Abstract
This report describes the case of a 62-year-old woman with type 2 diabetes and painful diabetic peripheral neuropathy, who, despite standard treatment, experienced persistent symptoms. Further investigation revealed erythromelalgia secondary to polycythemia vera, confirmed by bone marrow biopsy and JAK2 V617F mutation. The patient received a multidisciplinary regimen, including pregabalin, aspirin, and interferon-alpha, resulting in significant symptom relief and improved hematologic parameters. Follow-up over 2 years confirmed sustained symptom resolution, highlighting the importance of considering concurrent or secondary pain factors when diagnosing and treating refractory neuropathic pain to avoid misdiagnosis or missed diagnosis.
Introduction
Painful diabetic neuropathy (PDN), a prevalent form of diabetic peripheral neuropathy (DPN), affects approximately one-third of patients with diabetes. 1 This condition presents substantial management challenges due to its diverse symptomatology, which encompasses both paresthesias (numbness, tingling, prickling, and a feeling similar to ants crawling on the body) and various neuropathic pain phenotypes, such as burning pain, electric shock–like pain, stabbing pain, shooting pain, hyperalgesia (exaggerated response to noxious stimuli), and allodynia (pain evoked by normally innocuous stimuli).2–3 The pathogenesis of DPN is currently believed to be primarily associated with metabolic abnormalities, such as hyperglycemia, dyslipidemia, and insulin resistance, as well as microvascular dysfunction. These factors can lead to widespread neural damage, affecting neuronal cell bodies, small nerve fibers, and large nerve fibers and manifesting as hypoxia, DNA damage, endoplasmic reticulum stress, Schwann cell apoptosis, and impaired mitochondrial transport. 4 On this basis, the upregulation of voltage-gated sodium channels (Nav1.7, Nav1.8, and Nav1.9) in unmyelinated C fibers as well as the reduction of Kv channels in myelinated fibers can lead to neuronal hyperexcitability, thereby promoting the development of PDN. 4 The complexity of PDN pathogenesis has prompted the development of multiple pharmacological treatments, including duloxetine (a serotonin–norepinephrine reuptake inhibitor), pregabalin and gabapentin (anticonvulsants), amitriptyline (antidepressant), tapentadol and tramadol (opioid receptor agonists with norepinephrine reuptake inhibition properties), and 8% capsaicin patch. 1 These medications, either used alone or in combination, can provide some degree of pain relief for patients with PDN.
Erythromelalgia (EM) is a rare disorder characterized by intense burning pain, increased local temperature, and marked erythema of the skin, predominantly affecting the distal extremities. 5 The disorder can be classified into two types: primary and secondary. The latter occasionally occurs in association with polycythemia vera (PV), a myeloproliferative neoplasm involving clonal expansion of morphologically normal hematopoietic lineages and characteristic JAK2 mutations. 6 Current therapeutic options for PV-associated EM include low-dose aspirin, therapeutic phlebotomy, and cytoreductive agents such as hydroxyurea and interferon-alpha (IFN-α), and JAK2 inhibitors (ruxolitinib). These interventions have demonstrated efficacy in symptom control, disease modulation, and prevention of thrombotic sequelae. 7
We herein describe the diagnostic workup and multidisciplinary therapeutic approach for a rare case of concomitant PDN and secondary EM arising from PV, highlighting the clinical challenges posed by this complex comorbidity.
Case report
The reporting of this study was conducted in accordance with the Case Report (CARE) guidelines. 8 The patient provided written informed consent for publication of this case report, and her anonymity was strictly maintained.
A woman in her early 60s was admitted to the Department of Endocrinology and Metabolism at the First Affiliated Hospital of Zhejiang Chinese Medical University in Hangzhou, Zhejiang, China, in April 2022 due to poorly controlled plasma glucose and severe pain in her lower limbs. The patient had a history of type 2 diabetes for more than 10 years, with poor glucose control despite treatment with metformin (1 g twice daily), canagliflozin (0.1 g once daily), saxagliptin (5 mg once daily), pioglitazone (30 mg once daily), and acarbose (100 mg thrice daily).
