Abstract
Human parvovirus B19 infection in immunocompetent adults predominantly manifests as arthropathy or erythema infectiosum, with transient aplastic crisis being a rare hematological complication. We report a diagnostically challenging case of a man in his late 40s who developed recurrent severe aplastic anemia following mechanical valve replacement. Notably, the patient presented with progressive pancytopenia despite optimal anticoagulation and normally functioning prosthetic valves on serial echocardiograms. Critical diagnostic differentiation was achieved through comprehensive laboratory evaluation; elevated lactate dehydrogenase (232.00 U/L) and undetectable haptoglobin levels initially suggested mechanical hemolysis. However, suppressed reticulocyte production index (0.002) and the presence of human parvovirus B19 (DNA) confirmed virus-mediated marrow suppression. The absence of schistocytes on peripheral smear and stable hemoglobinuria levels further excluded prosthetic valve–related hemolysis. To the best of our knowledge, this study represents the first documented case of human parvovirus B19–induced aplastic anemia in a postcardiotomy patient with confirmed valve competence, highlighting three key clinical imperatives: 1. Human parvovirus B19 should be included in the differential diagnosis of postoperative pancytopenia regardless of immune status; 2. disproportionate reticulocytopenia with normal hemolysis markers may indicate viral marrow infiltration; and 3. serial echocardiographic surveillance cannot fully exclude subclinical hemolysis, necessitating multimodal laboratory correlation.
Introduction
Human parvovirus B19 (B19V), a single-stranded DNA virus of the family Parvoviridae and genus Erythrovirus, 1 poses distinct hematological challenges in specific clinical contexts. Although immunocompromised individuals exhibit heightened susceptibility to B19V infection, 2 its clinical significance in immunocompetent surgical populations remains underrecognized. Seasonal transmission patterns (winter/spring) and multiple routes of spread—including respiratory droplets, blood products, and vertical transmission 3 —underscore its epidemiological relevance. Although typically causing subclinical or self-limited manifestations in healthy hosts, B19V demonstrates tropism for erythroid progenitor cells through receptor-mediated invasion, leading to cellular lysis and erythropoietin receptor downregulation. 4 This pathophysiological mechanism underlies its capacity to induce transient aplastic crisis, particularly when superimposed on surgical stress.
The differential diagnosis of postoperative anemia following mechanical valve replacement presents unique diagnostic complexity. Since Rose’ et al.’s 5 seminal 1954 report on prosthesis-related hemolysis, advancements in valve design have reduced mechanical hemolysis incidence to <1% in contemporary series. 6 Current guidelines reserve reoperation for cases with demonstrable paravalvular leakage, refractory heart failure, or acute renal impairment.7,8 Nevertheless, the temporal overlap between postoperative recovery phases and B19V incubation periods (7–18 days) creates a critical diagnostic window in which hematological manifestations may be erroneously attributed to prosthetic valve dysfunction.
This clinical conundrum necessitates a paradigm shift in postoperative anemia evaluation. The coexistence of two mechanistically distinct etiologies—mechanical erythrocyte destruction versus virus-mediated erythropoiesis suppression—demands systematic laboratory differentiation. Heightened clinician awareness of B19V's hematological sequelae may prevent unnecessary interventions while optimizing antimicrobial stewardship, particularly given the virus's established association with pancytopenia in immunocompetent hosts.9,10
Case presentation
This case report adheres to the Declaration of Helsinki. Institutional review board exemption was granted by the Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology Ethics Committee (Wuhan, China; #TJ-IRB202407083) on 29 July 2024. Written informed consent was obtained from the patient for anonymized publication of clinical details and images. Reporting followed the Case Report (CARE) guidelines. 11 The completed checklist is available in Supplementary Materials. We have deidentified all patient details.
