Abstract
Angiomyofibroblastoma is a rare benign mesenchymal tumor typically found in the genital region of adult females, but its occurrence in the male scrotum is exceptionally uncommon. This report presents two cases of angiomyofibroblastoma in the male scrotum to enhance the understanding of this rare condition. In the first case, a man in his early 60s with a recent rectal cancer diagnosis was found to have a palpable right scrotal mass. Imaging suggested metastasis, but radical resection and pathological analysis confirmed angiomyofibroblastoma, with no evidence of testicular malignancy. The patient remained tumor-free for 7 years. In the second case, a man in his early 40s presented with a nontender, slowly enlarging right inguinal mass extending into the scrotum. Surgical resection revealed a 6 × 4-cm angiomyofibroblastoma, with no recurrence for 7 years. These cases underscore the importance of considering angiomyofibroblastoma in the differential diagnosis of scrotal masses in males. Accurate diagnosis relies on understanding the clinical, imaging, and histopathologic features of angiomyofibroblastoma to ensure appropriate management and favorable outcomes.
Introduction
Angiomyofibroblastoma (AMF) is a rare, well-circumscribed, blood vessel–rich benign myofibroblastoma. Most AMF cases involve the vulva, labia majora, vagina, cervix, and perineum of middle-aged women. 1 However, its occurrence in males is exceedingly rare, with only a few cases documented in the literature. 2 Owing to its rarity, nonspecific clinical and imaging features, and overlap with other soft tissue tumors, AMF poses significant diagnostic challenges, often leading to preoperative misdiagnoses as malignant entities such as sarcomas, metastatic tumors, or germ cell tumors.2,3 This can result in unnecessary surgeries, highlighting the need for heightened awareness and confirmatory histopathological evaluation. In this report, we present the imaging findings, clinicopathological profiles, and immunohistochemical results of two cases of AMF originating in the scrotum, which were managed at Guangdong Provincial People’s Hospital in 2017 and 2018, respectively.
Case presentation
The reporting of this study conforms to the Case Report (CARE) guidelines. 4 Institutional review board approval was obtained from the Ethics Committee of Guangdong Provincial People’s Hospital (Guangzhou, China; approval number: KY2024-726-01; date: 24 September 2024). Written informed consent for treatment and publication was obtained from both patients. We have deidentified all patient details.
Case 1
In early 2017, a man in his early 60s was referred to our hospital for the diagnosis of rectal cancer. Physical examination revealed a palpable mass, approximately 6 × 7 cm in size, with mild tenderness in the right scrotum. The levels of the tumor markers carcinoembryonic antigen (CEA), alpha-fetoprotein (AFP), cancer antigen (CA)-125, and CA19-9 were all within normal limits. Scrotal ultrasonography revealed a wide range of slightly hyperechoic areas in the right scrotum extending into the pelvis (Figure 1(a)). Pelvic magnetic resonance imaging (MRI) revealed a large soft tissue mass in the right scrotum, which was isointense on T1-weighted images (Figure 1(b)) and hyperintense on T2-weighted images (Figure 1(c)). Enhancement scans demonstrated significant enhancement, with a well-defined lesion boundary measuring approximately 56 × 70 mm (Figure 1(d)). Both testicles appeared normal in morphology. MRI findings suggested a lesion in the right scrotum, raising suspicion of a metastatic tumor. An enhanced pelvic computed tomography (CT) scan also revealed a suspected malignant testicular tumor on the right side. Subsequently, radical testicular resection and removal of the scrotal mass were performed. Intraoperatively, the right testis was poorly demarcated from a tumor measuring approximately 6 × 7 cm, which had an uneven surface. Postoperative pathological diagnosis confirmed the following. First, the scrotal mass was consistent with AMF. Second, no tumors were detected in the right testis or epididymis. Microscopically, the tumor cells were spindle-shaped, with minimal cellular variation and rare mitotic figures. Abundant thin-walled vessels were present within the collagenous stroma (Figure 1(e)). Immunostaining revealed that the tumor cells were positive for desmin, CD34, estrogen receptor, and progesterone receptor and negative for S-100 and smooth muscle actin (SMA) (Figure 2(a) to (g)). The pathological diagnosis was consistent with AMF. The patient experienced an uneventful recovery following the surgery. After 7 years of telephone follow-up, the patient remained asymptomatic with no evidence of tumor recurrence.

