Abstract
Objectives
To investigate whether intracytoplasmic sperm injection can achieve better embryonic and clinical outcomes compared with in vitro fertilization in women with non-male factor infertility and poor ovarian response.
Methods
This retrospective cohort study analyzed data from a single academically affiliated fertility clinic. The participants included couples with non-male factor infertility and poor ovarian response (normal male spermatozoa) who underwent oocyte retrieval with an oocyte yield of ≤4 between January 2016 and December 2022.
Results
A total of 766 oocyte retrieval cycles were included, with 318 cycles in the in vitro fertilization group and 448 in the intracytoplasmic sperm injection group. The in vitro fertilization group showed a higher fertilization rate (89.31% vs. 83.06%, p = 0.0002), while the intracytoplasmic sperm injection group had higher two-pronuclear fertilization rates and higher quality of embryos (76.90% vs. 67.44%, p < 0.001 and 54.86% vs. 43.94%, p < 0.001, respectively). However, no significant differences were observed in the clinical pregnancy rates, ongoing pregnancy rates, live birth rates, miscarriage rates, embryo implantation rates, and cumulative live birth rates between the two groups. In addition, multivariate logistic regression analysis, adjusted for potential confounding factors, showed no significant differences in the clinical pregnancy and live birth rates between the two groups.
Conclusion
In women with non-male factor infertility and poor ovarian response, intracytoplasmic sperm injection provides no significant improvements in embryonic or clinical outcomes compared with in vitro fertilization.
Keywords
Introduction
Assisted reproductive technology (ART) is a widely used technique, with significant advancements over the past few decades. In 1978, the first successful in vitro fertilization (IVF) therapy was performed. 1 Intracytoplasmic sperm injection (ICSI) treatment was established in 1992 to boost success rates, transforming the treatment method used for male factor infertility couples, allowing couples with severe male factor infertility to become pregnant. 2 ICSI has been increasingly used in patients with borderline or even normal symptoms over the past 20 years; 3 however, the Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology have noted that there is insufficient evidence to support the routine use of ICSI in patients with non-male factor infertility.3–4 In addition to male factor infertility, ICSI is widely used in patients with previous IVF failure, advanced age, low number of oocytes obtained, preimplantation genetic testing, and unexplained infertility; nonetheless, the routine use of ICSI for these purposes warrants further research. 5
Existing research indicates that ICSI is marginally less effective than IVF in other etiologies (e.g. endometriosis, tubal factor infertility, and unexplained infertility) and is significantly more effective in severe male factor infertility. Unlike conventional IVF, ICSI skips the sperm selection process during fertilization, increasing the chance of transferring genetic abnormalities to the progeny. Potential male or female factor infertility as well as other medical aspects of the ICSI technique for infertile couples are linked to an elevated risk. The incidences of chromosomal abnormalities during pregnancy,6–7 autism, 8 intellectual disability, 8 and birth defects9–10 associated with ICSI greatly increase with ICSI than with conventional IVF.
In patients with non-male factor infertility and poor ovarian response (POR), there are several plausible reasons for using ICSI. First, when the oocyte yield is low, ICSI can prevent the risk of fertilization failure that may occur with conventional IVF, thereby improving fertilization rates. 11 The extensive use of ICSI is linked to patients’ fear of total fertilization failure (TFF), and the therapeutic delusion propagated by mass media among patients raises societal pressure and makes it more challenging for physicians to discuss TFF with patients. 12 Second, in patients with advanced maternal age, ICSI may enhance the embryo quality because oocyte quality in older women is often compromised, and ICSI can directly inject sperms into the oocyte to increase the likelihood of fertilization. 13 Additionally, in patients with only a single oocyte retrieved, ICSI can maximize the use of the limited oocytes and improve the chance of obtaining transferable embryos. 14 Moreover, studies comparing the clinical outcomes of ICSI and IVF on sibling oocytes have shown that although the two methods have their own advantages and disadvantages in terms of fertilization rates and embryo quality, there are no significant differences in clinical pregnancy outcomes. 15 However, the financial burden for many couples receiving reproductive therapy increases because ICSI is far more expensive than conventional IVF. It remains debatable whether ICSI should always be the preferred insemination technique for patients with non-male factor infertility. A statistical analysis of the cumulative live birth rates (CLBRs) and cost-effectiveness ratio of IVF and ICSI for non-male factor infertility in Europe in 2014–2015 (total 2 years) suggests that ICSI has a high economic cost but does not improve the CLBR in patients with non-male factor infertility. 16
It remains debatable whether the indications for using ICSI for women with POR should be broadened. When the ovary is stimulated to produce additional follicles and/or oocytes, POR to stimulation occurs when less than four follicles and/or oocytes develop or are obtained. 17 Addressing this issue is a significant challenge because POR may occur with ART in 5%–35% of cases. 18 In this context, it is necessary to unravel the impact of ICSI on the clinical outcomes of POR patients and further clarify the scope of application of ICSI treatment.
