Abstract
The use of diabetes-specific nutrition formulas reduces postprandial glucose and insulin levels and promotes satiety, thereby improving glycemic control and contributing to weight loss. The effect of diabetes-specific nutrition formulas on the percentage of time in range (70–180 mg/dL) has not yet been investigated. This longitudinal pilot study was based on a case series of overweight or obese patients diagnosed with diabetes using continuous glucose monitoring and managed at the Hospital Universitario San Ignacio in Bogotá (Colombia). We compared continuous glucose monitoring data and anthropometric variables before and after 4 weeks of using a diabetes-specific nutrition formula as replacement for breakfast and afternoon snack. Eleven patients were included in this study; of these, 63.7% were men. The mean patient age was 55 ± 14.5 years, and 81.1% had type 2 diabetes. The mean percentage of time in range increased from 64% ± 24% to 75% ± 16% (mean difference: 10.63; 95% confidence interval: 2.13, 19.14; p = 0.01). The mean percentage of time above range (>180 mg/dL) decreased from 34% ± 25% to 23% ±14% (mean difference: −11.27; 95% confidence interval: −2.48, −20.05; p = 0.02), with no difference in hypoglycemia incidence or anthropometric variables. Our study suggests that the use of a diabetes-specific nutrition formula as replacement for breakfast and afternoon snack improves glycemic control, as measured via continuous glucose monitoring, in overweight or obese patients with type 2 diabetes. This adds to the emerging evidence on the effect of this intervention on continuous glucose monitoring metrics. Further evidence is needed from larger clinical trials to support the inclusion of this intervention in routine clinical practice.
Keywords
Introduction
Diabetes and obesity are major global health challenges affecting millions of people worldwide. The current prevalence of diabetes is 11%, which continues to rise.1,2 One of the main contributors to this trend is the high prevalence of obesity, which is 14% globally and exceeds 20% in some countries.1,2 These two conditions are closely linked; this association was demonstrated in previous studies where 86% of patients with type 2 diabetes (T2D) were also overweight or obese.1,2 This strong connection underscores the importance of preventing and treating obesity—not only to prevent diabetes but also to reduce cardiometabolic outcomes. The American Diabetes Association (ADA) emphasizes the importance of weight reduction in the treatment of T2D. 3 It recommends a weight loss of 5%–10% to improve glycemic control. 3 To achieve this goal, the ADA recommends interventions that include frequent counseling focused on nutritional changes, physical activity, and calorie restrictions. 3 Diabetes-specific nutrition formulas (DSNFs) can play a crucial role in achieving this goal, providing a practical and evidence-based approach to support diabetes management.
Overall, DSNFs attenuate the glycemic response, helping improve long-term glucose control and reduce cardiometabolic risk factors in individuals with diabetes. Although standard formulations vary by brand, DSNFs are generally designed with a macronutrient profile that supports glycemic response. 4 DSNFs have a lower carbohydrate content, which is an intentional reduction aimed at minimizing postprandial glucose spikes. 4 The inclusion of slow-digesting, low-glycemic carbohydrates helps slow down glucose absorption, reduce insulin demand, improve glycemic control, and promote satiety. 4 When carbohydrate intake is reduced, the proportions of fat and protein must increase to maintain the caloric balance. DSNFs often include monounsaturated fatty acids (MUFAs), which have been shown to be associated with improved insulin sensitivity and favorable lipid profiles. 4
Within glycemic control targets, continuous glucose monitoring (CGM) metrics are currently used as an adjunct to glycated hemoglobin (HbA1c) because they provide a more accurate and detailed approach to glycemic control and variability. 5 Among these metrics, a percentage of time in range (%TIR) between 70 and 180 mg/dL has been shown to be directly associated with the incidence of microvascular and macrovascular complications, cardiovascular mortality, and all-cause mortality. 6 However, evidence on the impact of DSNF use on %TIR is still emerging. In addition, there is insufficient evidence regarding the addition of DSNFs to standard medical therapy in patients with diabetes who have not achieved glycemic control. The aim of the present pilot study was to determine the impact of DSNF use on %TIR, other CGM metrics, and anthropometric variables in overweight or obese patients with diabetes by prospectively analyzing a series of patients for 4 weeks.
