Abstract
Background
The inherent brittleness of calcium phosphate cement has limited its clinical application. Polyvinyl alcohol fibers have been demonstrated to enhance the tensile toughness of concrete matrices. However, only a few international studies have investigated the modification of calcium phosphate cement using polyvinyl alcohol fibers. These studies have predominantly focused on the macroscopic and microscopic mechanical properties, often neglecting comprehensive evaluations of the composites’ osteoconductivity, degradation rate, and osteogenic properties.
Objective
To evaluate the cellular biocompatibility, bending strength, elastic modulus, and fracture toughness of polyvinyl alcohol/calcium phosphate cement composites, as well as to assess their degradability and osteoconductivity, providing a theoretical basis for their potential clinical application.
Method
Polyvinyl alcohol/calcium phosphate cement composite cement was prepared by incorporating polyvinyl alcohol fibers into the calcium phosphate cement solid phase. The degradability of the composite material was assessed via in vitro immersion experiments. Biocompatibility was evaluated by observing cell morphology and growth, using the cell counting kit-8 for assessing the cell viability and performing live/dead fluorescent staining. The impact of the composite on cellular alkaline phosphatase activity was determined using alkaline phosphatase assays. Finally, the composite material’s bending strength, elastic modulus, and fracture toughness were measured through three-point bending tests.
Keywords
Background
With the rising number of osteoporosis patients in China, addressing bone defects resulting from osteoporotic fractures has become a pressing challenge in the field of orthopedics. Bone transplantation is one of the effective solutions, with recent data indicating that bone transplantation is the second most common type of transfusion worldwide and the second most common form of tissue transplantation. 1 Currently, materials used for bone defect repair include autologous bone, xenogeneic bone, metal materials, bioceramics, polymers, and various composites. 2 While different materials exhibit robust bone repair capabilities, an ideal clinical bone repair material is yet to be established.
Calcium phosphate is the most critical inorganic component of bone tissue, and calcium phosphate cement (CPC) boasts superior biocompatibility, osteoconductivity, moldability, and injectability. 3 Consequently, CPC is considered one of the most promising bone graft materials for clinical application. However, the mechanical strength of CPC is usually insufficient to provide the support required for bone regeneration.
Polyvinyl alcohol (PVA) fiber, derived from PVA resin, is an organic synthetic fiber characterized by hydroxyl groups in its molecular chain, enabling it to form strong bonds with inorganic adhesive materials. 4 Incorporating PVA fibers enhances concrete’s bending strength (flexural strength (S)), flexibility, and toughness, 5 making it widely used in industries such as those involving porous materials and industrial cement.6,7 PVA fiber is non-toxic to humans, exhibiting good dispersion, hydrophilicity, high tensile strength, and a high elastic modulus. 8
Therefore, this study proposes to combine PVA fiber, known for its high elastic modulus, with CPC, recognized for its brittleness, to develop a composite cement with enhanced mechanical properties while preserving the inherent biocompatibility, osteoconductivity, and degradability of CPC. This type of research involves a multidisciplinary experimental design, requiring simultaneous evaluation of both mechanical (e.g. compressive strength, flexural strength) and biological (e.g. cytotoxicity, cell proliferation) properties of the material. Thus, although there has been considerable research focused on either mechanical properties or biocompatibility, very few studies have comprehensively covered both aspects. Furthermore, most studies on PVA/CPC have employed relatively low and single PVA fiber contents. For instance, Kucko 9 and Paknahad 10 used a PVA fiber mass fraction of only 2.5% in their studies. Without comparative tests of PVA fibers at different mass fractions, it is impossible to comprehensively explore the influence of PVA fiber content on the mechanical strength of CPC. In this study, we established three experimental groups with different fiber mass percentages and compared them with a negative control group. Our aim was to address the deficiencies in existing research and provide more comprehensive and accurate reference data for the further application of PVA/CPC materials in fields such as bone repair.
Materials and methods
Main instruments and equipment
CPC (Shanghai Ruibang Biomaterials Co, China), electronic universal testing machine (Instrong 5567, USA), microplate reader (Thermo Fisher Scientific, USA), and Zeiss inverted fluorescence microscope (Carl Zeiss AG, Germany) were used.
Cell selection
Mouse bone marrow mesenchymal stem cells (mBMSCs) were provided by Wuhan Procell Life Science & Technology Co., Ltd.