The patient reported experiencing numbness, tingling, and burning pain in both lower limbs, particularly in the feet, for the past 4 months. Following an initial diagnosis of PDN at a local hospital in March 2022, she underwent treatment with neurotrophic and neuropathic pain-relieving medications, including mecobalamin (0.5 g thrice daily), α-lipoic acid (0.6 g once daily), epalrestat (50 mg thrice daily), and pregabalin (75 mg twice daily). However, the symptomatic relief was unsatisfactory.
Additionally, the patient had a 10-year history of well-controlled essential hypertension and was maintained on felodipine sustained-release tablets (5 mg once daily). She reported no other significant medical history and no history of alcohol consumption or other medication use. Moreover, she denied any family history of infectious diseases or malignant tumors. No notable psychosocial history was reported.
The admission physical examination revealed mildly erythematous skin with elevated temperature over the bilateral anterior plantar region and toes, especially the left little toe, accompanied by slight toe swelling. Peripheral pulses were diminished in the dorsalis pedis, anterior tibial, and posterior tibial arteries bilaterally. Neurological examination revealed diminished ankle reflexes bilaterally; impaired vibration sense; and decreased sensitivity to pressure, temperature, and pinprick stimuli, with abnormal sensory threshold measurements. Laboratory investigations demonstrated abnormal hematologic parameters, including leukocytosis (10.3 × 109/L; reference range: 3.5–9.5 × 109/L), erythrocytosis (5.96 × 1012/L; reference range: 3.8–5.1 × 1012/L), thrombocytosis (355 × 109/L; reference range: 125–350 × 109/L), elevated hemoglobin (HGB) (163 g/L; reference range: 115–150 g/L), and increased hematocrit (HCT) (49.7%; reference range: 10.8%–28.2%). Additionally, HGB A1c was markedly elevated at 13.4% (reference range: 4.0%–6.0%). Immunologic workup, including antinuclear antibody (ANA), antineutrophil cytoplasmic antibody (ANCA), erythrocyte sedimentation rate (ESR), antistreptolysin O (ASO), anti-cyclic citrullinated peptide antibody (CCP), light chains, and cryoglobulins, returned negative results. Abdominal ultrasound revealed a slightly enlarged spleen. Cranial magnetic resonance imaging demonstrated chronic lacunar infarcts in the right semioval center and basal ganglia. Electromyography showed the following: (a) slowed conduction velocity in the left common peroneal motor nerve; (b) prolonged distal latency of left common peroneal nerve motor conduction compared with the contralateral side; (c) mildly reduced amplitude of left tibial nerve motor evoked potentials; (d) bilateral slowing of superficial peroneal sensory nerve conduction velocities; and (e) decreased sensory potential amplitudes in both superficial peroneal and sural nerves. Lower extremity computed tomography angiography revealed atherosclerotic stenosis affecting the distal abdominal aorta, bilateral common and internal iliac arteries, and distal right external iliac artery.
