Abstract
A wide spectrum of ocular manifestations have been documented in patients following infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Nevertheless, bilateral extraocular myositis specifically associated with natural SARS-CoV-2 infection has been rarely reported. We report the case of an East Asian adolescent female who presented with right ocular discomfort and diminished visual acuity for 2 weeks. A SARS-CoV-2 infection had been confirmed via reverse transcriptase–polymerase chain reaction 10 days ago. Magnetic resonance imaging of the orbits revealed bilateral eyelid swelling and diffuse, asymmetric thickening of the extraocular muscles, suggestive of myositis. Symptoms and imaging findings improved markedly with high-dose corticosteroids; however, recurrence occurred upon rapid tapering. The inflammation ultimately resolved with a more gradual tapering regimen, and no recurrence was observed during 12 months of follow-up. This case underscores that natural SARS-CoV-2 infection can trigger immune-mediated bilateral extraocular myositis, posing vision-threatening risks. Corticosteroids are effective, but gradual withdrawal is essential to prevent relapse.
Introduction
Even after >5 years since its emergence, coronavirus disease 2019 (COVID-19) continues to cause significant morbidity, with ocular complications affecting approximately 11% of patients worldwide. These include uveitis, retinal vascular occlusions, neuro-ophthalmic sequelae,1–3 and sight-threatening infections such as rhino-orbitocerebral mucormycosis in immunocompromised individuals. 4 Furthermore, since the introduction of COVID-19 vaccinations, numerous reports have described the adverse ocular events following vaccination.5–7 Nevertheless, bilateral extraocular myositis specifically associated with natural severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has been rarely reported. Here, we report the case of bilateral extraocular myositis associated with COVID-19.
Case report
The reporting of this study conforms to the Case Report (CARE) guidelines. 8 An adolescent female patient from East Asia presented in June 2023 with a 2-week history of discomfort and diminished visual acuity in her right eye. She had received three doses of a COVID-19 vaccine, with the first two doses received 18 and 12 months prior to presentation and a third dose received 6 months before the current assessment. She experienced fever and fatigue. Ten days prior to admission, she was diagnosed with conjunctivitis of the right eye at a local hospital and was treated with tobramycin and dexamethasone eye drops, administered three times daily. A SARS-CoV-2 reverse transcriptase–polymerase chain reaction (RT–PCR) test performed at the local hospital returned positive. The patient did not exhibit any other typical features of COVID-19. Her ocular symptoms worsened over the following week. Consequently, the patient was referred and admitted to Hebei Eye Hospital (Xingtai, Hebei Province, China) for specialized treatment.
Upon admission, the patient presented with fatigue, malaise, and a decreased appetite. The best-corrected visual acuity was 20/100 in the right eye and 20/33 in the left eye. She exhibited binocular eyelid swelling, pain, mild acquired ptosis, mild proptosis, and limitation of extraocular muscle movements in all directions, and an almost fixed binocular eyeball. Conjunctival congestion of the right eye was distinct (Figure 1). Hertel exophthalmometry readings were 15 mm in both eyes. The diameter of the bilateral pupils was 3 mm. The direct and consensual pupillary light reflexes showed a slow response. The tests of afferent pupillary defect were negative. No abnormalities were detected in the anterior segment and fundus examinations. The clinical findings did not reveal any signs of meningeal irritation or cranial nerve lesions. There was no additional relevant personal or family medical history.
