Abstract
Autosomal recessive congenital ichthyosis is a group of skin disorders characterized by abnormal keratinization. The collodion baby phenotype is a rare phenotype of autosomal recessive congenital ichthyosis characterized by a tight, translucent membrane that encases the newborn, which leads to significant medical challenges. This case report describes a male infant born at 35 weeks and 6 days of gestation who presented with the collodion baby syndrome. The present case exhibited a nonbullous congenital ichthyosiform erythroderma–like phenotype, characterized by diffuse erythema and fine scaling. Genetic analysis revealed two compound heterozygous mutations in TGM1: c.425G>T (p.Arg142Leu) in exon 3, previously reported as a pathogenic hotspot in nonbullous congenital ichthyosiform erythroderma, and a novel mutation, c.1198A>C (p.Asn400His) in exon 8, which has not only been associated with lamellar ichthyosis but has also been detected in nonbullous congenital ichthyosiform erythroderma. A comprehensive treatment strategy focused on symptom alleviation and supportive care led to significant improvement, and the infant was discharged after 6 days of hospitalization. This case highlights the importance of early diagnosis, multidisciplinary care, and genetic testing in managing autosomal recessive congenital ichthyosis. The identification of novel TGM1 mutations contributes to the expanding mutation spectrum and may inform future diagnostic and therapeutic approaches.
Introduction
Autosomal recessive congenital ichthyosis (ARCI) refers to a group of nonsyndromic phenotypes characterized by keratinization disorders. 1 The collodion baby phenotype is a rare manifestation of congenital ichthyosis, with an estimated prevalence of one in 300,000 live births. 2 These infants are typically born encased in a thick, taut, and translucent collodion membrane and often present with ectropion and eclabium. 3 Owing to impaired skin barrier function, affected infants are at high risk of dehydration, infections, electrolyte imbalances, feeding difficulties, and thermoregulatory issues, all of which contribute to increased mortality risk. 4 Close monitoring is essential to improve the quality of life of affected infants.
ARCI exhibits significant genetic heterogeneity and is associated with mutations in at least 13 genes. 1 Among these, mutations in TGM1 are the most frequently reported, accounting for 32%–51% of cases in different populations and often initially manifesting with the collodion baby phenotype.5,6 Genetic analysis is crucial for understanding the collodion baby phenotype, predicting the clinical severity of the disorder, and guiding targeted management for affected infants.
In this report, we present the case of a male infant with the collodion baby phenotype who demonstrated significant improvement following treatment. Exome sequencing revealed two novel mutations in TGM1 that are associated with ARCI.
Case presentation
A male infant was delivered prematurely at 35 weeks and 6 days of gestation in late 2023 at the Neonatology Division, Qingdao West Coast New Area People’s Hospital (Shandong, China). This was the mother’s fourth pregnancy and second live birth (G4P2). The delivery was spontaneous following signs of preterm labor. The infant weighed 2795 g at birth, with appearance, pulse, grimace, activity, and respiration scores of 10 at 1, 5, and 10 min after birth. There were no signs of intrauterine distress, premature rupture of membranes, or abnormalities in the amniotic fluid. The umbilical cord and placenta appeared normal. The mother, aged 41 years, was positive for Rh(D) blood type O. Her first delivery occurred at the age of 25 years and was followed by two miscarriages prior to this pregnancy. Laboratory findings before and after delivery revealed an elevated neutrophil-to-lymphocyte ratio; reduced red blood cell count and hemoglobin and hematocrit levels; and increased mean corpuscular volume and mean corpuscular hemoglobin level—all results were within twice the upper reference limit. However, her high-sensitivity C-reactive protein level was significantly elevated at 22.4 mg/L postdelivery—approximately four times the upper limit—warranting continued monitoring. Informed consent was obtained from the infant’s mother for treatment and publication of this case report. The reporting of this study conforms to the Case Report (CARE) guidelines. 7
Within 10 min of birth, the infant exhibited epidermal thinning, skin cracking, cyanosis, and irregular respiratory patterns (Figure 1). There was no mucosal involvement, nail abnormality, or bulla formation. Oxygen was immediately administered via face mask, and the infant was transferred to the neonatal intensive care unit (NICU). At NICU admission, the infant presented with erythema, cracked and peeling skin, a flat and soft anterior fontanelle, mild ectropion and eclabium, yellow–white crusting around the nose and mouth, ear malformations, small palpebral fissures, a flat nasal bridge, and syndactyly of the left hand. The torso and limbs were covered with diffuse erythema, dryness, extensive desquamation, and tense, shiny skin on the soles. Significant dry cracking was also observed at the popliteal fossa. These cutaneous findings were consistent with the collodion baby phenotype. Moreover, the infant had not initiated feeding or passed stool or urine. He remained responsive, with a strong cry and no significant respiratory distress or worsening cyanosis. Although the respiratory patterns were irregular, auscultation revealed no rales. Cardiac examination revealed strong, regular heart sounds; the abdomen was soft without abnormalities; and the limbs were warm, with only cyanosis. Dermatological consultation confirmed congenital ichthyosis. There was no parental consanguinity and no reported family history of the condition.