Ultrasonography, magnetic resonance imaging, and photomicrograph in case 1. (a) Ultrasound image revealed a wide range of slightly hyperechoic areas in the right scrotum. (b) Axial T1-weighted image revealed a large soft tissue mass in the right scrotum (indicated by the red arrow), which was isointense. (c) Axial T2-weighted image revealed a large soft tissue mass (indicated by the red arrow) in the right scrotum, which was hyperintense. (d) Coronal gadolinium-enhanced T1-weighted image demonstrated significant enhancement of the lesion (indicated by the red arrow), with a well-defined lesion boundary measuring approximately 56 × 70 mm and (e) The histopathological examination revealed spindle-shaped tumor cells with minimal cellular atypia and rare mitotic figures. Abundant thin-walled vessels were observed within the collagenous stroma.

Immunohistochemical staining of angiomyofibroblastoma. (a–g) Case 1: tumor cells were positive for CD34 (a), desmin (b), estrogen receptor (c), Ki-67 (5%+) (d), and progesterone receptor (e), but negative for S-100 (f) and smooth muscle actin (SMA) (g). (h–l) Case 2: tumor cells were positive for CD34 (h) and desmin (i), showed low proliferative activity with Ki-67 (1%+) (j), and were negative for S-100 (k) and SMA (l).
Case 2
In early 2018, a man in his early 40s was admitted for a right inguinal gradually enlarging mass that had been present for 4 years. On examination, a 6 × 5-cm mass was identified in the right inguinal region, extending into the scrotum. The mass was soft and nontender, and the transillumination test was negative. The levels of tumor markers, including CEA, AFP, prostate-specific antigen (PSA), β-human chorionic gonadotropin (HCG), CA15-3, CA19-9, and CA-125, were within normal ranges. Scrotal ultrasonography revealed a hyperechoic area measuring 6.5 × 2.3 cm above the right testicular sheath cavity, characterized by poorly defined borders and uneven echogenicity. This suggested the presence of an inguinal hernia. Pelvic MRI revealed a space-occupying lesion, approximately 5.1 × 2.2 cm in size, in the right scrotum. The lesion had clear borders and appeared hypointense on T1-weighted images (Figure 3(a)) and hyperintense on T2-weighted images (Figure 3(b)). MRI also revealed multiple shadows of hollow blood vessels. Enhancement scans revealed uneven enhancement (Figure 3(c)). The right scrotal lesion was subsequently excised. Intraoperative findings revealed a scrotal mass measuring approximately 6 × 4 cm, which did not invade the testicle. Additionally, a separate mass, approximately 3 cm in size, was noted outside the spermatic cord (Figure 3(d)). Microscopically, the tumor consisted of spindle-shaped cells arranged in hypocellular and hypercellular regions. The tumor was characterized by an abundance of thin-walled vessels distributed throughout the collagenous stroma (Figure 3(e)). Immunohistochemical staining revealed that the tumor cells were positive for vimentin, desmin, and CD34 but negative for S-100 (Figure 2(h) to (l)). Pathology confirmed the presence of AMF. Postoperatively, the patient had a favorable recovery and has remained recurrence-free for 7 years.
