Abstract
Objective
Reaching training with error augmentation has recently shown great promise for enhancing bimanual therapy training, using both robotic force feedback (haptics) and visually distorted display elements (graphics) to amplify motor learning.
Methods
In this two-arm, randomized controlled trial, we explored the effect of visual error augmentation alone by visually shifting the paretic limb’s cursor in the direction of error. We invited 38 chronic (>8 months postinjury) stroke survivors to practice bimanual reaching for approximately 40 min, 3 days per week for 3 weeks.
Results
The arm motor section of the Fugl–Meyer score (maximum score, 66 points) increased by an average of 2.2 and was retained at an average of 1.5 at a follow-up evaluation 7–9 weeks (approximately 2 months) later. For chronic stroke survivors, an improvement of ≥5.2 in the arm motor section of the Fugl–Meyer score is considered a clinically meaningful increase. No superiority was detected due to the error augmentation treatment, but other measures of composite abilities (range of motion, bimanual symmetry, and movement time) showed improvements favoring error augmentation.
Conclusions
Although removing robotic forces led to smaller gains than those reported previously, such touch-free bimanual therapy may prove to be an effective inexpensive automated rehabilitation tool for wider accessibility in therapeutic interventions.
Introduction
Stroke is the leading cause of disability. 1 Despite several advances in rehabilitation, stroke survivors continue to experience life-long reduction in function, necessitating the growing need for effective treatment. This is particularly true for the multifunctionality of the upper extremity. Repetitive reaching exercise has shown increases in various measures, 2 but the available therapy time is limited, and recovery is rarely complete. 3 Technology adjuncts can aid and often enhance rehabilitation with less economic demand. Strategies employing knowledge of the mechanisms of neuroplasticity can help reach the common goals of improved range of motion, coordination, accuracy, and efficiency. Herein, we explored bilateral exercises enhanced by a specialized form of augmented reality, error augmentation (EA).
Our research is based on previous studies on EA—a technique that amplifies errors during repetitive practice sessions. This technique capitalizes on the essential role of error in neural adaptation, enabling individuals to leverage their own mistakes to trigger change.4,5 However, individuals with compromised sensorimotor function may have difficulty noticing errors during tasks or activities. 6 Our group and others have clinically evaluated the outcomes of EA treatment, where errors were artificially enhanced during repetitive training. This may induce modest improvements in learning for both healthy individuals and those with a history of stroke.7 –11 This may be because magnified errors lead to heightened corrections, which is a form of error-driven or supervised neuroplasticity. 12 Such error-driven learning is an aspect of the neuroplasticity observed during skill acquisition.7,8,13 –16 If an error drives learning, then an artificially enhanced error can enhance learning.
This concept of EA was tested by our group in a blinded, randomized controlled study, where we employed the principle of EA in a collaborative therapist–participant–machine trio. 9 When a therapist provided a movement cue to the participants, an interactive robot system visually and haptically magnified their errors in real-time, without the participant’s knowledge. The EA group demonstrated significant advantages over the sham group, which underwent similar practices but no EA, showing a clear superiority of EA to robot-facilitated therapy. This was an important result because robotic treatments have not always shown superiority over regular practice or the typical standard of care.17 –19
The potentially high number of variations in this approach prompts us to explore which parts are most needed for its optimal efficacy. A follow-up study focused on bimanual tasks, wherein the participants provided cues to themselves to determine if similar results are possible from the participant’s own contralateral, less affected arm, allowing for self-rehabilitation with EA.10,12 Studies on bimanual training for stroke recovery have suggested that bimanual training engages additional cortical areas and can influence the formation of new task-relevant networks and connections in a reorganization plan associated with restored motor function.20 –24 An advantage of bimanual therapy is the possibility for additional “solo” training that can occur after daily one-on-one therapy has been completed. This self-telerehabilitation variation was again tested with EA, with the additional ability to compare our previous studies with similar protocols. This second study also demonstrated a clear superiority of EA to robot-facilitated therapy in a blinded, randomized controlled trial.