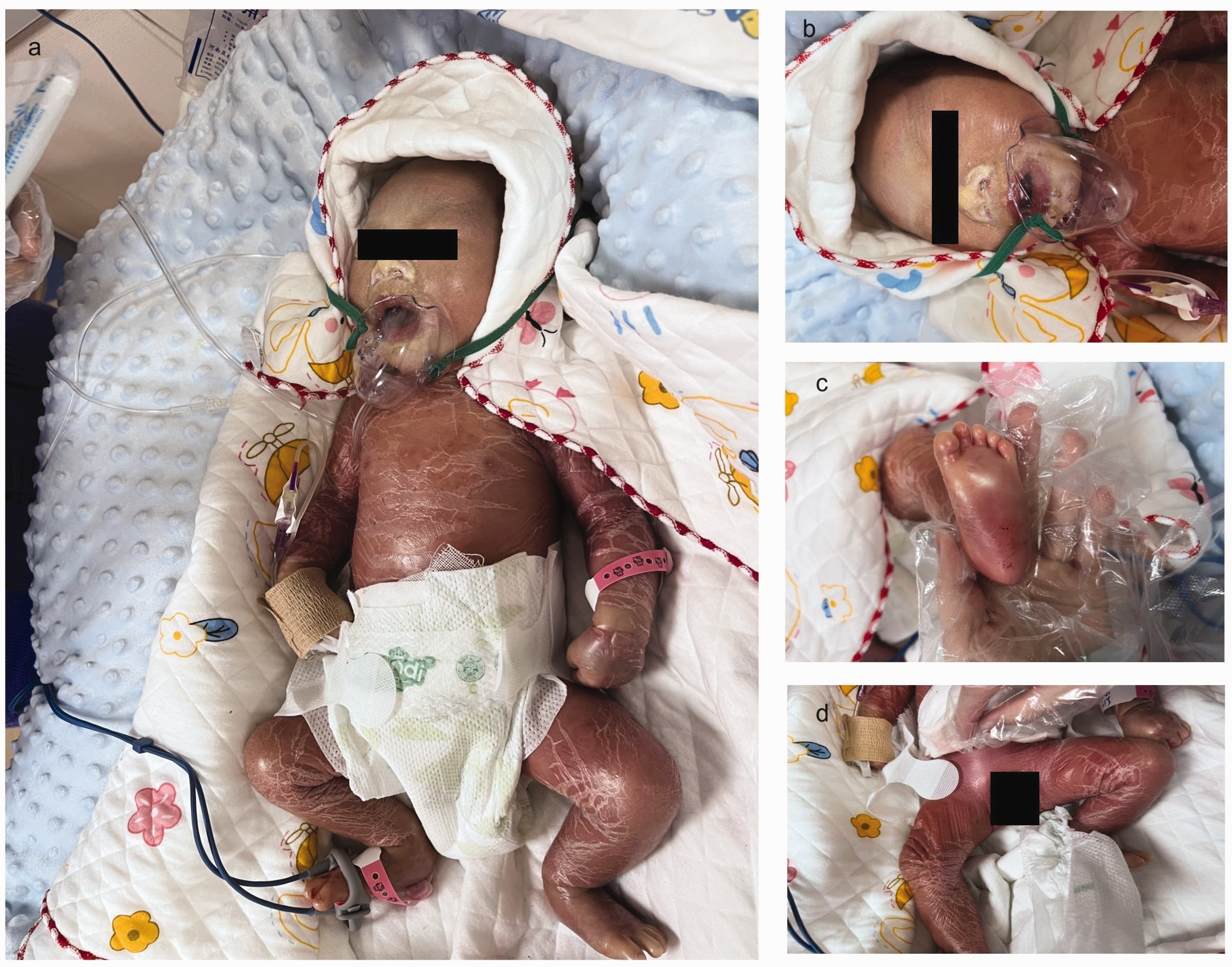
The baby with the collodion baby phenotype and generalized erythema, dry and cracked skin with peeling and tightness all over the body surface. (a) The full body frontal photograph. (b) The facial photograph. (c) The sole photograph and (d) the lower limb photograph. These pictures were taken on the day of birth. Written informed consent was taken from parents before image publication.