Based on the above findings and characteristic clinical manifestations, the patient was diagnosed with PDN complicated with diabetic macroangiopathy. A comprehensive treatment regimen was implemented, including the following: mecobalamin (500 μg thrice daily) for nerve regeneration, pentoxifylline (400 mg twice daily) to improve peripheral circulation, pregabalin (started at 75 mg twice daily and titrated up to 150 mg twice daily) for neuropathic pain control, and biphasic insulin aspart 30 (12 units before breakfast and 10 units before dinner) for glycemic control. Concurrently, we assessed the patient’s pain using the visual analog scale (VAS). 9 After 5 days of treatment, the patient’s VAS score for the toes showed improvement (Figure 1(a) and (b)) but remained around 4–5. The burning pain reportedly worsened at night and with heat exposure (e.g. warm water foot baths), while it improved with cold exposure. Given the patient’s skin and soft tissue abnormalities, including bilateral forefoot and toe erythema, increased skin temperature, and toe swelling, along with hematological abnormalities such as elevated HGB and HCT that could not be explained by the existing diagnosis and medical history, we consulted specialists in dermatology, rheumatology, and hematology. Further investigations, including cold water immersion tests, bone marrow aspiration, and genetic testing, were conducted. The cold water immersion test demonstrated a reduction in the patient’s VAS pain score after soaking her feet in cold water for 3–5 min (Figure 1(c) and (d)). Bone marrow biopsy revealed trilineage hyperplasia (involving erythroid, granulocytic, and megakaryocytic lineages) with polymorphic mature megakaryocytes, along with the detection of a JAK2 V617F mutation. Based on the patient’s clinical presentation and test results, a diagnosis of PV-induced EM was considered. Consequently, a low dose of aspirin (100 mg once daily) was added to the original treatment regimen. After 1 week, the patient’s foot pain had significantly improved (Figure 1(e)).

Changes in the patient’s pain intensity scores (measured using VAS) before and after interventions. (a) Status of the left fifth toe after 1 week of pharmacotherapy (including aspirin): pain score of 2/10. (b) Status of both feet after 5 days of neurotrophic and analgesic therapy: pain score of 4–5/10. (c) Assessment of the left foot after 5 min of cold water immersion revealed a skin temperature of 30°C and pain score of 3–4/10. (d) Assessment of the right foot after 3 min of cold water immersion revealed a skin temperature of 33°C and pain score 5–6/10 and (e) status of the left fifth toe before any pharmacotherapy: pain score of 7–8/10. VAS scores range from 0 (no pain) to 10 (worst imaginable pain). VAS: visual analog scale.
The patient subsequently attended regular outpatient follow-ups. During the follow-up visit at the hematology outpatient clinic 1 month later, it was noted that her HGB and HCT levels had progressively increased (HGB level: 176 g/L, HCT level: 56.6%). Thus, IFN-α therapy was added to the existing treatment regimen for cytoreduction (300 million units per dose, thrice a week, subcutaneously). The patient was followed up for over 2 years, primarily through outpatient visits to the departments of endocrinology and hematology. Initially, follow-ups were conducted monthly. With improvement in erythema, pain, blood glucose levels, and hematologic parameters, the intervals between visits were gradually extended to 3–9 months. The most recent follow-up in February 2025 showed complete resolution of pain symptoms, with hematologic parameters indicating mild anemia (HGB level: 104 g/L, HCT level: 31.6%) (Figure 2). The timeline of key events in the patient’s diagnosis and treatment process is presented in Figure 3.
