Abstract
Background
This study aimed to compare the expression patterns of hypoxia-inducible factor-1α in ovarian cyst capsules and endometrial tissue samples from patients with and without endometriosis to evaluate its potential as a biomarker of hypoxia in endometriosis.
Methods
This retrospective observational case–control study included 87 women of childbearing age (20–45 years) who underwent ovarian cystectomy at Balıkesir University Hospital between 2015 and 2020. Of these, 40 patients with confirmed endometriosis constituted the study group, while 47 patients with benign ovarian cysts comprised the control group. Immunohistochemical staining was used to assess the expression of hypoxia-inducible factor-1-alpha in ovarian cyst capsules and endometrial tissue samples. The relationship between the expression of hypoxia-inducible factor-1-alpha and clinical parameters, including age, body mass index, and endometriosis stage, was also evaluated.
Results
The expression of hypoxia-inducible factor-1-alpha was significantly higher in ovarian cyst capsules and endometrial tissues of patients with endometriosis than in those of controls (p < 0.05). No correlation was found between hypoxia-inducible factor-1-alpha expression and age, body mass index, or disease stage.
Conclusions
In this study, the expression of hypoxia-inducible factor-1-alpha in the ovarian and endometrial tissues was significantly elevated in patients with endometriosis compared with that in controls, independent of clinical parameters. These results support hypoxia-inducible factor-1-alpha as a stable biomarker of hypoxia in endometriosis and provide a basis for future research on hypoxia-targeted therapies for endometriosis management.
Introduction
Endometriosis (EMS) is a chronic gynecological disease affecting approximately 10%–15% of all women of childbearing age. It is characterized by the presence of endometrial-like tissue outside the uterine cavity, predominantly affecting the ovaries, fallopian tubes, and pelvic peritoneum. This ectopic tissue undergoes cyclic bleeding and elicits inflammatory responses similar to the uterine endometrium, resulting in chronic pelvic pain, dysmenorrhea, and infertility in many cases. 1 The pathogenesis of EMS is complex and multifactorial, involving genetic, immunological, hormonal, and environmental factors. 2 Despite extensive research, its exact etiology remains elusive. Certain underlying triggers may activate these complex mechanisms, initiating processes that contribute to the development and persistence of endometriotic lesions. The absence of a definitive understanding of its etiology poses significant challenges in developing targeted treatments, underscoring the need for further research into its pathogenesis.3,4
Hypoxia, characterized by reduced oxygen availability, is a critical factor in the pathogenesis of EMS. 5 To cope with low oxygen levels under hypoxic conditions, cells activate adaptive responses primarily mediated by the hypoxia-inducible factors (HIFs), particularly hypoxia-inducible factor-1-alpha (HIF-1α). HIF-1α is a transcription factor that regulates the expression of genes associated with angiogenesis, metabolism, cell survival, and invasion in EMS. 6 Furthermore, the hypoxic environment within endometriotic lesions may exacerbate inflammatory responses, contributing to increased pain and tissue damage, thereby influencing the severity of symptoms and the overall burden of the disease. 7
In this study, we hypothesized that hypoxia-induced HIF-1α expression is significantly higher in ovarian cyst capsules and endometrial tissues of patients with EMS compared with that in women without EMS. We aimed to investigate whether HIF-1α expression is independent of clinical variables such as age, body mass index (BMI), and EMS stage to assess its ability as a stable biomarker for EMS. Although the role of HIF-1α in EMS has been explored in previous studies, the present study provides new perspectives by directly comparing HIF-1α expression in ovarian cyst capsules and endometrial tissue samples from the same patient cohort. Few studies have employed this comparative approach, highlighting tissue-specific sensitivities to hypoxia in EMS. By focusing on the differential expression of HIF-1α in ovarian cyst capsules versus endometrial tissue samples from patients with and without EMS, our study aimed to provide previously unknown insights into the pathophysiology of EMS.
This approach contributes to identifying potential diagnostic markers and supports the development of hypoxia-targeted therapies, which could transform the management of EMS.
