Abstract
This current case report describes a Japanese woman in her 80s with xerostomia who presented with salivary gland dysfunction. She was positive for isolated anti-Ro52/SS-A antibody as determined by a chemiluminescent enzyme immunoassay and positive on a fluorescence enzyme immunoassay that recognizes both Ro52 and Ro60 antigens. A high serum concentration of anti-Ro52/SS-A antibody was determined by an enzyme-linked immunosorbent assay. A high anti-centromere antibodies (ACA) titre was also observed. Although Raynaud’s phenomenon or a high serum immunoglobulin G concentration were not observed, Masson-trichrome staining of the patient’s labial salivary glands showed considerable fibrosis. Her serum type I and type II interferon concentrations were normal. The present patient is the first with Sjögren’s syndrome (SS) to exhibit isolated anti-Ro52/SS-A antibody and ACA without anti-Ro60/SS-A antibody. This current case report presents her case together with those of four other SS patients who were positive for isolated anti-Ro52/SS-A antibody. The SS literature also includes cases positive for ACA and describes clinical characteristics. The other four SS patients with isolated anti-Ro52/SS-A antibody described here had no ACA; interstitial pneumonia and polyneuropathy were observed in these cases, although there was no consistent tendency regarding types I and II interferon.
Introduction
Autoantibodies that have been detected in individuals with Sjögren’s syndrome (SS) that involves glandular and extraglandular manifestations are reported to be related to specific organ damage.1,2 Although Ro antigen contains Ro52-kDa and Ro60-kDa subtypes, 3 individuals with SS who are anti-Ro/SS-A antibody-positive demonstrate higher disease activity compared with those without these autoantibodies. 4 Anti-Ro52/SS-A antibody in particular is reported to be associated with organ involvement such as myositis, interstitial lung disease (ILD)5,6 and congenital heart block. 7 As this current research group has shown previously, a high level of anti-Ro52/SS-A antibody coexists with anti-Ro60/SS-A antibody in SS; and anti-Ro52/SS-A antibody-seropositive SS shows high scores on the European Alliance of Associations for Rheumatology (EULAR) SS Disease Activity Index (ESSDAI). 8
The current authors speculated that since the disease activity or organ damage of SS patients with anti-Ro52/SS-A antibody might be influenced by coexisting anti-Ro60/SS-A antibody, it is possible that anti-Ro52/SS-A antibody-positive SS cases would directly show this antibody-specific disease condition. It was reported that isolated anti-Ro52-positive SS patients tended to have ILD or myositis with high disease activity.9,10 A previous study demonstrated that among individuals with SS, those with isolated anti-Ro52/SS-A antibody-seropositive SS exhibited a severe phenotype with organ involvement such as ILD or vasculitis. 10 In contrast, anti-centromere antibody (ACA), which is usually identified in individuals with limited cutaneous systemic sclerosis, was observed in patients who fulfilled the classification criteria for primary SS with clinical characteristics including a high frequency of Raynaud’s phenomena and/or a normal serum immunoglobulin G (IgG) concentration. 11 This current research group has previously shown that isolated anti-Ro52/SS-A antibody-seropositive patients with primary SS had no ACA. 8
For the present case series, the case of the current patient with isolated anti-Ro52/SS-A antibody-positive primary SS with ACA was compared with four cases of primary SS patients with isolated anti-Ro52/SS-A antibody-seropositive by determining their pathological characteristics.
Case report
In May 2023, a Japanese female in her 80s (patient 1; Table 1) visited the Department of Medicine, Nihon University School of Medicine, Tokyo, Japan with complaints of a dry mouth that had been present for approximately 1 month. Dry eyes were not described by the patient or detected in the physical examination. At another hospital, the results of Schirmer’s test and a fluorescein staining test were negative; however, the stimulated salivary secretion volume by a gum test was 2 ml (<10 ml is considered positive), the unstimulated salivary secretion volume was 0.013 ml/min and salivary gland scintigraphy revealed severe functional abnormality with a decreased 99mtechnetium uptake and washout rate (Figure 1). The general physical examination revealed no fever, lymphadenopathy or abnormalities in heart or respiratory sounds. There was also no Raynaud’s phenomenon, skin sclerosis, nail fold abnormalities, muscle weakness or peripheral neuropathy. The patient showed no symptoms found in CREST syndrome (i.e. calcinosis, Raynaud’s phenomenon, oesophageal dysmotility, sclerodactyly and telangiectasia). A chest X-ray did not reveal interstitial pneumonia or pleural effusion.
