Abstract
Objective
This study aimed to investigate the expression of the inflammasome-related molecules NOD-like receptor protein 3 and interleukin-18 in basal cell carcinoma tissues and to evaluate their potential as diagnostic biomarkers.
Methods
In this observational case–control study, tumor tissues and adjacent noncancerous tissue samples from 25 patients with basal cell carcinoma were analyzed. We quantified NOD-like receptor protein 3 and interleukin-18 expression at the mRNA and protein levels using quantitative real-time polymerase chain reaction and western blotting, respectively. The diagnostic performance of these molecules was assessed using receiver operating characteristic curve analysis.
Results
Our results revealed that both NOD-like receptor protein 3 and interleukin-18 were significantly upregulated in basal cell carcinoma tissues compared with adjacent noncancerous tissue samples at the mRNA and protein levels (p < 0.05). Receiver operating characteristic curve analysis demonstrated strong diagnostic performance, with an area under the curve of 0.931 for NOD-like receptor protein 3 (95% confidence interval: 0.845–1.00) and 0.860 for interleukin-18 (95% confidence interval: 0.739–0.980).
Conclusions
The marked upregulation of NOD-like receptor protein 3 and interleukin-18 suggests their involvement in the inflammatory tumor microenvironment and positions them as promising candidate biomarkers for basal cell carcinoma detection. Although these findings may suggest their relevance as future therapeutic targets, this role requires substantial validation through functional assays and in larger, more diverse study populations.
Keywords
Introduction
Basal cell carcinoma (BCC) is the most common type of skin cancer, representing approximately 80% of nonmelanoma skin cancers globally. Although BCC seldom metastasizes, its capacity for local invasion can result in considerable morbidity, especially in sun-exposed regions of the skin. Despite the growing volume of literature on BCC worldwide, there remains a significant deficiency in our comprehension of its underlying mechanisms, which hinders the advancement of more effective diagnostic and therapeutic strategies. 1
According to recent research, chronic inflammation plays a crucial role in forming the tumor microenvironment, impacting not only the development and spread of tumors but also their capacity to elude immune surveillance.2–5 Inflammasomes, especially the NOD-like receptor protein 3 (NLRP3) inflammasome, are crucial in regulating inflammatory responses by activating caspase-1, which results in the maturation and production of proinflammatory cytokines, including interleukin-1β and interleukin-18 (IL18).6–9 Numerous studies have indicated that deregulation of the NLRP3 inflammasome correlates with the onset of various malignancies, including colorectal, gastric, and breast cancers.10,11
IL18, triggered downstream of NLRP3, has multifaceted roles in cancer—occasionally facilitating antitumor immune responses, while at other times, exacerbating inflammation that promotes tumor proliferation. 7 These molecules have consequently emerged as intriguing candidates for biomarker identification in inflammation-driven malignancies.
Precise diagnosis and prognosis in dermatopathology depend significantly on the identification and validation of reliable molecular biomarkers. The expression profiles of NLRP3 and IL18 in BCC tissues may provide significant insights into the inflammatory pathways associated with BCC and act as possible diagnostic indicators.
This study evaluated the expression of NLRP3 and IL18 at the mRNA and protein levels in BCC tissues and adjacent nonmalignant tissues. Our objective is to elucidate the functions of these inflammasome components in the pathophysiology of BCC and to investigate their potential as diagnostic or therapeutic targets.
Methods
Twenty-five fresh-frozen tissue samples with BCC and 25 adjacent noncancerous tissue samples were procured from the Iranian Tumor Bank at Tehran University of Medical Sciences, Iran. This case-control study was approved by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.MED.REC.1401.302; approval date: 29 January 2023). All procedures were conducted in accordance with the principles of the Declaration of Helsinki (1975) and its latest amendment (2024). The samples were procured from patients with BCC according to inclusion criteria (patients with BCC) and exclusion criteria (patients who underwent chemotherapy, radiation, or antiinflammatory medications). Informed consent in writing was acquired from all tissue donors.