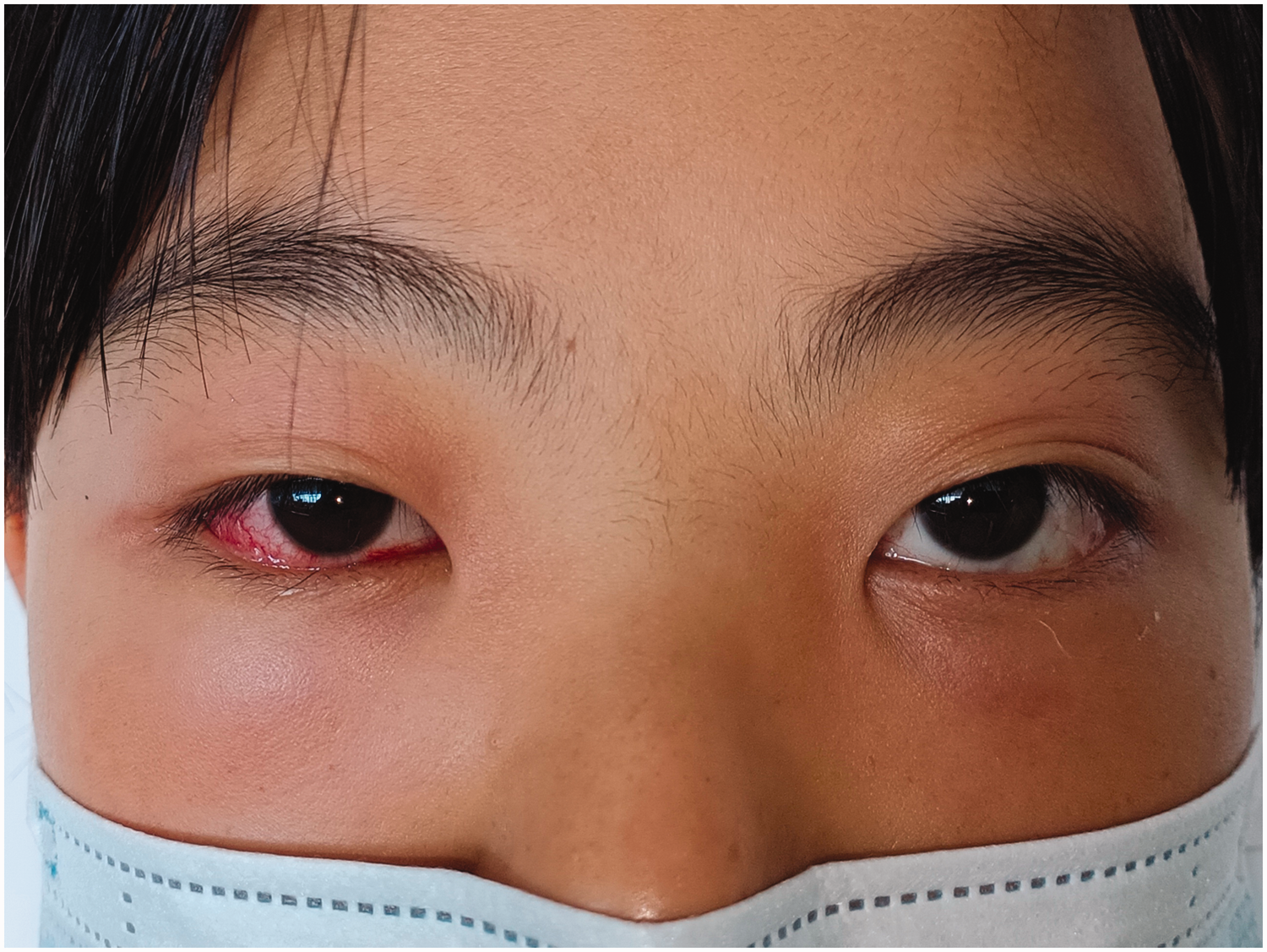
The patient presented with binocular eyelid swelling, mild ptosis, proptosis, limitation of extraocular muscle movements in all directions, and an almost fixed binocular eyeball.
A transient fever (38.0°C) developed on postadmission day 2, followed by subsequent normalization of temperature.
