Abstract
Objective
This study aimed to assess the feasibility and safety of using the ultrasound-guided axillary vein puncture technique to implant the totally implantable venous access device without a tunnel.
Methods
This study retrospectively analyzed data from 703 patients who underwent totally implantable venous access device implantation in the chest wall between January 2022 and March 2023. Ultimately, 685 patients underwent tunnelless totally implantable venous access device implantation via ultrasound-guided axillary vein puncture. We collected data regarding the patients’ age, sex, body weight, body mass index, axillary vein diameter, depth of the axillary vein from the body surface, surgical success rate, operation duration, and postoperative follow-up.
Results
Of the 703 patients who signed the consent forms for totally implantable venous access device implantation, 685 were included in the axillary vein puncture group. The follow-up period ended on 31 August 2023; the mean follow-up duration was 281.59 days. The following complications were observed: catheter retrograde to the internal jugular vein in seven patients, catheter occlusion in three patients, catheter-related infections in two patients, catheter-related vein thrombosis in two patients, and skin infection around the infusion port in one patient. The overall immediate and long-term complication rate was 2.13%.
Conclusion
The tunnelless totally implantable venous access device implantation technique via ultrasound-guided axillary vein puncture offers a highly effective, safe, and aesthetically pleasing alternative for chest wall totally implantable venous access device implantation.
Introduction
The totally implantable venous access device (TIVAD) was invented more than 40 years ago. 1 TIVADs are widely used in chemotherapy for oncology patients and for intravenous nutritional therapy; they provide higher safety and reliability than tunneled-cuffed silicone centrally inserted catheters and peripherally inserted central catheters. 2 Currently, TIVADs are mainly implanted in the chest wall in most hospitals in China, and the main venous accesses of choice are the internal jugular vein (IJV), subclavian vein, and axillary vein. Puncture of the subclavian vein based on anatomical landmarks may lead to additional complications, such as hemothorax, pneumothorax, pinch-off syndrome, and even catheter rupture, which can be severe and fatal.3–5 Therefore, few physicians prefer the subclavian vein as the puncture target. Currently, the IJV remains the preferred choice of most Chinese physicians. This is because the neck needs to be incised to create a tunnel for guiding the catheter to the chest wall; the complexity of the procedure is significantly high; pain is obvious; and most of the patients have postoperative adverse experiences such as pain in swallowing and neck movement, subcutaneous tunnel inflammation, 6 and the appearance of the catheter shape in the subcutaneous area after the procedure (Figure 1(a)). Some physicians puncture the axillary or subclavian vein, and the conventional method involves making an incision of approximately 2.5 cm below the puncture point and creating a short tunnel between the puncture point and the surgical incision before TIVAD implantation. However, our team adopted the tunnelless TIVAD implantation technique. After the success of ultrasound-guided puncture of the axillary vein and indwelling guidewire, physicians made a transverse incision of 2.5 cm on the medial side of the guidewire, directly creating a skin pocket and implanting TIVAD more easily. This technique simplifies the surgical steps by eliminating the need for tunnel creation. Currently, the technique is mature, and we have reviewed and analyzed previous medical records to summarize and share our surgical experience. A preprint version of this study has been published on ResearchSquare. 7

Photographs after implantation of the TIVAD. (a) Postoperative TIVAD implanted via the IJV and (b) postoperative TIVAD implanted via the axillary vein without tunnel. TIVAD: totally implantable venous access device; IJV: internal jugular vein.
Methods
All data were obtained from Shenzhen Cancer Hospital. This study was conducted in accordance with the principles of the Declaration of Helsinki (as revised in 2013). All patients signed an informed consent for undergoing surgery. The study was approved by the Ethical Review Committee of the Cancer Hospital Chinese Academy of Medical Sciences Shenzhen Center with approval NO.: KYLX2023-45. All patient details have been de-identified to protect confidentiality, ensuring that no individuals can be identified directly or indirectly through the presented data or case descriptions. In total, 703 patients who underwent chest wall TIVAD implantation from January 2022 to March 2023 were enrolled in the study. Among them, 290 were males and 413 were females. The study collected information on the patients’ sex, age, weight, height, body mass index (BMI), diagnosis, depth of axillary vein, axillary vein diameter, intraoperative problems, surgery results, surgery time, success rate, catheter access to the vascular pathway, and port implantation site. Additionally, the study reviewed the hospital information system and conducted follow-up calls to register complications related to TIVAD implantation, mainly focusing on wound recovery, catheter-related skin infection, catheter infection, catheter-related venous thrombosis, catheter rupture, pinch-off syndrome, catheter fracture, and reversal of the TIVAD. The follow-up period ranged from the date of TIVAD implantation to the date of TIVAD removal or to the cutoff follow-up end date, i.e. 31 August 2023.