Clinical characteristics of five cases of isolated anti-Ro52/SS-A antibody-positive Sjögren’s syndrome.
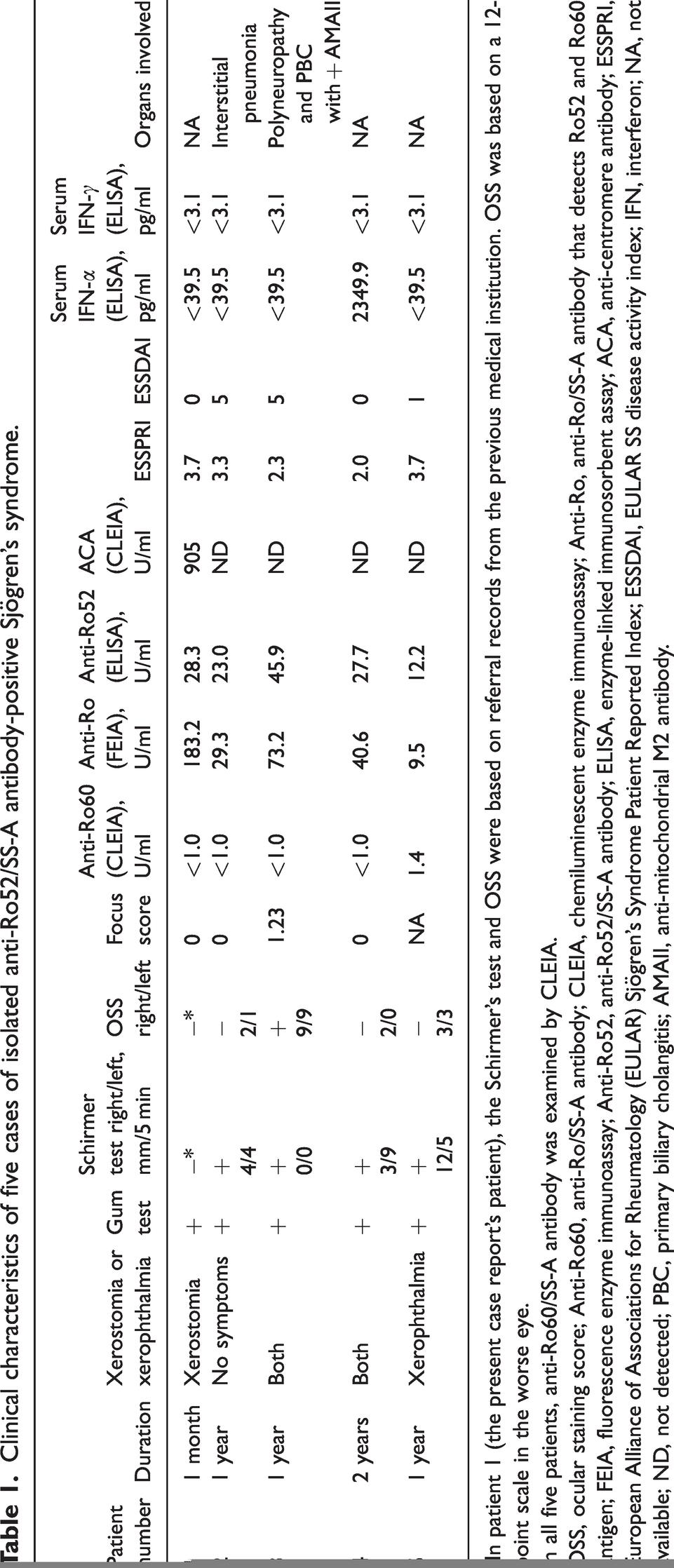
In patient 1 (the present case report’s patient), the Schirmer’s test and OSS were based on referral records from the previous medical institution. OSS was based on a 12-point scale in the worse eye.
