Abstract
Objective
Endovascular treatment has proven to be effective in improving the outcome of patients with large-vessel stroke. However, more than half of the patients experience poor outcomes despite successful recanalization, which was defined as futile recanalization. The present study aimed to identify the relationship between multiple inflammatory markers (before and after endovascular treatment) and futile recanalization.
Methods
Between November 2019 and September 2022, we retrospectively enrolled 456 patients according to the inclusion and exclusion criteria. Inflammatory variables, including neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, lymphocyte-to-monocyte ratio, systemic immune-inflammation index, systemic inflammatory response index, systemic coagulation-inflammation index, and white blood cell count to mean platelet volume ratio, were collected.
Results
Among the cohort of 456 patients, it was observed that 271 (59.4%) patients experienced futile recanalization. Univariate analysis revealed that patients with futile recanalization had higher neutrophil-to-lymphocyte ratio and systemic inflammatory response index but lower lymphocyte-to-monocyte ratio and systemic coagulation-inflammation index at admission compared with those without (p < 0.05). Additionally, at postoperation, patients with futile recanalization had higher neutrophil-to-lymphocyte ratio, systemic inflammatory response index, and systemic immune-inflammation index but lower lymphocyte-to-monocyte ratio and systemic coagulation-inflammation index compared with those without (p < 0.05). However, there were no significant differences in admission platelet-to-lymphocyte ratio, admission systemic immune-inflammation index, admission white blood cell count to mean platelet volume ratio, postoperation platelet-to-lymphocyte ratio, and postoperation white blood cell count to mean platelet volume ratio between the futile recanalization group and nonfutile recanalization group (p > 0.05). Furthermore, multivariate logistic regression analysis showed that only admission systemic inflammatory response index, postoperation neutrophil-to-lymphocyte ratio, postoperation systemic inflammatory response index, and postoperation systemic immune-inflammation index remained significantly correlated with futile recanalization (p < 0.05). Receiver operating characteristic curves revealed that postoperation neutrophil-to-lymphocyte ratio was the most predictive marker for futile recanalization (area under the curve = 0.623, 95% confidence interval: 0.572–0.675).
Conclusion
Admission systemic inflammatory response index, postoperation neutrophil-to-lymphocyte ratio, postoperation systemic inflammatory response index, and postoperation systemic immune-inflammation index were independently associated with futile recanalization, and postoperation neutrophil-to-lymphocyte ratio may have the highest ability in predicting futile recanalization in patients who underwent endovascular treatment.
Introduction
Stroke is one of the major causes of disability and death worldwide. 1 Since 2018, endovascular treatment (EVT) has become the standard treatment strategy for patients with large-vessel acute ischemic stroke (AIS) because the time window for EVT has been extended to 16–24 h.2,3 Recent studies have demonstrated that the proportion of patients with near-complete (modified Thrombolysis in Cerebral Infarction (mTICI): 2b) or complete reperfusion (mTICI: 3) is up to 80%.4,5 However, >60% of the patients experienced poor outcomes (modified Rankin scale (mRS) score ≥3) despite successful recanalization, 4 which was defined as futile recanalization (FR).
Multiple predictors of FR have been identified, including age, stroke severity, laboratory values (glucose, cystatin C, and creatinine), and cerebral small vessel disease burden.6–8 Numerous studies have shown that neuroinflammation plays an important role in the pathology of ischemic stroke.9,10 Neutrophil-to-lymphocyte ratio (NLR), platelet (PLT)-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR) have been reported as three inflammatory biomarkers and can provide additional information in the risk assessment of AIS.11,12 Furthermore, in recent years, some novel biomarkers related to inflammation have been discovered and could serve as outstanding predictors in patients with ischemic stroke. Two novel inflammatory markers, systemic immune-inflammation index (SII) and systemic inflammatory response index (SIRI), reflect the immune and inflammatory states and have been reported to be associated with the severity and atrial fibrillation in AIS patients.13,14 In addition, systemic coagulation-inflammation index (SCI), calculated as PLT count × fibrinogen level/white blood cell count, reflects coagulopathic and inflammatory pathways and serves as a good predictor of short-term mortality in patients with aortic dissection. 15 Moreover, as a combination of white blood cell count and mean PLT volume ratio, white blood cell count to mean platelet volume ratio (WMR) was demonstrated to be more stable and comprehensive regarding the pathophysiology of ischemic stroke.16,17 Moreover, several studies have demonstrated that inflammation markers, including NLR and PLR, play an important role in the pathogenesis and outcomes of patients receiving reperfusion therapy.18–20 Nevertheless, it is not clear which inflammation indicator best predicts FR. More importantly, the predictive ability of admission blood glucose and postoperative blood glucose for prognosis differs in patients with EVT.21,22 Similarly, the predictive abilities of inflammation markers before and after EVT may be different.
