Abstract
Based on the level of carcinogenic risk, human papillomavirus can be categorized into high-risk and low-risk types. High-risk human papillomavirus (such as human papillomavirus 16 and human papillomavirus 18) induces the inactivation of tumor suppressor genes p53 and retinoblastoma by encoding E6 and E7 proteins, leading to dysregulated cell cycles and excessive cell proliferation. In urothelial tumors, human papillomavirus infection is associated with the occurrence and progression of bladder cancer, playing a significant role in urothelial tumor development. This may be linked to specific molecular mechanisms triggered by human papillomavirus and its impact on treatment response. Furthermore, high expression of human papillomavirus-related molecular markers, such as p16, can serve as an important tool for predicting and monitoring the prognosis of human papillomavirus-associated tumors. Therefore, detecting and monitoring human papillomavirus infection status and related molecular markers can provide a more accurate assessment of bladder cancer prognosis and offer valuable guidance for formulating personalized treatment plans.
Introduction
Urological tumors, encompassing bladder, prostate, renal, and penile cancers, represent a major global health burden and have complex etiologies. In 2024, an estimated 169,360 new cases of urological tumor and 32,350 relevant deaths are projected in the United States alone, driven by genetic predispositions, environmental carcinogens, and microbial infections. 1 Among these factors, human papillomavirus (HPV) has emerged as a significant oncogenic agent. HPV infects over 80 million individuals in the U.S., with 14 million new global infections being reported annually. 2 While HPV is widely recognized for causing cervical cancer, its role in urological malignancies remains underexplored despite epidemiological links to bladder, penile, and prostate cancers.
High-risk HPV types (e.g. HPV16/18) evade immune clearance and integrate their DNA into host genomes, primarily through oncoproteins E6 and E7. E6 degrades the tumor suppressor p53, impairing DNA repair and promoting genomic instability, while E7 inactivates retinoblastoma (Rb), deregulating the cell cycle and driving uncontrolled proliferation. These mechanisms not only initiate carcinogenesis but also enhance tumor aggressiveness and immune evasion, particularly in immunocompromised microenvironments. However, the organ-specific dynamics of HPV-driven transformation in urinary systems, such as bladder urothelium versus prostate epithelium, remain poorly characterized.
Advancing HPV detection (e.g. polymerase chain reaction (PCR), serological assays) and prevention (e.g. vaccines targeting E6/E7) are critical for mitigating urological tumor risks. This review synthesizes the current evidence on HPV’s epidemiological patterns, molecular pathogenesis, and therapeutic vulnerabilities across the urological tumor spectrum. We further highlight emerging strategies, including immunotherapies and precision diagnostics, to bridge the gaps between experimental insights and clinical applications. By integrating multi-omics findings and clinical data, this study aimed to refine HPV-targeted interventions for improved patient outcomes.
The epidemiology of HPV in urinary system tumors
HPV is a double-stranded circular DNA virus with over 200 genotypes identified to date, 170 of which are classified into high-risk and low-risk types. Reports indicate that 610,000 cases of malignancy annually are attributed to high-risk HPV, with 69.89% of newly diagnosed malignancies being linked to HPV infection. This includes over 90% of cervical cancers, 88% of anal cancers, 24.9% of vulvar cancers, 50% of penile cancers, and 30.8% of oropharyngeal cancers. 3 The International Agency for Research on Cancer has highlighted the significant role of HPV infection in urological tumors, attributing 4.5% of urological malignancies to HPV infection. 4 Given the anatomical proximity of the urinary and reproductive systems, HPV infection rates in the urinary system are high. It is worth noting that although most current studies have proven the association between HPV infection and the risk of urinary system tumors, different influencing factors, such as geographical location, research design, sample size, and HPV detection methods, can lead to differences in the prevalence of HPV in bladder cancer (BC) in different studies. 3 The incidences of several common HPV-related urological tumors are described in Table 1.
Epidemiological overview of HPV prevalence in urological tumors.