Materials and methods
Study design and ethical considerations
The population of this case–control study consisted of women who underwent ovarian cystectomy at the Department of Obstetrics and Gynecology at Balıkesir University Hospital between 2015 and 2020. The study was conducted in accordance with the ethical standards of the Declaration of Helsinki (1975), as revised in 2024. Ethical approval was obtained from the Balıkesir University Faculty of Medicine Clinical Research Ethics Committee (Balıkesir, Turkey) on 14 October 2020 (Approval No: 2020/184). Written informed consent was obtained from all patients, including consent for the use of patients’ pathology slides containing the tissue samples collected during the surgery in future studies.
Inclusion and exclusion criteria
The study population consisted of 119 women of childbearing age (25–40 years) who underwent either open or laparoscopic surgery for ovarian cysts during the study period. The study excluded patients with complications arising from endocrine disorders (n = 17), autoimmune diseases (n = 5), or ovarian cancer (n = 1) as well as those who did not provide informed consent (n = 2). Additionally, women who had taken steroids or hormone medications within the past 3 months (n = 7) and those with a history of alcohol or drug use prior to surgery (n = 0) were excluded. Finally, 87 women were included in the study sample.
Grouping
Of the 87 patients who met the study inclusion criteria, 40 women who underwent surgery for EMS were included in the patient group, and 47 who underwent surgery for benign ovarian cysts other than EMS during the same period were included in the control group.
The two groups were compared for demographic, clinicopathological, and immunostaining profiles. Intra-group comparisons were also conducted for the recorded parameters.
Clinicopathological data
Clinicopathological data, including age at the time of surgery, BMI(body weight (kg) divided by height squared (m)), and EMS stage, were obtained from the hospital’s database. EMS staging was defined according to the Revised American Society for Reproductive Medicine classification of EMS. 8 Processed samples of ovarian cyst capsules and endometrial tissue samples were obtained from the database of the Department of Pathology. The ovarian cysts were classified as serous or mucinous; for each patient, an average of three sections per cyst were analyzed. Endometrial tissue samples were obtained via biopsy performed immediately prior to the surgery during the luteal phase.
Immunohistochemistry (IHC)
Immunohistochemical analyses were prospectively performed to determine the tissue expression of HIF-1α in ovarian cyst capsules and endometrial tissue samples. Samples that had been formalin-fixed and embedded in paraffin were divided into three to five sections and set on alcohol-cleansed glass slides. Subsequently, they were incubated overnight at 4°C with a primary rabbit polyclonal antibody against HIF-1α (Beyotime Biotechnology, Shanghai, China; Cat# AF7087, RRID: AB_2891397) at a dilution of 1:150. According to the manufacturer’s datasheet, this antibody has been validated for use in IHC, western blot (WB), immunofluorescence, and immunocytochemistry in human, mouse, and rat tissues. This validation suggests that the antibody has been tested to work with the natural HIF-1α protein under low oxygen conditions, and its use has been reported in peer-reviewed scientific articles.
Deparaffinized sections were rehydrated in graded ethanol. Antigen retrieval was conducted in a pressure cooker under high pressure for 3 min in citrate buffer (pH 6.0), followed by a 7-min treatment with 3% hydrogen peroxide to terminate the endogenous peroxidase activity. Followed by blocking in a solution of 0.01 mol/L Tris, 0.3% Triton X-100 (Sigma-Aldrich, St. Louis, MO, USA), and 10% normal goat serum for 30 min, the sections were incubated overnight at 48°C with the primary antibody. Glass slides were coated with Cell Conditioning 1 solution (Ventana Medical Systems, Tucson, AZ, USA) to reveal the antigen. Sections were washed with phosphate-buffered saline and then incubated with peroxidase-labeled anti-rabbit immunoglobulin G (IgG) for 30 min. Prior to dehydration and mounting, all slides were counterstained with hematoxylin and incubated with 3,3′-diaminobenzidine substrates (Beyotime, Wuhan, China). The slides incubated with IgG from healthy, isotype-matched rabbits served as negative controls. Entellan was used to coat the slides before immunohistochemical analysis.