In Figure 1, the real diagnosis and treatment process has been organized. A man in late 40s with a history of hypertension, diabetes mellitus, coronary artery disease, and chronic hepatitis B was admitted to the Rheumatology and Immunology Department of Tongji Hospital in mid-2020, presenting with a 6-month history of debilitating lumbar pain (visual analog scale score, 7/10), recurrent afternoon fever (peak, 38.5°C), night sweats, and unintentional weight loss of 20 kg. Physical examination revealed tenderness in the right second and third metacarpophalangeal joints and bilateral calf regions, although no neurological deficits were observed. Initial laboratory investigations demonstrated normocytic anemia (hemoglobin level, 103 g/L; erythrocyte count, 3.68 × 1012/L), neutrophilia (6.58 ×109/L; 75.7%), and a markedly elevated erythrocyte sedimentation rate (84 mm/h). Autoimmune evaluations, including antinuclear antibody, anti-neutrophil cytoplasmic antibody, and human leukocyte antigen-B27 testing, were unremarkable. Notably, blood cultures identified gram-positive cocci, and echocardiography revealed severe mitral valve regurgitation with valvular perforation (Figure 2(a)), consistent with subacute infective endocarditis.
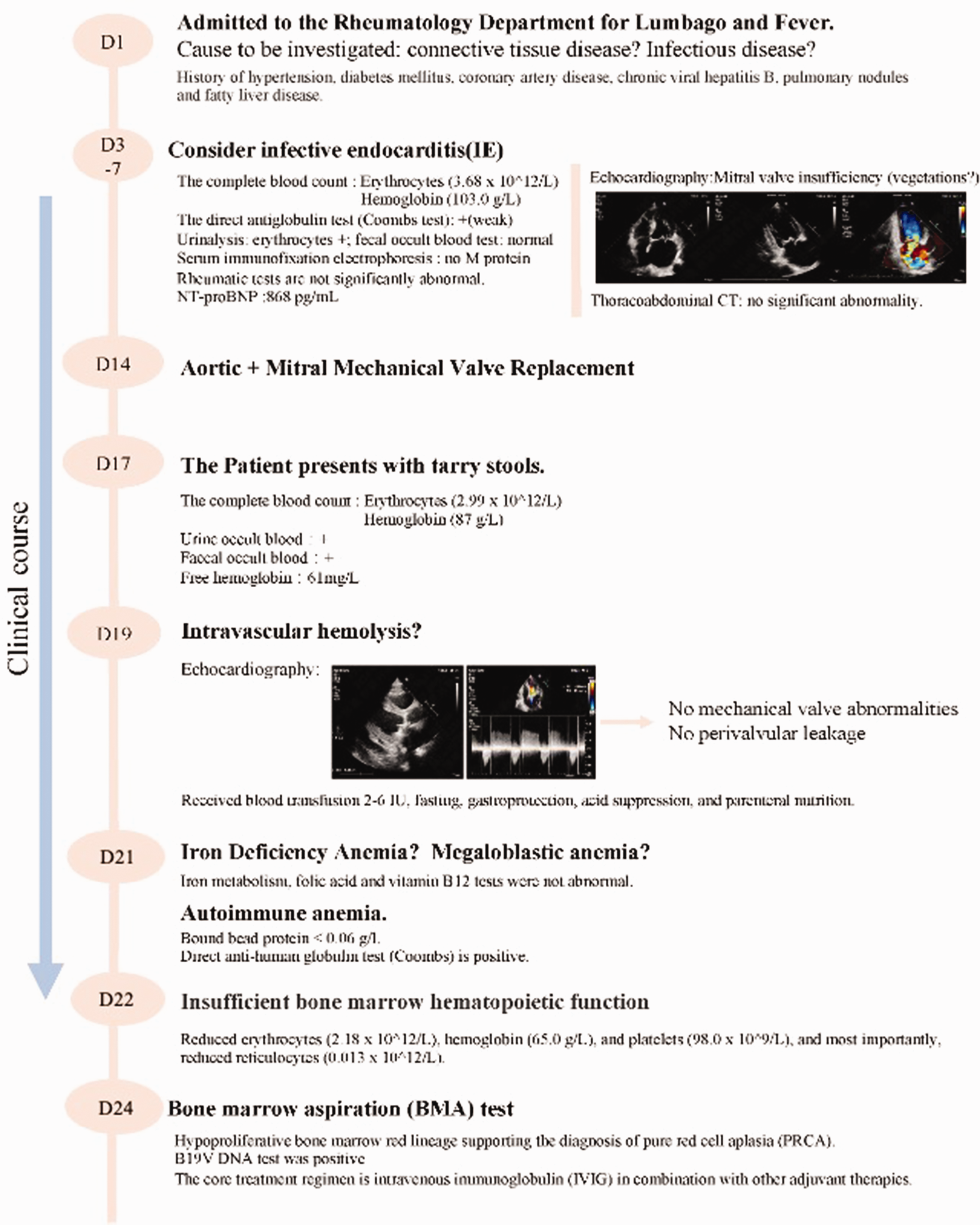
The clinical course of the patient (schematic representation).