To determine if the use of ICSI in POR patients improves laboratory and clinical outcomes, we investigated the fertilization outcomes, embryonic developmental potential, and assisted conception outcomes of POR patients who have undergone IVF or ICSI. The correct choice of the insemination technique is crucial, and this study provides a clinical basis for evaluating the choice of the insemination method for POR patients and clarifying the indications for ICSI application.
Materials and methods
Study population
We evaluated the files of all patients (#1367) from couples who had undergone oocyte retrieval with an oocyte yield of ≤4 between January 2016 and December 2022, totaling 766 POR cases. The data for this retrospective analysis were obtained from the Center for Reproductive Medicine, First Hospital of Jilin University (Changchun, China), and the demographic and clinical data were retrieved from the Reproductive Center’s electronic medical records. ART cycles are defined as fertility therapies that deal with (manipulate) oocytes, sperm, or embryos for pregnancy. The exclusion criteria were as follows: (a) abnormalities in the reproductive function of the partner, retrograde ejaculation disease, azoospermia, or the need for epididymal or testicular puncture; (b) polycystic ovary syndrome or infertility due to uterine factor; (c) unconsciousness or inability to express subjective symptoms of discomfort, accompanied with severe depression, anxiety symptoms, or mental illness; (d) acute infections of the genital or urinary system, sexually transmitted diseases, chromosomal abnormalities, or immunological infertility; (e) teratogenic radiation exposure; (f) use of donor oocytes/sperm, short-term insemination, or rescue ICSI; or (g) cancelation of oocyte retrieval during the ovulation cycle for personal reasons. Moreover, we used the ovarian response as a key criterion; females with fewer than four retrieved oocytes were considered. Figure 1 shows the patient inclusion process. The research program was approved by the institutional review board.

Flowchart of patient selection and inclusion criteria.
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. In accordance with ethical research practices, all patient details in this study have been de-identified to ensure that individuals cannot be recognized in any way. Any identifiers that could potentially disclose the identity of the patients, such as names, dates of birth, medical record numbers, and other personally identifiable information, have been removed from the datasets and reports used for this research. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 19
The center collected complete data on cycles in which frozen embryos were used for transfer prior to oocyte retrieval or cycles that were canceled (ovarian stimulation or monitoring was initiated; however, the cycle did not continue with oocyte retrieval). All patients underwent rigorous medical examinations and diagnosis, and detailed cycle information such as patient characteristics, clinical features, fertilization, embryo outcome, and pregnancy outcome were recorded. Only the initial oocyte retrieval data during the research period were included for patients who underwent repeat oocyte retrieval, and additional oocyte retrieval cycles between individual patients were not linked.
Selection and grouping of fertilization methods
The decision to perform IVF or ICSI on the retrieved oocytes is influenced by the couple’s preferences as well as the physician’s clinical approach. Couples are informed about the benefits and drawbacks of both treatments before making this decision. Two groups were established based on the method of fertilization: IVF non-male factor group (IVF group, n = 318) and ICSI non-male factor group (ICSI group, n = 448).
Stimulation protocol
The entire IVF process usually consists of five stages: (a) controlled ovarian hyperstimulation (COH); (b) oocyte retrieval; (c) fertilization; (d) embryo transfer; and (e) pregnancy test. 20 Ovarian reserve assessment was performed 1–3 months after the natural menstrual cycle prior to the start of stimulation. Depending on the patient’s ovarian function tests and other indications, the patient was treated with one of two COH regimens: gonadotropin (Gn)-releasing hormone (GnRH) antagonist or GnRH agonist. The initial dose was ≥150 IU. Ovulation was induced with human chorionic gonadotropin (hCG) when there were three or more follicles larger than 18 mm in diameter. Transvaginal ultrasound-guided oocyte retrieval was performed 34–36 h after administering hCG injection.