In all five patients, anti-Ro60/SS-A antibody was examined by CLEIA.
OSS, ocular staining score; Anti-Ro60, anti-Ro/SS-A antibody; CLEIA, chemiluminescent enzyme immunoassay; Anti-Ro, anti-Ro/SS-A antibody that detects Ro52 and Ro60 antigen; FEIA, fluorescence enzyme immunoassay; Anti-Ro52, anti-Ro52/SS-A antibody; ELISA, enzyme-linked immunosorbent assay; ACA, anti-centromere antibody; ESSPRI, European Alliance of Associations for Rheumatology (EULAR) Sjögren’s Syndrome Patient Reported Index; ESSDAI, EULAR SS disease activity index; IFN, interferon; NA, not available; ND, not detected; PBC, primary biliary cholangitis; AMAII, anti-mitochondrial M2 antibody.
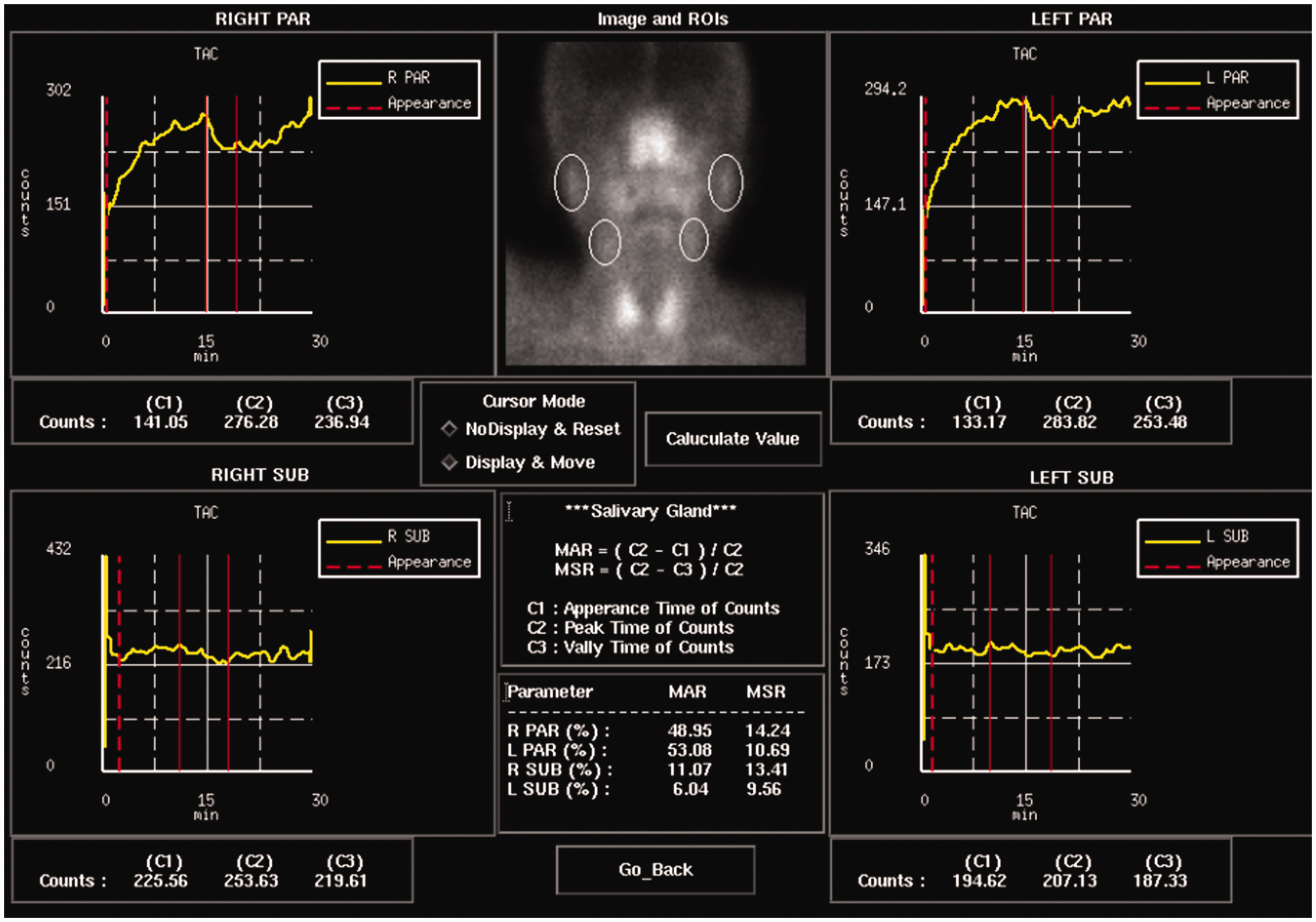
Results of salivary gland scintigraphy for a female in her 80 s (patient 1) who presented with complaints of a dry mouth that had been present for approximately 1 month who was subsequently diagnosed with Sjögren’s syndrome. The 99mtechnetium uptake in the upper centre of the image and the washout rate using acid stimulation as a time-activity curve are shown in the four panels. The top two images show the washout rates of the patient’s left and right parotid glands; and the bottom two images show the washout rates of the patient’s left and right submandibular glands. The colour version of this figure is available at: http://imr.sagepub.com.
Laboratory tests showed no urinary protein or occult blood and no leukocyte or thrombocytopenia, liver or renal dysfunction, or elevated myogenic enzymes, although the erythrocyte sedimentation rate was mildly elevated at 29 mm/h. The patient’s serum IgG was 1258 mg/dl, her complement C3/C4 was normal and cryoglobulins were negative. Antinuclear antibodies were 1:1280 in a discrete pattern and autoantibody