All patient data were anonymized, and no identifying information has been included in the study.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 9
Immunoreactivity assessment
The 87 prepared slides were examined for HIF-1α protein expression using an ergonomic headlight microscope (Nikon 300038, Tokyo, Japan) at 200× (20× objective lens, 10× ocular lens) magnification by two independent pathologists, who were blinded to the participants’ clinicopathological data. The degree of cytoplasmic and nuclear staining in the epithelium of the ovarian and endometrial cells was evaluated in antibody-treated tissue samples. The staining due to HIF-1α expression was scored as “0” for no staining, “1” for weak staining, “2” for moderate staining, and “3” for intense staining. Representative staining samples are shown in Figure 1.

(a) HIF-1α negativity in mucinous cysts in the control group (100×). (b) Weak HIF-1α expression in epithelial cells of the external endometriosis cyst (100×) (red arrow). (c) Moderate HIF-1α expression in epithelial cells of the external endometriosis cyst (200×) (red arrow) and weak HIF-1α expression in macrophages (200×) (yellow arrow). (d) Intense HIF-1α expression in epithelial cells of the external endometriosis cyst (200×) (red arrow) and intense HIF-1α expression in macrophages (200×) (yellow arrow) and (e) intense HIF-1α expression in stromal cells of the external endometriosis cyst (red arrow) and macrophages (yellow arrow), as also seen in Figure 1(d) (200×). The degree of cytoplasmic and nuclear staining in the epithelium of the ovarian and endometrial cells was evaluated in antibody-treated tissue samples. The staining due to HIF-1α expression was scored as “0” for no staining, “1” for weak staining, “2” for moderate staining, and “3” for intense staining. HIF-1α: hypoxia-inducible factor-1 alpha.
Statistical analyses
All collected data were analyzed using the Jamovi project 2.3.28 (Jamovi, version 2.3.28, 2023, retrieved from https://www.jamovi.org) and JASP 0.17.3.0 (Jeffreys’ Amazing Statistics Program, version 0.17.3.0, 2023, retrieved from https://jasp-stats.org) software packages. The normality assumption of the variables was tested using the Shapiro–Wilk, Kolmogorov–Smirnov, or Anderson–Darling test. Descriptive statistics obtained from the collected data were presented as mean ± SD values for normally distributed continuous variables, as median with minimum and maximum (range) values for non-normally distributed continuous variables, and as numbers (n) and percentages (%) for categorical variables.
Categorical variables were compared between the two groups using the Pearson’s chi-square and Fisher’s exact tests when the expected numbers of cells in the 2 × 2 tables were ≥5 and <5, respectively. Continuous variables were compared between two independent groups using the Student’s t-test or Mann–Whitney U test, as appropriate. Probability (p) values < 0.05 were deemed to indicate statistical significance.
The interobserver reliability for HIF-1α staining scores was assessed using Cohen’s kappa coefficient, yielding a κ value of 0.71, indicating a substantial agreement between the two pathologists (approximately 71% agreement beyond chance).
Results
Table 1 shows the distribution of patients’ data, including age at surgery, BMI, and tissue staining patterns in the patient and control groups. There was no significant difference between the two groups in terms of age at surgery and BMI. However, in terms of HIF-1α immunostaining, 26 (65%) ovarian cyst capsules and 33 (82.5%) endometrial tissue samples were stained in the patient group, whereas, no staining was observed in the control group, with p-values of 0.001 and 0.003, respectively. The distribution of HIF-1α immunostaining in ovarian cyst capsules and endometrial tissues in the patient and control groups is shown in Figure 2. The semiquantitative assessment revealed heterogeneous staining intensities, ranging from weak to intense, particularly in ectopic endometrial tissues. This variability is consistent with previous findings highlighting the complex nature of EMS-related hypoxia, as presented in Figure 1. In addition to epithelial cells and macrophages, HIF-1α expression was observed in stromal cells, as shown in Figure 1(d). Conclusively, this finding indicates a broader involvement of hypoxia-responsive pathways within the endometriotic microenvironment.
Comparison of age, BMI, and tissue staining patterns in cyst capsules and endometrial tissue samples across study groups.