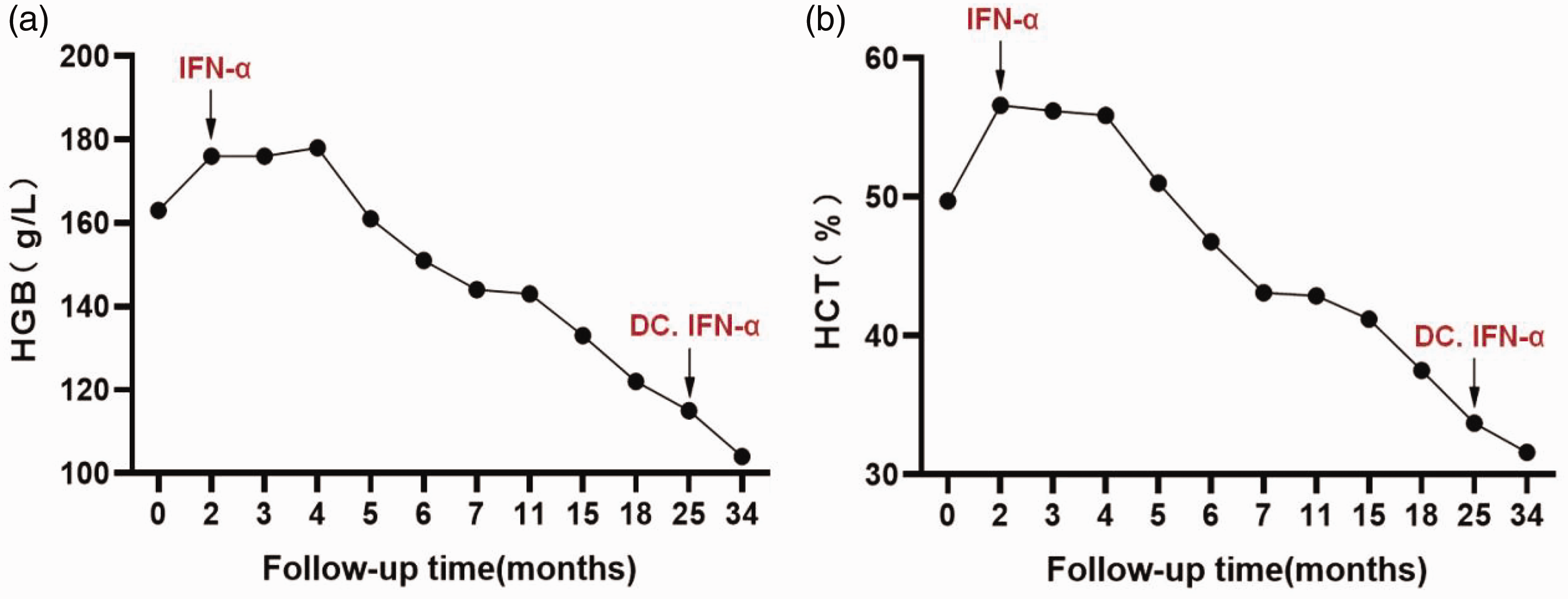
Monitoring of (a) hemoglobin and (b) hematocrit levels before and after IFN-α treatment.

Timeline of key diagnostic and therapeutic interventions in this case.
Discussion
The patient in this case had a long history of diabetes and presented with characteristic neuropathic symptoms, including burning pain, numbness, and tingling, predominantly affecting the bilateral lower extremities, particularly the feet. Based on neurological examination and electromyography findings and in accordance with the “Chinese Expert Consensus on the Diagnosis and Treatment of Diabetic Neuropathy (2021 edition),” 10 the diagnosis of PDN was established. The patient received treatments including mecobalamin, α-lipoic acid, ipratropium, and pregabalin for nerve nutrition and pain relief. However, symptomatic relief was unsatisfactory, suggesting the presence of overlapping or secondary pain causes. Further examination revealed redness, slightly elevated skin temperature, and mild swelling of the forefoot and toes, along with elevated blood cell counts. After consultation with rheumatology, hematology, and other relevant departments and following tests such as cold water testing, bone marrow examination, and genetic testing, the final diagnosis was established as PDN complicated by EM secondary to PV, in line with the World Health Organization (WHO) (2016) criteria for PV. 11 This suggests that secondary EM is a significant factor contributing to the persistence of pain despite standardized treatment for PDN.