The present study investigated the relationship between multiple inflammatory markers (before and after EVT) and FR in patients with large-vessel stroke and compared the predictive abilities of these inflammatory markers, aiming to determine the best inflammatory predictor of FR.
Methods
Patients
Between November 2019 and September 2022, consecutive patients admitted to the Stroke Unit of First Affiliated Hospital of Nanchang University were retrospectively assessed. We conducted this study in accordance with the Helsinki Declaration of 1975, as revised in 2024. All procedures were approved by the Ethics Committee of the First Affiliated Hospital of Nanchang University. In addition, all patient details have been deidentified. The reporting of this study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 23 Informed written consent was not required for this retrospective study. Patients were recruited if they met the following inclusion criteria: (a) age ≥18 years; (b) AIS with large-vessel occlusion in anterior circulation or posterior circulation; (c) underwent endovascular treatment with or without intravenous thrombolysis; and (d) achieved successful recanalization (mTICI: 2b or 3). Patients were excluded if they met the following exclusion criteria: (a) underwent standard medical treatment alone; (b) failed to achieve successful reperfusion (mTICI: <2b); (c) incomplete baseline or follow-up data; (d) pretreatment-modified Rankin scale (mRS) score >2; and (5) systemic inflammatory diseases or recent infections.
Definition
FR was defined as a poor outcome (mRS score: 3–6) at 3 months despite successful revascularization, and favorable recanalization (non-FR) was defined as functional independence (mRS score: 0–2) at 3 months with successful revascularization. 24 The stroke etiology was determined at discharge according to the classification of the Trial of Org 10172 in Acute Stroke Treatment (TOAST) and classified as “large-artery atherosclerosis,” “cardioembolism,” “undetermined causes,” and “other causes.” 25 Symptomatic hemorrhagic transformation (HT) was defined as hemorrhage detected on computed tomography (CT) with an increase in the National Institute of Health Stroke Scale (NIHSS) score (≥4 points). 26
Data collection
Demographic characteristics and clinical information, including age, sex, body mass index (BMI), history of hypertension, diabetes and atrial fibrillation, admission systolic blood pressure (SBP) and diastolic blood pressure (DBP), baseline NIHSS scores, onset-to-admission time, puncture-to-reperfusion time (PRT), and onset-to-reperfusion time (ORT) were recorded.
Laboratory data were divided into admission data and postoperative fasting data within 24 h after EVT. White blood cell (WBC), PLT, mean platelet volume (MPV), lymphocyte count, monocyte count, neutrophil count, and fibrinogen level were measured at admission before initiating any specific stroke treatment. Similarly, these parameters were collected within 24 h after EVT. Furthermore, the NLR, PLR, and LMR were calculated. SIRI, SII, WMR, and SCI were calculated using the following formulas, respectively: SIRI (109/L) = neutrophil count (109/L) × monocyte count (109/L)/lymphocyte count (109/L); 13 SII (109/L) = neutrophil count (109/L) × PLT count (109/L)/lymphocyte count (109/L); 13 WMR = WBC count (109/L)/MPV (fL); 17 and SCI = PLT count (109/L) × fibrinogen (g/L)/WBC count (109/L). 15
Imaging procedures and evaluation
All patients underwent standardized imaging evaluation with noncontrast CT and CT perfusion (CTP). Alberta stroke program early computed tomography score (ASPECTS) value was evaluated via noncontrast CT by two trained neurologists who were blinded to patients’ information. CTP images were acquired on the Siemens Somatom Force (Erlangen, Germany), and all scans were obtained with 40 mL of nonionic iodinated contrast (Iomeron, iomeprol, 400 mg iodine/mL; Patheon Italia S.P.A., Ferentino, Frosinone, Lazio, Italy). All CTP datasets were postprocessed using the software package (Syngo via CT Neuro Perfusion VB40). The thresholds for infarcted and hypoperfused tissue were defined as relative cerebral blood flow (rCBF) <30% and time to maximum of the tissue residue function (TMax) >6 s, respectively.