The diagnosis of BCC in malignant tissue samples (case group) was confirmed by a specialist pathologist. Additionally, a total of 25 adjacent noncancerous tissue samples were utilized as the control group, which was corroborated by pathological investigation and classified as noncancerous tissues. All tissue samples were maintained as fresh-frozen specimens until subsequent examination. In this investigation, we employed distinct numbering systems for the case and control groups; therefore, the investigators performing quantitative real-time polymerase chain reaction (qRT-PCR) and western blotting were not blinded to group allocation. All case and control samples were analyzed in a single qRT-PCR run for each gene, thereby minimizing the potential for bias.
RNA extraction, cDNA synthesis, and qRT-PCR
Fresh-frozen tissue samples were pulverized using liquid nitrogen. Subsequently, total RNA was extracted using the RNX Plus reagent (Sinaclone, Iran) in accordance with the manufacturer's guidelines. The subsequent stage involved assessing the purity and integrity of total RNA using a Nanodrop spectrophotometer (Thermo Fisher Scientific) and 1% Tris-acetate ethylenediaminetetraacetic acid-agarose gel electrophoresis. Complementary DNA (cDNA) was produced from 2 µg of total RNA utilizing the Pars Tous cDNA synthesis Kit (Iran). The qRT-PCR was performed utilizing the Amplicon SYBR Green PCR Kit (Denmark) on a Roche Real-Time Cycler (Germany). The qRT-PCR experiment for each sample was performed in duplicate. The primer sequences employed in the current study are presented in Table 1. Gene expression levels were standardized to β-actin, and relative quantification was executed utilizing the 2^–ΔΔCt technique.
Primers and related sequences used in present study.

Western blotting for NLRP3 and IL18
For western blotting assay, in the first step, total protein from fresh-frozen tissue samples were extracted. The tissue samples were homogenized in liquid nitrogen, and then tissue homogenates were incubated with 700 µL of radioimmunoprecipitation assay buffer containing protease inhibitor cocktail (Kiazist, Iran) for 15 minutes at 4°C. Subsequently, the tubes were centrifuged at 5000 rpm for 40 minutes at 4°C. Finally, the supernatant was collected and stored at −70°C. The total protein level was measured using BCA Kit (Kiazist, Iran).
For the western blotting assay, 40 µg of total protein from each sample were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis, following which the proteins were transferred to polyvinylidene fluoride (PVDF) membranes. Thereafter, the membranes were incubated with 5% nonfat skim milk powder (Sigma, Burlington, Massachusetts, USA) for 2 hours. Subsequent to the blocking procedure, the PVDF membranes were rinsed twice with tris-buffered saline with tween-20 (TBST) buffer and once with TBS buffer, each for a duration of 10 minutes. After the washing phase, the membranes were incubated overnight with the following primary antibodies: anti-NLRP3 (ab263899, Abcam; 1:1000), anti-IL18 (ab243091, Abcam; 1:1000), and anti-β-actin (ab8227, Abcam; 1:2500). The membranes were washed twice with TBST and once with TBS buffer, followed by a 2-hour incubation with horseradish peroxidase–conjugated goat antirabbit secondary antibody (ab6721, Abcam; 1:10,000). Ultimately, following three separate washes with TBST and TBS, the protein bands were detected with BioRad ECL choose western blotting detection reagent (BioRad, Hercules, California, USA). β-actin was utilized as an internal control, and Image J software was employed for densitometric analysis. The western blotting experiment was performed only once for each sample.
Statistical analysis
Data analysis was done using IBM Corp.'s SPSS version 16. Statistical significance was established at p < 0.05, and data were presented as mean ± SEM. The Kolmogorov-Smirnov test was employed to assess the normality of the data distribution. The independent t-test was utilized for data exhibiting normal distribution, whereas the Mann-Whitney U test was applied for data demonstrating non-normal distribution. Fisher's exact test was used to assess the association between IL18 and NLRP3 expression levels and the clinicopathological characteristics of the patients due to small sample sizes and low expected frequencies in contingency tables. Receiver operating characteristic (ROC) analysis was conducted using Prism 5.00 (GraphPad Software, La Jolla, California, USA) to evaluate NLRP3 and IL18 as possible biomarkers.