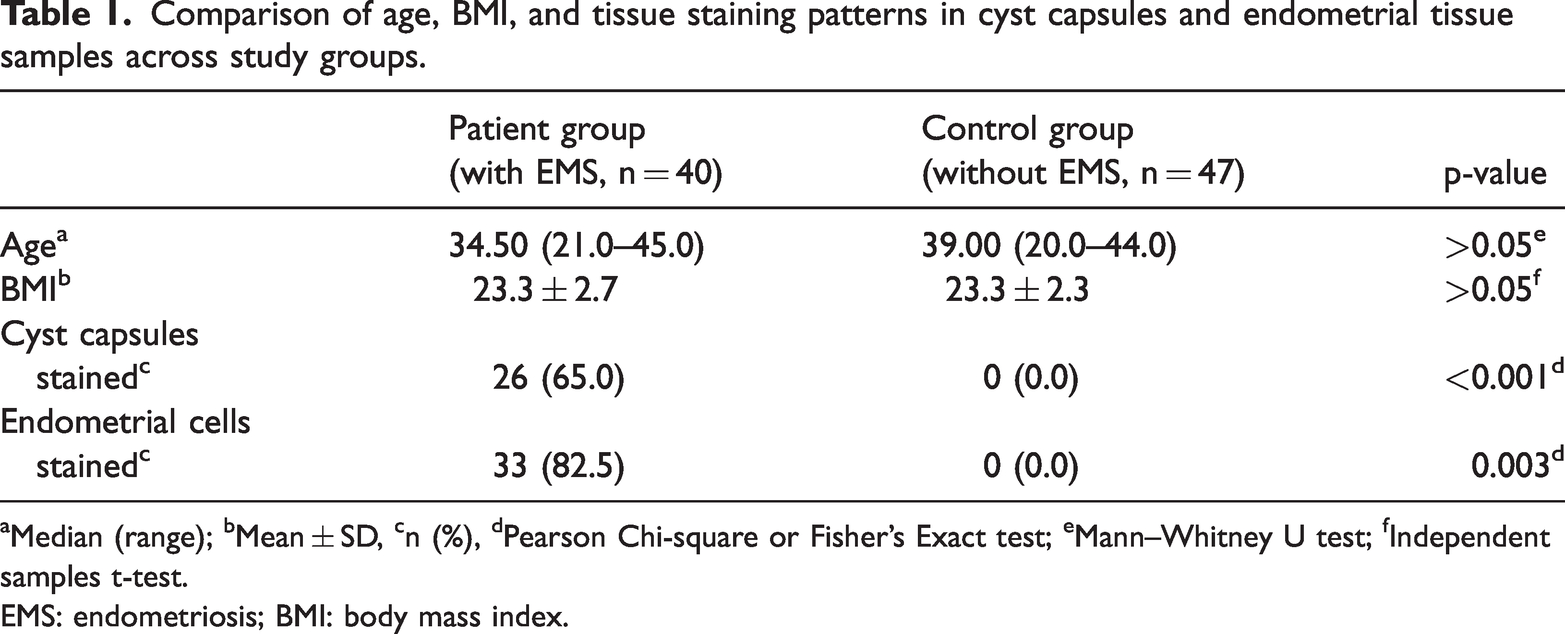
Median (range); bMean ± SD, cn (%), dPearson Chi-square or Fisher’s Exact test; eMann–Whitney U test; fIndependent samples t-test.
EMS: endometriosis; BMI: body mass index.

Distribution of the HIF-1α immunostaining profile of ovarian cyst capsule and endometrial cells in the patient (endometriosis) and control groups. HIF-1α: hypoxia-inducible factor-1 alpha.
Of the 40 patients in the patient group, 25 (62.50%) were identified as having stage III EMS and 15 (37.50%) as having stage IV EMS. HIF-1α immunostaining was observed in 60.0% of ovarian cyst capsules and 12% of endometrial tissue samples in patients with stage III EMS and 40.0% of ovarian cyst capsules and 26.7% of endometrial tissue samples in patients with stage IV EMS. Thus, there was no significant difference in HIF-1α immunostaining in ovarian cyst capsules and endometrial tissue samples between patients with stage III and IV EMS (p = 0.369 and p = 0.392, respectively, Table 2). Similarly, when analyzed across BMI groups, no significant difference was observed (Table 3). Representative staining in follicular cyst and endometrial samples from stage III and IV EMS patients is shown in Figure 3.
Comparison of staining patterns in cyst capsules and endometrial cells across disease stages in the patient group.

Pearson chi-square or Fisher’s exact test.