This was a retrospective study with data sourced from a single center, and the different groups were not randomized; this may have introduced selection bias. Statistical analyses were conducted using the Statistical Package for Social Sciences for Windows, Version 22.0 (IBM Corp., Armonk, NY, released in 2016). The data were presented as follows: continuous variables were reported as mean ± standard deviation, and categorical variables were presented as numbers with percentages. For independent normally distributed samples, the t-test was used; for paired samples, the paired samples t-test was employed; and for multiple independent samples that did not necessarily follow a normal distribution, the Kruskal–Wallis test was applied. A P-value <0.05 was considered to indicate statistical significance. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
Surgical procedure
Before the surgery, the axillary vein was explored via ultrasound to record its diameter and depth (Figure 2(a)). In the absence of special circumstances, the right axillary vein approach was preferred; however, if there were relative contraindications such as right breast cancer, right lung cancer, or the need for radiotherapy in the right chest wall or right lung, the left axillary vein approach was chosen. All patients were placed in the supine position, and the skin was disinfected. A maximized sterile barrier cavernous towel was applied, followed by local infusion anesthesia with 2% lidocaine (Figure 3(a)). The superficial ultrasound probe was coated with a coupling agent and then aseptically protected with a sterile luminal sleeve (Figure 3(b)).

Ultrasound image of the axillary vein. (a) Ultrasound image of a patient showing an axillary vein depth of 32.5 mm and axillary vein diameter of 9.6 mm; (b) ultrasonographic findings in a patient with poor axillary vein filling during calm breathing suggesting an axillary vein diameter of only 2.1 mm and (c) the axillary vein diameter was increased to 8.6 mm after increasing the intrathoracic pressure by exhaling forcefully and holding the breath.
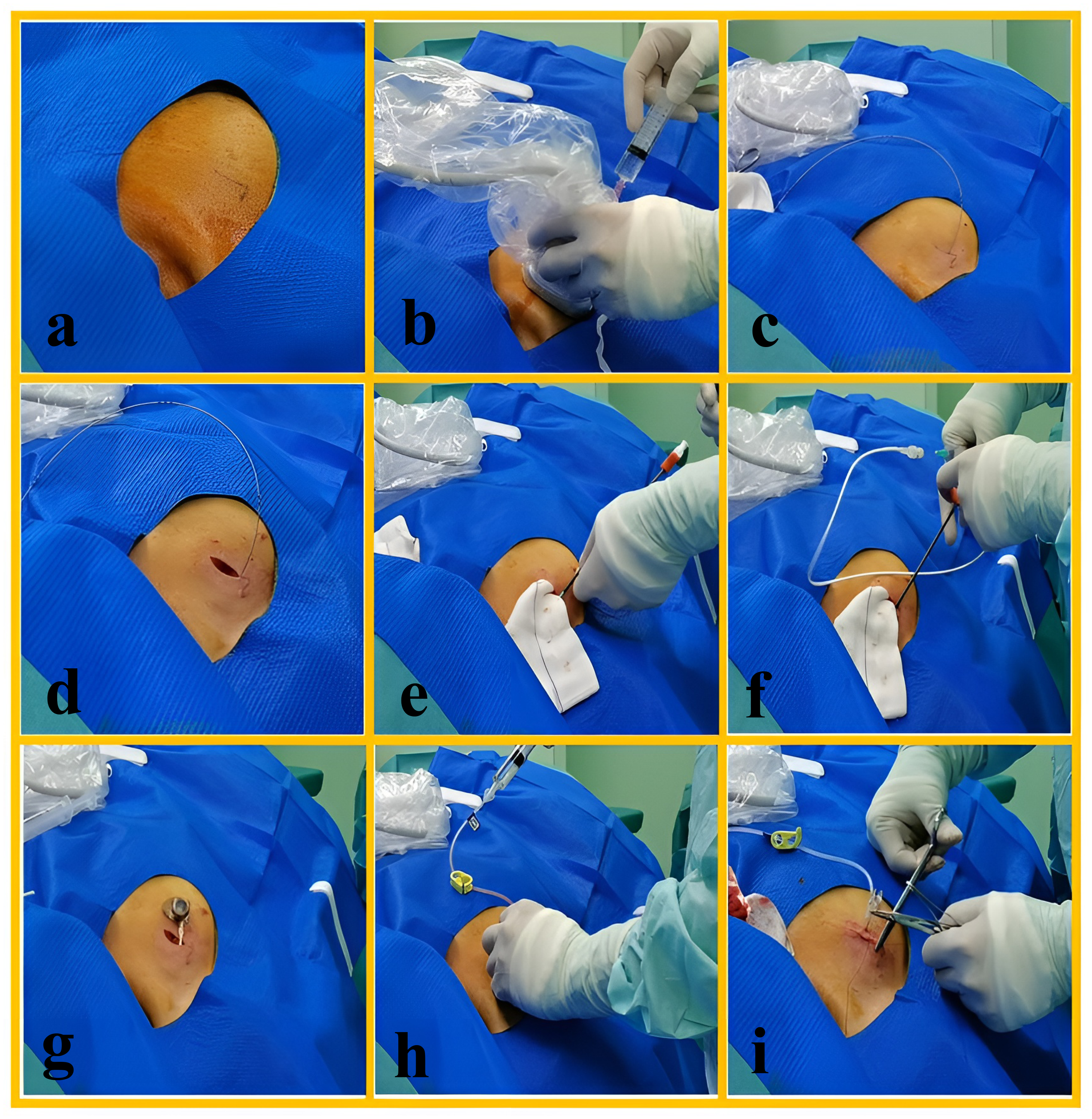
Surgical procedure of tunnelless TIVAD implantation via ultrasound-guided axillary vein puncture.
The puncture was performed using saline as the ultrasound medium by spraying a small amount on the skin over the axillary vein and puncturing the axillary vein under real-time ultrasound guidance (Figure 3(b)). Once the puncture needle broke through the vein and red blood was withdrawn, the needle and syringe were disconnected, and a guidewire was inserted through the axillary vein, subclavian vein, and brachiocephalic vein, ultimately reaching the superior vena cava (Figure 3(c)). After successful guidewire retention, ultrasonographic exploration of the neck was performed to confirm that the guidewire was directed downward toward the brachiocephalic vein and excluded from the IJV. If the tip of the guidewire was located in the IJV, it could be adjusted under direct ultrasonographic visualization.