Experimental preparation
Preparation of composite cement samples with PVA/CPC
The study included three experimental groups and one control group, each with a distinct CPC solid phase composition. PVA fibers were incorporated into each experimental group at specified mass ratios. Notably, 6-mm PVA fibers demonstrated optimal dispersion, flexural strength, and fracture toughness in cement-based materials. 9 A soluble phosphate aqueous solution was selected as the curing liquid, offering advantages of non-toxicity and neutral pH compared with highly alkaline pure water or strongly acidic dilute hydrochloric acid. The solid and liquid phases were quickly mixed with a metal spatula to ensure homogeneity. The resulting slurry was then poured into custom molds and allowed to cure at room temperature for 24 h. After curing, the samples were demolded. The compositions of the CPC solid phase, PVA fibers, and liquid phase, along with the corresponding sample designations, are detailed in Table 1.
Composite bone cement formulation.

CPC: calcium phosphate cement; PVA: polyvinyl alcohol.
The table presents the formulations of composite bone cement, with the solid phase consisting of varying mass fractions of CPC and PVA fibers (6 mm in length), and the liquid phase being a phosphate solution. The liquid:solid ratio was maintained at 1:2.
Preparation of the composite material extracts
Following the formulations detailed in Table 1, bone cement paste was prepared and transferred into custom-made silicone molds. After 24 h of curing, bone cement samples were obtained. These samples were washed with phosphate-buffered saline (PBS) for 15 min, and then sterilized in an autoclave at high temperature and pressure (0.15 MPa and 121°C, respectively) for 15 min. The sterilized samples were dried in an oven at 37°C.
The dried samples were immersed in an extraction medium consisting of GIBCO’s alpha-minimum essential medium (α-MEM) with 10% fetal bovine serum at a ratio of 10 mL/g. The samples were incubated at 37°C in a 5% carbon dioxide (CO2)–cell incubator (Thermo Fisher, USA) for 24 h. The supernatant was then extracted via centrifugation, filtered using a 0.22-μm filter (Millipore, USA) in a sterile workbench, and stored in a 50-mL centrifuge tube at 4°C for use within 24 h.
In vitro cell experiments
Evaluation of composite compatibility with mouse mBMSCs
Given that CPC has been widely used clinically and is shown to have no significant cytotoxicity, this study focused primarily on the effects of PVA fiber on cell growth and morphology. Therefore, only the 5% PVA/CPC group with the highest PVA fiber content was selected for analysis.
Sterilized circular composite blocks were taken and washed in PBS by shaking for 15 min. These blocks were soaked in α-MEM for 24 h and placed in 24-well plates. Cells were co-cultured with the composites at a concentration of 1 × 105 cells per well, with five composite wells set up. The cultures were incubated in a 37°C, 5% CO2 incubator. The medium was replaced every 24 h, and the cells were cultured for 3 days before being photographed.
The co-culture of mBMSCs with the real-time extracts of the composites and the composites themselves was carried out using identical procedures.
The mBMSCs proliferation activity was determined by cell counting kit-8 (CCK-8) assay
A cell suspension of 5 × 104 cells/mL was seeded in 96-well plates, with 100 μL per well. The experimental groups included 1% PVA/CPC, 2.5% PVA/CPC, and 5% PVA/CPC composite extracts. In comparison, the negative control group contained 0% PVA/CPC material extract, and the blank control group comprised α-MEM complete culture medium with 10% fetal bovine serum. After 24 h of culture, the original culture medium was aspirated, and the cells were treated with the respective real-time extraction solutions. This marked the first day of cell proliferation assessments.
On days 1, 2, and 3, 10 μL of CCK-8 reagent was added to each well. The absorbance was measured at a wavelength of 450 nm using a microplate reader to determine cell viability and proliferation.
Cytotoxicity was evaluated by live/dead fluorescent staining
Cells were seeded in 24-well plates at a concentration of 1 × 105 cells per well and cultured in an incubator. Sterilized composite samples were placed into Transwell chambers, which were then positioned in the corresponding wells, ensuring that the medium from each well contacted the composite sample (Figure 1). The Transwell chambers and composite blocks were removed at the designated time points. The cells at the bottom of each well were stained using live/dead fluorescent staining reagent to evaluate the cytotoxicity of the composite materials.

Cytotoxicity of each experimental group was detected using live/dead fluorescent staining (n ≥ 6).
Evaluation of the mobility of composites and mBMSCs cells
Cell scratch test
mBMSCs were cultured in 6-well plates with a cell suspension concentration of 2 × 105 cells/mL. On the second day, a 100-μL pipette tip was used to create three vertical scratch lines in each well, ensuring consistent, cell-free zones. The scratched cells were washed away with PBS. Subsequently, α-MEM without fetal bovine serum was added, and the cells were cultured in a 37°C, 5% CO2 incubator.
At the designated time points, images of the cells were captured and labeled. The cell migration rate was calculated using Image J software. The cell migration rate was determined using the following formula:
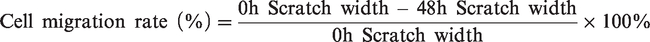
Transwell migration experiments
A cell suspension with 5 × 105 cells/mL concentration was prepared using serum-free α-MEM. An 8.0-μm Transwell chamber was selected and placed into a 24-well plate. In the lower chamber, 300 μL of serum-free extract from each experimental group and serum-free medium from the control group were added. In the upper chamber of each well, 200 μL of the serum-free cell suspension was added.
