Abstract
Objective
The objective of this study was to examine the applicability of shear wave elastography as a quantitative approach for measuring tissue stiffness in the differentiation of intra-abdominal abscesses from hematomas, which share similar radiological findings.
Methods
This prospective single-center study included patients with intra-abdominal abscesses or hematomas who underwent shear wave elastography prior to ultrasound-guided drainage in an interventional radiology clinic between 2021 and 2022. An independent samples t-test was used to compare the mean shear wave velocity between the abscess and hematoma groups.
Results
A total of 50 patients (33 with abscesses and 17 with hematomas) were included in this study. The elastography values of the patients were expressed as mean ± standard deviation (minimum–maximum). The mean shear wave velocity of hematomas (1.11 ± 0.41 (0.54–1.98) m/s) was significantly higher than that of abscesses (0.68 ± 0.16 (0.36–1.07) m/s) (p < 0.001). In the receiver operating characteristic analysis, the area under the curve was 84.8%. When the optimal cutoff value was defined as 0.72, the sensitivity and specificity were 82.4% and 67%, respectively.
Conclusion
Shear wave velocity can aid in differentiating intra-abdominal abscesses from hematomas using a simple and widely available method.
Introduction
Intra-abdominal fluid collection is a frequent complication of surgical procedures or trauma, which may present with various morphological and radiological features associated with its components. 1 Depending on their content, fluid collections are classified as seroma, abscess, lymphocele, hematoma, or biloma.2–5 Most intra-abdominal abscesses occur after a surgical procedure, and the abovementioned types of fluid collections can evolve into an abscess if they become infected. Although intra-abdominal abscesses most often occur secondary to gastric and intestinal surgery, other surgical interventions, including partial hepatectomy, pancreatectomy, and pelvic surgical procedures, can also lead to abscess formation. Hematomas, caused by intra-abdominal bleeding from lesions such as tubo-ovarian abscess or hemorrhagic corpus luteum cysts, are also common causes of pelvic fluid collection. 6
Ultrasonography is the first-line imaging modality for evaluating abdominal fluid collections. Hematomas may have different sonographic appearances, depending on the age of blood products. 7 Hematomas are anechoic in the acute phase but may become echogenic after organization and clot formation. During clot lysis, they appear heterogeneously hypoechogenic. After lysis is complete, they may appear anechoic again. Intra-abdominal abscesses usually present with irregular thick walls, heterogeneous content, and septations. They may occasionally contain gas, a highly specific finding, seen as echogenic foci with reverberation artifacts. 7 The presence of gas in an intra-abdominal fluid collection is suggestive of infection, although it is observed in only a small percentage of patients. 8 Differentiating hematomas from abscesses is challenging, particularly during the organization phase, which requires distinct diagnostic approaches.
Ultrasound elastography is a widely used method that has been increasingly adopted across various clinical fields. Elastography measures tissue stiffness through a dynamic examination in addition to conventional B-mode ultrasonography. Acoustic radiation force impulse (ARFI) elastography is a radiation force–based imaging technique combined with ultrasound for the quantitative measurement of tissue elasticity. Acoustic pulses are generated in real time by a B-mode ultrasound probe, leading to localized tissue displacement, and the resulting shear wave velocity (SWV) is displayed as an ARFI image. This technique has proven to be useful in differentiating tissue characteristics across various body parts.9–13
Although the differential diagnosis of intra-abdominal abscess and hematoma can usually be made based on clinical laboratory findings and imaging findings of computed tomography (CT) and magnetic resonance imaging (MRI), CT and MRI may not always be available. The determination of contrast enhancement in the walls after the administration of an intravenous contrast agent in CT and MRI can support the diagnosis of abscess, although contrast administration may be contraindicated in patients with allergies or a history of renal impairment, limiting the diagnostic performance of cross-sectional imaging methods. 14 However, ARFI elastography is a safer, noninvasive bedside method that involves no ionizing radiation and does not require contrast material administration.