At 21 min after birth, the infant’s blood glucose was low at 2.5 mmol/L, and a 10% intravenous glucose solution was administered to stabilize blood glucose levels. Intravenous access was secured through intact skin, and warm saline compresses were used instead of alcohol-based disinfectants to minimize skin injury. Sterile vaseline or paraffin oil was applied to reduce the risk of skin tearing during cannulation. Upon admission, the infant was placed in an incubator to maintain warmth, with continuous monitoring of heart function (electrocardiogram) and oxygen saturation. Initial laboratory tests revealed several abnormal indicators (Table 1). Based on the diagnosis, silicone oil cream was applied regularly for skin care, and the eyes were protected with vaseline oil gauze. As preventive measures, vitamin K1 was given to reduce the risk of neonatal hemorrhage, and piperacillin–tazobactam was administered for 2 days to prevent infection. Glucose infusion was continued, with regular monitoring confirming normal blood glucose levels.
Timeline of main symptoms, treatments, increased indicators, and decreased indicators. a

ADA: adenosine deaminase; ApoB: apolipoprotein B; AST: aspartate aminotransferase; C1q: complement component C1q; Ca2+: free ionized calcium; CK: creatine kinase; CK-MB: creatine kinase-MB isoenzyme; COHb: carboxyhemoglobin; CysC: cystatin C; GCA: glycocholic acid; GGT: gamma-glutamyl transferase;
Elevated WBC and monocytes may reflect infection or inflammation due to impaired skin barrier, though values normalized with supportive care. Increased RDW suggests early anemia or hemolysis. Elevated lactate and COHb indicate transient perinatal stress or hemolysis. Mild increases in AST, GGT, and LDH reflect nonspecific hepatic stress and resolved spontaneously. Mild decreases in sodium, free calcium, and bicarbonate likely resulted from fluid/electrolyte loss through the compromised skin barrier and improved with hydration and feeding.
Two days after admission, follow-up laboratory tests revealed persistently elevated lactate levels, although all other indicators returned to normal. No pathogens were detected; however, the presence of immunoglobulin G antibodies against cytomegalovirus, rubella virus, and herpes simplex virus indicated prior maternal exposure. Owing to irregular prenatal check-ups, it was unclear whether these infections occurred during pregnancy. Evaluation for hemolytic disease of the newborn (HDN) revealed that the infant had an Rh–positive blood type and a positive dilution test, indicating risk for HDN. Bilirubin levels were monitored dynamically, and appropriate phototherapy was administered as indicated (Table 1).
On the third day of hospitalization, cardiac and pediatric cranial ultrasound examinations revealed no abnormalities (conducted at the bedside, with no imaging records retained). Breastfeeding was initiated, with intake gradually increasing to 50 mL every 3 h. By that day, all oral feedings were successfully completed.