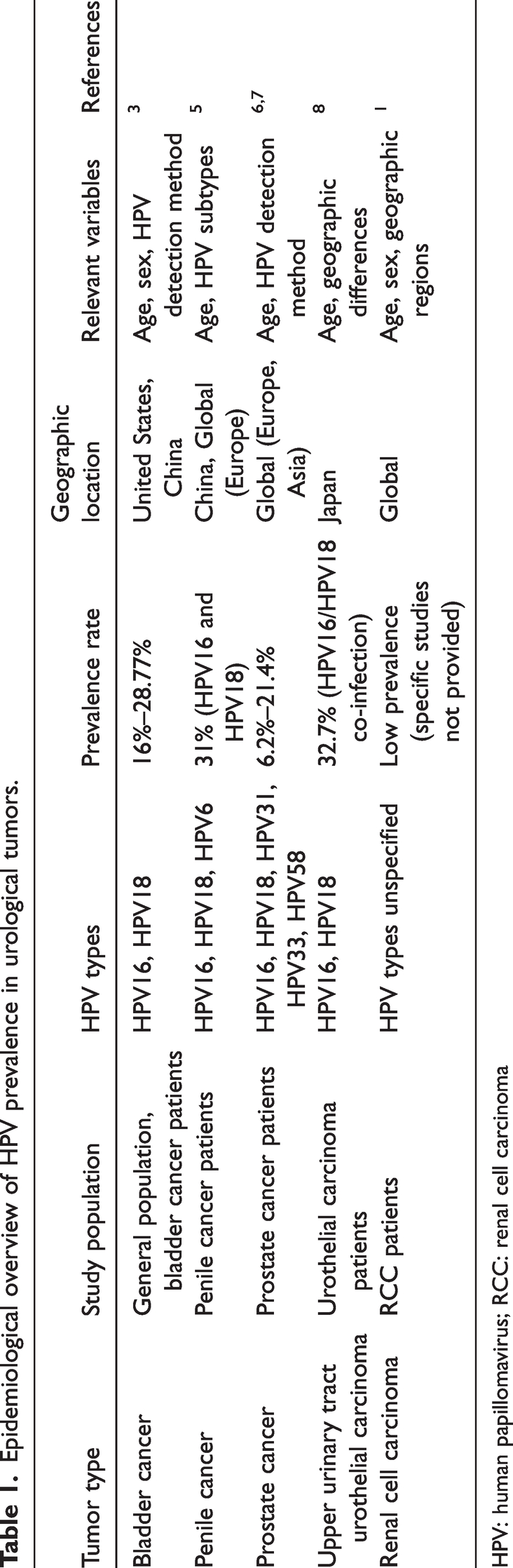
HPV: human papillomavirus; RCC: renal cell carcinoma
BC
The risk of BC is closely associated with HPV. A study showed an HPV prevalence of 16% among BC patients, with HPV16 (5.99%) and HPV18 (3.03%) being the most commonly detected types. 3 Otero-Murie et al. also demonstrated a significant association between HPV and BC diagnosis, with an odds ratio (OR) of 4.18 (95% confidence interval (CI): 2.63–6.66, I2 = 40%). 9 These findings suggest a strong correlation between HPV infection and BC development.
However, the role of HPV in causing BC remains controversial. For example, a study by Khatami et al. involving 2954 BC patients found no significant association between HPV infection and BC. 10 This indicates that while most studies suggest a link between HPV and BC, the conclusion is not uniformly accepted. Differences in study design, patient populations, and HPV detection methods are significant factors contributing to the variability in prevalence rates. Therefore, further large-scale, multicenter studies are needed to clarify the specific role of HPV in BC pathogenesis.
Although a meta-analysis by Sun et al. reported an HPV prevalence of 16% in BC (OR = 4.18), 3 Khatami et al. found no significant association in a cohort of 2954 patients. 10 This discrepancy may stem from methodological differences. Sun et al.’s study prioritized PCR-based detection in tumor tissues, whereas Khatami et al.’s analysis included urine samples with a lower sensitivity. Notably, HPV detection in urine is prone to false negatives due to low viral copy numbers and inhibitors in the urine matrix, suggesting tissue-specific sampling bias as a key confounder. It should also be noted that in schistosomiasis endemic areas (such as sub-Saharan Africa), the incidence of bladder squamous cell carcinoma (SCC) has significantly increased, which is closely related to the synergistic carcinogenic effect of chronic schistosomiasis infection and HPV. Chronic inflammation and fibrosis of the bladder mucosa caused by schistosomiasis infection can disrupt the epithelial barrier function and induce local immunosuppression, thereby promoting the persistent infection of high-risk HPV types (such as HPV16 and HPV18) and viral genomic integration. Studies have shown that in schistosomiasis endemic areas, schistosomiasis BC tissues are positively correlated with HPV infection factors, and patients with co-infection of HPV and schistosomiasis have stronger tumor invasiveness and poorer prognosis. 11 The discovery of this geographically specific co-factor emphasizes that HPV screening as well as schistosomiasis prevention and control need to be incorporated into the comprehensive management strategy of BC in epidemic areas.
Penile cancer
HPV infection is a significant risk factor for penile cancer, with 50% of cases being closely associated with HPV. A study by M.W. Yuan et al. analyzed HPV-associated malignancies in 124,772 patients and found that of the 7654 newly diagnosed male cancers, 61% (4669/7654) were penile cancers. 12 Additionally, research by Beatriz Serrano’s team indicated that 11.3% (13,000/115,000) of penile cancer cases were attributable to HPV infection. 13 Similarly, an evaluation of the association between HPV and penile SCC found that 46.1% (47/102) of such tumors were linked to HPV-related subtypes. 14 These findings underscore the critical role of HPV in the pathogenesis of penile cancer.