A lingering question from previous studies is whether a robot is necessary or whether a simpler setup of a tracking system might allow the illusion of distortion simply by displacing the visual feedback to look like the hand was being pushed further in the error direction. Here, we present results from a visual distortion environment that shifts the subject’s cursor in the direction of instantaneous error as if it is being pushed by a robot. We included the additional challenge of using both limbs to transport and balance a ball on a tray—TrayBall. As mentioned previously, the question was whether the observed adaptations can be generalized to movement ability outside the device, as similar research suggests that only 40% of adaptation transfers to motor ability after exiting the machine.5,25
Moreover, beyond the explicit rationale for the removal of robotic haptics, it is imperative to recognize the potential implications of this decision on the overall therapeutic approach. By shifting the focus from robotic interventions to a visual distortion environment, we aim to explore the fundamental principles underlying EA in stroke rehabilitation while minimizing the reliance on complex technological infrastructure. This methodological shift not only allows for a more cost-effective and scalable intervention but also fosters a deeper understanding of the mechanisms driving neuroplasticity and motor learning poststroke. Additionally, by embracing simplicity in our approach, we aim to empower both clinicians and patients with tools that are readily accessible and adaptable to diverse clinical settings and individual needs. Through this deliberate departure from conventional robotic-assisted therapies, we endeavor to pave the way for innovative, evidence-based rehabilitation strategies that prioritize efficacy, accessibility, and patient-centered care.
To test this, we again measured the effects of our training on clinical scores and hypothesized that bimanual training with EA can achieve the greatest gains in the arm motor section of the Fugl–Meyer (AMFM) score. This work was presented previously in its preliminary form focusing on feasibility and safety.25,26
Methods
Setting and participants
This two-arm, blinded randomized controlled trial was completed at the Robotics Lab within the Shirley Ryan AbilityLab (SRALab) rehabilitation hospital. The SRALab is an inpatient and outpatient facility where patients, clinicians, engineers, and scientists meet and conduct research within the clinical treatment areas.
We recruited participants through our Clinical Neuroscience Research Registry and through marketing via websites and available posters/flyers. The Northwestern University Institutional Review Board (Chicago, IL) approved this study on 2 March 2017 (STU00204661). All participants provided written informed consent, according to the Declaration of Helsinki of 1975, as revised in 2013, prior to commencing the study. The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) statements.27,28 This project has been registered at clinicaltrials.gov (ID#NCT03300141).
This standard randomized controlled trial tested a treatment group receiving visual EA feedback and a control group that received an equivalent amount of practice using the same device with veridical feedback (no EA). Based on the results of previous studies,5,9,10 we estimated that 15 participants per group were needed to detect significance.
The target population for the study was chronic stroke participants (at least 8 months postinjury) aged ≥18 years who had a history of a single stroke event. Individuals included in the study had mild-to-moderate upper extremity impairment (as confirmed by an AMFM score of 15–56 out of 66, completed by an occupational therapist) and residual active elbow flexion and extension when their arm was supported against gravity. Individuals were excluded from the study if they had multiple strokes, exhibited bilateral upper extremity paresis, and/or had severe sensory deficits in the affected limb. Participants were also excluded if they had severe spasticity in the arm or hand that prevents movement, measured as a Modified Ashworth score of 4. Individuals with aphasia, which may influence their ability to perform the experiment, were also excluded; this was determined by a score on the National Institutes of Health (NIH) stroke scale item #9 of >1. Individuals with cognitive impairment, determined by the Mini-Mental State Examination score of <23/30, were also excluded from the study. Participants were also excluded if they were currently receiving any other skilled arm rehabilitation, participated in similar robotics interventions previously, or had other neurological/medical issues that affected their safe participation. All participants were assessed by an expert occupational therapist during a preliminary screening session to ensure that they met the inclusion criteria.
Each qualified participant was randomly assigned to one of the two groups: (a) treatment group (or EA group; n = 19), trained with error-augmented visual feedback or (b) control group (n = 19) trained with no visual distortions. We used a block randomization method to ensure that an equal number of participants presenting with mild and moderate stroke symptoms were assigned to each group. Participants were randomized four at a time based on their AMFM scores assessed during the initial screening visit. The blocks were defined with an inclusion range of six AMFM points. Every other participant in the same group of four was assigned to a group such that they balanced that group; otherwise, the assignment was random.