As exudative fluids show higher elastography velocity values because of the more prominent proteinaceous and blood contents than transudative fluids in the pleural space, 12 we assumed that intra-abdominal hematomas would also have higher velocity values than abscesses. This study aimed to evaluate the efficacy of ARFI elastography in differentiating intra-abdominal abscesses from hematomas.
Material and methods
Patients
Patients who were referred to the tertiary university hospital interventional radiology department with a preliminary diagnosis of intra-abdominal abscess for imaging-guided aspiration between 2021 and 2022 were consecutively included in this study. Patients lacking laboratory results or elastography measurements of the lesion were excluded. In addition, patients with a history of antibiotic or immunosuppressive use within 2 weeks prior to aspiration were excluded. Patients with lesions located deeper than 8 cm from the skin surface were excluded due to technical failure of elastography measurement at this depth. Patients whose lesions lacked microbiological (presence of leukocytes/erythrocytes in the lesion or bacterial growth in culture) or macroscopic (pus formation in the lesion) evidence, which is compatible with seroma or lymphocele, and those who had a history of renal–hepatic diseases associated with ascites were not evaluated. Finally, the study included a total of 50 consecutive patients who met all the inclusion criteria.
This prospective cross-sectional study was approved by the Yüzüncü Yıl University Ethics Committee (approval number: 2021/10-10), and written informed consent was obtained from all patients. All patient details have been de-identified. This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. The reporting of this study conforms to the Standards for Reporting of Diagnostic Accuracy (STARD) guidelines. 15
Imaging
Sonographic examinations were performed by a radiologist, who had 15 years of ultrasonography and 7 years of elastosonography experience, using an Affiniti 70 G (Philips Healthcare, Bothell, WA, USA) ultrasound device with a C5-1 convex probe. Before drainage, the patients were evaluated in the supine position to identify the location and sonographic features of the fluid via B-mode ultrasonography. For each lesion, gain and focal depth adjustments were made for optimal visualization and measurements. Then, SWV measurements were performed using the ElastPQ option in ARFI elastography application via the same ultrasound probe. Before the elastography examination, all patients were instructed to hold their breath and remain still during the procedure. The probe was applied with gel and placed in slight contact with the skin, and the lesion site was identified via B-mode ultrasound. A region of interest box (10 × 5 mm) was placed in the most central and homogeneous area of the lesion, and at least four measurements were obtained for each lesion (Figures 1 and 2). Each ElastPQ value was expressed in meters per second (m/s) with the standard deviation (SD). The mean ElastPQ value for each lesion was calculated and recorded. Finally, ultrasonography-guided percutaneous drainage was performed. After performing microscopic examination of the fluid samples to determine the presence of leukocytes or erythrocytes, the lesions were classified as abscesses or hematomas.

Microscopic examination of a pelvic fluid collection in a 35-year-old woman who underwent a cesarean section showed multiple erythrocytes, which is compatible with a hematoma.

Microscopic examination of a pelvic fluid collection in a 33-year-old man who underwent appendectomy showed multiple neutrophils, which is compatible with an abscess.
Statistical analysis
The descriptive statistics for continuous variables were expressed as mean ± SD and minimum–maximum, while the categorical variables were presented as numbers and percentages. A Kolmogorov–Smirnov test was used to examine the distribution of variables. An independent samples t-test (unpaired) was used for the comparison of the mean values between the abscess and hematoma groups. The cutoff value for elastography data in the groups was determined using a receiver operating characteristic (ROC) curve analysis. The level of statistical significance was set at 5%. For all statistical analyses, SPSS software (IBM SPSS Statistics for Windows Version 21.0; IBM Corp, Armonk, NY, USA) was used.