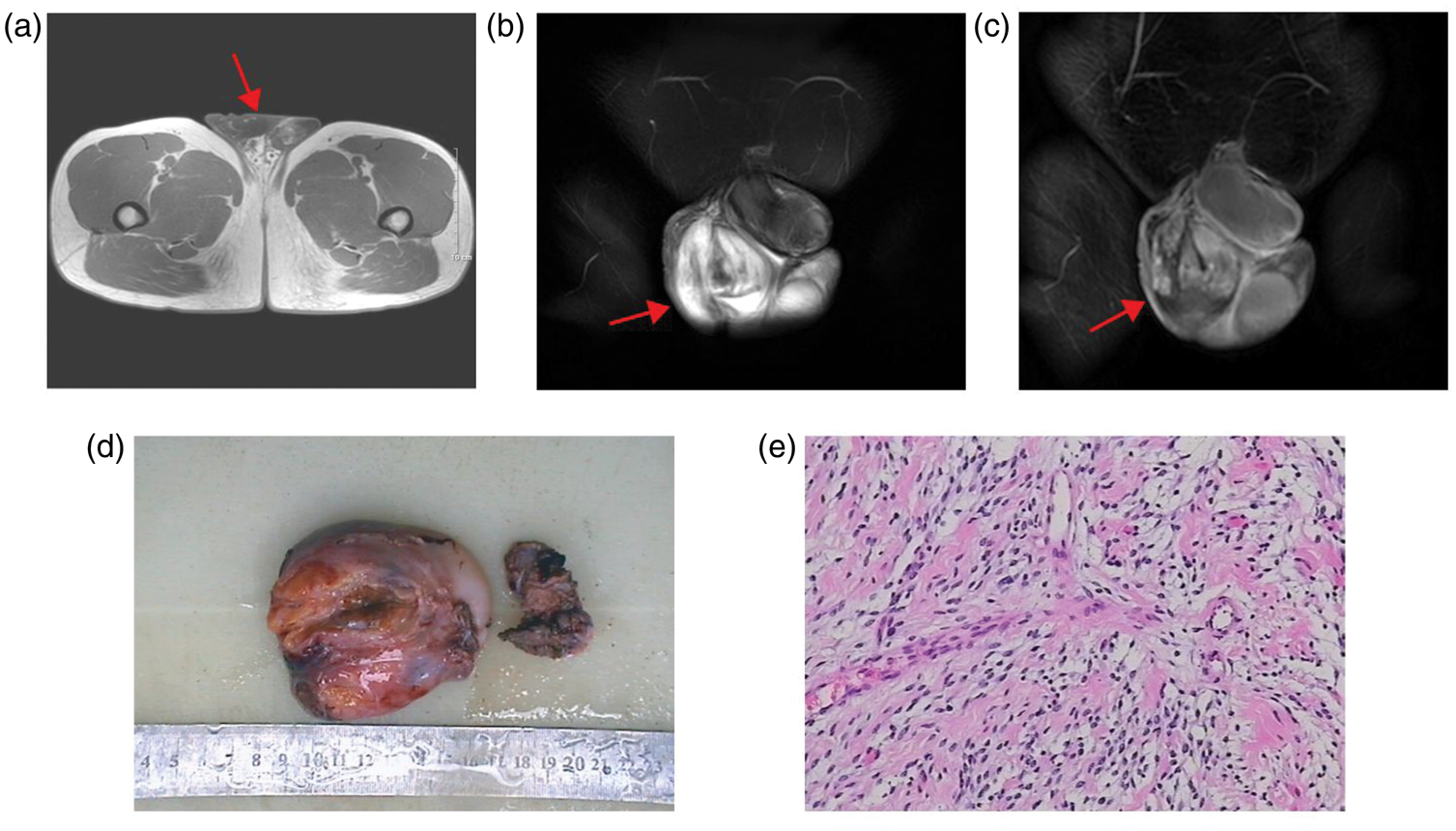
Magnetic resonance imaging, gross image, and photomicrograph in case 2. (a) Pelvic magnetic resonance imaging (MRI) showed a right scrotal mass (indicated by the red arrow) with hypointense signal on T1-weighted images and multiple vascular shadows within the mass. (b) Pelvic MRI identified a space-occupying lesion (indicated by the red arrow), approximately 5.1 × 2.2 cm in size, in the right scrotum. The lesion had clear borders and appeared hyperintense on T2-weighted images. (c) Enhancement scans revealed uneven enhancement (indicated by the red arrow). (d) Intraoperative findings revealed a scrotal mass, measuring approximately 6 × 4 cm, which did not invade the testicle. Additionally, a separate mass, approximately 3 cm in size, was noted outside the spermatic cord and (e) The histopathological examination revealed that the tumor consisted of spindle-shaped cells. The tumor was characterized by an abundance of thin-walled vessels distributed throughout the collagenous stroma.