A lateral incision of 2.5 cm was made on the medial side of the guidewire as a pocket incision, and the guidewire was adequately freed after incision (Figure 3(d)). The skin was separated subcutaneously downward to form a suitably sized skin pocket, and gauze tamponade was applied to facilitate hemostasis (Figure 3(d) and (e)). The tearable catheter sheath was gently placed along the guidewire; the sheath core and guidewire were then removed. The catheter was advanced 20 cm along the sheath, and the tearable sheath was withdrawn (Figure 3(f)).
Using the electrocardiographic positioning technique, if the peak of the P wave was observed, the catheter was withdrawn approximately 2 cm from the peak of the P wave. If no peak of the P wave was observed, it was recommended to withdraw the catheter to 10–12 cm and reintroduce it while checking for changes in the P wave until the peak of the P wave was observed. The same operation was then performed using the method described above. The catheter was trimmed, and the port was connected (Figure 3(g)). The port was buried into the skin pocket, and a noncoring needle was inserted into the port (Figure 3(h)). After confirming satisfactory blood drawing, the wound was closed with 4-0 absorbable sutures placed intermittently and sealed with tissue glue (Figure 3(h) and (i)).
Postoperative chest radiography was routinely performed to locate the tip of the catheter. No postoperative treatments such as wound disinfection, suture removal, or prophylactic antibiotics were required.
Results
Demographics and epidemiology
The total number of patients was 703. All patients were clearly diagnosed with malignant tumors pathologically before surgery and required intravenous chemotherapy. The three most common diagnoses were malignant breast tumors (28.7%), malignant colorectal tumors (24.6%), and malignant lung tumors (13.9%), with a cumulative ratio of these top three diseases accounting for 67.3% (Table 1) of all cases. There were 290 males and 413 females, with a mean age of 53.91 ± 12.43 years, mean weight of 60.29 ± 10.47 kg, mean BMI of 22.98 ± 3.43, mean axillary vein diameter of 8.31 ± 1.71 mm, mean axillary vein depth of 26.31 ± 4.51 mm, and mean operation time of 14.04 ± 3.43 min (Table 2).
Diagnosis of patients with TIVAD implantation between January 2022 and March 2023.