We considered an improvement in the AMFM score as the primary outcome measure for this study. An improvement of ≥5.2 is commonly believed to be clinically meaningful. 29
Apparatus
To implement the study design, we developed an immersive three-dimensional (3D) graphics display system known as LookingGlass. LookingGlass is a stereographic 3D visual display with a semi-silvered mirror that allows viewing of a large visual field and free movements of the hand in front of the user (Figure 1(a)). The LookingGlass was made in-house at the SRALab Robotics Lab. Stereographic liquid crystal shutter glasses synchronized separate left and right eye images with the TV, resulting in the perception of depth. Mobile arm supports attached to an armless, stationary chair assisted the participant in maintaining a partially flexed shoulder position and avoid fatigue during the exercise block (Figure 1(b)). In conjunction with the LookingGlass graphic display, a Leap® (UltraLeap, Mountain View, CA, USA) tracking tool was used, which allowed for a full field of view to track limb movement (Figure 1(c)). An in-house–constructed orthosis was used to assist the Leap® tracking tool if the participants had difficulty obtaining full-digit extension and abduction during the reaching movement.

(a) Schematic of the LookingGlass augmented reality system. (b) Apparatus in the clinical setting with a participant reaching in the augmented reality environment assisted by mobile arm supports and an orthosis (right hand) and (c) The Leap® Motion Controller hand-tracking sensor is positioned parallel to the ground on a rigid support under the hands of the participant.
Protocol
We asked chronic stroke survivors to practice a bimanual transport task in an augmented reality environment for approximately 40 min (excluding setup and breaks for rest), three times a week for 3 weeks. During the treatment sessions, the participants were seated in a stationary chair with both arms (hemiparetic and unaffected) supported by a gravity-balanced orthosis and their arms occluded by the screen (Figure 1(b)). The participant’s hand position was tracked using the Leap® tracking tool and displayed on the augmented display as two blue toy jacks (Figure 2(a)). In the augmented display, the participants controlled the two toy jacks with their left and right hands, respectively. Participants were instructed to move the toy jacks toward the green sphere targets (Figure 2(b)) while simultaneously maintaining level positioning between their two hands to prevent a ball from rolling off a tray (Figure 2(c)).

TrayBall software view. (a) The blue jacks are the position of the hands tracked by the Leap®. (b)The green balls are the targets that need to be reached by the participants (c) while maintaining symmetry of hand position to balance a white ball on a gray tray.
Figure 3 shows the training protocol timeline and the outline of each treatment session. Each treatment visit lasted approximately 75 min. It began with a 20-min setup to position the participant and set up the apparatus, followed by seven 6-min blocks of targeted reaching trials and 2 min of rest. Extra rest was provided as needed such that the participant reported little to no fatigue. Targeted reaching trials involved attempts to reach from a rest position with both arms to one of the 15 target pairs, followed by stopping for at least half a second. Participants had 10 seconds to make this motion, at which point the system provided a cue to return to the starting point and proceeded to the next motion. During the return to the starting point, the ball was frozen and therefore did not require tray balancing. The targets were spaced evenly on an invisible quarter sphere in front of the participant.

(a) Training protocol timeline. Treatment visits to the laboratory lasted 3 weeks with three visits per week, and clinical evaluations occurred at weeks 0, 5, and 12 and (b) Outline of each treatment session. Each visit consisted of seven 6-min blocks (orange), during which participants completed the reaching treatment with either EA feedback applied (treatment group) or veridical feedback (control group) with 2-min rest breaks (white), with an initial and final block included where movement ability was assessed (green) with veridical feedback applied.
If the participants were at least 70% successful in their attempts to reach the targets on any block, the targets were moved 0.05 m away from the rest position for the following blocks. If less than 30% of the attempts were successful, the targets were moved back 0.05 m closer to the rest position. This was performed to probe the participants’ range of motion as well as consistently provide them with an adequate level of challenge, in accordance with the challenging point theory.
Both the control and treatment (i.e. the group that received visual EA training) groups underwent the same amount of practice over 3 weeks of training, with three sessions per week (nine sessions total). Each session lasted approximately 40 min, including rest time. The EA treatment involved visual distortions applied at each time point by measuring the error vector, defined as the difference in position between the participant’s wrists, and visually magnifying the vertical component by a factor “g” of 1.3 on the paretic side.
As our primary outcome, the participants were evaluated by a blinded occupational therapist evaluator with clinical measures up to 1 week prior to treatment (pre), 4–7 days after the end of treatment (post), and at a follow-up performed 7–9 weeks post-treatment. The only exception was one participant whose return to follow-up occurred 3 years later due to researcher oversight.