After 24 h of culture, the medium was discarded, and the cells were washed thrice with PBS. Next, 200 μL of 4% paraformaldehyde was added to the lower chamber of each well to fix the cells for 30 min, followed by another PBS wash. The cells were stained with 0.1% ammonium oxalate crystal violet for 30 min in the lower chamber of each well. Non-migrated cells in the upper chamber were gently removed with a medical cotton swab. The migrated cells were then observed and photographed under an inverted microscope.
Evaluation of the osteogenic properties of the composite and mBMSCs in vitro
Osteoinductive and osteoconductive properties are critical for bone regeneration. Osteoinduction is the ability of progenitor cells to differentiate into osteoblast cell lines,11,12 while osteoconduction refers to the ability of phalanges to proliferate and integrate within materials. 13
Cells were seeded at a concentration of 1 × 105 cells per well and cultured in an incubator. The osteogenic induction medium was changed every other day. After 1 week of induction culture, the cells were washed with PBS. Subsequently, 25 μL of cell lysis buffer was added to each well, and the plates were placed in a 4°C refrigerator for 20 min. The cells on the surface of the composite material were then scraped off and collected into 1.5-mL EP tubes. The collected samples were centrifuged at 4°C for 10 min at 12,000 r/min. The resulting supernatant was collected as the sample solution for further analysis.
The activity of alkaline phosphatase (ALP) was assessed using the Bicinchoninic Acid Protein Assay kit (Thermo Fisher Scientific, USA) to determine the sample protein concentration. The ALP activity per unit protein content was then calculated using the ALP detection kit.
Qualitative staining of mBMSCs was performed using 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium (BCIP/NBT) chromogen. Cells were seeded at a concentration of 1 × 105 cells per well in a 12-well plate and cultured in an incubator. Following the sterilization of the composite samples, the osteogenic induction medium was changed, and the Transwell chambers containing the composites were placed into the corresponding wells, to ensure that the medium did not contain the composite samples.
After 1 week of osteogenic induction, the cells were fixed with 4% paraformaldehyde and washed with PBS. BCIP/NBT developer was added, and the cells were incubated at room temperature for 30 min. The stained cells were observed and photographed under an inverted microscope.
Material testing and characterization methods
Mechanical strength characterization of the composites
The failure of bone cement typically initiates from flexural microcracks, making S a critical predictor of clinical performance.
14
The International Bone Cement Research Society (ICRS) recommends flexural strength (measured using three-point bending) as a core mechanical property index for bone cement.
15
Consequently, the mechanical strength of the CPC and composite was tested using an electronic universal testing machine (Instron 5567, Instron, USA) with a loading speed of 0.5 mm/min and a span of 15 mm (Figure 2). The test samples were those cured for 24 h at room temperature, with dimensions of 25 mm in length, 5 mm in width, and 4 mm in height. The S, Young’s modulus (M), and fracture work (work of fracture (WOF)) were calculated using formulas (1), (2), and (3) respectively. Five samples were measured in each group.


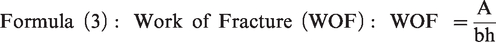
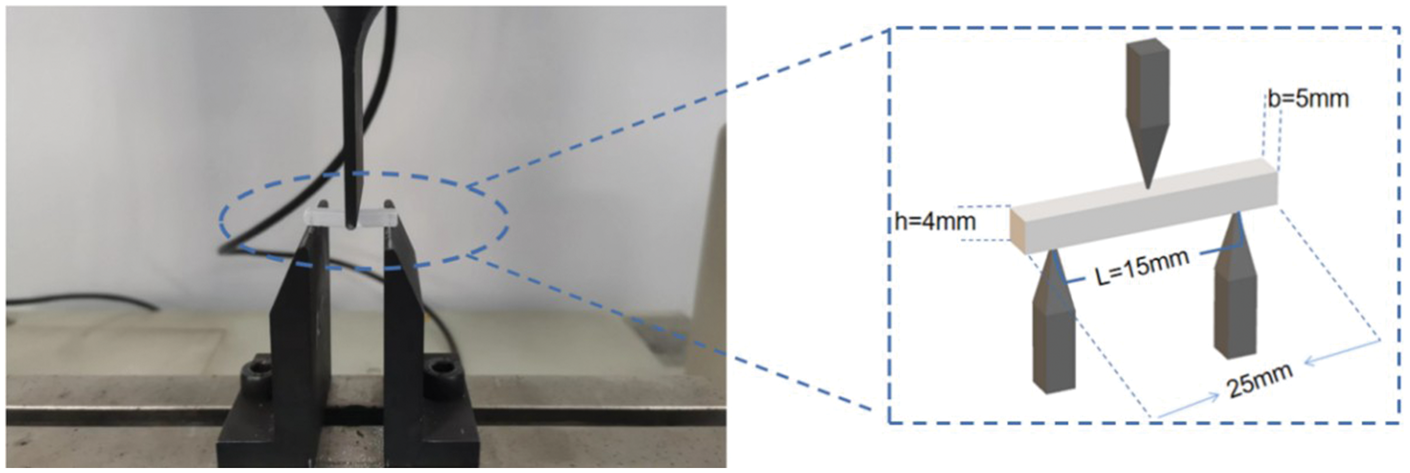
Three-point bending experiments were conducted to detect the mechanical strength of the composite material. The mechanical strength of the CPC and composite was tested using an electronic universal testing machine with a loading speed of 0.5 mm/min and a span of 15 mm. The test samples were those cured for 24 h at room temperature, with dimensions of 25 mm in length, 5 mm in width, and 4 mm in height. CPC: calcium phosphate cement.