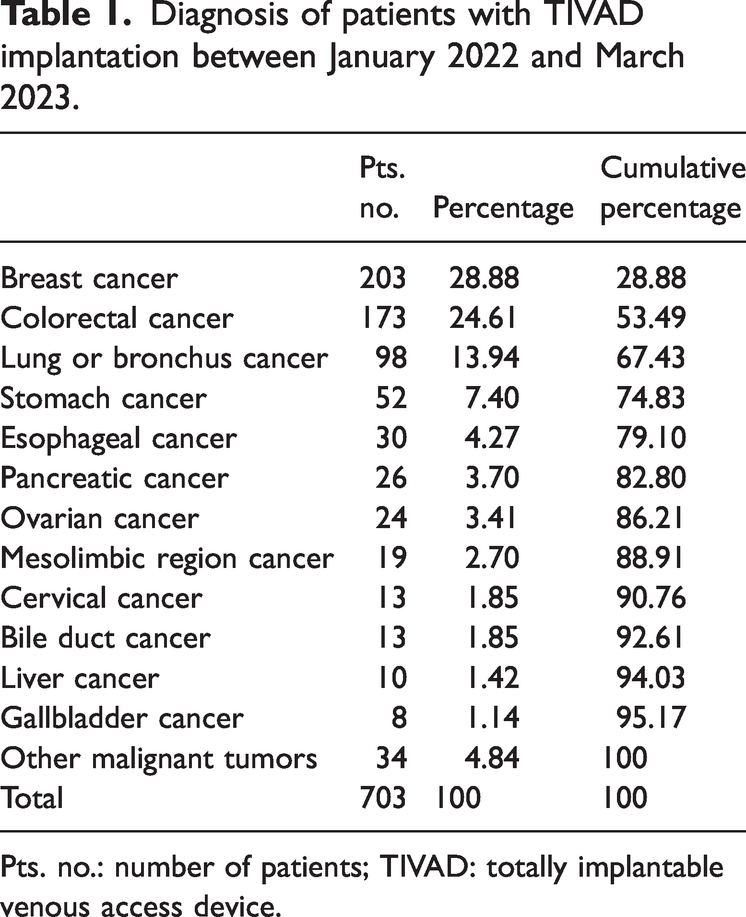
Pts. no.: number of patients; TIVAD: totally implantable venous access device.
Demographic characteristics of different venous access subgroups between January 2022 and March 2023.

IJV: internal jugular vein; BMI: body mass index.
Statistical Package for Social Sciences’ t-test was utilized to analyze the data between right and left axillary veins.
P-values marked with bold indicate statistically significant results.
Flow of patients
After 703 patients were initially evaluated using preoperative ultrasound measurement of axillary vein diameter and depth, it was found that axillary vein puncture was not feasible in 7 patients, primarily due to extreme obesity and inadequate axillary vein filling. The remaining 696 patients were deemed suitable for axillary vein puncture, resulting in an enrollment rate of 99% (Figure 4).

Chart flow of patients who underwent TIVAD implantation at the Cancer Hospital Chinese Academy of Medical Sciences Shenzhen Center between January 2022 to March 2023. TIVAD: totally implantable venous access device.
Of the 696 patients enrolled for axillary vein puncture, 685 underwent successful axillary vein puncture, while the procedure failed in 11 patients. Ultimately, 557 patients underwent tunnelless TIVAD implantation through the right axillary vein, and 128 patients underwent implantation through the left axillary vein, yielding an implantation success rate of 98.42% (Figure 4).
Axillary vein puncture failed in 11 patients (1.58%), including 7 patients with axillary artery injury. In four patients, access to the axillary vein with a guidewire was challenging, and ultrasound-guided adjustments were unsuccessful. For these 11 patients, tunneled TIVAD implantation was successfully performed by switching to IJV puncture (Figure 4).
Demographics of different subgroups
The baseline data of all patients were grouped according to the different venous accesses (Table 2), and statistical analysis was conducted using the Statistical Package for Social Sciences (SPSS) software. The mean age of the patients for whom the right axillary vein approach was used was 54.39 ± 12.63 years; their mean BMI was 22.75 ± 3.33, and the mean operation time was 13.57 ± 2.43 min. The mean age of the patients for whom the left axillary vein approach was used was 51.57 ± 11.37 years; their mean BMI was 23.56 ± 3.36, and the mean operation time was 14.28 ± 2.55 min.