Outcome metrics
Our primary outcome measure was the AMFM score to evaluate performance-based impairments, with the maximum score being 66 points.29 –31 We recorded the AMFM score at three stages during the experiment. First, we obtained scores during screening and pre-evaluation and then averaged them to establish a (a) baseline value. Then, we recorded the (b) score at postevaluation after the participant completed all nine treatments. Finally, we obtained the (c) follow-up scores 7–9 weeks (approximately 2 months) postevaluation. We then compared the postevaluation and follow-up values with baseline values to reveal whether training resulted in clinical improvement in AMFM scores.
We then assessed participants’ task performance using a Composite Error, a comprehensive analysis of kinematic performance using our technology. Composite Error was the sum of three normalized error metrics. The first metric was the range of motion deficit, which was calculated as follows:
Other tertiary outcome measures collected at pre-, post-, and follow-up evaluation were as follows: (a) Action Research Arm Test (ARAT), a 19-item evaluation of upper extremity functional performance, where a higher score indicates greater movement function (a maximum score of 57); 32 (b) Wolf Motor Function Test (WMFT), a 17-item quantitative measure of upper extremity ability through unilateral and bimanual functional activities, where a higher score indicates greater movement proficiency (a maximum score of 75); 33 (c) Grip strength assessed using a hand-held dynamometer (cutoff for disability: <62 lbs for males and <41 lbs for females); 34 and (d) Box and Blocks Test (BBT) that evaluates unilateral gross motor skills by measuring the number of blocks moved from one side of a box to another within 60 s. 35
Statistical analysis
To test the effect of the visual EA treatment across time, we used a nonparametric generalized linear mixed-effects model with two factors: group (EA treatment and control) and time (pre-, post-, and follow-up evaluation). The threshold for significance was p = 0.05. Nonparametric approaches were employed if chi-square goodness-of-fit revealed that the distributions were non-normal. Analyses were conducted using SPSS software (SPSS Inc. Chicago, IL, USA) as well as MATLAB (MathWorks. Natick, MA, USA).
To determine the sample size needed to reach statistical significance, we performed a simple power analysis of the simple two-arm clinical trial data from a previous study. 10 Based on a power >80%, two-sided type I error of 0.025 (adding to 0.05), and estimated variance of 2.5, 36 we obtained a sample size of 11 participants per group. Considering a 10% participant dropout rate and a safety factor of 1.25, we finally calculated 15 individuals per group, totaling 30 participants in 2 groups.
Results
In this two-arm randomized controlled trial, 69 participants were screened based on inclusion/exclusion criteria (Figure 4) from March 2017 to December 2021. Of those assessed, 27 were excluded for not meeting the inclusion criteria, and 4 declined to participate. Thirty-eight participants were randomized into two groups: (a) 19 participants in the treatment group, where visual distortion (EA) was performed and (b) 19 participants in the control group, with no EA applied (veridical feedback). In both treatment and control groups, three participants each withdrew from the study for personal or unrelated medical reasons. The treatment group included 16 participants who completed post- and follow-up evaluations for clinical analysis and only 9 participants who completed evaluation for kinematic analysis (seven participants’ data were lost due to technical failure). The control group included 15 participants who completed evaluation for clinical analysis and 12 participants who completed evaluation for Composite Error analysis (one participant’s data were lost due to technical failure); one participant was unavailable and lost to follow-up. One participant from the control group was not followed up until 3 years later, but their data were included in all analyses.