Results
Demographics and clinicopathological data
The demographic and clinical characteristics of the participants are delineated in Tables 2 and 3. The study cohort comprised 11 women and 14 men, the majority of whom abstained from smoking and alcohol consumption. Histopathological evaluation revealed that 22 tumors were categorized as BCC-not otherwise specified (BCC-NOS), 2 as infiltrative BCC, and 1 as mixed histology. According to the tumor, node, metastasis (TNM) staging, four patients were classified as stage 0, five as stage I, eight as stage II, and eight as stage III. Bone invasion was identified in two instances and perineural invasion in seven instances.
Demographic characteristics of participants.

Clinicopathological information of participants.
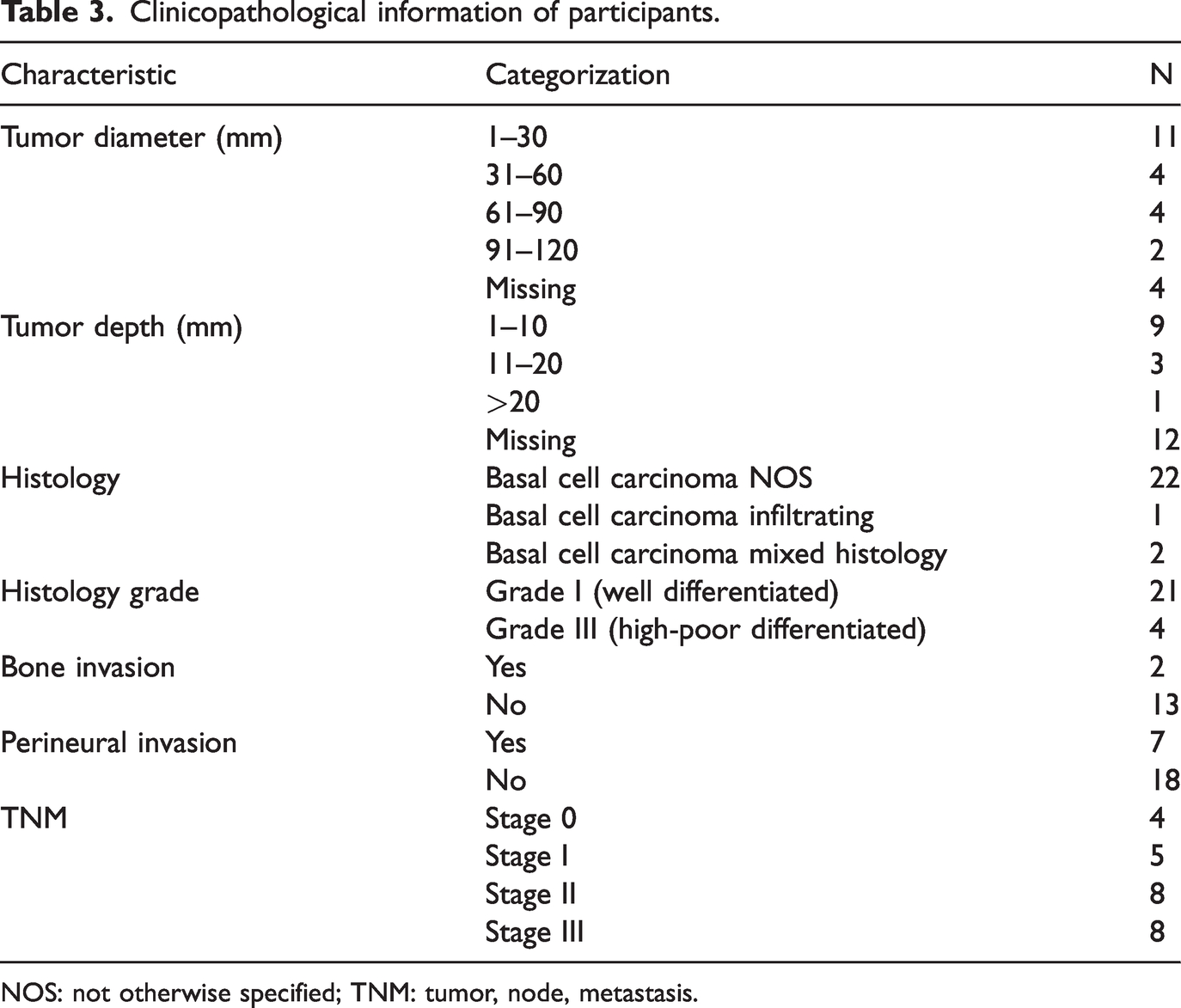
NOS: not otherwise specified; TNM: tumor, node, metastasis.