After 6 days of hospitalization, the infant’s skin condition significantly improved. Tightness had decreased, although dryness, cracking, and desquamation persisted (Figure 2). Desquamation appeared in patches of varying sizes with free edges, but the extent of scaling and cracking had significantly decreased compared with that at admission. Overall skin thickness had also decreased. His parents requested his discharge on these improvements. Before discharge, the parents received detailed guidance on skin care, including frequent daily application of emollients and immediate moisturization after bathing. With parental consent, whole blood samples were collected from both the infant and parents for clinical exome sequencing and copy number analysis. Genetic testing identified heterozygous mutations at positions 425 and 1198 of TGM1 (Table 2),8–21 associated with ARCI. Specifically, the mutation c.425-G > T was maternally inherited, and c.1198-A > C was paternally inherited on the basis of the sequencing results of the parents.

Skin picture of the infant after 6 days of hospitalization.
TGM1 gene variants and associated ichthyosis subtypes.

BSI: bathing suit ichthyosis; LI: lamellar ichthyosis; NBCIE: nonbullous congenital ichthyosiform erythroderma; SICI: self-improving collodion ichthyosis.
The infant has been followed monthly since discharge. Follow-up evaluations included assessments of scaling severity, fissure healing, signs of infection, and the effectiveness of emollient therapy. Growth parameters—weight, length, and head circumference—were also regularly monitored. To date, the infant has maintained good skin condition, with no signs of infection, and has demonstrated normal growth and developmental progress. We deidentified all patient details.
Discussion
ARCI manifests at birth and persists throughout the lifetime, encompassing multiple subtypes with various clinical presentations that range from mild forms, such as ichthyosis vulgaris, to more severe forms, such as lamellar ichthyosis (LI).15,22 Diagnosis of ichthyosis usually requires a combination of clinical presentation, skin biopsy, genetic testing, and family history. In the present case, the infant demonstrated transient abnormalities in several clinical indicators during the first 6 days, including elevated white blood cell/monocytes, red cell distribution width, lactate, carboxyhemoglobin, aspartate aminotransferase, gamma-glutamyl transferase, and lactate dehydrogenase, along with mildly decreased sodium, calcium, and bicarbonate (Table 1). According to the Chinese National Health Industry Standard (WS/T 779-2021): Reference Intervals of Blood Cell Analysis for Children, these changes reflected secondary responses such as inflammation, stress, or electrolyte loss due to the impaired skin barrier, all of which normalized with supportive care. Although not specific to collodion babies, such findings highlight the importance of close monitoring for complications in this high-risk population. The diagnosis of the collodion baby phenotype was based on characteristic clinical features and further confirmed by genetic testing. The infant exhibited signs of congenital ichthyosis immediately after birth, with fine scaling and diffuse erythema supporting a diagnosis of nonbullous congenital ichthyosiform erythroderma (NBCIE). The distribution involving the central face and extremities was not consistent with bathing suit ichthyosis (BSI), 21 the absence of a thick collodion membrane excluded harlequin ichthyosis, the lack of spontaneous improvement made self-improving collodion ichthyosis (SICI) unlikely, 23 and the absence of bullae ruled out epidermolytic ichthyosis. 24 In our case, both LI and NBCIE were considered. The predominance of diffuse erythema and fine scaling may be more consistent with NBCIE, whereas LI usually manifests with large, dark, thick, plate-like scales. 25 Genetic findings were discussed to complement the clinical diagnosis; nevertheless, as skin biopsy can increase diagnostic accuracy in ichthyosis, we recommend including it in similar cases in the future.