results were negative, including anti-cyclic citrullinated peptide antibody at 0.6 U/ml (normal value: <4.5 U/ml), anti-double-strand deoxyribonucleic acid antibody <10 IU/ml, anti-ribonucleoprotein antibody <2.0 U/ml, anti-Scl-70 antibody <1.0 U/ml, anti-Sm antibody <1.0 U/ml and an anti-ARS antibody index <5.0.
The patient’s anti-Ro60/SS-A antibody measured by a chemiluminescent enzyme immunoassay method was <1.0 U/ml, which was negative, but the result was positive by a fluorescence enzyme immunoassay (FEIA) that can identify both Ro60 antigen and Ro52. Based on this observation, it was speculated that the patient was isolated positive for anti-Ro52/SS-A antibody. A biopsy of the patient’s minor salivary gland revealed mild interstitial fibrosis but no focus (Figure 2).

The minor salivary gland biopsy results for a female in her 80 s (patient 1) who presented with complaints of a dry mouth that had been present for approximately 1 month who was subsequently diagnosed with Sjögren’s syndrome: (A) haematoxylin and eosin staining; and (B) Masson trichrome staining to evaluate fibrosis. The scale bar (100 µm) in panel A also applies to panel B. The colour version of this figure is available at: http://imr.sagepub.com.
Based on the presence of positive anti-Ro52/SS-A antibody and the unstimulated salivary secretion volume, the patient was classified in accordance with the 2016 American College of Rheumatology (ACR)/EULAR Classification criteria as having primary Sjogren’s syndrome. 12 The result of an enzyme-linked immunosorbent assay (ELISA) for Ro52/SS-A antigen (Orgentec Diagnostika, Mainz, Germany) in the patient’s serum was positive at 28.3 U/ml (the control dilution medium concentration was 0.4 U/ml). Anti-La/SS-B antibody was negative.