NLRP3 and IL18 gene expression
The qRT-PCR study demonstrated that NLRP3 mRNA levels were significantly elevated in BCC tumor tissues relative to adjacent noncancerous tissues (p < 0.0001, Figure 1(a)). Correspondingly, IL18 mRNA expression was markedly elevated in tumor samples (p < 0.0001), exhibiting a 6.2-fold increase relative to controls (Figure 1(b)).

The mRNA expression of NLRP3 (a) and IL18 (b) genes in tumor tissue samples and adjacent noncancerous tissue samples of patients with BCC. The data are presented as mean ± SEM. p value < 0.05 was considered as significant. BCC: basal cell carcinoma; IL18: interleukin-18; NLRP3: NOD-like receptor protein 3.
NLRP3 and IL18 protein expression
Western blot examination indicated that NLRP3 protein levels were markedly increased in tumor tissues relative to adjacent noncancerous tissues (p = 0.017; Figure 2(b)). The expression of IL18 protein was markedly elevated in tumor samples (p = 0.0006; Figure 2(c)). Protein band intensity was quantified via densitometry and normalized to β-actin as the loading control.

The protein expression of NLRP3 and in tumor tissue samples and adjacent noncancerous tissue samples of patients with BCC. The band of NLRP3, IL18, and β-actin (a), the band intensity of NLRP3 (b) and IL18 (c) in tumor tissue samples and adjacent noncancerous tissue samples of patients with BCC. The data are presented as mean ± SEM. p value <0.05 was considered as significant. BCC: basal cell carcinoma; IL18: interleukin-18; NLRP3: NOD-like receptor protein 3.
Association of NLRP3 and IL18 genes expression with clinicopathological characteristics
Table 4 illustrates the correlation between NLRP3 gene expression and the clinicopathological characteristics of patients. Table 4 demonstrates that histology grade (p > 0.999), bone invasion (p = 0.528), and perineural invasion (p > 0.999) revealed no significant correlation with NLRP3 gene expression. Table 5 demonstrates that histology grade (p = 0.548), bone invasion (p = 0.528), and perineural invasion (p > 0.999) exhibited no significant correlation with IL18 gene expression.
The association between NLRP3 with clinicalpathological characteristics in patients with basal cell carcinoma.

Because of small sample numbers, Fisher's test p values were considered.
The association between IL18 with clinicalpathological characteristics in patients with basal cell carcinoma.

Because of small sample numbers, Fisher's test p values were considered.
ROC curve analysis
We conducted ROC curve analysis to evaluate the diagnostic significance of NLRP3 and IL18 expression. The area under the curve (AUC) for NLRP3 was 0.931 (95% confidence interval (CI): 0.845–1.00), demonstrating a sensitivity of 95% and a specificity of 85% (p < 0.0001; Figure 3(a)). The AUC for IL18 was 0.860 (95% CI: 0.739–0.980), indicating a sensitivity of 70% and a specificity of 95% (p = 0.0031: Figure 3(b)).