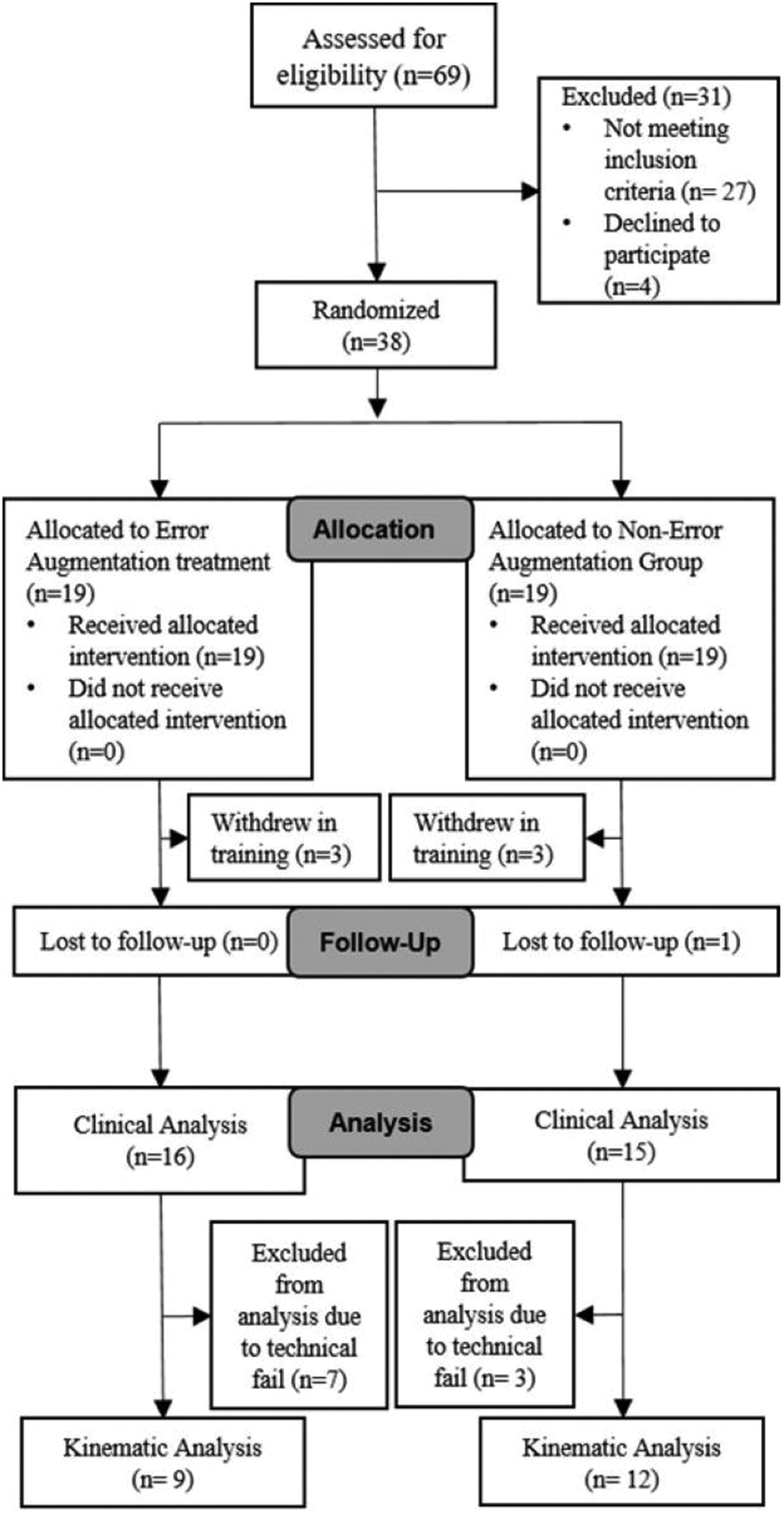
Participant recruitment flow diagram, based on the CONSORT 28 protocols. CONSORT: Consolidated Standards of Reporting Trials.
Demographic information of all participants in the treatment and control groups who completed clinical and kinematic performance analyses is presented in Table S1. Individual information for all participants included in clinical and kinematic data analyses is shown in Table S2. Technical failures, detected only after study completion, led to the loss of kinematic performance data for 10 participants, who were excluded from the analysis (Figure 4; Table S2).
Data distributions for pre-to-post and pre-to-follow-up differences in the control and treatment groups met the assumption of normality, as confirmed by the Shapiro–Wilk test (control: p = 0.6, p = 0.3 and treatment: p = 0.1, p = 0.9, respectively). We observed improvement in the clinical outcome, AMFM score, across time for both groups (p = 0.005, F = 5.612). However, our hypothesis that EA treatment would show superior improvement was not supported (time × group interaction, p = 0.069, F = 2.772). Interestingly, unlike the control group, the average gains observed in the treatment group were significant at postevaluation (Figure 5). We considered this as underpowered evidence suggesting that some individuals may respond better to this treatment (Figure 5). Nevertheless, both groups showed an improvement in AMFM score by <5.2, the minimal clinically important difference. 29 We also did not detect superiority when the participants were divided into mild (AMFM score >24.5) and severe impairment groups, as performed in a previous study 18 (mild: time × group interaction, p = 0.887; severe: p = 0.658).