SPSS’s t-test was utilized to analyze the data regarding age, BMI, and operation time between the two groups (right and left axillary veins). The results indicated P-values of 0.021, 0.014, and 0.003, respectively. These statistical results suggested significant differences in age, BMI, and operation time between patients for whom right and left axillary vein access was used. This was a retrospective study; therefore, the grouping of the left versus right axillary veins was nonrandomized. Thus, the observed differences between the two groups could be attributable to selection bias. However, the disparity in the procedural times between the left and right axillary approaches primarily stems from variations in the operating surgeons’ familiarity with patient positioning.
Patients with right axillary vein access had a mean weight of 60.16 ± 10.42 kg, mean axillary vein diameter of 8.49 ± 1.674 mm, and mean depth of 26.16 ± 4.5 mm. Patients with left axillary vein access had a mean weight of 59.99 ± 9.22 kg, mean axillary vein diameter of 8.31 ± 1.65 mm, and mean depth of 26.52 ± 4.22 mm.
SPSS’s t-test was also applied to analyze the data for mean weight, axillary vein diameter, and axillary vein depth between the two groups. The results indicated P-values of 0.845, 0.285, and 0.416, respectively. These statistical results suggested no statistically significant differences in the mean body weight, axillary vein diameter, or depth of axillary vein between patients with right and left axillary vein access. These data suggested comparable objective puncture difficulty between the left and right axillary vein groups. The choice of implanting the TIVAD on either side may be primarily determined by the surgeon’s habitual preference. These findings indicate that the dominant-hand position facilitates better operational maneuverability for physicians.
Effect of BMI on axillary veins and surgical time
Using the Chinese BMI standard, the patients were grouped into the following four categories: wasting (BMI < 18.5), normal (18.5 ≤ BMI < 24), mild-to-moderate obesity (24 ≤ BMI < 28), and severe obesity (BMI ≥ 28). These groupings were based on BMI statistics and were used to analyze axillary vein diameter, axillary vein depth from the body surface, and the time of surgical operation (Table 3).
Effect of BMI on axillary veins and surgical time.

BMI: body mass index.
Wasting: BMI <18.5; Normal: BMI between 18.5 and 23.99; mild-to-moderate obesity: BMI of 24–27.99; severe obesity: BMI >28.
Statistical methods: Kruskal–Wallis test.
P-values marked with bold indicate statistically significant results.
The wasting group included 64 patients with a mean BMI of 17.21 ± 1.00. Their mean axillary vein depth from the body surface was 21.5 ± 3.5 mm, mean axillary vein diameter was 8.5 ± 1.1 mm, and mean operation time was 13.64 ± 2.62 min.
The normal group consisted of 376 patients with a mean BMI of 21.56 ± 1.49. Their mean axillary vein depth from the body surface was 25.0 ± 3.4 mm, mean axillary vein diameter was 8.4 ± 1.7 mm, and mean operation time was 13.91 ± 3.39 min.
The mild-to-moderate obesity group had 216 patients with a mean BMI of 25.57 ± 1.08. Their mean axillary vein depth from the body surface was 28.3 ± 3.6 mm, mean axillary vein diameter was 8.3 ± 1.8 mm, and mean operation time was 14.04 ± 2.97 min.
The severe obesity group comprised 47 participants with a mean BMI of 30.29 ± 2.61. Their mean axillary vein depth from the body surface was 33.4 ± 4.8 mm, mean axillary vein diameter was 8.3 ± 2.4 mm, and mean operation time was 15.64 ± 5.68 min.
Using the SPSS Kruskal–Wallis test for multiple independent samples, the data showed statistically significant differences in the depth of the axillary vein between the different groups (P < 0.001) and in the duration of the surgical maneuver (P = 0.031). However, there was no statistically significant difference in the diameter of the axillary vein between the groups (P = 0.98).
Effect of thoracic pressure on axillary veins
Patients with axillary vein diameters of <5.5 mm were categorized as the group with poor axillary vein filling. To ensure successful puncture, the patients were instructed to inhale deeply, exhale forcefully, and then hold their breath. This action increases intrathoracic pressure, improving the filling of the axillary veins (Figure 2(b) and (c)) and enhancing the success rate of the puncture. This subgroup consisted of 41 patients, representing 5.83% of the total patient population.
In the calm breathing state, the mean diameter of the axillary vein for this subgroup was 3.52 ± 1.56 mm. When the patients were instructed to hold their breath after forceful exhalation, the mean diameter of the axillary vein increased to 7.73 ± 1.95 mm. A paired samples t-test using SPSS was conducted, yielding a t-value of −13.138 and a P-value of <0.001 (Table 4). These results indicate that increasing the intrathoracic pressure significantly increases the axillary vein diameter.