IVF or ICSI was used to fertilize the collected oocytes, and the retrieved oocytes were fertilized 70–72 h later to assess embryo morphology and select embryos ready for transfer or freezing. On day 3, oocytes were judged to be of high-quality if they had 6–9 cleaved globules, 5% nucleus-free fragments, and no notable morphological defects. Transfer of 1–2 embryos or blastocysts in a fresh cycle was performed on day 3 or 5. After the embryo transfer, the remaining embryos were frozen. Patients with unprepared endometrium, as indicated by increased progesterone levels (i.e. >1.5 ng/mL), thin endometrium, intrauterine fluid, and endometrial polyps early on the day of hCG administration, underwent selective freezing. Endometrial preparation was performed using natural cycles, hormone replacement therapy (HRT), or downregulated HRT cycles, followed by thawing of frozen embryos and their subsequent transfer. The serum hCG level was measured 14 days after the transfer. If the conception was successful, the luteal phase assistance was continued until 10 weeks of gestation. Following up from gestational testing until live birth, other pertinent obstetric and neonatal outcomes were examined. Measured parameters included birth weight, length, sex, gestational age, congenital anomalies, and 1-year developmental outcomes.
Basal circulating anti-müllerian hormone (AMH), follicle-stimulating hormone (FSH), luteinizing hormone (LH), and estradiol (E2) levels were measured on day 2 or 3 of the menstrual cycle. The results of the sex hormone assay were obtained from the Reproductive Center’s electronic medical records.
Fertilization, embryo transfer, and pregnancy assessment
Variables in the analysis included patient sociodemographic characteristics, personal health history, female baseline hormone levels, AMH levels, cycle type, embryo transfer status, and pregnancy outcome. Miscarriage, ectopic pregnancy, or stillbirth were all considered adverse pregnancy events. Fertilization, embryo transfer, and pregnancy were the focus of the study.
To assess the actual reproductive prospects of ICSI and IVF, it is necessary to conduct a clinical comparison of the two insemination methods using laboratory data. The fertilization rate is defined as the ratio of the number of fertilized oocytes to the number of metaphase II (MII) oocytes utilized for IVF/ICSI. The two-pronuclear (2PN) fertilization rate was calculated as the ratio of 2PN fertilized oocytes to IVF/ICS-treated MII oocytes. The 2PN cleavage rate was defined as the ratio of cleavage stage embryos developed from 2PN zygotes on day 3 to the total number of 2PN zygotes. High-quality embryos were defined as 2–5 oocytes with <20% fragmentation on day 2 and 6–9 oocytes with <20% fragmentation on day 3. The high-quality embryo rate was defined as the ratio of high-quality embryos on day 3 to the total number of embryos during the oogenesis stage. Blastocyst formation rate was defined as the ratio of blastocysts that had been cultured for 3 days and were still being cultured to the number of embryos. The available blastocyst rate was defined as the ratio of available blastocysts to the number of blastocysts obtained from day 3 embryos that were cultured until day 5 or 6.
The implantation rate was calculated as the number of gestational sacs divided by the number of transferred embryos. The pregnancy rate was the percentage of positive pregnancies per initiated cycle in a fresh embryo transfer cycle. A positive pregnancy was defined as plasma hCG level >5 IU/L on day 14 after embryo transfer. The biochemical pregnancy rate was defined as the proportion of biochemical pregnancies among positive pregnancies. Biochemical pregnancy signifies an early spontaneous abortion following a positive pregnancy test. The clinical pregnancy rate was calculated as the proportion of clinical pregnancies among all fresh embryo transfer cycles. Clinical pregnancy was defined as fetal heartbeat at 6–8 weeks on ultrasound. The miscarriage rate was defined as the percentage of miscarriages in clinical pregnancies. The ongoing pregnancy rate was defined as the proportion of ongoing pregnancies in fresh embryo transfer cycles. Ongoing pregnancy was defined as a pregnancy lasting >12 gestational weeks. The ongoing pregnancy rate was calculated as the ongoing pregnancy ratio of fresh embryo transfer cycles. The live birth rate was defined as the ratio of live births per initiated cycle to fresh embryo transfer cycles. CLBR was defined as the total number of births (including fresh embryo transfers and subsequent transfer ART cycles with frozen/frozen–thawed embryos) in which at least one live birth was obtained in addition to the ongoing pregnancy cycle in an ART cycle of oocyte retrieval.
Statistical analysis
The distribution and clinical characteristics of patients undergoing traditional IVF and ICSI were compared. All statistical data were analyzed using GraphPad Prism 9 software. Continuous variables were expressed as mean ± standard deviation or median (interquartile range) values and compared between the two groups using independent samples Student’s t-test or Mann–Whitney U test according to the distribution characteristics of the variables. Categorical variables were expressed as numbers (%) and tested using the chi-square test or Fisher exact test. A two-sided significance level of p < 0.05 was considered to indicate a statistically significant difference.