The SARS-CoV-2 RT–PCR test was negative, whereas the rapid SARS-CoV-2 antigen detection test remained positive. Blood tests revealed an increase in the neutral granular cell count (8.86 × 109/L; reference range: 1.8–6.3), neutrophil granulocyte percentage (85.1%; reference range: 40%–75%), aspartate aminotransferase level (82 U/L; reference range: 0–40 U/L), and lactic dehydrogenase (LDH; 283 U/L; reference range: 109–245 U/L). The erythrocyte sedimentation rate (ESR) was 60 mm/h (reference range: 0–20 mm/h), and the C-reactive protein (CRP) level was 114.9 mg/L (reference range: 0–10 mg/L). Thyroid function tests showed normal levels of free triiodothyronine (FT3), free thyroxine (FT4), thyroid-stimulating hormone, thyroid peroxidase antibodies (TPOAb), and thyroglobulin antibodies (TGAb) (Supplementary Material).
Chest computed tomography (CT) showed pleural effusion, with no other abnormalities in either lung field (Figure 2). Magnetic resonance imaging (MRI) revealed bilateral eyelid swelling, and all extraocular muscles of both eyes were thickened to varying degrees. T2-weighted images revealed that the thickened extraocular muscles demonstrated significantly increased signal intensity compared with normal extraocular muscles. Contrast-enhanced imaging revealed significant enhancement of the extraocular muscles. Inflammatory exudative changes were observed in the retrobulbar space and around the optic nerve (Figure 3). The right optic nerve thickness was 0.40 × 0.58 cm, while that of the left right optic nerve was 0.46 × 0.63 cm. Brain MRI revealed no abnormalities. No abnormalities were observed in standard pattern visual evoked potentials (VEP). The patient was diagnosed with COVID-19–associated bilateral extraocular myositis.

Chest CT showed pleural effusion, and no other abnormalities were found in both lungs. (a) Lung window; (b) mediastinal window. CT: computed tomography.

MRI revealed bilateral eyelid swelling, and all extraocular muscles of both eyes were thickened to varying degrees. Contrast-enhanced imaging demonstrated that the extraocular muscles showed a significant enhancement in intensity. (a) T1WI; (b) axial fat-suppressed T2WI; (c) coronal fat-suppressed T2WI and (d) coronal contrast-enhanced T1WI. MRI: magnetic resonance imaging; T1WI: T1-weighted images; T2WI: T2-weighted images.
The patient was started on intravenous methylprednisolone sodium succinate injection (80 mg daily). Calcium and potassium supplements were administered to mitigate the drug-related side effects. After 5 days, her symptoms and pleural effusion resolved (Figure 4). Treatment was continued with an intravenous injection of methylprednisolone sodium succinate (40 mg daily) for another 5 days, followed by a switch to oral administration of prednisone acetate tablets (20 mg once daily).

The patient’s pleural effusion disappeared (a) lung window and (b) mediastinal window.
Unfortunately, the patient showed recurrence 5 days after initiating oral steroid therapy. Her blood tests revealed an increase in the white blood cell count (16.37 × 109/L; reference range: 5–12 × 109/L), neutral granular cell count (12.74 × 109/L; reference range: 1.8–6.3 × 109/L), neutrophil granulocyte percentage (77.8%; reference range: 40%–75%), aspartate aminotransferase level (14 U/L; reference range: 0–40 U/L), and LDH level (188; reference range: 109–245 U/L). The ESR was 14 mm/h (reference range: 0–20 mm/h), and the CRP level was 1.30 mg/L (reference range: 0–10 mg/L). Methylprednisolone sodium succinate was reinitiated at a dose of 80 mg once daily for 3 days, followed by 60 mg for 3 days and 40 mg for 3 days. Therapy was switched to oral prednisone acetate tablets (40 mg once daily), and the dose was tapered by 5 mg per week until discontinuation. The uncorrected visual acuity was 20/20 in both eyes. Direct and consensual pupillary light reflexes and testing for relative afferent pupillary defect were normal. Hertel exophthalmometry readings were 13 mm in both eyes. Orbital CT revealed a near-normal appearance of all extraocular muscles of both eyes (Figure 5), and no recurrence or complications were observed during the 12-month follow-up period (Figure 6).