The primary outcome measure, AMFM score, failed to show the superiority of our customized EA treatment. Associated gains (bottom) were pooled for each group for the major regions of time and shown as thicker lines. Wings indicate variance across each treatment group. For the above graphs, time was aligned with each participant’s initial training session (time zero). AMFM: arm motor section of the Fugl–Meyer.
The secondary metric, Composite Error, revealed a benefit to our training by reducing errors between the two groups (p = 0.031, F = 3.70). However, EA treatment did not show superior improvement (time × group interaction, p = 0.575, F = 0.56), despite the EA group exhibiting the greatest error reduction (Figure 6, lower figure, red bars). We failed to notice any differences after separating the three components of Composite Error. We also investigated the correlation between changes in Composite Error and AMFM (r = 0.2) and the correlation between postevaluation and follow-up evaluation (r = −0.35). This low correlation suggests that this technology may measure different aspects of improvement in ability and function.

Using the same format as in Figure 5, a secondary metric from our technology was our Composite Error, which normalized speed, accuracy, and difficulty level. This was more sensitive to changes but did not show a significant advantage for the treatment group.
We failed to notice any difference in ARAT (p = 0.48), WMFT (p = 0.70), grip strength (p = 0.71), and BBT (p = 0.41) between the two groups from training through the final follow-up visit (Figure S1).
Discussion
In this two-arm blinded randomized controlled trial, the principal objective was to explore and ascertain the efficacy of EA learning integrated with visual distortions concerning motor learning and performance in a bimanual task. The primary outcome was assessing the effectiveness of these interventions using the AMFM score. The AMFM outcome did not show any significant difference between the treatment (visual EA) and control groups. However, all participants showed improvement in a general metric that inspected a composite of three aspects of movement error (range of motion, vertical hand asymmetry, and average movement time), providing slightly clearer results, where the treatment group showed a decrease in Composite Error.
A possible reason for the nonsignificance of this study, compared with our previous study, is that the special treatment (EA) was only applied to vision, rather than performing haptic plus visual EA. 10 This is an important finding, as it suggests that functional motor learning only occurs when the participants’ visual and haptic environments are augmented, not when only the visual environment is augmented. Consequently, the comparatively weaker outcomes observed in this study may be attributed to the fact that the treatment group received solely visual feedback without the concurrent inclusion of haptic feedback.
A study participant did not complete the follow-up until 3 years later. Although we acknowledge the deviation of methodology (follow-up period was 7–9 weeks post-treatment), we opted to include the participant in our analyses to ascertain potential insights gained from longitudinal analysis. It is also worth noting that the number of participants in the study was relatively small, and the study was powered to detect a difference between the groups within a short time (3 weeks of treatment and three visits per week). Hence, the detected effects likely do not reflect the full magnitude that might have been obtained in a full course of treatment (up to 8 weeks). Therefore, it remains unclear whether other implementations of bimanual EA may lead to better outcomes and insights on responsiveness. Nonetheless, training improved average AMFM scores in both groups. Detectable differences from EA treatment (vs. control) may emerge with more sustained treatment regimens or with a more focused cohort of participants who are better capable of engaging in such therapy.
EA holds promise as a rehabilitative technique to be used in conjunction with typical treatment, but the underlying neurological mechanisms that make some individuals respond to EA remain clear. This study highlights the sensory contribution to learning, considering that weaker results occurred when we removed haptic feedback and applied only visual distortion. Although we previously reported that a combination of haptic and visual EA feedback resulted in superior outcomes to controls in arm and hand abilities, 10 similar improvement was observed after providing only visual feedback in the current study; however, we could not distinguish the superiority of EA. The consequent additional sensory sources that are part of haptic EA (cutaneous and proprioceptive) may lead to superior recovery of motor ability.
This study differs from previous EA studies5,9,10 in several ways, including the replacement of free exploration with a tray balancing task, addition of the bimanual component, and removal of robotic forces (haptics) with hands-free. Additionally, in this study, the error is augmented only in the vertical direction for tray balancing. This is because the Z-axis is the most relevant for poststroke patients as it represents the most challenging direction for movement. This choice was made to keep the exercise simple for patients, as overly complex tasks could reduce attention and engagement.