Results
Among the 50 patients (26 women and 24 men) with a mean age of 51 ± 15.4 (18–79) years, 33 (66%) had abscesses and 17 (34%) had hematomas. All hematomas included in this study were in the acute stage. Chronic hematomas were excluded as they cannot be drained because of their prominent clot components. In addition, chronic intra-abdominal hematomas may become infected, leading to misinterpretation of the results. The mean age of women (46.7 ± 16 (18–79) years) was significantly lower than that of men (55.7 ± 14 (21–79) years) (p = 0.0402) (Table 1). This difference was attributed to the inclusion of relatively younger female patients of reproductive age who underwent obstetric and gynecological interventions.
The features of the patients in the abscess and hematoma groups.

SWV: shear wave velocity.
An evaluation of the localizations and etiologies of the hematomas revealed that 13 hematomas developed following various operations in the pelvic region (cesarean section, hysterectomy, salpingectomy, and myomectomy), 3 after intra-abdominal operations in the upper abdominal quadrants (gastric cancer, gastric perforation, and cholecystectomy), and 1 following treatment for ureteral calculus in the perirenal area (Figure 1).
Among the 33 abscesses, 17 developed in the upper quadrants of the abdomen (postoperatively or spontaneously), 9 in the pelvis (postoperatively or spontaneously), and 7 in the liver (spontaneously) (Figure 2). Bacterial growth in culture was detected in 12 of the 33 (36.4%) abscesses: 6 gram-negative, 4 gram-positive, and 2 mixed (gram-positive and gram-negative).
There was no statistically significant difference in the volume of the fluid contents between abscesses (91.06 ± 47.96 mL) and hematomas (119.29 ± 57.97 mL) (p = 0.0726).
Patients in the abscess group (55.2 ± 15.1 (18–79) years) were significantly older than those in the hematoma group (42.9 ± 13.4 (20–68)) (p = 0.0062) (Table 1). We believe that this difference may be because older patients are more prone to infectious conditions due to increased frequency of various diseases impairing the immune system, such as malignities and diabetes. In addition, relatively older people more frequently undergo gastrointestinal surgeries, which may be complicated with abscess, while younger patients undergo pelvic surgeries, such as cesarean section and myomectomy, which are more often associated with bleeding. Additionally, as a supporting finding, 76.5% (13/17) of the patients with hematoma and only 24.2% (8/33) of those with abscess had effusion in the pelvic region (p = 0.00039), and 42.4% (14/33) of the patients in the abscess group and 70.6% (12/17) of those in the hematoma group were women (p = 0.05899).
The mean SWVs were 0.68 ± 0.16 (0.36–1.07) m/s (Figure 3) in patients with abscesses and 1.11 ± 0.41 (0.54–1.98) m/s in those with hematomas (Figure 4), and the difference was found to be statistically significant (p < 0.001). In the ROC analysis, the area under the curve was 84.8%. The optimal cutoff value was defined as 0.72, with a sensitivity of 82.4% and specificity of 67% (Figure 5).

Elastography image of a 69-year-old patient who had a heterogeneous hypoechoic fluid collection in the right upper quadrant. The fluid obtained by drainage and laboratory examination results of this fluid were consistent with an abscess.