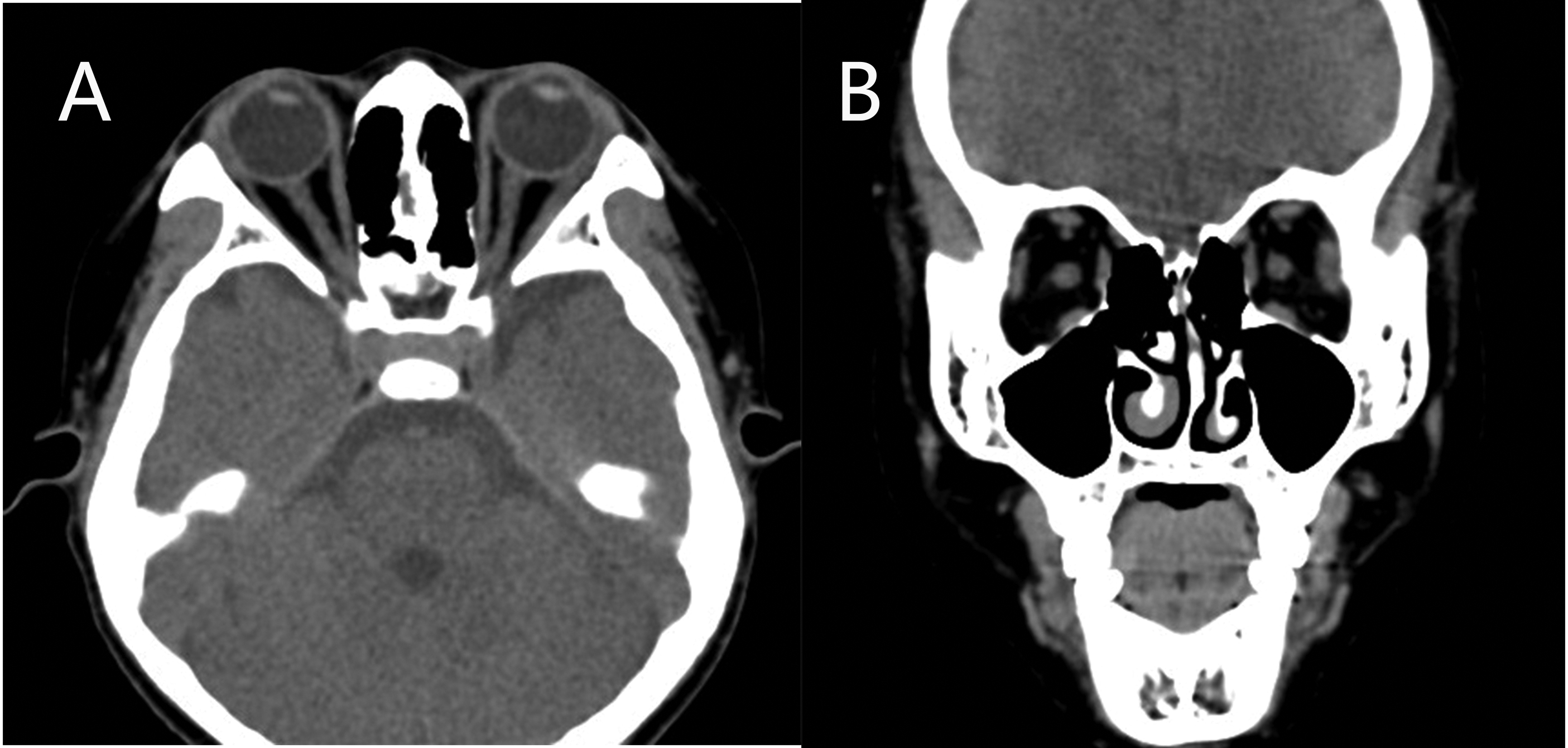
Orbital CT revealed that all extraocular muscles of both eyes were close to normal (a) axial CT scan and (b) coronal CT. CT: computed tomography.