Error-augmented learning treatment may not always be the most effective for all training conditions, and other forms of guidance may provide better results.2 –9 It remains unclear which of these factors might be the reason for the lack of clinical superiority for EA treatment seen in this study.
Our secondary measure, Composite Error, although not validated, is more comprehensive in assessing a broad, balanced metric that does distinguish EA as superior. Although we failed to detect a significant difference in the effect of treatment (time × group), we observed a significant change only for the EA treatment, and the most remarkable changes were observed in the visualization of the Composite Error (Figure 6). It remains unclear whether such metrics, when objective, appropriately normalized, and comprehensive, may offer a more sensitive measure of motor deficit than their clinical counterparts.
Our tertiary metrics, using validated clinical measures, did not detect any significance between the two groups from training through the final follow-up visit. Interestingly, the treatment group demonstrated a wider range of changes in scores from pre-evaluation to follow-up evaluation for ARAT, grip strength, and BBT (Figure S1). This variability within the treatment group suggests potential individualized responses to the intervention, warranting further investigation into personalized treatment approaches. This study represents only our analysis of a priori questions in the context of this completed study. Numerous other clinical metrics and motion-tracking data within this extensive dataset await exploration and investigation, such as smoothness or lateral error. We anticipate that shared data from this study can help the community in performing further mining and resolving novel questions in the future.
In this study, we aimed to explore if error-driven learning with visual distortions resulted in improved motor learning and performance. Although previous studies have used robotics, we restricted the EA treatment to be only visual, using an augmented reality display. Although this visual EA training does not show comparable results to haptically implemented EA in upper extremity rehabilitation, it offers a simple, inexpensive, and robot-free alternative in the clinic for chronic stroke patients that leads to improvement in function. Expanding the scope of analysis through longer-term interventions involving a larger participant pool holds the potential to provide a more nuanced understanding and differentiation of this specialized form of EA treatment.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251361115 - Supplemental material for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605251361115 for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial by Courtney Celian, Teresa Puzzi, Martina Verardi, Erica Olavarria, Federica Porta, Alessandra Pedrocchi and James L Patton in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251361115 - Supplemental material for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial
Supplemental material, sj-pdf-2-imr-10.1177_03000605251361115 for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial by Courtney Celian, Teresa Puzzi, Martina Verardi, Erica Olavarria, Federica Porta, Alessandra Pedrocchi and James L Patton in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251361115 - Supplemental material for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial
Supplemental material, sj-pdf-3-imr-10.1177_03000605251361115 for Does visual error augmentation offer advantages during bimanual therapy in individuals poststroke? A randomized controlled trial by Courtney Celian, Teresa Puzzi, Martina Verardi, Erica Olavarria, Federica Porta, Alessandra Pedrocchi and James L Patton in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Eyad Hajissa, Felix Huang, Robert Kenyon, Francois Kade, Kelly Thielbar, Yazan Abdel Majeed, and Piper Hansen for their earlier help on this project. We also thank the Robotics Lab group at the Shirley Ryan AbilityLab for their insights and comments during the development of this project.
Author contributions
CC, MV, FP, and JLP collectively played pivotal roles in shaping the study’s conception and design. AP was involved in conducting protocol refinement and defining outcome metrics. CC, TP, MV, FP, AP, and JLP actively participated in data interpretation. The acquisition of data and their subsequent analysis involved the collaborative efforts of CC, MV, FP, and JLP. The manuscript drafting and critical revisions were conducted with significant contributions from CC, TP, MV, EO, FP, ALGP, and JLP, ensuring the inclusion of substantial intellectual content. All authors have provided their unanimous final approval for the version intended for publication.
Data availability statement
Declaration of conflicting interests
AP is the co-founding partner of two startups: Agade srl and AllyArm srl.
AP has conducted seminars for Novartis and has collaborated in various research and educational projects funded by international entities (EU funds- FP7 program; Horizon, HE; NIH; ESA), national public institutions (MIUR PRIN, INAIL, Lombardy Region, ASI), private institutions (Cariplo Foundation, Telethon Foundation, Valduce Foundation, FORST Foundation), and companies (Kayser Italia srl; Generali Wellion; Tecnobody srl; EMAC srl; Merz Therapeutics; AllyArm srl).
CC, TP, MV, EO, FP, and JLP declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the National Institute on Disability, Independent Living, and Rehabilitation Research, RERC 90REGE0005.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