Statistical analysis
Continuous variables were presented as mean (±SD) or median (interquartile range (IQR)), and categorical variables were presented as frequency (percentages). Analysis of baseline characteristics between the FR group and non-FR group was performed using Student’s t-test or Mann–Whitney U test for continuous variables and chi-square test or Fisher exact test for categorical variables. Furthermore, potential variables with raw values (p < 0.05) were included in multivariate logistic regression analysis to identify predictors of FR. Moreover, we determined whether the associations between multiple inflammatory markers and FR were nonlinear by assessing the fit of models with restricted cubic splines using the likelihood ratio test. In addition, receiver operating characteristic (ROC) curve analysis was used to compare the predictive abilities of multiple inflammatory markers, and a comparison of area under the curve (AUC) values between inflammatory markers was conducted through the DeLong test.
Statistical significance was defined as p < 0.05. All statistical analyses were performed using SPSS (version 26.0) and R software package (version 4.2.1).
Results
Between November 2019 and September 2022, 669 patients who met the inclusion criteria were screened. Of those, 213 patients were excluded, 47 because of unsuccessful reperfusion (mTICI: <2b) and 166 because of missing complete baseline or follow-up data. Thus, 456 patients were enrolled in the analysis, with 104 (22.8%) patients with a TICI grade of 2b, and 352 (77.2%) with a TICI grade of 3. The 3-month mRS scores of 456 patients were as follows: 0 (14.0%), 1 (17.1%), 2 (9.4%), 3 (9.6%), 4 (11.2%), 5 (5.7%), and 6 (32.9%). Finally, 271 patients were included in the FR group and 185 in the non-FR group.
The baseline demographic characteristics, clinical data, neuroimaging information, and stroke etiology between the FR and non-FR groups are listed in Table 1. There were no significant differences in sex, BMI, history of hypertension, diabetes, admission SBP, admission DBP, onset-to-admission time, ORT, and affected vessel territory (p > 0.05). However, patients with FR were older and had a higher frequency of atrial fibrillation, higher baseline NIHSS scores, higher PRT, lower ASPECT scores, lower perfusion mismatch ratio, and higher incidence of HT complications, especially symptomatic HT than those with non-FR (p < 0.05). According to the TOAST classification, stroke etiology in the FR group was determined as large-artery atherosclerosis in 104 patients (38.4%), cardioembolic in 125 patients (46.1%), undetermined causes in 36 patients (13.3%), and other causes in 6 patients (2.2%). Conversely, stroke etiology in the non-FR group was determined as large-artery atherosclerosis in 89 patients (48.1%), cardioembolic in 57 patients (30.8%), undetermined causes in 22 patients (11.9%), and other causes in 17 patients (9.2%). The distribution of stroke etiology differed between the FR and non-FR groups (p < 0.001).
Comparison of baseline demographic, clinical, neuroimaging, outcome, and stroke etiology data between FR and non-FR groups.

ASPECTS: Alberta stroke program early computed tomography score; BMI: body mass index; DBP: diastolic blood pressure; FR: futile recanalization; HT: hemorrhagic transformation; IQR: interquartile range; NIHSS: National Institute of Health Stroke Scale; ORT: onset-to-reperfusion time; PRT: puncture-to-reperfusion time; SBP: systolic blood pressure.
p < 0.05.
The baseline laboratory data between the FR and non-FR groups are listed in Table 2. Patients with FR were admitted with lower PLT count, lymphocyte count, LMR, and SCI (p < 0.05) than those without. In contrast, patients with FR were admitted with higher MPV, neutrophil count, NLR, and SIRI (p < 0.05) than those with non-FR. Regarding postoperative laboratory data, the WBC count, MPV, neutrophil count, NLR, SIRI, and SII in the FR group were higher than those in the non-FR group (p < 0.05). In contrast, the PLT count, lymphocyte count, LMR, and SCI in the FR group were lower than those in the non-FR group (p < 0.05).
Comparison of baseline laboratory data between FR and non-FR groups.