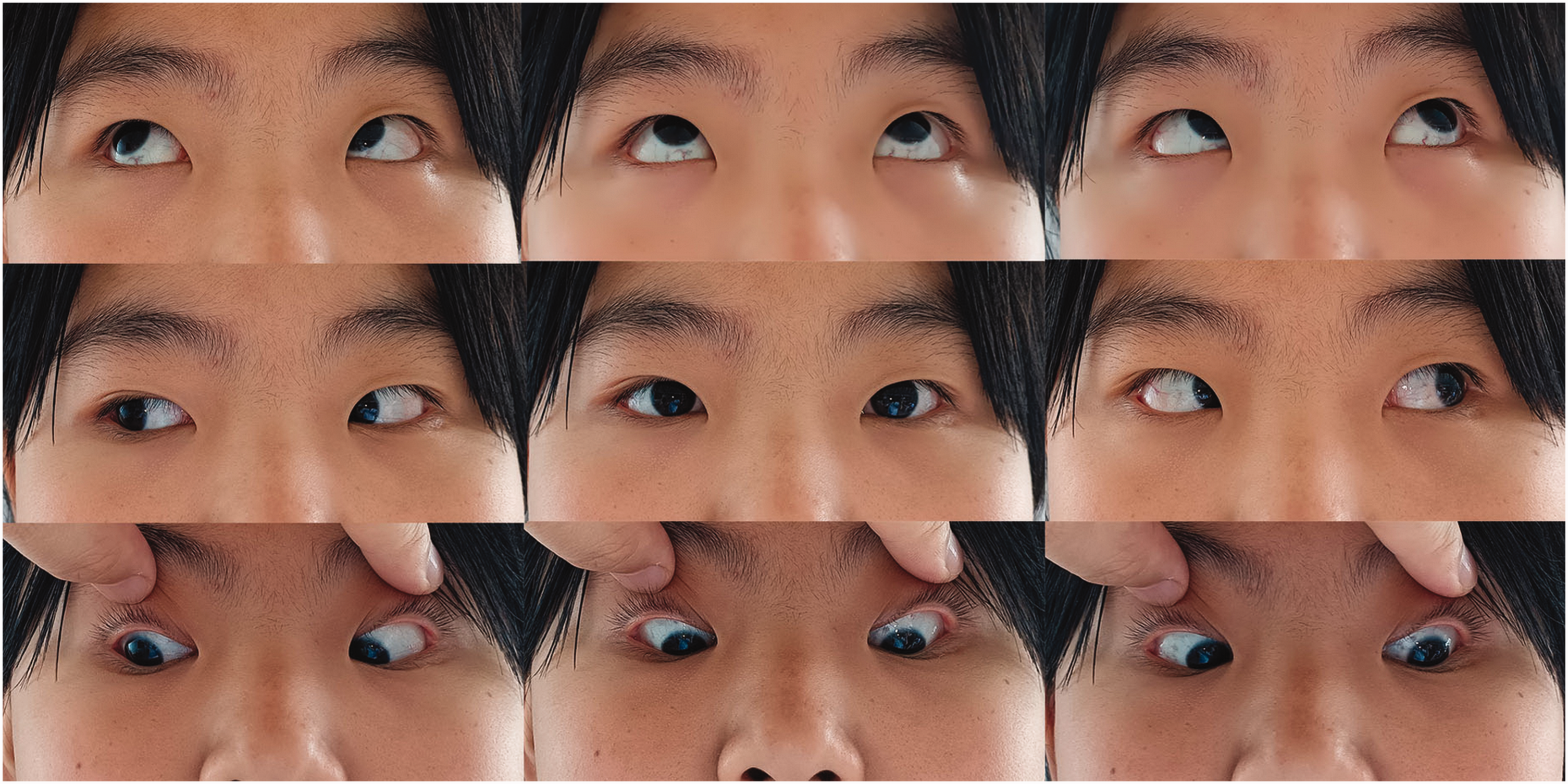
After a 3-month follow-up, extraocular muscle movements were normal.