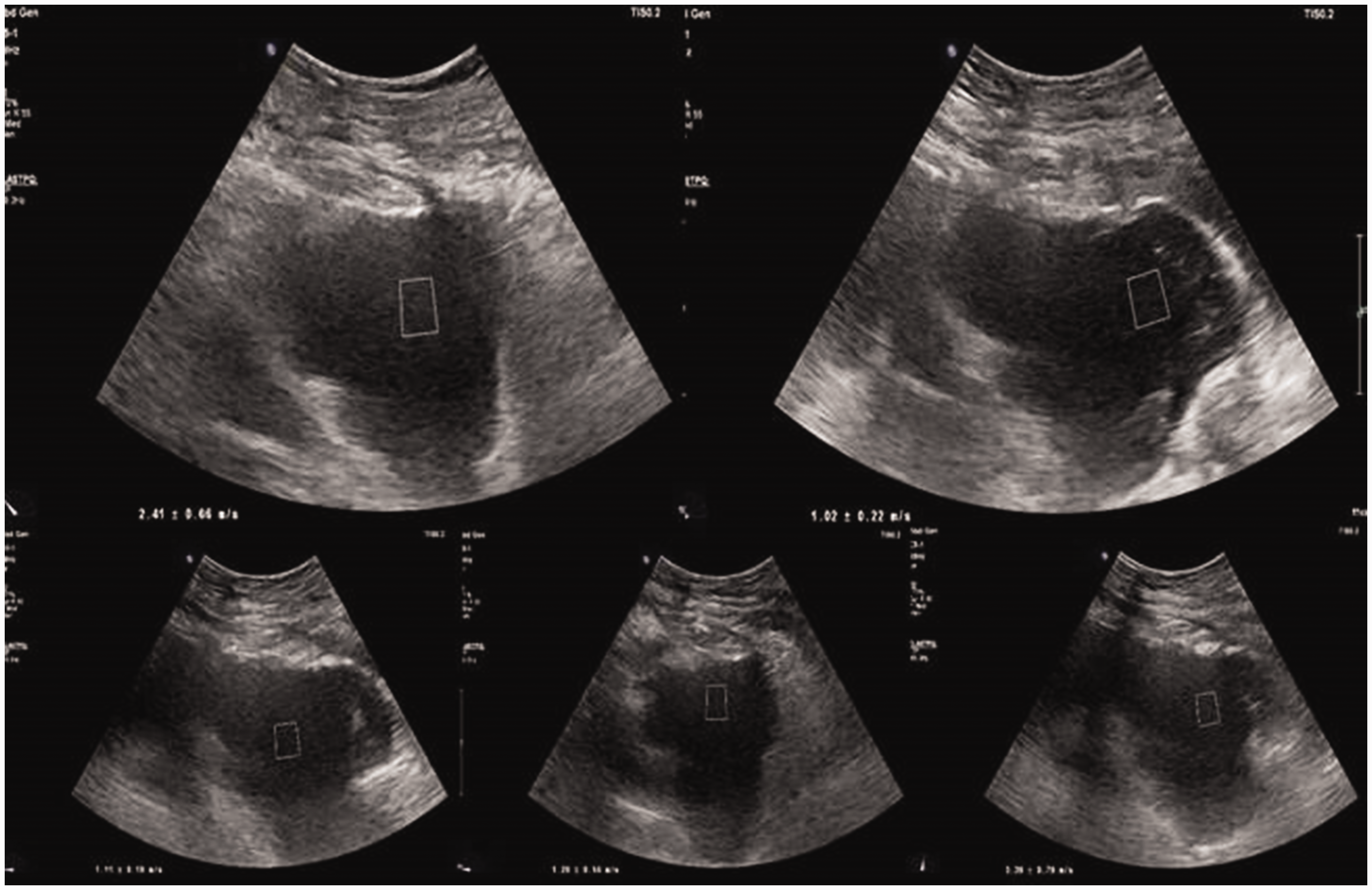
Elastography image of a 34-year-old patient who had a heterogeneous hypoechoic postoperative fluid collection in the pelvis. The laboratory examination results and macroscopic appearance of the aspirated fluid were consistent with a hematoma.