Prostate cancer
Clinical studies have suggested an association between prostate cancer pathogenesis and HPV infection. The high-risk HPV types HPV16, HPV18, HPV31, HPV33, and HPV58 are most commonly detected in prostate tumors. A meta-analysis by Binbin Yin and colleagues on 971 prostate cancer cases reported an HPV infection rate of 21.4% (208/971). 15 Additionally, research has indicated that 14.9% (53/356) of prostate cancer cases are closely associated with HPV infection. 16 Notably, a study by Nalisson Marques Pereira and colleagues involving 162 prostate cancer patients revealed HPV infection in 6.2% (10/162) of the patients. However, the authors suggested that the results might have been influenced by rectal contamination. On the one hand, HPV infection was detected in the rectum, and transrectal transducers were used during biopsy collection. Conversely, no expression of the L1 gene or significant levels of HPV L1 protein were detected in nontumorous or tumorous prostate tissues. While HPV infection is considered an important risk factor for prostate cancer, detection bias may have arisen from the methods used. Therefore, further research on more accurate and reliable detection technologies is needed.
Upper urinary tract urothelial carcinoma
Although the pathogenesis of upper urinary tract urothelial carcinoma is similar to that of BC, there is a lack of extensive clinical research on HPV infection rates and the role of HPV in renal, pelvis, and ureteral carcinomas. A Japanese study used in situ hybridization to investigate the presence of HPV16 and HPV18 DNA in patients with renal pelvis and ureter transitional cell carcinoma (TCC). Among 98 TCC patients, 32.7% (32/98) tested positive for HPV infection, with 18.8% (6/32) co-infected with HPV16 and HPV18. 17
Testicular and renal tumors
Although existing research has paid less attention to the role of HPV in testicular and renal tumors, recent evidence suggests a potential association. However, epidemiological data on HPV infection in testicular tumors remain controversial. A systematic review including 17 studies showed that the detection rate of HPV DNA in testicular tumor tissues is approximately 3.1%–7.4%, but most studies have small sample sizes and high heterogeneity in detection methods. Thus, further verification of their biological significance is warranted. The association between renal cell carcinoma (RCC) and HPV is even weaker. A recent multicenter study performed next-generation sequencing analysis on tissue samples from 1203 RCC patients, and only 0.8% (10/1203) detected HPV16/18 integration events, with no significant association with tumor grade or prognosis. 18 Notably, the choice of HPV detection method may affect the accuracy of results. For example, Sarier et al. pointed out that HPV typing in male genital samples after circumcision requires optimized sampling strategies to prevent false-negative results; this methodological challenge may also apply to HPV detection in deep urinary system tumors (such as renal tumors). 19 In the future, standardized detection processes and large-sample studies are needed to clarify the pathogenic role of HPV in testicular and renal tumors.
Mechanisms of HPV-induced urological tumor development
HPV is primarily transmitted through skin contact and nonsexual contaminants. Around 200 HPV types have been identified, classified into high-risk and low-risk types based on their carcinogenic potential. High-risk HPV types are key contributors to carcinogenesis. They enter the human body through microabrasions in the skin or mucosa, disrupt normal cellular regulatory mechanisms, and integrate their DNA into the host genome. This leads to uncontrolled cell proliferation and excessive expression of viral early-region genes related to viral replication, ultimately resulting in malignant transformation into cancer (Figure 1). Approximately 90% of cervical cancers and most of anal and vulvar cancers are linked to high-risk HPV infections. 20
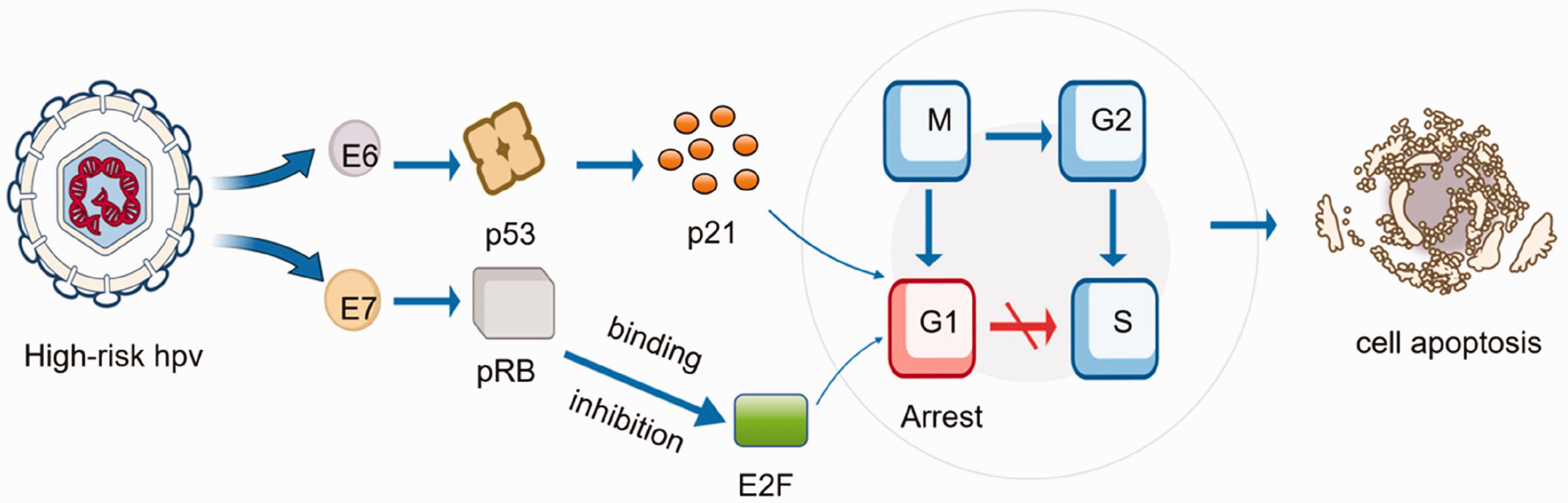
Mechanisms of HPV-induced urological tumor development. HPV: human papillomavirus.