FR: futile recanalization; IQR: interquartile range; LMR: lymphocyte-to-monocyte ratio; MPV: mean platelet volume; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; PLT: platelet; SCI: systemic coagulation-inflammation index; SII: systemic immune-inflammation index; SIRI: systemic inflammatory response index; WBC: white blood cell; WMR: white blood cell count to mean platelet volume ratio.
p < 0.05.
As shown in Table 3, multivariate logistic regression analysis revealed that admission NLR, admission LMR, admission SCI, postoperation LMR, and postoperation SCI did not show significant association with FR after adjusting for age, atrial fibrillation, baseline NIHSS scores, PRT, ASPECTS classification, perfusion mismatch ratio, HT complications, and stroke etiology (p > 0.05). However, admission SIRI (odds ratio (OR): 1.311; 95% confidence interval (CI): 1.022–1.252; p = 0.018), postoperation NLR (OR: 1.042; 95% CI: 1.009–1.076; p = 0.013), postoperation SIRI (OR: 1.041; 95% CI: 1.000–1.083; p = 0.048), and postoperation SII (OR: 1.000; 95% CI: 1.000–1.000; p = 0.011) remained significantly correlated with FR after adjusting for variables with p < 0.05.
Multivariate logistic regression analysis of inflammatory variables for futile recanalization.

CI: confidence interval; LMR: lymphocyte-to-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; OR: odds ratio; SCI: systemic coagulation-inflammation index; SII: systemic immune-inflammation index; SIRI: systemic inflammatory response index.
Adjusted for age, atrial fibrillation, baseline NIHSS scores, puncture-to-reperfusion time, ASPECTS classification, perfusion mismatch, hemorrhagic brain complications, and stroke etiology.
p < 0.05.