The global prevalence of DPN is approximately 30%. 12 DPN is associated with dysfunction of small-fiber sensory neurons, impairing the detection of temperature and pain. 13 Consequently, approximately a quarter of individuals with DPN may present with hyperalgesia or allodynia, a condition known as PDN. 14 PDN is characterized by sensations such as burning, electric shock–like pain, tingling, and lancinating pain, primarily affecting the toes and feet. 14 The pain intensity of PDN ranges from moderate to severe and may persist for several years, often worsening at night or with heat exposure. 15
Similar to PDN, EM presents with intense burning pain in the distal limbs, often affecting the feet. The pain typically occurs at night and can be triggered by movement, heat, or the affected limb hanging down. Cooling the affected limb, either through cold water immersion or using a fan, or elevating the limb can provide relief. 16 In addition to pain, erythema and elevated skin temperature are key features of EM and can help differentiate it from PDN. Erythema typically appears on the palms and soles but may extend to the dorsal surfaces of the hands and feet, often in a bilateral and symmetric distribution. In some cases, erythema may extend proximally or remain unilateral. It is worth noting that in patients with darker skin tones, these skin manifestations may be less pronounced or more difficult to detect, as observed in the current case. 17
Although the erythema in this patient was atypical due to the darker skin color, the clinical features observed—such as increased skin temperature, pain relief with cold exposure, and elevated blood cell counts—suggest secondary EM. Secondary EM is associated with various diseases and drugs, including connective tissue diseases (systemic lupus erythematosus and rheumatoid arthritis), infectious diseases (human immunodeficiency virus (HIV) infection and poxvirus infection), and certain medications (bromocriptine and cyclosporine). Among these, myeloproliferative disorders such as PV and essential thrombocythemia are the most common well-established causes.17–18 PV is a myeloproliferative neoplasm characterized by increased red blood cell count and a higher risk of thrombosis, with over 95% of patients carrying a JAK2 mutation. Studies have shown that EM occurs in approximately 5.3% of PV patients. 19 Following the diagnosis of PV in the current case, the patient’s lower limb pain significantly improved with treatment using aspirin and IFN-α, further suggesting that the pain was not solely caused by PDN.
It is noteworthy that a previous case report described an association between PV, EM, and peripheral sensorimotor axonal neuropathy, with the proposed pathogenesis involving tissue hypoxia secondary to blood hyperviscosity. 6 In this report, the patient had no history of diabetes, and phlebotomy therapy proved effective. 6 Although the EM in our patient was considered secondary to PV, previous reports have indicated that diabetes indirectly contributes to the development of EM through the onset of neuropathy.20–22 In contrast to our case, gabapentin has been reported to produce significant pain relief in diabetes-related secondary EM, 22 possibly because neurological dysfunction plays a key role in EM pathogenesis. This dysfunction includes damage to both distal small fibers and large fibers, such as lesions affecting C-type nociceptive fibers.23–25 Consequently, pharmacologic agents commonly used to manage neuropathic pain in PDN, including gabapentin, pregabalin, sertraline, amitriptyline, and capsaicin, have also been shown to be effective in treating EM.20,26 The pathogenesis of EM is complex; in addition to neuropathy, it is associated with mutations in the Nav1.7 sodium channel gene, vascular dysregulation, and microcirculatory disturbances. 16 In addition to the abovementioned medications, sodium channel blockers such as mexiletine, carbamazepine, and lidocaine as well as vasodilators such as nitroprusside and misoprostol can be used to treat EM.20,26 Notably, different subtypes of EM, including primary, secondary, and idiopathic EM, may have distinct dominant pathophysiological mechanisms. Studies have shown that in cases of EM related to PV, platelet dysfunction is a key pathogenic mechanism: platelets are activated and aggregate in small arteries, leading to thrombotic occlusion, inflammation, and smooth muscle cell proliferation. Aspirin can irreversibly inhibit cyclooxygenase, reducing the production of prostaglandins and thromboxane. This also explains why the patient’s pain symptoms significantly improved after adding aspirin to the treatment regimen for PDN (Table 1).
Comparison between painful diabetic neuropathy and erythromelalgia.

EM: erythromelalgia; PV: polycythemia vera; HIV: human immunodeficiency virus; IFN-α: interferon-alpha.