Statistics of patients in the group with poor axillary venous filling.
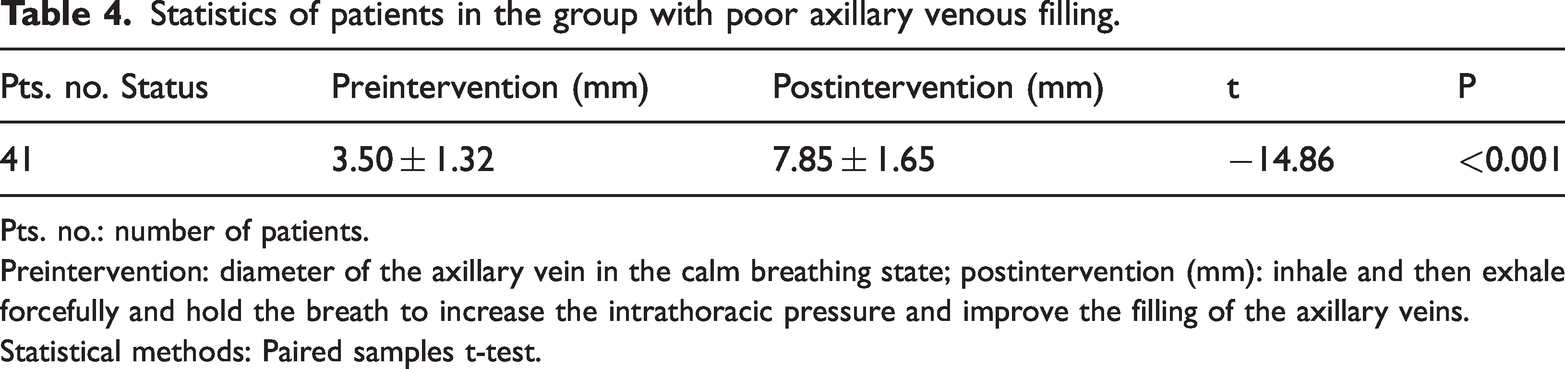
Pts. no.: number of patients.
Preintervention: diameter of the axillary vein in the calm breathing state; postintervention (mm): inhale and then exhale forcefully and hold the breath to increase the intrathoracic pressure and improve the filling of the axillary veins.
Statistical methods: Paired samples t-test.
The data revealed that for patients with poor axillary vein distension, surgeons can improve puncture success rates by encouraging patients to increase intrathoracic/intra-abdominal pressure (e.g. through Valsalva maneuver), thereby reducing venous return and enhancing axillary vein filling.
Operational process problems
After successful venous puncture, the guidewire failed to enter the superior vena cava and instead entered the IJV in 47 cases, accounting for 6.69% of the total cases. After withdrawing the guidewire to a length of 8–10 cm, the direction of the guidewire was adjusted under direct ultrasound visualization, with a 100% success rate for these adjustments.
In 13 cases (1.90% of the total), the guidewire in the axillary vein was folded. In nine patients, the guidewires were successfully adjusted under ultrasound guidance, while the procedure failed in four patients, who were subsequently switched to IJV puncture.
In electrocardiographic positioning of the catheter tip, 14 individuals (1.99% of the total) failed to show a high-acuity sinus P wave. After withdrawing the catheter to a position of 10–12 cm and then reintroducing it, a high-acuity sinus P wave was finally visible in these patients.
In seven patients (1.02% of the total), the artery was mistakenly penetrated during the procedure. Compression was applied after the arterial puncture, and no serious arterial injury or severe hematoma occurred. Subsequently, these patients were switched to IJV puncture, and TIVAD placement was ultimately successful (Table 5).
Operational process difficulties.

Pts. no.: number of patients; IJV: internal jugular vein.
IJV subgroup
TIVAD implantation via the IJV route was performed in 18 patients, including 8 males and 10 females. The mean age of the patients was 55.83 ± 11.90 years, mean height was 160.94 ± 6.72 cm, mean weight was 67.07 ± 16.90 kg, mean BMI was 25.72 ± 5.14, mean axillary vein depth was 29.3 ± 5.60 mm, and mean axillary vein diameter was 5.65 ± 1.62 mm.