In the stress–strain curve analysis, p represents the maximum bending stress load, L is the sample span, b is the sample width h, is the sample height, m is the slope of the linear elastic region of the stress–strain curve, and A is the area under the stress–strain curve.9,16,17 M is a physical quantity that reflects the ability of a composite material to resist elastic deformation. 18 A higher M value indicates a greater resistance to elastic deformation, corresponding to increased material stiffness. This property is a crucial performance parameter for materials.
In vitro degradation rate characterization of the composites
Dried composite material samples from each group were weighed and soaked in PBS (pH = 7.35) at a mass ratio of 30 mL/g in plastic containers. These containers were then placed in a 37°C electric blast drying oven. At fixed daily intervals, the samples were removed, washed several times with ultrapure water, and dried in a 60°C oven to measure the weight loss rate.
The weight loss rate was calculated using the following formula:

Statistical methods
Quantitative test results were analyzed using GraphPad Prism 9.5.0 (USA) software. Quantitative data are presented as mean ± SD values, with the number of parallel samples indicated in the figure legends. Significance between the two groups was determined using Student’s t-test, with p < 0.05 indicating statistical significance.
The unpaired t-test was used to assess the S, M, and WOF between the CPC control and PVA/CPC experimental groups. One-way analysis of variance was employed to analyze cell proliferation, cytotoxicity, cell migration rate, and osteogenic ability.
Result
Morphological observations of mBMSCs after co-culture with composites and their extracts
After 3 days of co-culture of 5% PVA/CPC composite samples with mBMSCs, the growth of cells at the edge of the mBMSCs normal culture group (Figure 3(a)) and the 5% PVA/CPC composite samples (Figure 3(b)) showed no significant differences. Cells were in good contact with the composite edges and grew tightly to the edge of the material, exhibiting normal spindle-shaped morphology. Similarly, cells in the real-time extract experimental (Figure 3(c)) and control groups displayed comparable growth and were in good condition.

Cell morphology and growth of each group after 72 h of culture. (a) α-mBMSCs cultured in normal culture with complete medium. (b) mBMSCs co-cultured with 5% PVA/CPC composite. (c) mBMSCs cultured with 5% PVA/CPC composite extract. α-mBMSCs: alpha-mouse bone marrow mesenchymal stem cells; CPC: calcium phosphate cement PVA: polyvinyl alcohol.
In vitro cell proliferation of PVA/CPC compound in cement
The cell proliferation in the extract cultures of each group is shown in Figure 4(a). The number of cells increased with extended culture time, indicating that the composite cement exhibited no obvious cytotoxicity. There were no significant differences in the cell numbers between the blank control, CPC control, and experimental groups at 24 h. At 48 h, cell proliferation in the experimental and CPC control groups was slightly lower than that in the blank control groups; however, the differences were not statistically significant (p > 0.05). At 72 h, the cell numbers in the experimental and CPC control groups were significantly lower than those in the blank control group, suggesting that CPC bone cement inhibits cell proliferation to some extent. However, there were no significant differences in the cell numbers between the experimental and CPC control groups at 24, 48, and 72 h (p > 0.05), indicating that the incorporation of PVA fibers did not increase the cytotoxicity of CPC bone cement. The viability of each group was calculated based on the optical density value (Figure 4(b)), demonstrating that the cell proliferation activity of each group was close to 100%.