Methods
Study design and settings
This longitudinal study was performed in a series of overweight or obese patients who were diagnosed with diabetes using CGM and treated at the Endocrinology Unit of the Hospital Universitario San Ignacio in Bogotá, Colombia, between March and April 2023. The Strengthening the Reporting of Observational Studies in Epidemiology-Nutritional Epidemiology (STROBE-nut) statement was used to guide the reporting of the study. 7 The study was approved by the Ethics and Research Committee of the Hospital Universitario San Ignacio (FM-CIE-1171-23); all participants were informed about the studies aims, and written consent was obtained.
Participants
Adult patients were included if they demonstrated proper use of the CGM device and adherence to sensor use at least 70% of the time. Patients were excluded if they had acute decompensation of diabetes or changes in hypoglycemic therapy within the past 3 months, had used dietary supplements in the past month, were pregnant or breastfeeding, were using or planned to use glucocorticoids in the upcoming months, or had significant cognitive impairment.
Variables
The intervention consisted of replacing the patients’ breakfast and afternoon snack each with a dose of a DSNF. Throughout the study, the formula used was Glucerna GL (Abbott Nutrition Inc., Columbus, OH, USA) at a dose of 8 fl oz (237 mL); the nutritional content of this formula is provided in Appendix 1. The main outcomes were based on the CGM data, wherein we compared basal values (measured during the 14 days before the intervention) with those obtained after 4 weeks of intervention. CGM data included a TIR of 70–180 mg/dL, time above range (TAR) >180 mg/dL, time below range (TBR) <70 mg/dL, and coefficient of variation (CV) calculated by dividing the standard deviation of sensor glucose values by the mean of those values. The CGM device used was the FreeStyle Libre (Abbott Diabetes Care, Alameda, CA, USA); data review was performed using the LibreView platform (Abbott Diabetes Care, Alameda, CA, USA). Secondary outcomes included body composition variables measured using electric bioimpedance and the incidence of hypoglycemic events (level 1: glycemia <70 mg/dL and >54 mg/dL, level 2: glycemia <54 mg/dL, and level 3: a state where the patient requires assistance from another person due to changes in their physical or mental status). 8
Data sources: measurements
The initial visit involved the collection of data on demographic and clinical variables. This included anthropometric variables such as weight, height, abdominal circumference, and body mass index (BMI), which was used to classify patients as overweight (25.0–29.9 kg/m2) and obese (≥30 kg/m2). 9 Additionally, a single frequency bioelectrical impedance analysis device (OMRON® HBF-514C, Omron Healthcare, Inc., Illinois, USA) was used to measure whole-body electrical impedance at 50 kHz between the hand and foot. This device was used to measure bioelectrical impedance and provided an estimate of the percentage of body fat, visceral fat, and muscle mass using the manufacturer’s unpublished equation. During this assessment, the CGM data collected over the past 14 days were reviewed. The measurement of anthropometric and body composition variables, in addition to the evaluation of CGM metrics, were repeated at the final visit, which occurred after 4 weeks. Adjustments in drug therapy were made, and diabetes and nutrition education were provided at both visits according to ADA guidelines. 10
Sample size
The sample size was calculated for detecting a 5% change over time in %TIR, from 70% to 75%, assuming repeated measurements with a correlation coefficient of 0.6, an alpha-type error of 5%, a power of 80%, and a common SD of 5%. The final sample size was determined to be 10 patients. A 10% loss to follow-up was assumed, resulting in the enrollment of 11 patients in the study.
Statistical methods
Data were described using means and standard deviations or as medians and interquartile ranges for continuous variables, according to the distribution of the data. A Shapiro–Wilk test was used to assess the normality assumption. Categorical variables were described using absolute numbers and percentages. Analysis was performed by comparing baseline measurements with follow-up measurements (after 4 weeks of intervention) using paired t-tests for normal data and the Wilcoxon test for non-normal data. McNemar's chi-square test was used to compare categorical variables. Subgroup analysis was performed to determine whether the effect differed by the type of diabetes. All tests were two-tailed at a significance level of 0.05. STATA version 16.0 (Release 16. StataCorp LLC, College Station, TX) was used for statistical analysis.