Baseline information for this study included age (continuous), body mass index (BMI) (continuous), ethnicity, education, female infertility diagnosis, infertility duration in years (continuous), total motile sperm count (TMC), COH regimen (antagonist, prolonged agonist, extra-long duration agonist, or other), starting dose of Gn (continuous), total antral follicle count (AFC) (continuous), AMH (continuous), FSH (continuous), LH (continuous), and E2 (continuous). To handle the missing values indicated in Table 1, we applied median mode imputation. To illustrate the relationship between ICSI and reproductive outcomes in patients with POR, we compared the laboratory outcomes, pregnancy outcomes, and live births between the two groups. The number of retained oocytes (continuous), fertilization-related indicators (continuous), and embryo-related indicators (continuous) constituted the laboratory outcomes. We evaluated the rates of implantation, biochemical pregnancy, clinical pregnancy, ongoing pregnancy, miscarriage, delivery, CLBR, and fetal birth abnormalities to analyze pregnancy outcomes. As continuous data, basic information on live births was tracked and evaluated, including the number and sex of fetuses delivered, height, weight, birth outcomes, and the number of anomalies during the first year of life. A multivariate logistic regression model was employed to compare the effects of ICSI and conventional IVF on clinical pregnancy and live birth, after adjusting for potential confounding factors.
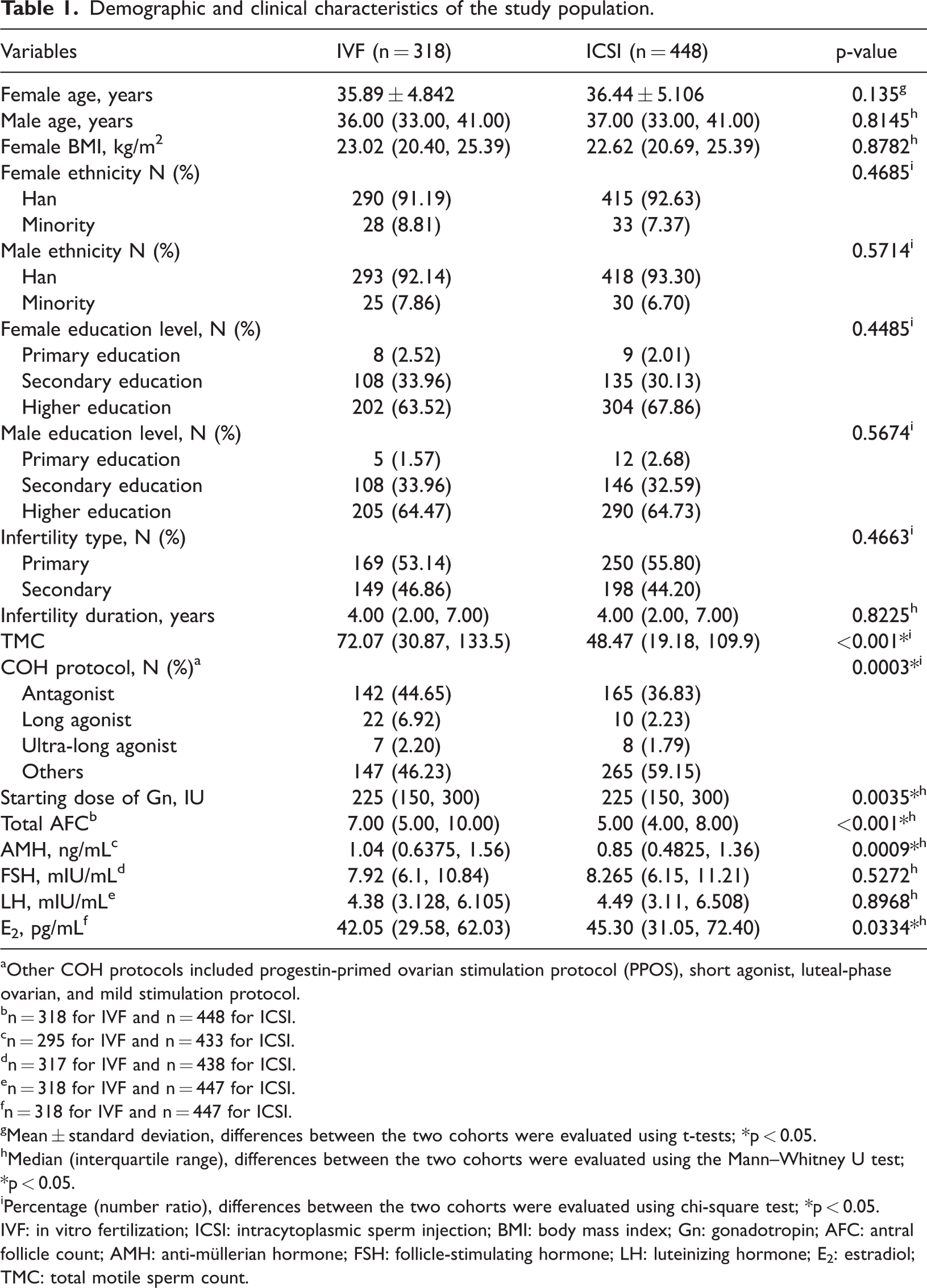
Demographic and clinical characteristics of the study population.