Among these patients, five had poor axillary vein filling, making puncture extremely difficult; they were therefore referred to the IJV group. In seven patients, the axillary artery was mistakenly punctured, leading to failure of axillary vein puncture. In four patients, the guidewire folded in the axillary vein and could not be adjusted to the superior vena cava under ultrasound guidance, resulting in the inability to perform tunnelless TIVAD implantation via axillary vein puncture. Two patients had extremely severe obesity and poor axillary vein filling, coupled with deep axillary vein depths, making puncture difficult, and were therefore directed to IJV puncture (Table 6).
Demographics of patients converted to internal jugular vein puncture.

BMI: body mass index; IJV: internal jugular vein.
Follow-up data
In all patients, after surgery, the TIVAD catheters functioned well, with their tips located in the middle and lower sections of the superior vena cava. There were no surgery-related complications such as pneumothorax, hemothorax, subcutaneous hematoma, postoperative wound infections, or wound ruptures.
At the end of the follow-up period on 31 August 2023, the status was as follows: 471 TIVADs were still in regular service, 128 TIVADs were removed at the end of the treatment cycle, 68 patients visited third hospitals after TIVAD placement resulting in incomplete information, 34 patients died during the follow-up cycle, and 2 TIVADs were unintentionally removed. The mean duration of follow-up was 281.59 (range: 1–721) days.
During follow-up, retrograde migration of the catheter into the IJV was observed in seven patients (0.99%). Catheter occlusion occurred in three patients (0.426%), and the TIVAD became functional again after catheter recanalization with urokinase thrombolysis. There were two cases (0.28%) of catheter-related infections leading to unintentional removal of the TIVAD. Additionally, two patients (0.28%) developed catheter-related vein thrombosis, and one patient (0.14%) had a mild skin infection around the infusion port; the patient recovered after anti-infective treatment.
No other complications such as port turnover, catheter fracture, pinch-off syndrome, or the need for secondary surgical adjustments were reported. The overall immediate and long-term complications rate was 2.13%.
Discussion
Most physicians in China prefer the IJV for TIVAD implantation. This preference is mainly due to the superficiality of the IJV, which allows for safe puncture and has a very high success rate, accompanied by a very low probability of pneumothorax.4–5 However, the IJV approach has obvious disadvantages. For instance, the IJV approach significantly prolongs the procedure, necessitating increased injection of local infiltration anesthetics, thereby intensifying patient pain. 6 Additionally, the wound in the neck and the tubular shape of the catheter (Figure 1(a)) as it traverses the clavicle and courses subcutaneously through the neck compromise patient privacy and aesthetics. Especially, young breast cancer patients who are concerned about aesthetics may express dissatisfaction with the tubular shape of the catheter associated with IJV-access TIVAD.
Some physicians prefer penetrating the subclavian vein using bone anatomical landmarks as guides, with high incidence of pneumothorax and even serious complications such as hemothorax. 9 Additionally, the pinch-off syndrome may occur, posing a risk of catheter rupture. 3
The implantation technique of tunnelless TIVAD, which lacks a subcutaneous tunnel in the neck, results in a more aesthetically pleasing postoperative outcome (Figure 1(b)) and is thus more favored by patients. The overall immediate and long-term complications rate was 2.13%. Seo et al. reported an immediate and long-term complication rate of 1.4%. 10 These studies suggest that this technique demonstrates good safety and high reliability.
This study can help surgeons simplify the surgical procedure, shorten the operation time, reduce the extent of local anesthetic injection, alleviate postoperative pain, and facilitate the separation of the fascia around the catheter. By enhancing the skill level of physicians in performing real-time ultrasound-guided puncture, axillary vein puncture may be selected for 99% of patients. If the axillary vein is poorly filled, intrathoracic pressure can be increased to improve vascular conditions and consequently enhance the success rate of the puncture.
There was a significant difference in patient age and BMI between the two groups of patients for whom right and left axillary vein access was used. The primary reason for this significant difference was that the subgroups were not randomly assigned. Moreover, there was a selection bias. In obese patients, the axillary vein diameter does not increase with higher BMI; however, greater BMI is associated with increased axillary vein depth. This anatomical variation may lead to technical difficulties during puncture, elevating the risk of puncture failure. Therefore, for morbidly obese patients, surgeons should emphasize the potential need to convert to an IJV approach in case of axillary puncture failure. For surgeons who perform pocket creation first to venous access, the incision site should be carefully planned according to the patient’s BMI to accommodate anticipated tissue depth.