A figure showing (a) preoperative echocardiogram and (b) postoperative echocardiogram.
Following stabilization of cardiac function and completion of preoperative evaluations, mechanical mitral and aortic valve replacement was performed. Intraoperative findings confirmed friable vegetations on both valves. The patient was extubated within 24 h and transferred from the intensive care unit with stable hemodynamics.
On postoperative day 3, the patient developed melena, lethargy, fever (38.8°C), and progressive pallor. Laboratory studies revealed worsening anemia (hemoglobin level, 65.0 g/L) with reticulocytopenia (0.013 ×1012/L), thrombocytopenia (98 ×109/L), and elevated free hemoglobin level (132 mg/L). Serial echocardiograms confirmed normal prosthetic valve function without paravalvular leakage (Figure 2(b)). Despite empiric corticosteroid therapy and intravenous immunoglobulin (IVIG) administration for suspected immune-mediated hemolysis, pancytopenia persisted. Bone marrow biopsy performed on postoperative day 17 demonstrated erythroid hypoplasia (granulocyte-to-erythroid ratio, 50:1), scattered hemosiderin-laden macrophages, and absence of dysplastic changes, confirming pure red cell aplasia (PRCA) (Figure 3(a) to (c)). Definitive diagnosis was established through a positive result for B19V DNA.

The bone marrow biopsy: (a) aspirate smear, (b) immunohistochemistry, and (c) chromosome test.
After initiating treatment for PRCA associated with human papilloma virus B19 infection, the core therapeutic regimen includes IVIG, supplemented by other adjuvant therapies: (a) high-dose IVIG (total dose 1.5–2.0 g/kg); (b) immunosuppressive therapy with cyclosporine A or mizoribine; (c) recombinant human erythropoietin injection (Chinese hamster ovary cells), 4000 IU, administered three times weekly; and (d) supportive measures such as blood transfusion and oxygen therapy. As shown in Table 1, following treatment, significant improvements were observed in complete blood count parameters, including increased reticulocyte count and stable hemoglobin levels, prompting consideration for outpatient discharge.
Patient's laboratory test results.

B19V: human parvovirus B19; CMV: cytomegalovirus; EBV: Epstein–Barr virus; HBV: hepatitis B virus; hsTnI: high-sensitivity troponin I; NT-proBNP: N-terminal pro-B-type natriuretic peptide.
Review
This case series elucidates a critical diagnostic paradigm shift in postoperative hematological complications following mechanical valve replacement. Our findings demonstrate that B19V-induced PRCA, although rarely documented in cardiothoracic surgical populations, represents a potentially fatal etiology of hemolytic anemia that demands heightened diagnostic vigilance. The clinical trajectory underscores two fundamental misconceptions in postoperative care: (a) the erroneous assumption that mechanical valve integrity excludes concurrent infectious pathologies and (b) the underappreciation of B19V's capacity to exploit perioperative immune dysregulation. The pathophysiological convergence of mechanical erythrocyte trauma and virus-mediated erythropoietic suppression creates a unique diagnostic conundrum. Although intravascular hemolysis following cardiac procedures typically prompts evaluation for paravalvular leakage or prosthesis malfunction, our findings reveal that sustained hematological deterioration (particularly reticulocytopenic anemia) should immediately trigger investigation for opportunistic viral pathogens.