EMS: endometriosis.
(p-values represent comparisons between stage III and IV EMS for each tissue type).
Comparison of staining patterns in cyst capsules and endometrial cells across BMI in the patient group.
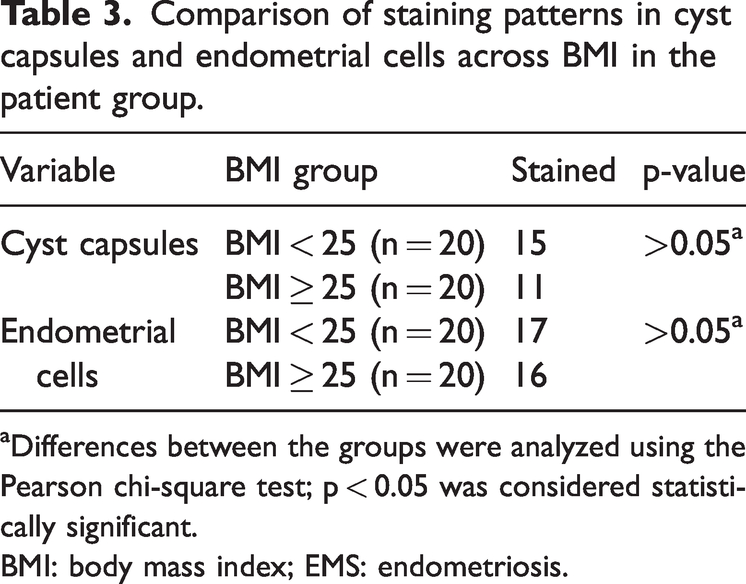
Differences between the groups were analyzed using the Pearson chi-square test; p < 0.05 was considered statistically significant.
BMI: body mass index; EMS: endometriosis.
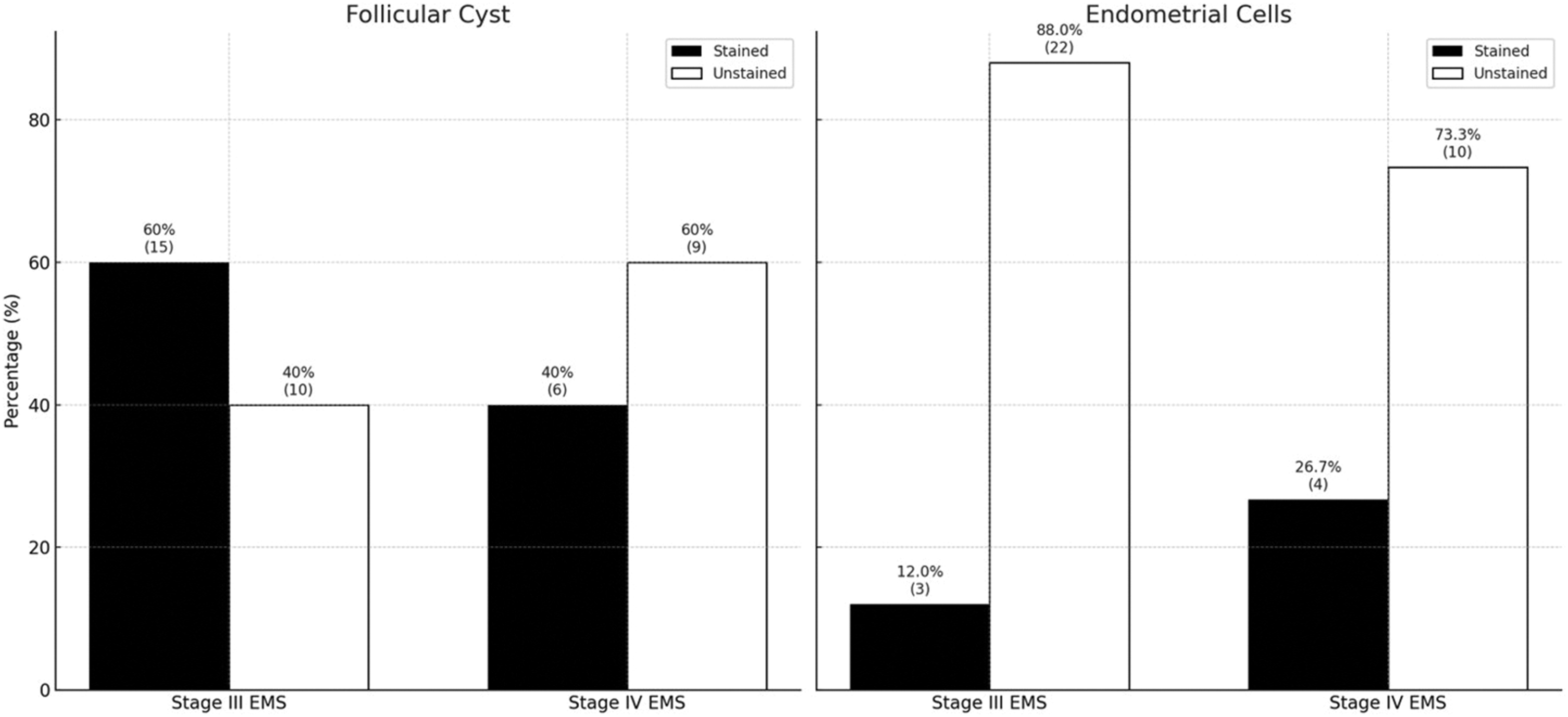
HIF-1α staining in samples of ovarian cysts (a) and endometrial cells (b) in patients with stage III and IV endometriosis. HIF-1α: hypoxia-inducible factor-1 alpha.
Discussion
This study demonstrated that HIF-1α expression in ovarian cyst capsules and endometrial tissue samples was significantly higher in patients with EMS than in those without EMS. In addition, HIF-1α expression did not show a significant relationship with age, BMI, or disease stage in patients with EMS.
Consistent with our findings, Zhang et al. 10 found that the expression of HIF-1α was significantly higher in endometriotic tissues than in normal endometrial tissues. The outcome of the study supported the role of hypoxia in EMS progression based on an examination of the relationship between HIF-1α and vascular endothelial growth factor (VEGF). They suggested that VEGF could trigger angiogenesis in response to hypoxia. Similar to our study, the study conducted by Bo and Wang 11 demonstrated elevated levels of HIF-1α in ectopic endometrial tissues, emphasizing