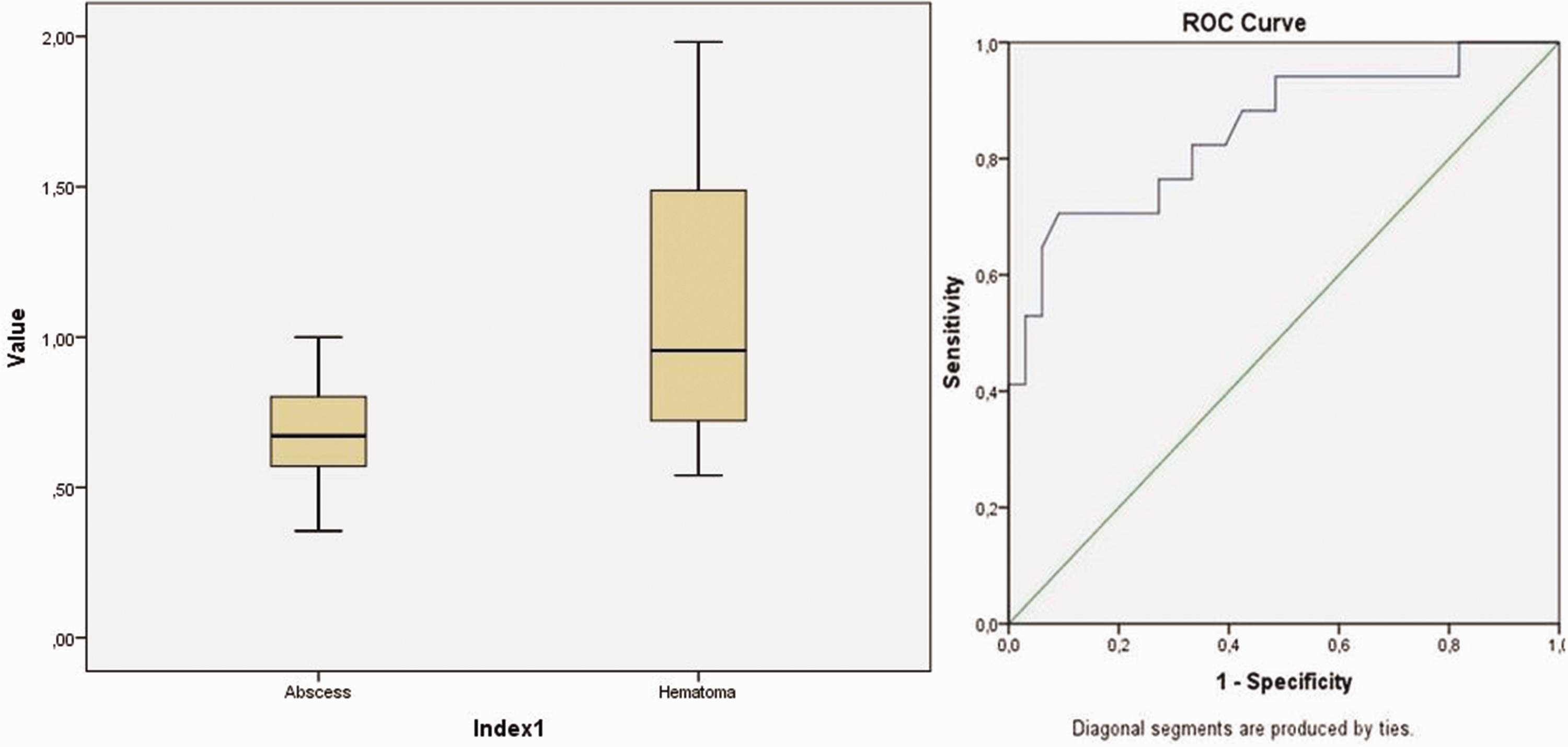
Box-plot graphs of the SWE scores for the abscess and hematoma groups and ROC curve.