Results
Baseline demographic and clinical characteristics are shown in Table 1. In total, 11 patients were included in the analysis. Of these, 81.1% were diagnosed with T2D and 18.2% with type 1 diabetes (T1D). Furthermore, 36.4% of the patients were obese and 63.3% were overweight. All patients reported full compliance with the recommended doses of the DSNF.
Demographic and clinical characteristics of the patients included in the study.

DPP4: dipeptidyl peptidase-4; SGLT2: sodium–glucose cotransporter 2; GLP1: glucagon-like peptide-1; BMI: body mass index.
Metrics of glycemic control
The mean baseline %TIR (70–180 mg/dL) was 64% ± 24%. The mean post-intervention %TIR was 75% ± 16%, with a statistically significant increase observed after the intervention compared with that at baseline (mean difference: 10.63; 95% confidence interval (CI): 2.13, 19.14; p = 0.01) (Table 2 and Figure 1). Subgroup analysis was performed to determine whether the change in %TIR differed by the type of diabetes; we found that the increase in %TIR after the intervention was significantly higher in patients with T2D (mean difference: 12.66; 95% CI: 2.51, 22.81; p = 0.02); however, this change was not significant in those with T1D (mean difference: 1.5; 95% CI: −20.55, 17.55; p = 0.50). The proportion of patients with %TIR >70% was higher after the intervention than at baseline; however, this difference was not statistically significant (p = 0.53). Similarly, the increase in %TIR after the intervention was significantly higher in obese patients (mean difference: 21.0; 95% CI: 2.48, 39.51; p = 0.03), but not in overweight patients (mean difference: 4.7; 95% CI: −3.9, 13.3; p = 0.22). Change in %TIR was similar for both sexes.
Glycemic control variables before and after the use of DSNF.

Data presented as mean and standard deviation.
DSNF: diabetes-specific nutrition formula; GMI: glucose management indicator; TIR: time in range; TAR: time above range; TBR: time below range; CV: coefficient of variation
p-values were calculated using paired t-tests for comparison with baseline values.
p-values were calculated using McNemar's chi-square test for comparison with baseline values.

Bar graph of glycemic control metrics as per CGM before and after DSNF use. CGM: continuous glucose monitoring; DSNF: diabetes-specific nutrition formula.
With respect to other CGM metrics, the mean %TAR (>180 mg/dL) was 34% ± 25% at baseline and significantly decreased to 23% ± 14% after the intervention (mean difference: −11.27; 95% CI: −2.48, −20.05; p = 0.02), with substantial changes observed in the subgroup of patients with T2D (mean difference: −13.3; 95% CI: −2.8, −23.8, p = 0.02) and obesity (mean difference: −21.3; 95% CI: −1.1, −41.4, p = 0.04). No significant differences were noted in %TBR (<70 mg/dL), CV, or incidence of hypoglycemic events, with an incidence ratio of 1.39 events/patient-day (95% CI: 0.87, 2.25; p = 0.14).
Anthropometric and body composition measures
No significant changes were observed in the following anthropometric measures: weight (p = 0.37), BMI (p = 0.25), abdominal circumference (p = 0.11), and body composition variables measured using electric bioimpedance, including fat percentage (p = 0.23), visceral fat (p = 0.58), and muscle mass (p = 0.38) (Table 3).
Anthropometric variables before and after DSNF use.

Data are presented as mean and standard deviation (±). p-values were calculated using paired t-tests for comparison with baseline values.
Data extracted from the analysis of 11 patients. bData extracted from the analysis of 5 patients
BMI: body mass index; DSNF: diabetes-specific nutrition formula.