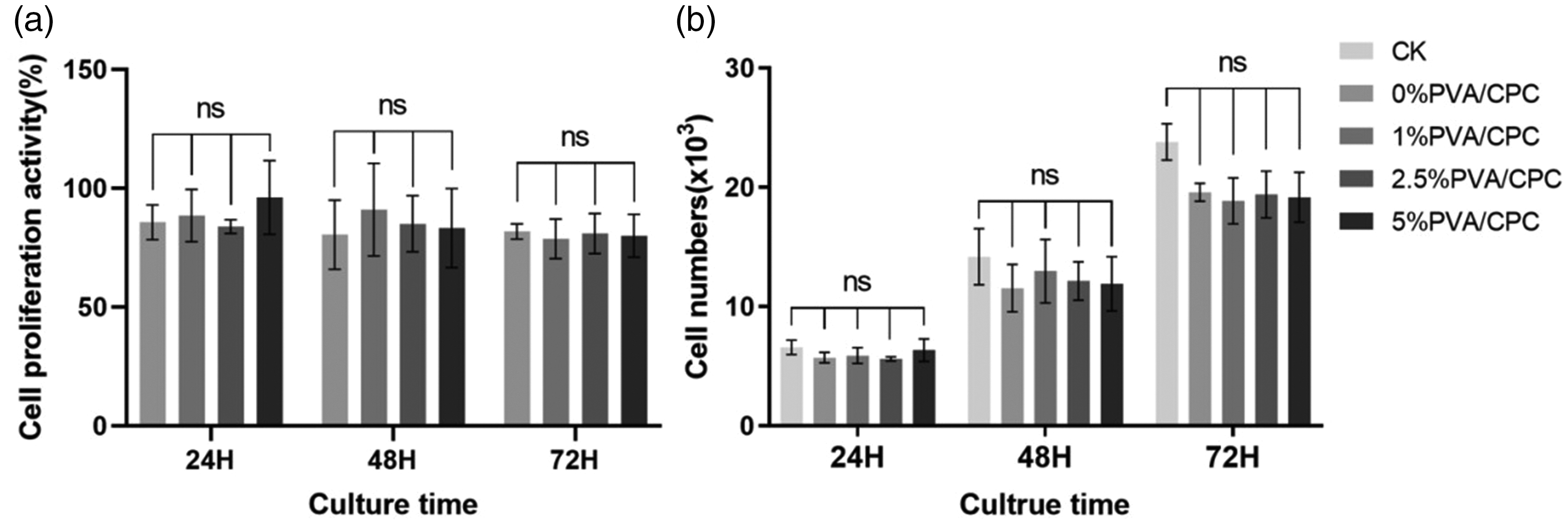
CCK-8 reagent was used to detect cell proliferation in the blank control, CPC control, and experimental groups. Statistical analyses for multiple group comparisons were conducted using t-test. (a) Cell proliferation on CPC and composite cement wafers with different PVA fiber content. (b) Proliferation viability of cells on CPC and composite cement wafers with varying content of PVA fiber. CK served as the control group (p > 0.05, n = 6). CCK-8: cell counting kit-8; CPC: calcium phosphate cement; PVA: polyvinyl alcohol.
In vitro cytotoxicity profile of PVA/CPC composite bone cement
Cell proliferation in the blank, CPC control, and all experimental groups was assessed using the CCK-8 reagent. Figure 4(a) shows the proliferation of cells on CPC and composite cement wafers with varying PVA fiber contents, while Figure 4(b) illustrates the cell proliferation viability on these wafers.
Cell migration behavior of PVA/CPC composite bone cement was examined by scratch assay
The migration area ratio of each group was analyzed using the ImageJ software after 48 h (Figure 5). The cell migration rate was relatively low for samples with 1% and 5% PVA content. However, statistical analysis revealed that the differences in the migration rates between each experimental group and the CPC control group were not statistically significant (p > 0.05).

The cell migration rate of each group was assessed at 48 h with the cell scratch assay. (a) Image J was used to precisely measure the cell-free area at different time points and (b) quantitative analysis of cell mobility shift in each group cultured for 48 h (p > 0.05, n = 9).
Transwell assay to test the cell migration behavior of PVA/CPC composite bone cement
Figure 6 shows that the lower chamber’s cell density gradually increased with extended culture time. There was no significant difference in the density of cells that migrated to the lower chamber between the CPC control group and each experimental group, consistent with the results of the cell scratch assay.

The effect of PVA fibers on the migration ability of mBMSCs was determined using the Transwell method (n ≥ 6). PVA: polyvinyl alcohol; mBMSCs: mouse bone marrow mesenchymal stem cells.
In vitro osteogenic properties of PVA/CPC composite bone cement
As shown in Figure 7(a), the ALP activity in each experimental group was not statistically significant (p > 0.05). The qualitative ALP staining (Figure 7(b)) was consistent with the quantification results, showing no significant difference in ALP staining intensity between the CPC control group and each experimental group.