However, the patient’s ACA value was high at 905 U/ml (cutoff value: 10 U/ml). Physically, tongue dryness was marked, Raynaud’s phenomenon was not observed, the modified Rodnan total skin thickness score of 17 sites was 0 and nail epithelial extension or nail fold bleeding at the same site was not observed. The patient’s EULAR Sjögren’s Syndrome Patient Reported Index (ESSPRI) was 3.7 points (dryness: 6 points; fatigue: 5 points; pain: 0 points) and her ESSDAI score was 0 points (Table 1). No cytopenia, elevated myogenic enzymes, KL-6, cryoglobulinaemia or low complement was observed. The IgG level was normal at 1258 mg/ml (normal: 861–1747 mg/ml). Examinations by chest X-ray and chest computed tomography (CT) including high-resolution CT showed no interstitial changes. The patient’s serum concentrations of interferon-alpha (IFN-α) and interferon-gamma (IFN-γ) (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) measured by ELISA were both below the assay’s detection sensitivity. After treatment including gargling with sodium gualenate hydrate sodium bicarbonate once or twice a day and the use of artificial saliva was administered to the patient, the subjective symptoms of xerostomia improved.
The cases of four individuals with isolated anti-Ro52/SS-A antibody-positive SS without ACA were documented at the same institution after April 2022 (patients 2–5; Table 1). All four were female and their ages ranged from 55 to 73 years. Three patients had either xerophthalmia or xerostomia. The result of the stimulated saliva secretion test was positive in all four patients, but salivary gland scintigraphy was positive in all cases (data not shown). Schirmer’s test was positive in each of the four cases and fluorescein staining determined by the ocular staining score was positive in patient 3. A minor salivary gland biopsy was performed on two patients and the focus score of one of these two patients was positive at 1.23. In the series of four patients, the levels of anti-Ro52/SS-A antibodies that had been measured by an ELISA ranged between 12.2 and 45.9 U/ml. The IgG level was elevated to 1837 mg/dl in only patient 5 and a decrease in C4 was observed only in patient 4. The patients’ ESSPRI scores were 2.0–3.7 and their ESSDAI results were 0–5. Two of the five patients presented with polyneuritis and interstitial pneumonia, respectively; and the former patient had primary biliary cholangitis and was positive for anti-mitochondrial M2 antibody. The serum IFN-α and IFN-γ measurements showed that only patient 4 had a high IFN-α value at 2349.9 pg/ml, but no organ damage was observed.
The study was conducted with the approval of the Ethics Committee (Human Studies) of Nihon University Itabashi Hospital affiliated to Nihon University School of Medicine, Tokyo, Japan (approval no. RK-220510-5). The reporting of this case report conforms to the CARE guidelines. 13
Discussion
A literature search indicated that the current case (patient 1) is the first observed case of SS in which both isolated anti-Ro52/SS-A antibody and ACA were positive without anti-Ro60/SS-A antibody. No organ damage specific to each autoantibody was observed, but the patient’s salivary gland dysfunction determined by salivary scintigraphy was severe compared with her lacrimal gland function. A decline in saliva quantity and quality that is associated with aging is widely observed even among individuals without SS. 14 In addition, considerable fibrosis was detected by a staining method specific for fibrosis. In individuals with systemic sclerosis (SSc), ACA positivity among the Ro52-positive cases has been observed at a high rate of 61.9%. 15 In contrast, a review of 16 patients with ACA-positive and anti-Ro60/SS-A antibody-negative primary SS did not observe anti-Ro52/SS-A antibody in any of the cases. 8 Although the number of cases examined was low, the difference in the complication rate of ACA to anti-Ro52/SS-A antibody was significantly different between the SSc and SS cases. 8 The autoantibody profile may explain the characteristics of this current case. The frequency of isolated anti-Ro52/SS-A antibody in SS was reported as 12.0% in two previous studies.8,10
Regarding the high disease activity of isolated anti-Ro52/SS-A antibody-positive cases, two patients had organ damage, which was consistent with a previous report. 10 Regarding Ro52/SS-A and organ damage, it was reported that excessive expression of TRIM21/Ro52 suppresses cell proliferation and enhances CD40-mediated apoptosis. 16 Apoptosis further increases the amount of Ro52 antigen, inducing a stronger autoimmune response. 16 Regarding the relationship between anti-Ro52/SS-A antibody titres and the degree of organ damage, a previous study demonstrated that although there were few positive cases of organ damage, the antibody-positive cases tended to have higher antibody titres. 8
The current patient (patient 1) showed the highest anti-Ro52/SS-A antibody level among the five cases by FEIA, but no significant value was shown by ELISA. In her case, the anti-Ro52/SS-A antibody was measured by both ELISA, which measures enzyme activity; and FEIA, which measures the intensity of the fluorescent substrate. Although these two assays are quantitative measurement methods, they are not interchangeable. Since our patient’s serum IgG level was normal, it is unlikely that there was an interference effect due to the presence of heterophilic antibodies; in addition, her rheumatoid factor (which can bind to foreign antibodies) value was also normal, and thus excessive positivity due to the absence of antigen is unlikely.