Discussion
The present study evaluated the change in %TIR (70–180 mg/dL) 4 weeks after the introduction of a DSNF as replacement for breakfast and afternoon snack in overweight or obese patients with diabetes who were treated with standard medical therapy. The study found a clinically and statistically significant increase in %TIR (70–180 mg/dL) and a significant decrease in %TAR (>180 mg/dL). In this study, the %TIR (70–180 mg/dL) increased by an average of 10.6 percentage points post-intervention. According to the consensus on CGM, an increase in %TIR of >5% is associated with better glycemic control and a reduction in complications, 11 making this a clinically significant change.
In 2020, the glycemic response of participants with T2D was evaluated using CGM. The results showed better postprandial glycemic control and a significant decrease in nocturnal glycemic variability in the group that replaced their breakfast and afternoon snack with the DSNF. 12 Although no data on %TIR were reported, these findings could explain the improvement in metabolic control observed in the current study. In contrast, a randomized controlled trial assessing the effect of lifestyle interventions alone and in combination with DSNF administration for 4 weeks (as replacement for breakfast snack) showed a significant reduction in the CV and an increase in %TIR of 6.1 percentage points with the use of DSNF. 12 However, in the present study, the increase in %TIR was greater, possibly due to the replacement of two meals, including the breakfast and afternoon snack.
A 5% rise in %TIR has been shown to reduce HbA1c levels by up to 0.8%,13,14 which is consistent with previous findings linking DSNF to improved HbA1c and mean glycemia and reduced glycemic variability and postprandial glucose.15–20 These findings are supported by a systematic review and meta-analysis that compared the efficacy of DSNF with that of standard formulations in terms of glycemic control in patients with T2D. The analysis revealed a significant reduction in fasting glycemia and HbA1c levels (−0.67%) associated with DSNF use. 21 Another systematic review and meta-analysis included 18 randomized controlled trials comparing the effect of MUFA-rich DSNFs with lifestyle interventions on glycemic control and reported a reduction in postprandial peak glucose and insulin levels, mean glycemia, and HbA1c (−0.63%) with the use of DSNFs. 22 These findings are consistent with those of our study, showing that the increase in %TIR was associated with a substantial reduction in hyperglycemia exposure. On an average, there was an 11.2 percentage-point decrease in %TAR >180 mg/dL following the intervention.
The long-term effects of DSNF use, as demonstrated in studies such as DIRECT23–25 and RESET, 26 focus on diabetes remission. These studies incorporate nutritional formulas as part of therapeutic lifestyle changes involving low-calorie diets. However, it remains unclear whether using DSNFs for partial meal replacement, as demonstrated in this study, impacts metabolic control and cardiometabolic risk factors in obese patients undergoing standardized pharmacological treatment. This study found that the increase in %TIR after the intervention was significantly higher in obese patients, suggesting major benefits for this group. Although we found no significant changes in anthropometric or body composition variables after the intervention, previous studies have reported significant weight loss in overweight or obese patients with T2D using DSNFs.15,16,27 The assessment of these variables in our study could have been limited by the length of follow-up (4 weeks) because previous studies with longer follow-up have reported significant improvements in weight and body composition variables. For example, a recent randomized controlled trial compared the use of DSNF (as meal replacement) in combination with standard care (DSNF group) vs. standard care only (control group) over 3 months. Compared with the control group, they found two-fold greater weight loss and an almost two-fold reduction in percent body fat and increase in fat-free mass in the DSNF group; the DSNF group also exhibited a percentage reduction in visceral adipose tissue that was several times greater than that in the control group. 28
In the subgroup analysis, the increase in %TIR was significant in patients with T2D but not in patients with T1D. This is consistent with existing evidence on the efficacy of DSNF in glycemic control, which was almost exclusively observed in patients with T2D. 17 The greater net insulin deficit experienced by patients with T1D could be the primary factor influencing the observed differences in the effects of DSNF on postprandial glycemic and insulin responses. However, no statistically significant differences were found for %TAR <70 mg/dL and the incidence of hypoglycemia, suggesting the safety of DSNF use in terms of hypoglycemia risk.