The ROC curve was used to compare the predictive abilities of multiple inflammatory markers for FR (Figure 1). We observed that the AUC values of admission NLR, admission SIRI, postoperation NLR, postoperation SIRI, and postoperation SII to discriminate FR were 0.585 (95% CI: 0.532–0.638), 0.589 (95% CI: 0.537–0.642), 0.623 (95% CI: 0.572–0.675), 0.592 (95% CI: 0.540–0.644), and 0.571 (95% CI: 0.519–0.624), respectively, indicating that postoperation NLR was the strongest predictor of FR. Comparing the areas under these correlated ROC curves through the DeLong test, our results found a significant difference in AUC values between postoperation NLR and postoperation SII (p = 0.0003). Furthermore, the probability of an outcome based on continuous admission SIRI, postoperation NLR, postoperation SIRI, and postoperation SII levels showed significant noncubic associations for FR (Figure 2).
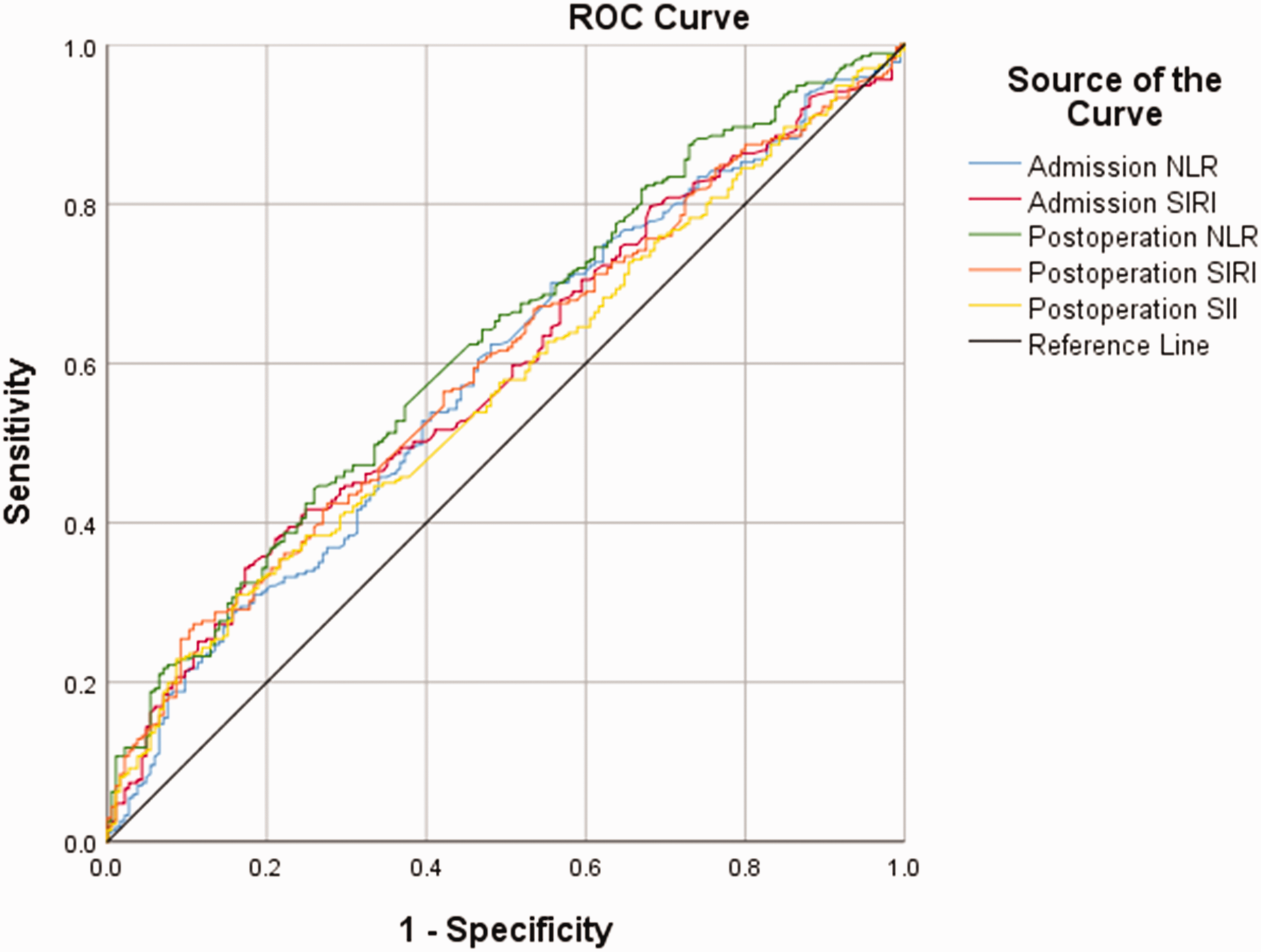
Receiver operating characteristic curves for multiple inflammatory markers to predict futile recanalization. NLR: neutrophil-to-lymphocyte ratio; SIRI: systemic inflammatory response index; SII: systemic immune-inflammation index.

Restricted cubic splines showing noncubic associations for futile recanalization based on continuous admission SIRI (a), postoperation NLR (b), postoperation SIRI (c), and postoperation SII (d).
Discussion
The main innovation of this study lies in the comprehensive analysis of the correlation between inflammatory indicators (NLR, PLR, LMR, SCI, SIRI, SII, and WMR at admission and postoperation) and FR in patients with large-vessel stroke. Our results revealed that patients with FR versus those without had higher levels of NLR and SIRI but lower levels of LMR and SCI at admission. Meanwhile, patients with FR were observed to have higher levels of NLR, SIRI, and SII but lower levels of LMR and SCI at postoperation compared with those without. Furthermore, the findings in our study suggested that FR is associated with admission SIRI, postoperative NLR, postoperative SIRI, and postoperative SII but not with other inflammatory markers. It is worth noting that postoperative NLR can predict FR with better performance than other inflammatory indicators.