Discussion
AMF is a rare benign tumor of mesenchymal origin that predominantly affects the genitourinary system in both men and women. In 1992, Fletcher et al. 5 first reported 10 cases of AMF in the soft tissues of the pelvic cavity in females. In 1998, Laskin et al. 6 reported similar cases in males, with AMF located in the scrotum in six cases and in the inguinal region in five cases. The tumor was well-defined but not encapsulated. 7 A review of 17 male cases, including the two reported herein, reveals a wide range of clinical presentations (Table 1).8–22 Ages at diagnosis ranged from 26 to 83 years (median, 43 years), with tumors primarily located in the scrotum (seven cases), inguinal region (five cases), and spermatic cord (two cases) and less commonly in the glans penis, abdomen/pelvis, or bladder. Presenting symptoms varied from painless swelling to dysuria or hematuria, and tumor sizes ranged from 3 to 25.5 cm. Notably, preoperative diagnoses were incorrect in most cases, with AMF misdiagnosed as metastatic tumors, inguinal hernias, germ cell tumors, or sarcomas, highlighting its diagnostic challenge. In a larger series of 137 cases, Wolf et al. 23 reported a female-to-male ratio of 10:1, with vulvar predominance in women and no recurrences after excision, although the follow-up period was limited to 12 months. This observation may be attributed to the relatively short follow-up period. The present study provides a longer follow-up period of 7 years with no recurrences. Clinically, differential diagnosis should include cellular angiofibroma, aggressive angiomyxoma (AAM), and inguinal hernia.
Clinical characteristics of angiomyofibroblastoma in male patients.

Data were compiled from referenced studies and the current report.
MRI: magnetic resonance imaging; NA: not available.
The pathogenesis of AMF remains unclear; current hypotheses propose that local irritation or chronic injury may induce abnormal proliferation of smooth muscle cells within vessel walls, culminating in tumor formation. 24 Some studies have indicated that estrogen influences the growth and development of AMF,25,26 which could account for the higher prevalence of the condition in females than in males. In our cases, the positive expression of estrogen and progesterone receptors in case 1 supports this hypothesis, suggesting a sex steroid–dependent mechanism.
Imaging manifestations of AMF typically include well-demarcated lesions with inhomogeneous echogenicity and multiple hypoechoic areas within an echogenic stroma on ultrasonography, as reported in the literature. 27 Ultrasonography in our cases showed hyperechoic areas, differing from typical descriptions. On MRI, AMF lesions are generally slightly inhomogeneous and hyperintense on T2-weighted images, with T1-weighted images showing signal intensity comparable to or lower than that of muscle. 14 Contrast-enhanced MRI in our cases demonstrated marked progressive enhancement, consistent with prior reports; however, these radiologic features lack specificity, limiting their utility in differential diagnosis. In case 1, preoperative MRI and CT findings, combined with the patient’s rectal cancer history, led to a misdiagnosis of metastatic tumor, prompting radical orchiectomy. This highlights the limitations of imaging in distinguishing AMF from malignant lesions, underscoring the need for needle biopsy to prevent unnecessary interventions.
AMF exhibits distinctive morphological features that facilitate its identification, although similarities with other soft tissue tumors pose diagnostic challenges. Typically, AMF arises in superficial soft tissues and is characterized by well-defined boundaries. Histologically, it displays spindle cell proliferation with alternating regions of high and low cellularity, prominent thin-walled blood vessels, and abundant loose, edematous stroma. 28 These histomorphological similarities to other mesenchymal tumors, such as cellular angiofibroma or AAM, necessitate immunohistochemical analysis for accurate diagnosis. Immunohistochemical studies consistently demonstrate positivity for vimentin and desmin, variable expression of SMA and CD34, and frequent reactivity for estrogen receptor (ER) and progesterone receptor (PR), while S-100 protein is typically negative.29,30 Table 2 summarizes the immunohistochemical profiles of 17 male AMF cases, showing consistent positivity for desmin (14/15 cases) and CD34 (14/16 cases), frequent vimentin positivity (9/10 cases), and variable SMA expression (7/13 cases). ER and PR positivity was observed in 5/10 and 4/10 cases, respectively, supporting AMF’s myofibroblastic origin and potential hormonal dependency. These markers were critical in confirming AMF in our cases, where case 1 was positive for desmin, CD34, ER, and PR and case 2 for vimentin, desmin, and CD34. Laskin et al. 6 proposed that AMF is derived from perivascular stem cells capable of myofibroblastic differentiation, a hypothesis supported by ultrastructural evidence of myofibroblastic features. 7 Overall, 50% of AMF cases in males are SMA-positive, while only 15% of AMF cases in females show SMA positivity. 31 Moreover, the positive expression of both ER and PR in tumor cells suggests that AMF is a sex steroid–dependent neoplasm. 30 The consistent morphological and immunophenotypic features observed in our cases align with the established literature, reinforcing the diagnostic utility of these characteristics.