Discussion
The reported COVID-19–related ophthalmic conditions include conjunctivitis, scleritis, uveitis, endogenous endophthalmitis, retinal artery and vein occlusions, nonarteritic ischemic optic neuropathy, glaucoma, and neurological and orbital sequelae. These manifestations arise through the following three principal mechanisms: (a) immune dysregulation causing ocular inflammation; (b) hypercoagulability leading to thrombotic events; and (c) immunosuppression predisposing to secondary infections. 1 Extraocular myositis is a rare clinical finding, with etiologies including systemic inflammation, autoimmune diseases, and infection. Potential mechanisms include the following: (a) direct viral entry via angiotensin converting enzyme (ACE-2) receptors on muscle cells 9 or (b) T cell–mediated inflammation and cytokine-driven damage. 10 The susceptibility of extraocular muscles to inflammation may be increased by their unique microanatomy—characterized by rich vascularity and high mitochondrial density—which facilitates inflammatory cell recruitment. 11
The primary presentation in this patient was diffuse enlargement of the extraocular muscles, accompanied with restricted ocular mobility and visual disturbances. Bilateral extraocular muscle inflammation necessitates consideration of thyroid-associated ophthalmopathy (TAO) and orbital inflammatory pseudotumor as primary differential diagnoses. These conditions, although more common in adults, should be ruled out in children and adolescents. However, patients with TAO typically present with ocular signs such as eyelid retraction or abnormal thyroid function.
Orbital cellulitis, an acute infection secondary to sinusitis, trauma, or skin infection, was ruled out based on the absence of fever, leukocytosis, and rapid steroid response. Crucially, myositis pain localizes to eye movements rather than diffuse warmth/erythema, with imaging demonstrating selective tendon–myofiber involvement without fat stranding or abscess.
Anti-neutrophil cytoplasmic antibody (ANCA)–associated vasculitis (AAV), which may cause granulomatous orbital masses, was excluded by negative serology (p/c-ANCA) and absence of multiorgan involvement. ANCA plays both pathogenic and diagnostic roles in AAV, making serologic testing essential for confirmation. 12
IgG4-related ophthalmic disease typically presents with bilateral lacrimal gland enlargement, infraorbital nerve thickening, and compressive optic neuropathy and is histologically characterized by IgG4+ plasma cell infiltration and elevated serum IgG4 levels. 13 This contrasts sharply with our case, which showed no lacrimal involvement, no normal serum IgG4 levels, and no mass effect on imaging.
Similarly, lupus orbitopathy—a rare complication of systemic lupus erythematosus (SLE) characterized by painful proptosis and restricted motility—is commonly associated with systemic manifestations (e.g. malar rash and serositis) and serologic markers (antinuclear antibody (ANA)/anti-dsDNA). 14 Our patient did not meet the SLE diagnostic criteria and had no serologic evidence, excluding this etiology.
Sarcoidosis—a multisystem granulomatous disorder—may involve the orbit, often presenting as anterior quadrant masses. Bilateral hilar lymphadenopathy on chest imaging and elevated serum ACE levels support the diagnosis. 15 In this case, the absence of pulmonary abnormalities on imaging, along with normal serum ACE levels, excluded sarcoidosis as a potential diagnosis.
Lymphoproliferative disorders encompass reactive lymphoid hyperplasia, atypical lymphoid hyperplasia, and orbital mucosa-associated lymphoid tissue (MALT) lymphoma. These typically manifest as painless proptosis, eyelid or conjunctival swelling, palpable masses, or diffuse muscle enlargement mimicking myositis. In contrast, orbital myositis typically exhibits a rapid and significant response to corticosteroid therapy, whereas lymphoma demonstrates poor or only transient response, ultimately requiring biopsy for diagnosis. 16
Ocular myositis falls within the spectrum of overall classification of idiopathic orbital inflammatory diseases, which are defined as noninfectious, nonspecific orbital inflammations without identifiable local or systemic causes. Orbital myositis may also occur as part of more widespread systemic inflammatory processes such as Crohn’s disease and the more recently described IgG4-related disease. 17 This condition is an exclusive diagnosis and requires the exclusion of ocular inflammation with a clear cause. As the patient had a well-defined course of viral infection, a diagnosis of idiopathic orbital inflammatory disease was not considered appropriate.