Limitations
This study has certain limitations that need to be acknowledged. First, as a pilot case series study, it had a limited sample size, which allowed us to detect clinically and statistically significant differences in %TIR and %TAR but did not allow us to control for all possible covariates such as insulin-regimen and antidiabetic medications that could have influenced the results or made the results generalizable to similar populations. Second, this study lacked a control group, which did not allow the exclusion of the effect of other confounding factors, such as different pharmacological management and lifestyle recommendations that patients received according to the ADA recommendations. Third, the study population was heterogeneous; the study included T1D patients who have greater net insulin deficit and whose glycemic control depends on insulin intake before and after the intervention meal; this introduces variations in the effect of DSNF on the postprandial glycemic and insulin response and does not allow conclusions to be drawn in this specific subgroup. Additionally, HbA1c, fasting glucose/insulin, and lipid profile measurements were not performed before or after DSNF; therefore, the potential impact on other metabolic outcomes could not be measured. Finally, the relatively short follow-up period of 4 weeks limits the detection of changes in some variables such as anthropometric parameters. Controlled clinical trials with larger sample sizes and longer follow-up period are needed to confirm the preliminary results of our pilot study. Our research group is currently working on a randomized controlled trial to consolidate this evidence.
Conclusions
Our results suggest that the use of DSNF as replacement for breakfast and afternoon snack in overweight or obese patients with T2D can clinically and significantly increase %TIR levels (70–180 mg/dL) at the expense of decreasing %TAR levels (>180 mg/dL) without increasing the risk of hypoglycemia. These findings add to the available evidence suggesting that DSNF improves glycemic control in this patient population. However, larger clinical trials are needed to consolidate this evidence.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251382375 - Supplemental material for Impact of diabetes-specific nutrition formulas on time in range in overweight or obese Colombian patients with diabetes: A case series study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251382375 for Impact of diabetes-specific nutrition formulas on time in range in overweight or obese Colombian patients with diabetes: A case series study by Ana-M Gomez, Maria-P Chicue, Diana-C Henao and Oscar-M Muñoz-Velandia in Journal of International Medical Research
Footnotes
Acknowledgment
We would like to acknowledge Angélica Veloza, Adriana Gómez, and Marisol Vergara who participated in patient enrollment and data collection.
Author contributions
AMG, MPC, and OMM conducted data analysis and drafted the manuscript. DCH was involved in patient enrollment, data collection, and manuscript draft review.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of conflicting interests
AMG reports speaker fees from Novo Nordisk, Elli Lilly, Boehringer Ingelheim, Abbott, and Medtronic. DCH reports speaker fees from Novo Nordisk, Medtronic, Servier, and Abbott. Other authors have no conflicts of interest to report.
Ethics approval
Ethical approval for reporting this case series was obtained from the Ethics and Research Committee of the Hospital Universitario San Ignacio (FM-CIE-1171-23).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Prior presentation of abstract statement
No.