Although several randomized controlled trials have confirmed the effectiveness of mechanical thrombectomy in patients with large-vessel stroke, more than half of the patients experience FR and cannot achieve a good functional prognosis.2,3,27 The pathophysiological mechanisms of FR are complex and unclear. No-reflow (a phenomenon referred to as impaired capillary reperfusion despite large-vessel recanalization), early arterial reocclusion, HT, ischemia–reperfusion injury, and poor collateral circulation may play essential roles in FR. 28 Numerous studies have demonstrated that inflammation was associated with poor outcomes in AIS patients undergoing EVT.29–32 However, the precise mechanism by which inflammation participates in FR remains unclear. The recanalization processes in the penumbral region may enhance the inflammatory cascade, leading to leukocyte aggregation and microvascular thrombus formation, thereby causing no-reflow.31,33 Conversely, following the onset of reperfusion, the extensive inflammatory response may release proteolytic enzymes, consequently resulting in blood–brain barrier disruption, thereby causing HT and ischemia–reperfusion injury.31,34
Classic inflammatory factors play different roles in the inflammatory process, such as neutrophils mainly acting on initiating inflammation, lymphocytes exerting a regulatory impact on inflammation, and monocytes contributing to inflammation and thrombosis by interacting with PLTs and endothelial cells. 31 In recent years, various novel inflammatory indicators (NLR, PLR, LMR, SCI, SIRI, SII, and WMR) have emerged mainly for the following two reasons: (a) the levels of individual blood cells such as neutrophils may be biased because the measurements are affected by different conditions such as hydration and dehydration 31 and (b) incorporating two or more markers into a consolidated index could encompass the intricacies of the immune system, inflammation, and coagulation, such as NLR incorporating neutrophils and lymphocytes, PLR incorporating PLTs and lymphocytes, LMR incorporating lymphocytes and monocytes, SCI combining coagulation with inflammation, SIRI combining the immune system with inflammation, SII combining the immune system with inflammation, and WMR combining coagulation with inflammation.12–17 Therefore, new inflammatory markers may reflect the inflammatory state more reliably and comprehensively compared with individual inflammatory cells.
Recently, two studies have explored the association between inflammatory factors and FR.31,35 Guan et al. 31 demonstrated that admission LMR acted as a prognostic marker for predicting FR in patients with AIS undergoing EVT. Meanwhile, Lattanzi et al. 35 revealed that admission SIRI increased the risk of poor outcome at 3 months despite successful recanalization. In our present study, we found that patients with FR versus those without FR had higher levels of SIRI but lower levels of LMR at admission, which were consistent with the findings of previous studies.31,35 However, few studies have systematically analyzed the correlation between various inflammatory indicators, including classical indicators and emerging markers, and FR, comparing the impact of admission and postoperative inflammation on FR. This is the first study to summarize the correlation between existing reported inflammatory markers and FR and compare the predictive abilities of preoperative and postoperative inflammation. Finally, we revealed that postoperative NLR had the strongest predictive ability for FR.
Although our study presents interesting novel findings, there are some limitations. First, our study suggested that the predictive powers of inflammatory factors for FR were modest. Although postoperative NLR was identified as the strongest predictor of FR in our study, its AUC value was only 0.623, limiting its clinical utility. Previous studies have underscored the complexity of FR and reported the diverse factors associated with FR.36,37 Therefore, our future studies should combine inflammatory markers with other factors to predict the severity risk of FR, improving accuracy. Second, this is a single-center retrospective study of patients with a single ethnic background, which may lead to a possibility of patient selection bias, statistical analysis bias, and limited generalizability. However, the detection of laboratory parameters using the same method in a stroke center results in better uniformity. Third, we did not measure the levels of inflammatory markers at other time points. Future studies should further recruit multicenter and multiethnic patients to identify the correlation between dynamic changes of inflammatory markers and FR.
Conclusion
Admission SIRI, postoperation NLR, postoperation SIRI, and postoperation SII are associated with FR in patients with EVT, and postoperation NLR may be a better indicator to predict FR than other inflammatory indicators.
Footnotes
Acknowledgment
None.
Authors’ contributions
Concept and design: ZX, LG, XL, XL; acquisition of data: XL, ZX, LG, YC, DC, HZ, XX, YG, QY, CZ, LZ; analysis or interpretation of data: QH, JZ, KZ, PF, DH, XL, JL; drafting of the manuscript: XL, ZX, LG; critical revision of the manuscript for important intellectual content: XL; and funding: JL, DH. All authors have read and approved the final manuscript.
Data availability statement
Data are available upon reasonable request from the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval
This study was approved by the Ethics Committee of the First Affiliated Hospital of Nanchang University (IIT 2023 Clinical Ethic Review No. 364).
Funding
This study was supported by the National Natural Science Foundation of China (No. 82101405), the Natural Science Foundation of Jiangxi Province (No.20212BAB216023), the Double Thousand Talents Program of Jiangxi province (No. jxsq2019101021), and the Educational Science Foundation of Jiangxi Province (No. GJJ2200136).