the role of hypoxia as a key factor in promoting the survival and proliferation of ectopic cells. In addition to the findings reported by Zhang et al., 10 Bo and Wang 11 demonstrated that HIF-1α, released in response to hypoxia, stimulates VEGF expression not only directly but also through an indirect inflammatory pathway, wherein HIF-1α promotes angiogenesis via interleukin-8 (IL-8) and cyclooxygenase-2 (COX-2). Although our findings rely on the intensity of HIF-1α expression, future studies incorporating quantitative assessments, such as enzyme-linked immunosorbent assay (ELISA) or reverse transcription polymerase chain reaction (RT-PCR), would provide more robust evidence for its role in hypoxia-driven pathophysiology. Although HIF-1α is not a disease-specific biomarker, its persistent activation in hypoxic microenvironments, characteristic of endometriotic tissues, supports its relevance in this context.
Future analyses should include complementary biomarkers such as VEGF and inflammatory mediators such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) to further validate their specificity. In our study, the primary reason for not examining indirect mechanisms, such as those involving inflammatory factors IL-8 and COX-2, was to specifically assess the impact of HIF-1α on EMS development. Moreover, assessing indirect pathways would require a more extensive experimental design and additional resources. Therefore, we focused on HIF-1α to enhance the clarity of our findings.