Other COH protocols included progestin-primed ovarian stimulation protocol (PPOS), short agonist, luteal-phase ovarian, and mild stimulation protocol.
n = 318 for IVF and n = 448 for ICSI.
n = 295 for IVF and n = 433 for ICSI.
n = 317 for IVF and n = 438 for ICSI.
n = 318 for IVF and n = 447 for ICSI.
n = 318 for IVF and n = 447 for ICSI.
Mean ± standard deviation, differences between the two cohorts were evaluated using t-tests; *p < 0.05.
Median (interquartile range), differences between the two cohorts were evaluated using the Mann–Whitney U test; *p < 0.05.
Percentage (number ratio), differences between the two cohorts were evaluated using chi-square test; *p < 0.05.
IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection; BMI: body mass index; Gn: gonadotropin; AFC: antral follicle count; AMH: anti-müllerian hormone; FSH: follicle-stimulating hormone; LH: luteinizing hormone; E2: estradiol; TMC: total motile sperm count.
Results
Basic information
This study involved 766 oocyte retrieval cycles, with 318 cycles in the IVF group and 448 in the ICSI group. Table 1 summarizes the characteristics of the study population. The TMC was significantly higher in the IVF group than in the ICSI group (72.07 (30.87, 133.5) vs. 48.47 (19.18, 109.9); p < 0.001). The COH protocol and starting dose of Gn differed significantly between the two groups (p < 0.01). The AFC was higher in the IVF group than in the ICSI group (7.00 (5.00, 10.00) vs. 5.00 (4.00, 8.00); p < 0.001). Patients in the IVF group had greater basal circulating levels of AMH and lower levels of E2 (1.04 (0.6375, 1.56) vs. 0.85 (0.4825, 1.36); p = 0.0009 and 42.05 (29.58, 62.03) vs. 45.30 (31.05, 72.40); p = 0.0334, respectively). Age, BMI, ethnicity, education level, cause of infertility, duration of infertility in years, and baseline FSH and LH levels did not differ between the two groups. A small portion of baseline data were missing, and the reasons for nonparticipation were not systematically recorded.
Laboratory outcomes
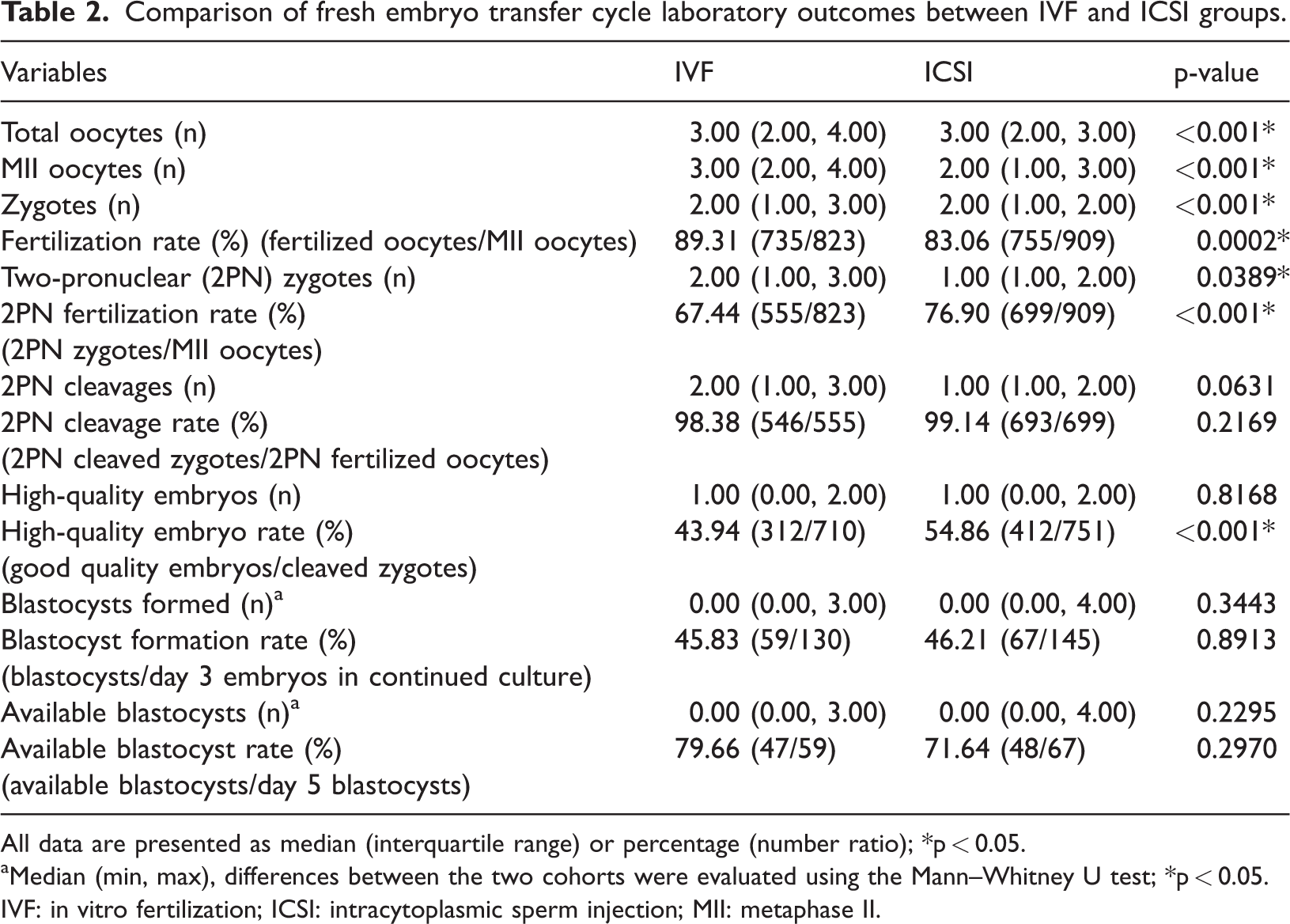