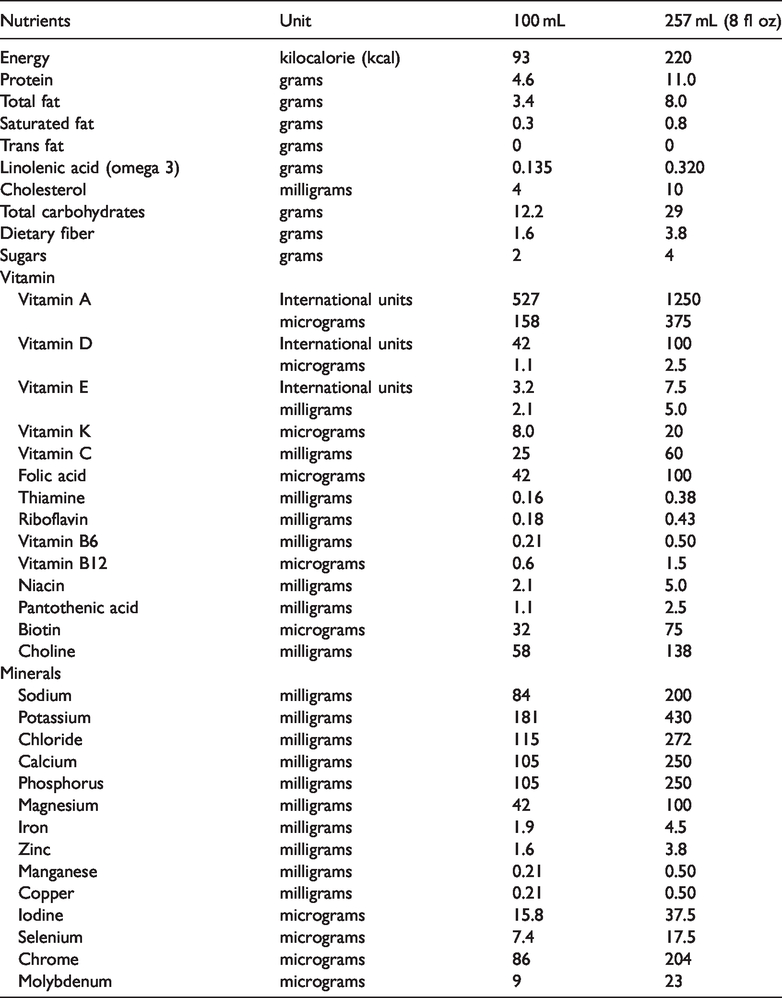
Appendix 1. Glucerna GL (Abbott Nutrition Inc.,Columbus,OH,USA) administered at a dose of 8 fl oz (237 mL),nutritional content.
| Nutrients | Unit | 100 mL | 257 mL (8 fl oz) |
|---|---|---|---|
| Energy | kilocalorie (kcal) | 93 | 220 |
| Protein | grams | 4.6 | 11.0 |
| Total fat | grams | 3.4 | 8.0 |
| Saturated fat | grams | 0.3 | 0.8 |
| Trans fat | grams | 0 | 0 |
| Linolenic acid (omega 3) | grams | 0.135 | 0.320 |
| Cholesterol | milligrams | 4 | 10 |
| Total carbohydrates | grams | 12.2 | 29 |
| Dietary fiber | grams | 1.6 | 3.8 |
| Sugars | grams | 2 | 4 |
| Vitamin | |||
| Vitamin A | International units | 527 | 1250 |
| micrograms | 158 | 375 | |
| Vitamin D | International units | 42 | 100 |
| micrograms | 1.1 | 2.5 | |
| Vitamin E | International units | 3.2 | 7.5 |
| milligrams | 2.1 | 5.0 | |
| Vitamin K | micrograms | 8.0 | 20 |
| Vitamin C | milligrams | 25 | 60 |
| Folic acid | micrograms | 42 | 100 |
| Thiamine | milligrams | 0.16 | 0.38 |
| Riboflavin | milligrams | 0.18 | 0.43 |
| Vitamin B6 | milligrams | 0.21 | 0.50 |
| Vitamin B12 | micrograms | 0.6 | 1.5 |
| Niacin | milligrams | 2.1 | 5.0 |
| Pantothenic acid | milligrams | 1.1 | 2.5 |
| Biotin | micrograms | 32 | 75 |
| Choline | milligrams | 58 | 138 |
| Minerals | |||
| Sodium | milligrams | 84 | 200 |
| Potassium | milligrams | 181 | 430 |
| Chloride | milligrams | 115 | 272 |
| Calcium | milligrams | 105 | 250 |
| Phosphorus | milligrams | 105 | 250 |
| Magnesium | milligrams | 42 | 100 |
| Iron | milligrams | 1.9 | 4.5 |
| Zinc | milligrams | 1.6 | 3.8 |
| Manganese | milligrams | 0.21 | 0.50 |
| Copper | milligrams | 0.21 | 0.50 |
| Iodine | micrograms | 15.8 | 37.5 |
| Selenium | micrograms | 7.4 | 17.5 |
| Chrome | micrograms | 86 | 204 |
| Molybdenum | micrograms | 9 | 23 |
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