A prior case report documented a 12-year-old Caucasian male who developed bilateral extraocular muscle and lacrimal gland enlargement following SARS-CoV-2 infection, without vision loss, characteristic systemic manifestations, or pleural effusion. 18 In the present case, the patient developed isolated pleural effusion without pulmonary inflammation or other systemic manifestations—an uncommon finding in COVID-19. 19 Pleural effusions have been most frequently observed in critically ill patients and those with multisystem inflammatory syndrome. COVID-19–related pleural effusions were identified 5–7 days after hospital admission and approximately 11 days after the onset of COVID-19 symptoms. 20 Cytomorphologic changes in effusion fluid specimens from COVID-19 patients may help cytopathologists better understand and manage effusion cytology cases, ultimately improving diagnostic triage. 21 In our patient, thoracentesis was not performed promptly because of worsening ocular symptoms. However, these symptoms improved, and when cytologic analysis was planned, imaging revealed complete resolution of the effusion. No abnormalities were detected on blood testing. After discussion and consultation with experts in pediatrics and respiratory medicine, we believed that the effusion was most likely an immune-related exudate. A meta-analysis revealed that COVID-19 patients with pleural effusion had significantly lower blood lymphocyte counts and elevated platelet, CRP, LDH, and D-dimer levels compared with those without pleural effusion, suggesting that a sustained inflammatory response and cytokine storm underlie this complication. 22
The patient presented with bilateral reduction in visual acuity and sluggish direct/consensual pupillary responses. VEP testing was performed to exclude retrobulbar optic neuritis. Due to weekend admission constraints, VEP testing was delayed until post-treatment day 3, by which time ocular symptoms had significantly improved. Imaging revealed inflammatory exudative changes in the retrobulbar space and around the optic nerve, suggesting that the vision loss was likely caused by inflammation affecting the optic nerve. Therefore, prompt disease control is essential to prevent continuous progression and deterioration, which can result in severe damage to visual function.
Systemic inflammatory biomarkers (e.g. CRP and ESR) reflect the magnitude of systemic inflammation. 23 Although nonspecific for ocular pathology and never diagnostic, serial measurements provide valuable adjunctive data: persistently elevated levels indicate suboptimal disease control, necessitating treatment reassessment. Ophthalmic findings and clinical symptoms remain primary determinants of immunomodulatory therapy, with CRP/ESR serving supplemental roles.
Corticosteroids remain the only medication conclusively shown in clinical trials to be effective in the treatment of COVID-19. 24 Although widely used in combating chronic inflammatory diseases, corticosteroid therapy carries significant trade-offs, most notably an increased risk of infection. 25 Despite concerns regarding the side effects, we cannot reduce the corticosteroid doses or discontinue their administration abruptly. In this case, rapid reduction (as in our initial oral switch) provoked recurrence at day 5.
This case establishes SARS-CoV-2 infection as a trigger for severe bilateral extraocular myositis. Corticosteroids are effective in the management of such patients. However, corticosteroids should not be withdrawn too rapidly to prevent a relapse. Concurrently, close monitoring for adverse side effects of high-dose therapy is essential.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251384077 - Supplemental material for Immune-mediated extraocular myositis in an adolescent following coronavirus disease 2019: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605251384077 for Immune-mediated extraocular myositis in an adolescent following coronavirus disease 2019: A case report by Yixiang Wu, Xiao Chen, Zheng Fan and Mingyu Ren in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank the patient for providing consent for publication of this case report.
Author contributions
Collection and management of the data: Y.W. and X.C.; preparation of the manuscript: Y.W. and Z.F.; review and approval of the manuscript: M.R. All authors have read and approved the manuscript.
Data availability statement
The data supporting the findings of this study are available within the article and its supplementary materials.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest regarding the publication of this study.
Ethics statement and informed consent
The study was approved by the Ethics Committee of Hebei Eye Hospital, under registration number (2025LW03). The participant and their guardians provided informed consent. The reporting of this study conforms to the Case Report (CARE) guidelines.
Funding
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