Laboratory outcomes are summarized in Table 2. The total number of acquired and fertilized oocytes differed significantly between the two groups (p < 0.001). The number of MII oocytes was higher in the IVF group than in the ICSI group (3.00 (2.00, 4.00) vs. 2.00 (1.00, 3.00); p < 0.001). Fertilization rates were higher in the IVF group than in the ICSI group (89.31% vs. 83.06%; p = 0.0002). The 2PN fertilization rate and high-quality embryo rate were lower in the IVF group than in the ICSI group (67.44% vs. 76.90%; p < 0.001 and 43.94% vs. 54.86%; p < 0.001, respectively). However, the 2PN cleavage counts/rate, blastocyst formation counts/rate, and available blastocyst counts/rate were not significantly different between the two groups.
Comparison of fresh embryo transfer cycle laboratory outcomes between IVF and ICSI groups.
All data are presented as median (interquartile range) or percentage (number ratio); *p < 0.05.
Median (min, max), differences between the two cohorts were evaluated using the Mann–Whitney U test; *p < 0.05.
IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection; MII: metaphase II.
Clinical outcomes
By the follow-up cutoff date, the IVF and ICSI groups had completed 86 and 80 fresh embryo transfer cycles, respectively. Table 3 shows a comparison of the pregnancy outcomes of the two groups. The number of embryos transferred was significantly different between the groups. Double embryos were transferred in 65.12% of the IVF group participants and single embryos were transferred in 53.75% of the ICSI group participants. The embryo stage at transfer was not significantly different between the two groups. The implantation rate (25.35% vs. 32.48%, p = 0.2064), pregnancy rate (45.35% vs. 50.00%, p = 0.5488), biochemical pregnancy rate (17.95% vs. 17.50%, p = 0.0522), clinical pregnancy rate (37.21% vs. 41.25%, p = 0.5941), miscarriage rate (34.38% vs. 45.45%, p = 0.362), ongoing pregnancy rate (24.42% vs. 22.20%, p = 0.7708), and live birth rate (26.74% vs. 22.50%, p = 0.5264) did not show intergroup differences. We also analyzed the CLBR. In the IVF group, there were eight ovarian stimulation cycles in which a clinical pregnancy was achieved but no delivery was made before the cutoff date. The corresponding number of cycles in the ICSI group was 23. After excluding null cases (cycles with no live birth outcome), the CLBR in the IVF group was higher than that in the ICSI group (19.03% vs. 14.82%, p = 0.1299); however, the difference was not statistically significant.