Discussion
Hematoma, abscess, biloma, urinoma, seroma, anastomotic leak, or ascites may present as loculated or diffuse intra-abdominal fluid collections, either spontaneously or secondary to trauma or surgery. 1 As imaging modalities have lower specificity in differentiating intra-abdominal fluid collections, distinguishing between hematoma and abscess is challenging, as they require different treatment approaches. Although clinical and laboratory findings are important, they may not always be adequate for early diagnosis. Therefore, it is important to determine whether an intra-abdominal fluid collection is an abscess, particularly in cases when rapid aspiration is required for treatment. 3
Ultrasonography remains the first-line radiological modality for evaluating intra-abdominal fluid collections and guiding drainage procedures, as it is noninvasive, inexpensive, and widely available. Ultrasound elastography is a relatively new technology that provides quantitative information on tissue elasticity. 7
Various CT studies on the differentiation of intra-abdominal fluid collections have reported contradictory results. In a study by Karcaaltıncaba and Akhan, 4 the mean Hounsfield units (HU) were <40 for pelvic abscess and >60 for pelvic hematomas on CT, and the authors concluded that CT-HU was beneficial for differentiating abscesses from hematomas. Another study suggested that infected intra-abdominal collections requiring drainage tend to have HU values above 20, and the authors reported that CT has limited value in differentiating infected from noninfected intra-abdominal fluid collections. 8 In another study, Skusa et al. 16 examined the CT findings and clinical parameters for differentiating infected from noninfected intra-abdominal fluids and reported that these methods have low diagnostic accuracy in distinguishing infected from sterile fluid collections.
Previously, diffusion-weighted MR imaging (DWI) was explored for differentiating abdominopelvic abscesses from noninfected fluid collections. It was suggested that DWI is superior to T2-weighted imaging and that quantitative apparent diffusion coefficient measurements has a potential diagnostic value for this differentiation. 14 It has been reported that older (>3 weeks old) hematomas tend to demonstrate a concentric-ring configuration, most prominent on pulse sequences, appearing as a thin dark peripheral rim on T2-weighted images and a bright inner circle on T1-weighted images. It was concluded that the concentric-ring sign on MRI is an important feature suggesting a chronic hematoma. 17 Although MRI can aid in the diagnosis of hematomas, it is expensive and time-consuming and may require contrast administration.
The ultrasonographic features of intra-abdominal fluid collections may vary depending on the nature of their contents. In a study by Akhan et al., 18 gray-scale ultrasound was used to differentiate intra-abdominal abscesses from other fluid collections. They reported that gray-scale findings may appear very similar, and in many cases, the nature of the fluid cannot be determined via conventional ultrasonography alone. In addition, elastography has been used for differentiating transudative from exudative pleural effusions, with SWV values of the transudative pleural fluid found to be significantly lower. 12 In this study, the mean SWV of hematomas was significantly higher than that of abscesses, with a cutoff value of 0.72 m/s. Zhi et al. 19 investigated the use of elastosonography in differentiating benign from malignant breast lesions and suggested that hemorrhagic contents of the lesions contribute to increased stiffness. Similarly, Batur et al. 20 compared the stiffness of hemorrhagic ovarian cysts and endometriomas using elastosonography. They reported that the clot areas within hemorrhagic cysts were significantly stiffer than cystic areas, similarly to endometriomas. This increased stiffness of hemorrhagic lesions on elastosonography is attributed to degraded blood products, elevated protein levels, and high iron concentrations.20,21
Gaspari et al. 22 examined the stiffness of skin and subcutaneous abscesses using elastosonography. They concluded that elastosonography could detect only 58% of abscess cavities. Among these abscesses, 38% showed mixed red (most soft) and yellow (soft) signals, while 62% showed mixed yellow (soft) and green (medium stiffness) signals. They stated that in contrast to abscess cavities, the surrounding indurated tissues were detected in almost all (98%) of the lesions. In addition, in all these lesions, the surrounding indurated tissue showed blue (firm) and dark blue (stiffest) signals. They concluded that abscess cavities tend to be generally softer, while the surrounding indurated tissue is consistently stiffer on elastosonography.
Awadallah et al. 23 compared the stiffness of focal liver lesions using shear wave elastosonography and found the lowest stiffness in hepatic abscess when compared with focal fatty infiltration, hemangioma, hepatocellular carcinoma, and metastasis. Consistent with these findings, El-Gazzarah et al. 24 measured the stiffness of focal liver lesions in 135 patients. They found the lowest stiffness values in simple cysts (1.4 kPa), abscesses (2.71 kPa), and hydatid cysts (2.8 kPa).