ARCI is genetically heterogeneous and has been associated with at least 13 genes to date, with TGM1 gene mutations being among the most common causes. 26 TGM1 encodes transglutaminase 1 (TGase 1), an enzyme crucial for the formation of the cornified envelope by facilitating the calcium-dependent cross-linking of various proteins. Over 110 mutants in TGM1 have been reported across different populations and are account for some cases of ARCI, 1 particularly NBCIE, LI, BSI, SICI, and phenotypes that exhibit overlap between LI and NBCIE (Table 2).8–21 Mutations associated with BSI and SICI are predominantly located in exons 6 and 7, whereas those linked to LI are primarily found in exons 4, 5, 7, and 8. Moreover, variants associated with NBCIE tend to cluster in exons 3 and 13. In our patient, we detected compound heterozygous mutations in TGM1: c.425-G > T (p.Arg142Leu) in exon 3 and c.1198-A > C (p.Asn400His) in exon 8 (Table 2). Notably, a mutation at amino acid 142 (p.Arg142Cys) in exon 3 has been previously reported as a pathogenic hotspot in NBCIE, lending further support to the clinical diagnosis in this case. The site is considered to be strongly associated with the disorder, 27 and functional studies suggest that missense mutations at amino acid 142 can impair TGM1 activity. 28 Although a different mutation pattern (p.Arg142Leu) has not been previously reported, its location at a known pathogenic site supports its potential clinical relevance and underscores the importance of this region for TGase 1 function. The second mutation, p.Asn400His in exon 8, has not been reported previously in any study. Although the mutations detected in exon 8 are frequently associated with LI and may result in mild or severe phenotypes,16–18 a homozygous mutation in this exon (c.1165C>T [p.Arg389Ser]) has also been reported in an NBCIE case. 12 This highlights that although exon-level clustering provides clues to genotype–phenotype correlations, phenotype determination depends on both the exon involved and the precise mutation site and its functional impact. Establishing definitive mutational hotspots and genotype–phenotype correlations requires large-scale studies. Further research involving larger cohorts and functional analyses is required to confirm its biological consequences and clarify its role in ARCI phenotypes.
Owing to their compromised skin barrier, collodion babies are at an increased risk of complications, including skin infections, dehydration, electrolyte imbalance, mobility limitations, and feeding difficulties, 3 making comprehensive management strategies challenging. In this case, the infant’s skin, encased in a thick, tight membrane, began to crack and peel shortly after birth, increasing the risk of infection. Softening the skin was crucial to prevent injury and subsequent infection. Silicone oil cream, primarily containing dimethicone, was applied for its lubricating and protective properties, forming a barrier that softened the skin and reduced the risk of bacterial invasion. In addition to skin-related complications, collodion babies are prone to ectropion, which exposes the conjunctival mucosa and markedly increases the risk of ocular infection and ulceration. To prevent this, we used sterile vaseline oil gauze to cover the eyes, and sterile saline eye drops were administered four times daily until the skin loosened and the eyelids could close properly. To date, the infant has maintained healthy skin with no signs of infection and has shown normal growth and developmental progress.
The management strategy in this case was comprehensive, focusing on both symptom relief and preventive care, including correction of hypoglycemia, stabilization of blood glucose levels, use of an incubator for warmth, and continuous monitoring of heart and oxygen levels. These approaches aimed to minimize complications and align with previous research by Paller and Butala, 29 who emphasized the importance of enhancing clinical care to improve the quality of life, preserve organ function, and reduce the risk of complications. 4 Beyond the measures used in our case, recent studies have also proposed additional strategies that may further improve patient outcomes. For example, monitoring vitamin D levels and administering oral vitamin D have been shown to help alleviate persistent skin dryness. 30 Pauline Besonhe proposed the systemic use of acitretin or vitamin A (retinoids) from an orthopedic perspective, presenting promising results for future clinical treatments. 31 Although these approaches were not implemented in our case, they may still provide valuable insights and therapeutic possibilities for managing similar cases in the future.
A multigene panel encompassing genes associated with ARCIs is considered the preferred diagnostic approach. 32 With an enhanced understanding of disease-related mutations, prepregnancy genetic testing could enable the prevention of collodion baby phenotype by guiding parental reproductive decisions. Furthermore, identification of specific mutations forms the foundation for the development of gene- and cell-based therapies for ARCI. 33 In the present case, genetic screening of the parents could support informed choices regarding future pregnancies. Specifically, knowing their carrier mutations enables the possibility of embryo screening if they choose in vitro fertilization for their next pregnancy. This proactive strategy highlights the critical role of genetic screening in managing and potentially reducing the prevalence of hereditary conditions.
Footnotes
Acknowledgments
We used Grammarly and ChatGPT to assist with grammar correction and language polishing during manuscript preparation. We sincerely thank the patient and the patient’s family for their cooperation and for providing consent to publish this case.
Author contributions
Study conception and design: LL and LW. Supervision: LL. Project administration: LW. Resources: HX and XL. Investigation: LY and LW. Visualization: HX and XL. Draft manuscript preparation: LY. Review and editing: LL. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
No conflict of interest.
Funding
The authors declare that the study received no funding.