Immunohistochemical profiles of angiomyofibroblastoma in male patients.

Data were compiled from referenced studies and the current report.
ER: estrogen receptor; NA: not available; PR: progesterone receptor; SMA: smooth muscle actin.
For intrascrotal masses, it is essential to be aware of AMF, which is benign and typically does not recur following complete surgical resection. A thorough understanding of the patient’s condition should be obtained based on their presenting symptoms and preoperative imaging data, including MRI and ultrasound. In addition, needle biopsy may be helpful in the preoperative diagnosis of AMF; however, owing to the lack of sufficient literature and case reports, the decision to perform a biopsy should be made based on the specific condition, tumor characteristics, and the patient’s overall health status. If the diagnosis cannot be confirmed before surgery, the surgical approach should be determined based on intraoperative findings to prevent misdiagnosis and overtreatment. AMF allows the complete resection of the lesion with testicular preservation and has a favorable prognosis.
In line with previous AMF studies, we revisited a case of misdiagnosis involving an older male patient with a history of rectal malignancy. Preoperative MRI and CT suggested that the scrotal mass was a metastatic tumor. During intraoperative exploration, the right testis was poorly demarcated from the tumor and appeared as a mass with an uneven surface suggestive of malignancy. However, postoperative pathology confirmed it as a benign lesion—AMF. After reviewing the relevant literature and conducting a detailed analysis of this case, we determined that AMF is unrelated to rectal cancer. The two conditions have distinctly different pathogenic mechanisms and pathological characteristics. The misdiagnosis in this case was primarily due to the preoperative imaging presentation and rare intraoperative appearance of the mass. Other cases presented in Table 1, such as those misdiagnosed as inguinal hernias or germ cell tumors, further illustrate the diagnostic pitfalls. These errors often result from AMF’s nonspecific imaging features and rarity in males, emphasizing the need for heightened clinical suspicion and histopathological confirmation to guide appropriate management.
Management strategies for AMF emphasize conservative surgical approaches, primarily complete local excision with negative margins, while preserving adjacent organs such as the testis in scrotal cases. This is contradictory to the approach used for more aggressive tumors such as AAM or sarcomas, which may require wide resection, adjuvant therapy, or even radical procedures owing to their infiltrative nature and higher recurrence risk. Distinguishing AMF from these entities is crucial, as accurate diagnosis allows for organ-sparing surgery and minimizes postoperative complications. In cases of diagnostic uncertainty, preoperative needle biopsy or frozen section analysis during surgery can facilitate this differentiation, potentially averting overtreatment. Long-term follow-up, as in our 7-year observation study with no recurrences, further supports the efficacy of these strategies for AMF.
Conclusion
AMF is a rare benign mesenchymal tumor that is difficult to distinguish from other malignant tumors invading the male genital tract based solely on clinical features and imaging. Nonetheless, when a mass appears in the scrotum of a middle-aged man with distinct demarcation from the testis, along with a high signal on T2-weighted imaging and persistent enhancement on contrast-enhanced scans, AMF should be strongly suspected. Definitive diagnosis remains dependent on pathological examination.
Footnotes
Acknowledgements
AI tools were used for minor language polishing.
Authors’ contributions
SH, QFX, CJY, and XYP contributed to the conception and design of the study. HZC, SH, HBX, and FQYZ were responsible for data acquisition. CJY, QFX, ZYL, HBX, and KW performed the data analysis. QFX, CJY, SH, and XYP drafted the manuscript. SH, XYP, and QFX made critical revisions to the manuscript. All authors have read and approved the final manuscript.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This research was supported by the Guangzhou Basic and Applied Basic Research Foundation (No. 202201011030), the Ganzhou Science and Technology Program (No. 2023NS227010 and 2023LNS26879), and the Foundation of the Department of Finance of Guangdong Province (No. KS0120220269).