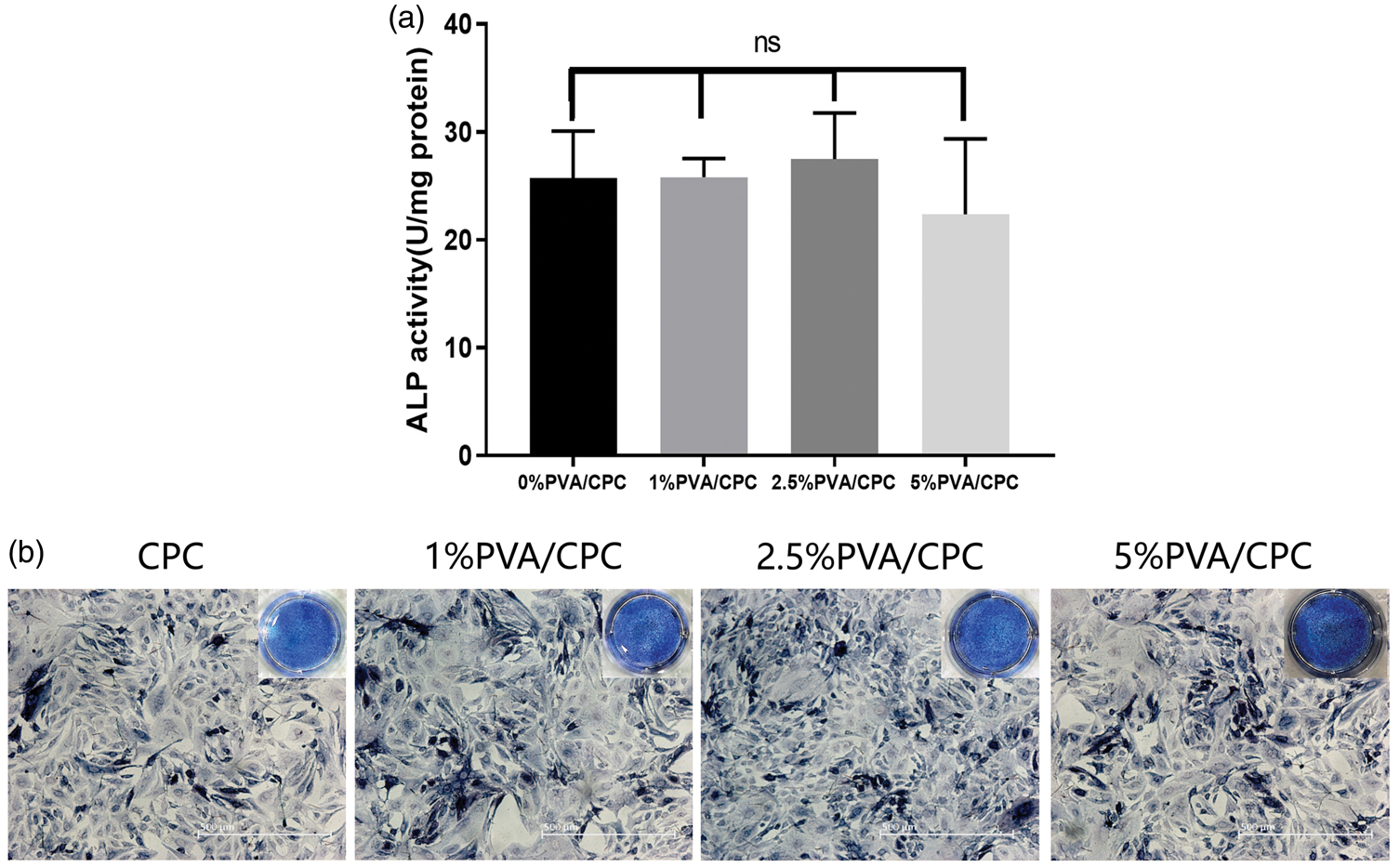
Comparison of ALP activity between the CPC control group and each experimental group. (a) Quantitative ALP detection and (b) qualitative staining for ALP (p > 0.05, n = 6). ALP: alkaline phosphatase; CPC: calcium phosphate cement.
Mechanical strength of the PVA/CPC composite bone cement
As shown in Figure 8(b), incorporating PVA fibers significantly improved the S of CPC, with S increasing proportionally to the PVA fiber content.

Effect of different PVA fiber mass scores on the representative stress–strain curve (a), CPC bending strength (b), Young’s modulus (c), and fracture work (d). ap < 0.05; bp < 0.01; and cp < 0.001 indicate statistically significant differences (n ≥ 3).
Figure 8(c) illustrates that the CPC control group exhibited the highest M, indicating that CPC bone cement has high stiffness and brittleness. In contrast, the M value of experimental groups containing PVA fibers was 50% of that in the control group, suggesting that PVA fibers enhance the toughness of CPC bone cement and reduce its brittleness.