In the literature, only a few studies have investigated sonoelastography for cystic lesions, and data on hemorrhage and abscess are extremely limited. Previous studies have suggested that SWV values are higher in hemorrhagic lesions and lower in abscesses. Consistent with the literature, we found that the SWV values were significantly higher in hematomas than in abscesses. In addition, we found a cutoff value of 0.72 m/s with a sensitivity of 82.4% and specificity of 67% for differentiation. We did not include chronic hematomas in this study because of the increased risk of infection and the likelihood of aspiration failure due to their semi-solid clot contents. As increased stiffness of hematoma is associated with degraded blood products, high protein levels, and high iron concentrations, we believe that chronic hematomas would show higher SWV values than acute hematomas. Consistent with this hypothesis, another study conducted at our institute revealed that the mean SWV values were significantly higher in subacute deep venous thrombosis than in acute deep venous thrombosis. 9 A study focused on the differentiation of elastography values in chronic/subacute and acute hematomas would be very useful; if this theory is confirmed, measurement of elastography values can be potentially used to identify chronic hematomas in cases where diagnostic aspiration is usually unsuccessful. In addition, establishing a cutoff value could help differentiate acute from chronic hematomas and may aid in diagnosing ongoing bleeding through serial sonoelastographic measurements.
Because abscesses have thick, enhancing walls and show restricted diffusion, contrast agent administration and additional sequences are required to differentiate hematomas from abscesses on sectional radiological examinations. An advantage of this method is that it does not require contrast administration and does not expose patients to ionizing radiation. In addition, CT and MRI may not be available in certain centers and conditions. Moreover, it takes only 1–2 min to obtain quantitative results. Another advantage is that it is a very feasible method that can be performed at the bedside, even in patients who have undergone surgical interventions that restrict their movement. The main limitation of elastography for this differentiation is that not all ultrasound devices include the elastography modality, and operators must be well-trained in the technique as this is a dynamic method in which the patients are asked to hold their breath and remain still for a short time. Calculating the mean SWE values in such lesions provides rapid information through a safe and readily available method, allowing aspiration of the content in the same procedural session in cases of abscess.
The major limitation of this study was that a single observer performed the calculations, as elastosonography is an operator-dependent technique. Moreover, the sample size in the hematoma group was relatively small. Furthermore, as only acute hematomas were included, elastographic features were not analyzed according to hematoma age. Because of the technical failure of elastography in lesions with a depth of >8 cm, we could not evaluate deeper lesions. Confounding factors, including localization of effusion, amount of effusion, depth of the effusion, age of the effusion, and sex and age of the patients, which may influence SWE values, were not examined in this study. These factors should be evaluated in further studies with larger sample sizes, particularly in hematoma patients. In addition, future studies should assess the response to antimicrobial treatment by comparing pre- and post-treatment SWV measurements.
Conclusion
Radiological differentiation between intra-abdominal abscesses and hematomas, which have different treatment options and complications, is essential and challenging in most cases, as gray-scale ultrasound, CT, and MRI have some disadvantages and limited diagnostic accuracy. The results of this study support our hypothesis, demonstrating that intra-abdominal abscesses have significantly lower SWV values than hematomas, thereby aiding differentiation between these entities. We believe that if further studies with larger sample sizes confirm these findings, SWE measurements may become a routine radiological tool for differentiating intra-abdominal abscesses from hematomas.
Footnotes
Acknowledgements
None.
Author contributions
SÖ: Conceptualization, data curation, formal analysis, methodology, writing, and editing.
MÖ: Data curation, review, supervision, and editing.
FD: Review and editing.
MBA: Data curation and review.
MA: Review and editing.
İD: Review and writing.
CG: Supervision and editing.
AÖ: Data curation, editing, and review.
İÇ: Data curation and editing.
SB: Data curation and editing.
Data availability statement
The dataset of the study may be availed from the corresponding author upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest in this study.
Funding
There is no funding or financial support for this study.