The ROC curve analysis for NLRP3 (a) and IL18 (b) genes expression as potential tumor markers. ROC: receiver operating characteristic; IL18: interleukin-18; NLRP3: NOD-like receptor protein 3.
Discussion
The notable increase of NLRP3 and IL18 gene expression in BCC tissues underscores the possible significance of inflammatory signaling in the pathogenesis of this prevalent skin cancer. NLRP3, a pivotal modulator of innate immunity, facilitates the maturation of proinflammatory cytokines by caspase-1 activation, resulting in the secretion of IL1β and IL18, all of which have been demonstrated to affect tumor-associated inflammation.6,11 The increase of IL18, a cytokine that activates Th1 and Th2 immune responses, signifies a shift in the immunological context of the tumor microenvironment. 9 This inflammatory milieu may enhance tumor cell proliferation, allow immune evasion, and encourage remodeling of the extracellular matrix. Although inflammation is commonly acknowledged as a characteristic of malignancy, our findings contribute to the expanding evidence that the dysregulated activation of inflammasome pathways, especially those associated with NLRP3 and IL18, may significantly influence the immunopathology of BCC. These molecules may engage in a self-sustaining cycle, wherein stress signals from the tumor or its surroundings activate the inflammasome, thus extending chronic inflammation that facilitates carcinogenesis and local tumor proliferation.
Our observation of significant upregulation of NLRP3 and IL18 genes in BCC tissues aligns with previous research emphasizing the function of inflammasome components in different malignancies. Our findings align with those of Ahmad et al., 12 who identified elevated NLRP3 expression in human BCC specimens and suggested a possible involvement of inflammasome components in keratinocyte carcinogenesis. Comparable trends have been documented in other cutaneous malignancies; for instance, sustained activation of NLRP3 in melanoma cells has been demonstrated to facilitate tumor proliferation. Okamoto et al. 13 demonstrated, for the first time, that melanoma cancer cells, particularly in advanced stages, autonomously and continuously secrete IL1β without external stimuli, attributing this secretion to the persistent activation of the NLRP3 inflammasome and the IL1 receptor pathway. In advanced stages, melanoma adopts an autoinflammatory state and utilizes the inflammasome to sustain this condition.6,13 Moreover, pan-cancer investigations have revealed substantial upregulation of NLRP3-related genes in many cutaneous malignancies, including cutaneous melanoma. 11 These data collectively suggest that dysregulated NLRP3 activation may be a common mechanism underlying the formation of skin cancers. Nonetheless, it is crucial to acknowledge that the function of NLRP3 in cancer seems to be context-dependent. Research conducted by Allen et al. 14 and Wei et al. 15 has demonstrated a tumor-suppressive role of NLRP3 in colitis-associated and hepatic malignancies, respectively, highlighting the intricate and dualistic nature of inflammasome signaling in tumor biology.
Our findings concerning IL18 correspond to prior research that has documented its overexpression in squamous cell carcinoma and melanoma, where IL18 is believed to facilitate the formation of a proinflammatory tumor microenvironment. 9 Simultaneously, evidence supports the antitumor function of IL18, notably via the activation of natural killer cells and the augmentation of adaptive immune responses,16,17 highlighting the complex and context-dependent nature of its function in cancer.
Our data illustrate the concurrent upregulation of NLRP3 and IL18 in BCC tissues, contributing to the literature indicating that these molecules may synergistically influence the inflammatory environment of malignancies. These discoveries not only corroborate findings from associated skin malignancies but also offer novel insights into the specific roles of NLRP3 and IL18 in BCC.
Our analysis presents various novel contributions in comparison to prior investigations. Initially, we concurrently evaluated the mRNA and protein expression levels of NLRP3 and IL18 in matched BCC and adjacent noncancerous tissues, offering a more thorough understanding of their role in tumor biology. Secondly, to our knowledge, this is the first publication investigating IL18 expression in human BCC tissues. Third, we employed ROC curve analysis to assess the diagnostic efficacy of these markers, introducing a quantitative aspect that has not been previously investigated in this context. These features establish our findings as a significant preliminary step in comprehending the diagnostic and possibly therapeutic significance of inflammasome-related chemicals in BCC.