Comparison of fresh embryo transfer cycle pregnancy outcomes between IVF and ICSI groups.

Data are presented as percentage (number ratio); ∗p < 0.05.
IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection.
Table 4 shows the number of live births in the IVF and ICSI groups. The IVF and ICSI groups had 70 and 68 live births, respectively. A comparison of the groups revealed no significant differences in any of the variables except the birth weights of the neonates.
Comparison of live birth fetuses between the IVF and ICSI groups.

Mean ± standard deviation; all data are presented as median (interquartile range) or percentage (number ratio); ∗p < 0.05.
IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection.
Multivariate logistic regression for effect comparison
After adjusting for potential confounding factors (Table 5), we compared the effects of ICSI and conventional IVF on clinical pregnancy and live birth rates. The crude odds ratios (ORs) showed no evidence of an association between insemination method and clinical pregnancy or live birth (OR = 1.027, 95% confidence interval (CI): 0.554–1.902; OR = 0.788, 95% CI: 0.530–1.172, p > 0.05). All ORs remained statistically nonsignificant after adjusting for confounding factors, indicating no significant differences in clinical pregnancy and live birth rates between ICSI and IVF.
Multivariate logistic regression analysis of the impact of ICSI versus IVF on clinical pregnancy and live birth.

Factors for adjustment: Female age, Female BMI, and Infertility duration.
Factors for adjustment: Female age, Female BMI, Infertility duration, COH protocol, Starting dose of Gn, Total AFC, AMH, and E2.
Factors for adjustment: Female age, Female BMI, Infertility duration, COH protocol, Starting dose of Gn, Total AFC, AMH, E2, the number embryo transferred, and the stage of the embryo transferred.
IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection; OR: odds ratio; CI: confidence interval.
Discussion
The effect of the insemination method on oocyte fertilization and embryo quality in separate IVF or ICSI cycles is difficult to compare because the differences between infertile couples may affect oocyte fertilization and embryo development. 21 Thus, this study aimed to compare ART outcomes following conventional IVF and ICSI in couples with normal sperm and POR.
In this study, we used IVF or ICSI for insemination in patients with POR and non-male factor infertility and compared the laboratory and clinical outcomes as well as neonatal-related indicators of delivery between the two modalities. The fertilization rate was higher in the IVF group than in the ICSI group; however, the 2PN fertilization and high-quality embryo rates were lower in the ICSI group. The variations in other laboratory outcomes between the two groups were not statistically significant. Embryo transfer differed between the two groups, with the IVF group transferring a higher number of double embryos and the ICSI group transferring a higher number of single embryos. There was no statistically significant difference between the two groups in terms of clinical pregnancy rate, ongoing pregnancy rate, live birth rate, miscarriage rate, embryo implantation rate, or CLBR. Except for fetal weight, there was no statistically significant difference between the two groups in the number of fetuses delivered, fetal sex, or length.
Consistent with our findings, some studies have shown that in non-male factor infertility, there are no significant differences in clinical pregnancy and live birth rates between ICSI and IVF. 11 Additionally, research has indicated that in poor responders with only a single oocyte, ICSI does not result in higher live birth rates than IVF. 14 This supports our conclusion that in patients with non-male factor infertility and POR, ICSI does not improve clinical outcomes. Similarly, it has been pointed out in relevant literature that in patients with non-male factor infertility and POR who have fewer than four oocytes, ICSI does not significantly improve fertilization rates and embryo quality. 12 This aligns with our results, where despite the higher fertilization rate in the IVF group and the greater high-quality embryo rate in the ICSI group, no significant differences were observed in clinical pregnancy and live birth rates. Furthermore, in patients with advanced maternal age, 13 ICSI does not increase the live birth rate. This is consistent with our findings that in patients with non-male factor infertility and POR, IVF and ICSI have similar clinical pregnancy and live birth rates. Finally, consistent with our study, previous studies comparing the clinical outcomes of ICSI and IVF on sibling oocytes have shown that although the two methods have their own advantages and disadvantages in terms of fertilization rates and embryo quality, there are no significant differences in the clinical pregnancy outcomes. 15
Data published by the European Association for In Vitro Fertilization Monitoring showed that in European countries, the proportion of ICSI in total ART cycles increased steadily from 1999 to 2006 and has remained stable since then at a level close to 70%. Data from the United States show a similar tendency, with the usage of ICSI growing from 36.4% in 1996 to 76.2% in 2012, with patients with non-male factor infertility experiencing the greatest increase. 22 ICSI is widely used in reproductive medicine, and both the American Society of Reproductive Medicine and the European Society of Human Reproduction and Embryology guidelines recommend routine ICSI for all oocytes and reserve ICSI for patients with male factor infertility or a history of TFF. 23 Based on data from the Society of Assisted Reproductive Technology Clinical Outcome Reporting System, live birth rates are higher with IVF than with ICSI for non-male factor infertility. A meta-analysis of four randomized controlled trials revealed that ICSI did not outperform IVF in terms of fertilization rates, clinical pregnancy rates, or clotting rates. 24 A meta-analysis of 22 cohort studies showed that IVF 24 had higher rates of clotting and live births. The results of the largest cohort research to date, comprising approximately 1.4 million ART cycles, are consistent with the findings of this meta-analysis. The study used the Human Fertilization and Embryology Authority’s extensive database to assess reproductive outcomes after IVF and ICSI in POR patients with normal sperm parameters.