Case discussion
Postoperative intravascular hemolysis remains a critical complication following mechanical valve replacement, with contemporary studies reporting incidence rates below 1% in optimized surgical settings. 7 Although technological advancements have mitigated traditional mechanical etiologies such as paravalvular leakage and chordal abnormalities, 12 the pathophysiological interplay between shear stress dynamics and erythrocyte fragility persists as a fundamental mechanism. 13 Elevated shear forces exceeding 3000 dyn/cm2 induce not only direct erythrocyte membrane fragmentation but also platelet glycoprotein Ibα cleavage, creating a prothrombotic milieu that exacerbates hematological compromise.14,15 This mechanistic understanding necessitates rigorous postoperative surveillance, particularly in patients requiring transfusion support or developing secondary organ dysfunction, given the 10% increased mortality risk associated with reoperation for refractory hemolysis. 16
Definitive diagnosis requires multimodal integration of prosthetic valve imaging and hematological profiling. Although transthoracic echocardiography remains the first-line examination for assessing transvalvular gradients and leaflet mobility, 17 the emergence of four-dimensional flow cardiac magnetic resonance imaging permits quantitative analysis of turbulent kinetic energy—a parameter correlating strongly with hemolytic severity (r = 0.82; P < 0.01). Laboratory differentiation hinges on recognizing phenotype-specific markers: schistocyte counts of >1% and haptoglobin level of <0.1 g/L strongly suggest mechanical destruction, whereas reticulocytopenia (<40 × 109/L) with normal fragmentation indices indicate marrow suppression etiologies.
The emergence of B19V-associated PRCA in this context introduces a novel diagnostic dimension. As an erythrotropic parvovirus targeting P antigen–positive erythroblasts, 18 B19V induces not only direct cytolysis but also erythropoietin receptor downregulation through nonstructural protein 1 protein–mediated JAK/STAT pathway inhibition. 1 This dual mechanism explains the paradoxical combination of hemolytic markers (elevated lactate dehydrogenase (LDH) level >500 U/L) and reticulocyte production index of <2% observed in our case—a hematological profile distinct from classical mechanical hemolysis.
Diagnostic criteria for hemolytic anemia require reinterpretation in postcardiotomy patients. Although interpretation of traditional markers (reticulocytosis, LDH level elevation, and indirect hyperbilirubinemia) remains valid, it must account for surgical context: 1.Reticulocyte dynamics: postoperative erythropoietin resistance may blunt reticulocyte response despite hemolytic activity; 2. LDH specificity: levels of >2.5 × upper limit of normal (ULN) show 94% specificity for hemolysis versus 68% for 1.5–2.5 × ULN ranges; and 3. hemoglobinuria thresholds: concentrations of >50 mg/dL strongly correlate with intravascular hemolysis (positive predictive value, 91%).
Therapeutic challenges in B19V-associated PRCA underscore the importance of early virological identification. Although no Food and Drug Administration–approved antivirals exist, IVIG (2 g/kg over 2–5 days) demonstrates 78% response rates by neutralizing viral particles through donor-derived anti-viral protein 2 immunoglobulin (Ig) G. 19 Our protocol's success (hemoglobin level, Δ + 47 g/L at 4 weeks) aligns with emerging evidence, suggesting enhanced efficacy when combining IVIG with erythropoiesis-stimulating agents in immunocompromised hosts. 19 Paradoxically, the absence of seroconversion in our patient despite clinical resolution highlights the complex interplay between surgical stress and humoral immunity—only 62% of postcardiotomy patients develop protective IgG titers versus 93% of immunocompetent controls. 20
This paradigm necessitates protocolized postprocedural virological surveillance, particularly in patients with persistent anemia despite adequate valve function. Future research should address the temporal relationship between cardiopulmonary bypass duration and B19V reactivation rates as well as the potential role of preemptive IVIG in high-risk populations.
Conclusion
Postprocedural hemolysis following valvular surgery, although rare, demands meticulous etiological discernment to prevent life-threatening complications. This case confirms B19V as a critical differential for postvalvular anemia with normal imaging. Early polymerase chain reaction testing and IVIG may avert unnecessary interventions, although generalizability requires multicenter validation.
Footnotes
Acknowledgments
Patient consent was obtained and no artificial intelligence tools were used.
Author contributions
XM was a trainee who participated in the management of the patient and drafted the manuscript. NH was the main consultant for the patient's admission, and residents CL and YF together with XM assisted in the management of the patient. SL played a key role in all stages of the patient’s management and provided supervision and guidance for the writing and review of the manuscript. CC reviewed the manuscript.
Data availability statement
Data provision: IRB-archived datasets (TJ-IRB202407083) available upon request.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
This study involving a human participant was reviewed and approved by Ethics Committee of Tongji Medical College, Huazhong University of Science and Technology (Institutional Review Board document number TJ-IRB202407083). Written informed consent was obtained from the patient for treatment as well as for the publication of the case report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