The shorter procedure time for right axillary vein puncture during TIVAD implantation compared to that for the left side may be attributed to physicians’ habitual right-sided positioning for puncture procedures. However, all patients who underwent guidewire ectopic placement into the IJV were in the right axillary vein puncture subgroup. If we only considered the proportion of patients for whom the right axillary vein approach was used, it accounted for as much as 8.43%. In terms of anatomy, the angle between the right axillary vein and the right brachiocephalic vein is larger, making it easier for the guidewire to retrograde into the IJV. In contrast, the left axillary vein and the left brachiocephalic vein are more directly connected to the superior vena cava, and the guidewire rarely retrogrades into the IJV after a successful puncture. Thus, some surgeons prefer the left axillary vein approach for puncture. 10 There was no statistically significant difference in the diameter and depth of the right and left axillary veins, indicating a consistent objective difficulty of puncture for both sides. Therefore, the preference for the left axillary vein in puncture procedures may be clinically advocated.
In a retrospective study of 216 axillary vein port implantation procedures, Seo et al. 10 reported the following intraoperative complications in descending order of frequency: advancement of device into unintended vein (n = 33), catheter kinking (n = 13), tract bleeding (n = 5), sheath bending (n = 3), axillary artery puncture (n = 3), and preexisting stenosis of the brachiocephalic vein (n = 1). In our study, the most frequently encountered issues were guidewire malposition and aberrant advancement (n = 60), followed by arterial injury (n = 7). Notably, we did not observe any cases of sheath bending, catheter kinking, or tract bleeding. While both studies employed similar surgical protocols, the key difference lies in the incision orientation; Seo et al. utilized a longitudinal incision, whereas our approach involved a transverse incision. Transverse incision may be more favorable for tissue separation around the guidewire and could avoid the occurrence of catheter kinking and sheath bending. It is noteworthy that despite ultrasound guidance, arterial injury occurred with a low incidence. 10 Fortunately, none of the studies reported any severe hemorrhagic complications resulting from arterial puncture.
The disadvantages of the tunnelless TIVAD implantation technique via the axillary approach include the smaller diameter of the axillary vein compared to that of the IJV, the requirement of a skilled and experienced surgeon for performing ultrasound-guided puncture procedures, the significantly greater depth of the axillary vein compared to that of the IJV, increased difficulty in puncture due to deeper vein placement in obese patients, greater influence of venous filling by respiratory movements, and a tendency for the guidewire to retrograde into the IJV when puncturing the right axillary vein.
This study has certain limitations. As a single-center retrospective study, some data were unavailable, and the TIVAD site was not randomized. Furthermore, patient follow-up was inconsistent. The incidence of complications during follow-up may have been underestimated due to the short duration of TIVAD retention, patient mortality, and loss to follow-up.
Conclusions
This technique may achieve a high puncture success rate with minimal complications and excellent safety, thereby reducing the required dosage of local anesthetics, alleviating patient pain and discomfort, and enhancing postoperative aesthetic results. TIVAD implantation via ultrasound-guided axillary vein puncture may be the preferred choice for chest wall TIVAD implantation.
In summary, the tunnelless TIVAD implantation technique utilizing ultrasound-guided axillary vein puncture may offer a highly effective, safe, and aesthetically pleasing alternative for chest wall TIVAD implantation.
Footnotes
Acknowledgments
We thank Dr. Haiguan Luo and Dr. Shuchen Zhu from the Cancer Hospital Chinese Academy of Medical Sciences Shenzhen Center for their extensive surgical experience and support. We also thank Lihui Huang and Yuying Pan for assisting us with follow-up registration and data recording.
Authors’ contributions
Study concept, design and writing: Haiguan Luo and Yuying Pan. Data collection: Haiguan Luo, Shuchen Zhu Lihui Huang, and Yuying Pan. Statistical analysis: Haiguan Luo and Lihui Huang. All authors contributed significantly to the writing, translation, and data recording of the article. All authors contributed to the article and approved the submitted version.
Availability of data and materials
The datasets used and analyzed in this study are available upon reasonable request from the corresponding author.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics and consent to participate
The project was approved by the Ethical Review Committee of the Cancer Hospital Chinese Academy of Medical Sciences Shenzhen Center with approval NO.: KYLX2023-45.
Funding
This study was supported by the Guangdong Province Health and Wellness Appropriate Technology Promotion Program, Project Investigator: Haiguan Luo, Project No. 202303252151441107. The publication of this article was supported by the Shenzhen High-level Hospital Construction Fund.