In Figure 8(a), the stress–strain curve of the CPC control group displayed a clear brittle fracture pattern, while the experimental groups with PVA fibers showed evident tough fracture patterns. The area under the curve, corresponding to the fracture toughness (WOF) of CPC, increased with higher PVA fiber content, indicating enhanced fracture toughness.
The WOF for each group was calculated by substituting the area under the stress–strain curve into Formula (3). 15 As depicted in Figure 8(d), the WOF of the fibreless CPC control group was very low, while that of the 5% PVA/CPC experimental group was nearly 250 times higher than that of the control group. A summary of the S, bending modulus, and toughness of the CPC control group and each experimental group is provided in Table 2.
Bending strength, Young’s modulus, and fracture work (mean ± SD) of the CPC control group and each experimental group.

CPC: calcium phosphate cement; PVA: polyvinyl alcohol.
Statistical significance was observed between each experimental group and the control group.
p < 0.05; bp < 0.01; cp < 0.001.
In vitro degradation of PVA/CPC composite bone cement
As shown in Figure 9, the composite cement degraded rapidly within the first 10 days of soaking, and the weight loss rate stabilized after day 13. Incorporating PVA fibers reduced the weight loss rate of the composite cement. When the cement samples were immersed in PBS for 10 days, the weight loss rates for 0% PVA/CPC, 1% PVA/CPC, 2% PVA/CPC, 2.5% PVA/CPC, and 5% PVA/CPC were 11.93% ± 0.6714%, 10.97% ± 0.6083%, 9.864% ± 0.4957%, 9.366% ± 0.6118%, and 8.522% ± 0.5592%, respectively.

Weight loss rates of 1% PVA/CPC, 2.5% PVA/CPC, and 5% PVA/CPC samples immersed in PBS (n ≥ 3). PVA: polyvinyl alcohol; CPC: calcium phosphate cement; PBS: phosphate-buffered saline.
Discussion
In this study, we investigated the effects of varying mass percentages of PVA fibers on the mechanical properties of CPC. The results indicated that the incorporation of PVA fibers significantly enhanced the flexural strength, toughness, and WOF of CPC, while simultaneously reducing its M value. These findings are consistent with existing literature and further confirm the potential of fiber-reinforced composites in the field of bone repair.17,19–21
The control CPC group exhibited typical brittle fracture behavior, with a nearly linear stress–strain curve that fractured immediately after reaching the yield point, showing a flexural strength of only 1.244 ± 0.0804 MPa. However, after the incorporation of PVA fibers, the composite material displayed significantly tough fracture behavior. 19 The 1% PVA/CPC group demonstrated a flexural strength of 4.061 ± 0.7505 MPa, 3.2 times that of the control group; the 5% PVA/CPC group reached a flexural strength of 5.758 ± 0.8661 MPa, which was 4.6 times that of the control group. These data suggest that the addition of PVA fibers can significantly improve the flexural strength of CPC, and as the fiber content increases, the flexural strength further increases. However, the difference between the 2.5% and 5% PVA/CPC groups was not significant, suggesting that a fiber content of 2.5% achieves the maximum enhancement in flexural strength for the composite cement. This is consistent with conclusions from other studies, indicating that the mechanical properties of composites will reach saturation at a specific fiber content. 22
The incorporation of PVA fibers significantly reduced the M value of CPC. In the 1% PVA/CPC group, the M value was the lowest at 1008 ± 56.12 MPa, which represents a 60% lower value than that in the control group. This result aligns with findings reported in the literature, suggesting that the addition of fibers increases the toughness of the material by reducing the rigidity of the matrix. 23 However, when the PVA fiber mass fraction exceeded 1%, there was no further reduction in the M value, likely due to the aggregation of fibers within the matrix. 24 These aggregates failed to distribute uniformly within the CPC matrix, leading to a decrease in the frictional interface between the fibers and the matrix, thereby limiting the elastic deformation capacity of the composite. 22
The most significant improvement was observed in the WOF of the composite material. The WOF of the control group was only 0.0478 ± 0.0074 KJ/m2, while that of the 1% PVA/CPC group was 119.1 times that of the control group, significantly mitigating the brittleness of CPC. The WOF of the 5% PVA/CPC group reached 11.92 ± 4.66 KJ/m2, nearly 250 times that of the control group. Similarly, some researchers 9 have incorporated PVA fibers into CPC to create a composite bone cement, achieving a nearly 400-fold increase in fracture toughness. This enhancement is attributed to further surface modification of the PVA fibers, which strengthens the interfacial friction between the fibers and CPC matrix, thereby significantly improving the fracture resistance of the composite bone cement. This significant enhancement is attributed to the crack-bridging and energy-dissipation effects of PVA fibers, 9 which transformed the fracture mode of CPC from brittle to tough, providing a stable mechanical environment for bone regeneration. Similar studies have also demonstrated that fiber-reinforced composites can improve the fracture toughness of materials by hindering crack propagation.9,25,26
Scanning electron microscopy examination of fracture surfaces within the PVA/CPC composite revealed that their morphological characteristics exhibited marked variations dependent on fiber content, as detailed in the Supplementary Materials.