To investigate the clinical significance of our findings, we analyzed the correlation between the expression levels of NLRP3 and IL18 and clinicopathological features, including histological grade, bone invasion, and perineural invasion. Our findings indicated no significant association between gene expression levels and the specified parameters. This may result from the study's small sample size, and we advise that future research reassess these correlations in bigger cohorts.
Although this study provides insightful information, a number of limitations should be noted. The limited sample size (n = 25) constrains the generalizability of our results and may diminish statistical power, especially in identifying nuanced changes in expression or clinical associations. Conversely, most of the BCCs were categorized as NOS, and their subtypes remained ambiguous, despite the confirmation of BCC through pathological evaluation.
Despite the statistically significant changes in NLRP3 and IL18 gene expression between tumor and adjacent normal tissues, bigger multicenter investigations are required to validate these findings and enhance their therapeutic significance. Moreover, functional tests, including gene knockdown and caspase-1 inhibition, were excluded from this study. Consequently, a conclusive causal relationship between inflammasome activation and tumor growth cannot be demonstrated. Prior research has demonstrated that NLRP3 activation can affect tumor behavior by regulating immune responses and cytokine signaling,6,11 but these effects require direct experimental validation specifically within BCC. The lack of longitudinal clinical data, including tumor recurrence or treatment response, limits our capacity to assess the prognostic or predictive significance of these indicators. Subsequent research integrating functional assays and clinical follow-up data will be essential to ascertain if NLRP3 and IL18 just signify inflammatory processes or play an active role in carcinogenesis and therapeutic resistance in BCC.
In conclusion, this study demonstrates that NLRP3 and IL18 (at the mRNA and protein levels) are significantly upregulated in BCC tissues when compared with adjacent noncancerous tissue samples. The strong diagnostic performance observed in our ROC analysis highlights the potential of these inflammasome-related molecules as promising biomarkers for BCC detection. From a translational perspective, these findings provide a solid foundation for further research into their roles as both diagnostic tools and potential therapeutic targets.
It is suggested that future research evaluates these expression patterns in larger and more diverse study populations, ideally across multiple clinical centers. Such studies could also include functional assays to causally link NLRP3 and IL18 activity to BCC pathogenesis. Furthermore, determining the prognostic and predictive significance of these markers requires longitudinal data that correlate their expression levels with key clinical outcomes, including tumor recurrence and response to therapy. Ultimately, this line of research will be essential to determine whether modulation of these inflammatory pathways could provide a novel therapeutic avenue for the management of BCC.
Footnotes
Acknowledgments
The present article was extracted from MSc dissertation of the first author at Kermanshah University of Medical Sciences that the authors would like to appreciate this center and Deputy for Research and Technology, Kermanshah University of Medical Sciences.
Author contributions
Zahra Mehrabi: Conceptualization, Methodology, Investigation, Writing—Original Draft, Writing—Review & Editing; Zohreh Rahimi: Data Curation, Supervision, Writing—Review & Editing; Parisa Mohammadi: Methodology, Writing—Original Draft, Writing—Review & Editing; Nazanin Jalilian: Investigation, Data Curation; Lida Haghnazari: Formal Analysis, Visualization, Writing—Review & Editing; Farhad Salari: Investigation, Validation; Soheila Asadi: Supervision, Project Administration, Writing—Review & Editing.
Consent to participate
Written informed consent was obtained from all participants.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Ethical considerations
This study was approved by the Research Ethics Committee of Kermanshah University of Medical Sciences (ethics approval number: IR.KUMS.MED.REC.1401.302).
Funding
This research was supported by Deputy for Research and Technology, Kermanshah University of Medical Sciences, 4020217 (29 January 2023).