The mechanism of oocyte fertilization is complex and primarily related to oocyte quality, sperm–oocyte conjugation process, culture environment, and in vitro manipulation. In the population, the likelihood of IVF fertilization failure is 5%–15%. 25 Because of the smaller number of oocytes recovered and the decreased fertilization potential in POR women, it has been proposed that ICSI be used as the preferred method of insemination in women with POR to prevent overly low fertilization rates or fertilization failure. It is noteworthy that in this study, a higher number of MII oocytes were acquired in the IVF group than in the ICSI group, the overall fertilization rate of IVF was higher than that of ICSI in patients with POR, and the 2PN fertilization rate was lower in the IVF group. This could be attributed to immature oocyte meiotic development and late fertilization events following normal insemination. In the study, ICSI produced high-quality embryos from a small number of MII oocytes more efficiently than IVF. For patients, the stimulation of one ovary to produce more high-quality embryos boosts the chances of pregnancy and live birth. This trial, however, demonstrated no effect of ICSI in patients with non-male factor infertility and POR in terms of clinical pregnancy or live birth rates. In another retrospective multicenter study conducted in 15 tertiary centers in Europe, which involved approximately 5000 patients who underwent IVF or ICSI for non-male factor infertility, IVF and ICSI showed similar live birth rates in poor-, suboptimal-, normal-, and high-response patients. This shows that the number of received oocytes is not a determining factor in the choice of insemination technique in the case of non-male factors. 26 Thus, enhancing the oocyte quality is the key to fertilization and clinical success in women with POR, and POR in women should not be regarded as an indication of ICSI. 27
When performing IVF among POR patients, we must convey the safety of ICSI to the patient. A large cohort study comparing birth defects among more than 300,000 natural-born infants and over 6000 infants born via ART found that the rate of birth defects in the IVF group was comparable to that of the general population, whereas the risk of birth defects was higher in the ICSI group. 9 There was a higher incidence of multiple pregnancies in the IVF group in this study and correspondingly higher incidence of low birth weight. In this study, due to the small sample size, there were only two newborns with congenital disorders; of these, one died 6 months after birth, and both infants died prematurely. There were no differences in the birth abnormalities between the two groups of infants.
The additional costs and financial burden that would be incurred by ICSI treatment are unclear, especially when no male factor is present and executing ICSI treatment does not contribute to the pregnancy outcome.
Our study included 766 oocyte retrieval cycles, encompassing diverse laboratory and clinical outcomes, which ensures the reliability of the results. However, there are certain limitations. As a single-center study, the results may not be generalizable to all populations or settings. The data were collected retrospectively, which may have introduced potential biases. Additionally, the study did not assess the long-term outcomes, such as the health and development of the offspring.
In conclusion, choosing ICSI for fertilization in patients with advanced non-male factor infertility and POR does not improve fertilization; does not prevent fertilization failure; and does not improve embryo quality, embryonic developmental potential, or pregnancy outcome. ICSI is not routinely recommended for women with non-male factor infertility and POR, and IVF remains the preferred method of insemination for these patients.
Footnotes
Acknowledgements
The authors thank the patients enrolled in this study.
Author contributions
All authors have read and approved the submitted version of the manuscript. QHW and YQ were involved in data collection and interpretation and manuscript generation. HJY performed statistical analysis. DRL reviewed the manuscript and edited it.
Data availability statement
The data will be made freely available upon request to the corresponding author.
Declaration of conflicting interests
All authors report no conflicts of interest with the generation or publication of this manuscript.
Funding
This work was supported by the Science and Technology Department of Jilin Province, China (NO. 20210101354JC).