Individuals with anti-Ro/SS-A antibody-positive SS may have a strong activation of the innate immune system and an increase in IFN-α, but the serum level of IFN-α was increased as a statistical outlier in only one of the five patients, and no such increase was observed in the current patient 1. Her IFN-γ was measured because it is thought that IFN-γ is a representative type II interferon that is increased in the salivary glands, but no increase was observed although she was positive for ACA. A previous study measured cytokine levels in the serum of primary SS patients, but they did not detect IFN-α or IFN-γ. 17
It has become clear that in the pathology of SS, not only the adaptive immune system that is composed mainly of IFN-γ from type 1 helper T cell, but also natural immunity accompanied by the release of type I IFN plays a role in priming the disease at an early stage. 18 It has also become clear that antigen presentation and T/B cells interaction due to IFN release are involved in autoantibody production. Measuring these interferons at the serum level may be difficult. We speculate that ACA counteracts the high type I/II interferon signature with isolated anti-Ro52/SS-A antibodies. In terms of phenotype, we suspect that it involves little organ damage such as interstitial pneumonia and that immunoglobulins are unlikely to increase.
Regarding fibrosis in ACA-positive SS patients, a previous study evaluated fibrosis in the salivary glands of ACA-positive SS patients by applying Azan-Mallory staining, which revealed a significant increase in fibrosis. 11 Staining collagen fibres using Azan-Mallory staining or Masson trichrome staining is required in the evaluation of fibrosis because using haematoxylin and eosin staining alone is insufficient.
This current study had several limitations. First, it was a case report describing a small patient series and it is unclear how many SS patients exist in whom ACA is associated with isolated anti-Ro52/SS-A antibody. Secondly, Ro52 can cause extensive organ damage including interstitial pneumonia and myopathy, but this was not investigated. Regarding the low frequency of positive minor salivary gland biopsy results, if there is a sufficient amount of minor salivary gland tissue, there remains a possibility that a focus indicating sialadenitis could have been obtained. In Japan, the Ministry of Health, Labor and Welfare (MHLW) criteria are used in the daily clinical practice for SS patients, and thus evaluations such as the unstimulated saliva secretion test in the 2016 ACR/EULAR Classification criteria were not performed for all of the patients in this case series. Regarding the staining test, the MHLW standards consider corneal staining as positive, which differs from the other countries that belong to the EULAR and the ACR. These study limitations will need to be reconciled as clinical research progresses. Future research aims to address the following: (i) aquire further data concerning the frequency of this new phenotype including both isolated anti-Ro52/SS-A antibody and ACA-positive SS cases that have not been reported before; (ii) determine the clinical pathological symptoms of these individuals by conducting observational studies with non-SS controls; and (iii) determine whether ACA-positive SS progresses to SSc over the long term.
In summary, this is the first report of a case of SS that was positive for both isolated Ro52/SS-A antibody and ACA. The patient had no organ damage such as interstitial pneumonia, Raynaud’s phenomenon or elevated serum IgG; and pathologically, her case was unprecedented, showing significant fibrosis in the minor salivary glands. When considering the treatment approach for patients with SS that is positive for both isolated Ro52/SS-A antibody and ACA, this current case is informative when considering how the lack of organ damage and fibrosis in the salivary glands affect the outcome.
Footnotes
Author contributions
Study conception and design: H.N., M.T.; acquisition of data: K.N., H.N., H.K., J.S., Y.I., T.I., H.H.; analysis and interpretation of data: K.N., M.T., Y.N., K.A., H.I., M.N., S.A., N.K., H.N.; writing manuscript: H.N.
Availability of data
All data analysed in this study are included in this article.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