In another study, Dai et al. 12 demonstrated that HIF-1α enhances cellular invasion in EMS and promotes epithelial–mesenchymal transition, a key process contributing to the progression of the disease. Furthermore, their work highlighted how matrix metalloproteinases, which play a crucial role in tissue remodeling, are regulated by HIF-1α under hypoxic conditions, emphasizing the role of hypoxia in driving extracellular matrix degradation and tissue invasiveness in endometriotic lesions. 12 Many studies have examined HIF-1α expression in EMS; however, none have directly compared HIF-1α expression levels between endometrial tissue samples and ovarian cyst capsules. Therefore, this direct comparison in the present study represents a key aspect of our work. We observed that the rate of HIF-1α immunostaining was higher in ovarian cyst capsules than in endometrial tissue samples in patients with EMS. These findings demonstrate differential expression of HIF-1α in these tissue types, suggesting that EMS exhibits tissue-specific sensitivity to hypoxic microenvironments, as also demonstrated by Wu et al. 13 This heterogeneity in HIF-1α expression levels aligns with the multifactorial and variable presentation of EMS, suggesting that hypoxia-induced pathways may manifest differently depending on individual pathological conditions. We conducted the endometrial sampling during the luteal phase, which represents another strength of our study. Research has shown that HIF-1α plays an important role in endometrial function and pathology, with its expression fluctuating throughout the normal menstrual cycle and peaking during the secretion and menstruation phases. 8 Based on this, we anticipated that HIF-1α expression would be highest during the luteal phase, allowing us to distinctly observe the differences in its expression between ovarian cyst capsules, endometrial tissues, and control samples. This finding suggests that HIF-1α is regulated by hormonal fluctuations in normal endometrial tissues. In contrast, Ren et al. 14 demonstrated that HIF-1α expression remains stable throughout the menstrual cycle in normal, ectopic, and eutopic endometrial tissues, independent of the cycle phase. Although both abovementioned studies report that HIF-1α is expressed in nonpathological normal endometrial tissue, we did not observe any HIF-1α staining in the endometrial tissue samples of patients without EMS in our study. Differences in the methodology or sample selection criteria between the studies may account for this discrepancy. Negative control slides confirmed the specificity of staining, reducing the likelihood of a technical error.
In our study, we observed that the endometrial tissue samples from patients with EMS showed significantly higher staining than normal endometrial tissues. Thus, we concluded that the significant staining of HIF-1α observed in both ovarian cyst capsules and endometrial tissue samples in patients with EMS indicate that these tissues were exposed to a hypoxic microenvironment, which maintained HIF-1α persistently active in response to hypoxia.
Furthermore, we observed that HIF-1α expression was not correlated with clinical parameters, such as age, BMI, or EMS stage. This finding indicates that HIF-1α may act as a stable biomarker in the pathophysiology of EMS, irrespective of disease progression or the physical characteristics of the patient. Thus, the consistency in HIF-1α expression across different stages and demographics implies that its hypoxia-driven effects may be uniformly present in patients with EMS, independent of these factors.
Previous studies on HIF-1α expression in EMS have yielded varied results regarding its correlation with disease severity. Although some studies have demonstrated increased HIF-1α expression in advanced stages of EMS, others have shown a more complex relationship. For example, in their prospective study, Karakus et al. 15 found that women with more severe stage III/IV EMS had higher serum levels of HIF-1α than those with less severe stage I/II EMS. They also demonstrated higher CD95/FAS expression along with HIF-1α expression in patients with advanced-stage EMS, concluding that these two markers potentially serve as biomarkers for diagnosing EMS and assessing its severity. In a similar study, Badary et al. 16 observed that endometriotic glands showed significant cytoplasmic expression of HIF-1α in patients with poor ovulation, severe EMS, and infertility lasting >2 years. They also found that patients with EMS who experienced intense pain showed significant immunohistochemical expression of HIF-1α and mechanistic target of rapamycin markers. In contrast to the findings reported by Karakus et al. 15 and Badary et al, 16 Filippi et al. 17 observed heterogeneity in HIF-1α expression among different EMS phenotypes, with higher levels observed in ovarian endometriomas but not in deep infiltrating EMS. Furthermore, they found that in ovarian endometriomas, the expression of hypoxia-related genes (HIF-1/2α) was positively correlated with the expression of the angiogenic gene VEGF-A. These findings suggest that although HIF-1α plays a role in EMS pathogenesis, its direct correlation with disease severity remains inconclusive and may vary depending on the type of endometriotic lesion.
Most previous studies have consistently shown an inverse relationship between BMI and EMS risk. Women with lower BMI are more likely to develop EMS, particularly deep infiltrating EMS.18–20 Zhou et al. 21 and Ozmen et al. 22 examined the relationship between BMI and HIF-1α expression and showed that high BMI and obesity are associated with increased HIF-1α expression, particularly in the fat tissue in the omental region. Ozmen et al. 22 also stated that the complex interplay between BMI, HIF expression, and methylation patterns suggests a connection between obesity, hypoxia-related molecular pathways, and adipose tissue dysfunction. Our study found no significant difference in BMI between the patient group and control group. Furthermore, in the patient group, BMI did not have a statistically significant effect on HIF-1α staining in ovarian cyst capsules or endometrial tissue samples.
