptoms while still allowing fresh embryo transfer. In addition, Lyons et al. 14 reported that late OHSS is related to multifetation, so the lower multifetation rate in the D5ET group may have helped to reduce the occurrence of moderate or severe OHSS in these patients.
The present study demonstrated that blastocyst transfer can obtain good clinical outcomes in patients at high risk of developing OHSS; however, blastocyst culture is not always successful. Whether blastocyst culture can be carried out often depends on the developmental status of the day 3 embryos. Langley et al. 15 reported that the blastocyst formation rate from day 3 eight-cell embryos was 76%, while that of day 3 six-cell embryos was only 54%, suggesting that day 3 eight-cell embryos have greater developmental potential. Therefore, the number of day 3 eight-cell embryos is regarded as an important indicator of the success of blastocyst culture. Milki et al. 16 reported that blastocyst culture was appropriate if at least four day 3 eight-cell embryos were present, whereas Levitas et al. 17 suggested that the presence of more than two day 3 eight-cell embryos was required. In the present study, the presence of more than four day 3 eight-cell high-quality embryos was necessary for blastocyst culture. A total of 1003 day 3 eight-cell high-quality embryos were used for blastocyst culture; the blastocyst formation rate was 69.0% and the excellent-quality blastocyst formation rate was 19.3%.
Factors affecting the excellent-quality blastocyst formation rate in the present study were analysed using logistic regression. Female age was shown to be negatively associated with excellent-quality blastocyst formation, which is consistent with the results of Thomas et al. 5 The in vitro insemination method was also shown to affect excellent-quality blastocyst formation. Miller and Smith 18 reported that ICSI had a detrimental effect on blastocyst development because this method bypasses the selection processes of the zona pellucida, increasing the possibility that an abnormal sperm will enter the oocyte. Vanderzwalmen et al. 19 have reported that abnormal sperm fertilization does not affect the morphology of day 3 embryos, but does affect their developmental potential, and therefore affects blastocyst formation.
Gonadotrophin dosage was another factor shown to affect the quality of blastocyst formation. Thomas et al. 5 reported that gonadotrophin dosage was negatively related to excellent-quality blastocyst formation and that the excellent-quality blastocyst formation rate was higher in patients who were sensitive to gonadotrophin. Patients in the present study were at high risk of developing OHSS and were usually sensitive to gonadotrophin. In order to reduce the incidence of OHSS in such patients, the duration and dosage of gonadotrophin treatment is usually decreased. 2 However, gonadotrophin is essential for oocyte development. Yoldemir et al. 20 reported that decreased gonadotrophin dosage and duration could affect day 3 embryo quality and increase embryo fragmentation. Therefore, in patients at high risk of developing OHSS with many ovarian follicles after ovarian stimulation, the use of an appropriate gonadotrophin dose is very important in maintaining oocyte quality and embryo developmental potential.
In conclusion, the present study showed that in patients with a high risk of developing OHSS, D5ET can decrease the multifetation rate and the incidence of moderate or severe OHSS, but does not affect the clinical pregnancy rate or live birth rate. Patients with a high risk of developing OHSS who are relatively young, who receive an appropriate gonadotrophin dose for ovarian stimulation and who undergo IVF are suitable candidates for blastocyst transfer. However, these conclusions require further confirmation in larger studies.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by the National Science Foundation (31271605), the Innovation Talent Grant from the Health Department of Henan Province and the Youth Fund from the First Affiliated Hospital of Zhengzhou University.