In summary, this study demonstrates that the incorporation of PVA fibers significantly improves the mechanical properties of CPC, broadening its application prospects in bone repair. In addition, we evaluated the degradation rate of the composite materials in a simulated physiological environment using in vitro immersion experiments. Generally speaking, the incorporation of PVA fibers reduces the weight loss rate of the composite bone cement to a certain extent. However, the differences in the weight loss rates among the composite material groups are relatively small, and the trends of changes in the weight loss rate are similar. This may be because PVA fibers are relatively stable, resistant to acids and alkalis, and not easily degradable. 27 Moreover, the mass proportion of PVA fibers in the composite materials is relatively small; therefore, their influence on the degradation rate of the composite materials is limited.
Although the above-mentioned research has clarified the enhancement of PVA fibers on the mechanical strength of CPC bone cement and their influence on the degradability, we cannot ignore the changes in the biocompatibility of the composite materials. In this study, direct/indirect contact experiments confirmed that the PVA/CPC composite materials are non-cytotoxic in short-term culture and support the normal proliferation and migration of mBMSCs, consistent with the in vivo inert characteristics of PVA reported by Petre 28 and Kucko. 9 Notably, although PVA fibers are nondegradable, their uniform dispersion in the CPC matrix does not trigger an inflammatory response and retains the inherent osteoconductivity of CPC, as demonstrated by scratch and Transwell experiments. However, the fact that the materials lack osteogenic induction ability29,30 suggests that future optimization can be achieved through surface functionalization, such as bone morphogenetic protein-2 loading.
In addition, although the three-point bending test is widely used for material strength detection, it has certain limitations. Bone cement mainly bears axial compressive loads in spinal surgery, with compressive strength being the core index. 14 Therefore, in future research, we will improve the compression test. We will also explore the optimization of the interface between the fibers and CPC matrix as well as the influence of different fiber types and morphologies on the properties of composite materials, with the aim of developing higher-quality bone repair materials.
Conclusion
Incorporation of PVA fibers led to a comprehensive enhancement of the mechanical properties of CPC. This modification significantly improved the S and M values of CPC while markedly enhancing its fracture toughness, thereby addressing the inherent brittleness of CPC. Moreover, the addition of PVA fibers augmented the mechanical strength of CPC without compromising its inherent biocompatibility and osteoconductivity. Furthermore, this modification did not affect the osteogenic activity or the degradability of CPC bone cement.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251389734 - Supplemental material for Preparation and properties of polyvinyl alcohol/calcium phosphate composite bone cement
Supplemental material, sj-pdf-1-imr-10.1177_03000605251389734 for Preparation and properties of polyvinyl alcohol/calcium phosphate composite bone cement by Shengliang He, Yi He, Xuancheng Fang, Shiwen Liao, Jinhuang Xu, Minhui He, Cong Zheng, Lifen Zhou, Dawei Xu and Jianrong Huang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251389734 - Supplemental material for Preparation and properties of polyvinyl alcohol/calcium phosphate composite bone cement
Supplemental material, sj-pdf-2-imr-10.1177_03000605251389734 for Preparation and properties of polyvinyl alcohol/calcium phosphate composite bone cement by Shengliang He, Yi He, Xuancheng Fang, Shiwen Liao, Jinhuang Xu, Minhui He, Cong Zheng, Lifen Zhou, Dawei Xu and Jianrong Huang in Journal of International Medical Research
Footnotes
Acknowledgment
Not applicable.
Author contributions
Conceptualization: He Shengliang and Huang Jianrong; Methodology: He Shengliang and Xu Dawei; Software: He Shengliang; Validation: He Shengliang, Xu Dawei, and He Yi; Formal Analysis: He Shengliang and He Yi; Investigation: He Shengliang and Fang Xuancheng; Resources: He Shengliang; Data Curation: He Shengliang and Liao Shiwen; Writing–Original Draft Preparation: He Shengliang; Writing–Review & Editing: He Shengliang, Huang Jianrong, and Xu Dawei; Visualization: He Shengliang and Xu Jinhuang; Supervision: He Shengliang, Zheng Cong, and He Minhui; Project Administration: He Shengliang and He Minhui; Funding Acquisition: Huang Jianrong.
Availability of supporting data
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare no conflict of interest.
Funding
This research was funded by the Guangzhou Science and Technology Plan Project, grant number 201707010081.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