Our research has certain limitations. The exclusion of patients with stage I–II EMS restricts the generalizability of our findings across the entire disease spectrum. Future studies should aim to include all EMS stages to assess the dynamic range of HIF-1α expression throughout disease progression. The small sample size of our study restricts the generalizability of some findings. Studies on larger populations may be necessary to further evaluate the relationship between HIF-1α and clinical parameters. Moreover, the exclusion of additional biomarkers, such as inflammatory and angiogenic markers (e.g. IL-6, TNF-α, and VEGF), limits the ability to explore the potential connections between hypoxia and inflammation in greater detail. Another limitation is the retrospective nature of our study, which may have introduced selection bias, thereby affecting the interpretation of our findings. Additionally, our study only examined ovarian cyst capsules and endometrial tissue samples, without assessing how HIF-1α expression varies across different types of EMS, such as peritoneal or rectovaginal EMS.
Although HIF-1α is not a disease-specific biomarker, its consistent activation in hypoxic microenvironments, characteristic of endometriotic tissues, supports its relevance in this context. The lack of HIF-1α staining in our control tissue samples indicates low baseline expression levels that fall below the detection threshold of semiquantitative IHC. The sample collection during the luteal phase may have affected this outcome, which should be regarded as a possible methodological limitation. Although semiquantitative IHC provides a comparative assessment of HIF-1α expression, the lack of confirmatory methods such as quantitative PCR, ELISA, or WB limits the accuracy of our results. This study requires additional quantitative techniques to support the immunohistochemical findings. Future research using quantitative techniques such as ELISA and RT-PCR could enhance the validity of our biomarker-related findings.
Despite being aware of these limitations, we could not address them owing to the constraints of available resources as well as factors inherent to the scope of our study design. Factors such as sample size, retrospective design, and exclusion of additional biomarkers were challenging to address because of time and financial limitations. In future studies, we plan to conduct more comprehensive investigations to overcome these limitations and validate our findings in larger populations.
Conclusions
Although EMS is a widely prevalent condition, its pathogenesis remains partially understood. This case–control study demonstrated that HIF-1α expression is significantly increased in both ovarian cyst capsules and endometrial tissues in women with EMS. Our findings support the critical role of hypoxia in EMS pathophysiology. Furthermore, the lack of a significant association between HIF-1α expression and clinical parameters such as EMS stage, age, or BMI suggests that HIF-1α acts as a stable biomarker and that hypoxia has a strong influence on disease progression and symptomatology. Our study provides a significant foundation for the development of hypoxia-based therapeutic strategies. Further examination of HIF-1α expression and other hypoxia-related markers in larger sample groups across different types of EMS (e.g. peritoneal and rectovaginal EMS) may contribute to the available treatment options. More comprehensive research is needed to better understand the complex role of HIF-1α in EMS pathophysiology.
Footnotes
Acknowledgments
A certified translator with academic English training improved the language of the manuscript, and QuillBot’s advanced grammar correction tool was used to improve the clarity of our manuscript.
Authors’ contributions
Conceptualization: E.T.C. and C.S.U; methodology: E.T.C and A.U; software: E.T.C; validation: E.T.C, C.S.U, and G.T; formal analysis: A.U. and C.S.U; investigation: E.T.C; resources: C.S.U; data curation: N.A.S; writing–original draft preparation: E.T.C; writing–review and editing: G.G, S.A, and M.I.T; visualization: C.S.U; supervision: G.G. and A.U; project administration: A.U. and C.S.U; funding acquisition: E.T.C. and C.S.U; project administration: A.U. and C.S.U; funding acquisition: E.T.C. and C.S.U.
Availability of data and materials
Data are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The study was approved by the Balıkesir City Hospital Ethics Committee (Approval No: 2020/184; 14 October 2020) and conducted in accordance with the principles of the Declaration of Helsinki.
Funding
This research received no external funding.