In this case, the patient was regularly treated with IFN-α for PV-associated secondary EM, and follow-up assessment over 2 years showed sustained remission of peripheral blood counts (bone marrow histological examination was not repeated due to patient refusal). Concurrently, her lower limb pain symptoms were alleviated. Previous studies have suggested that interferons play a role in neuroprotection and neurological repair.27–28 For instance, IFN-γ has been shown to enhance the ability of human umbilical cord mesenchymal stem cells to alleviate damage to diabetic nerve fibers and Schwann cells, thereby improving neurological function in diabetic mice. 27 Similarly, high-dose, long-term treatment with IFN-α-2b has demonstrated neuroprotective effects on retinal pigment epithelial cells. 28 These findings suggest that IFN therapy ameliorates neurological damage associated with both PDN and EM. Nevertheless, the specific therapeutic efficacy and underlying mechanisms of IFN-α in alleviating pain symptoms in patients with EM and concomitant PDN remain to be further validated with additional clinical evidence. Other research has indicated that injecting the JAK2 inhibitor AG490 into the dorsal horn of the spinal cord of diabetic neuropathy rats may effectively alleviate neuropathic pain by inhibiting the JAK2/STAT3-CAV-1-NR2B pathway. 29 The traditional Chinese medicine Jinmaitong can regulate the activation of microglia by inhibiting the JAK2/STAT3 signaling pathway, effectively improving diabetic neuropathic pain. 30 Based on these findings, JAK2 inhibitors such as ruxolitinib may be considered for patients with PV-related secondary EM combined with PDN if IFN therapy is not tolerable, as they can effectively treat PV and potentially improve neuropathic pain in these patients.
Limitations
Although this report describes a rare case of PDN complicated by PV-associated secondary EM, with potential clinical diagnostic and therapeutic implications, several limitations should be acknowledged. First, as a single-case observation, these findings may reflect individualized characteristics influenced by patient-specific factors and the authors’ subjective judgment. Therefore, the diagnostic and therapeutic experience reported here requires validation through more relevant cases. Second, some clinical data were incomplete. Specifically, the documentation of cutaneous erythema during hospitalization was insufficiently detailed, and post-discharge follow-up lacked a comprehensive and systematic assessment of pain and lower extremity erythema; the evaluations were limited to verbal inquiry and visual observation.
Conclusions
In summary, we report a rare case of PDN complicated by EM secondary to PV. This case highlights the need to consider overlapping or secondary pain etiologies when standard treatments for PDN, such as neurotrophic therapy and analgesics, provide insufficient relief. Comprehensive clinical evaluation and targeted investigations are essential to avoid misdiagnosis or missed diagnosis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251382382 - Supplemental material for Painful diabetic neuropathy complicated with secondary erythromelalgia in polycythemia vera: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605251382382 for Painful diabetic neuropathy complicated with secondary erythromelalgia in polycythemia vera: A case report by Hong Zhang, Zhengran Qin and Xiaohong Feng in Journal of International Medical Research
Footnotes
Acknowledgments
The authors acknowledge the assistance of ChatGPT (OpenAI, USA) for language refinement in this manuscript. The AI tool was used exclusively for grammatical and fluency enhancement; all academic content and interpretations were independently formulated by the authors, thus preserving the originality of the study.
Author contributions
All authors participated in the clinical management of the patient. Hong Zhang and Zhengran Qin drafted the manuscript, and Hong Zhang and Xiaohong Feng revised it. All authors reviewed and approved the final version of the manuscript.
Consent for publication
Written informed consent for publication of this case report was obtained from the patient.
Data availability statement
All relevant data are included in this article.
Declaration of conflicting interest
The authors declare no conflict of interest regarding the publication of this study.
Ethical approval
This case report was approved by the Ethics Committee of The First Affiliated Hospital of Zhejiang Chinese Medical University, Hangzhou, China (Approval No. 2025-KLS-410-01; Date: 12 June 2025).
Funding
This study was supported by the Traditional Chinese Medicine Science and Technology Plan Project of Zhejiang Province (2022ZB102).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