Integration of HPV genes into host chromosomes
HPV DNA integrates preferentially into transcriptionally active or fragile genomic regions (e.g. FRA3B, 13q14), often disrupting tumor suppressor loci such as Rb. During integration, viral early proteins orchestrate replication and host genome manipulation: E1 recruits replication machinery (topoisomerase I, DNA polymerase α) to fragment host chromatin, while E2 binds viral control sequences to regulate replication initiation. Crucially, intact E2 suppresses E6/E7 expression in episomal HPV. However, integration-induced E2 loss abolishes this repression, unleashing E6-mediated p53 degradation and E7-driven Rb inactivation, which collectively impair cell cycle checkpoints and promote genomic instability. 21
Beyond p53/Rb axis disruption, HPV proteins exhibit tissue-specific oncogenicity. E6 stabilizes telomerase via MYC-dependent hTERT activation, enabling cellular immortalization, while E5 enhances proliferation through lysosomal pH modulation and immune evasion via major histocompatibility complex I (MHC-I) trafficking interference. 22 Notably, HPV integration sites dictate organ-specific pathogenesis. In BC, E6-APOBEC synergy drives mutagenesis, whereas prostate tumors show E6-androgen receptor (AR) crosstalk amplifying MYC oncogenicity. 23 These findings underscore the need for organotypic models to dissect context-dependent HPV.
HPV and the p53
The p53 gene, often referred to as the “guardian of the genome,” is a key tumor suppressor gene that plays a critical role in the cell cycle. It encodes the nuclear transcription factor p53 protein, which prevents genomic damage and mutations. High-risk HPV types such as HPV16 and HPV18 encode E6 protein, which interacts with and degrades the p53 protein. This degradation disrupts cell cycle control and increases the risk of malignancy. 24
The E6-mediated degradation of p53 also allows cells to evade apoptosis, enhancing the likelihood of cancerous transformation. However, without functional p53, E6 cannot inhibit p53 in the conventional sense. Loss of p53 function promotes genomic instability in HPV-infected cells, driving tumorigenesis. 25 Notably, p53 mutations frequently occur in BC where mutant p53 proteins drive metabolic reprogramming of cancer cells, promoting progression. 23
Analysis of HPV integration sites and epigenetic effects
HPV integration events have significant genomic site preferences; they mainly affect common vulnerable sites in the host genome (such as FRA3B and FRA16D). These regions are rich in AT repeat sequences and are prone to breakage under replication pressure, becoming “hotspots” for viral integration. 26 In urinary system tumors, the selection of HPV integration sites shows organ specificity. The integration of HPV16 in BC often leads to the deletion of tumor suppressor genes FHIT and WWOX, inducing genomic instability and driving tumor progression, while in prostate cancer, HPV18 prefers to integrate into AR signaling pathway genes (such as MYC and PTEN), synergically activating hormone-dependent carcinogenesis. 27 Long-read sequencing techniques (such as PacBio and Oxford Nanopore) further revealed that HPV16 integration in BC can truncate the epithelial differentiation regulatory gene TP63 and induce epithelial–mesenchymal transition. Further, HPV integration and TMPRSS2–ERG fusion events in prostate cancer are mutually exclusive, suggesting that it promotes cancer through substitution mechanisms. At the epigenetic level, after HPV integration, demethylation of the CpG island in the locus control region (LCR) region of the virus eliminates E2-mediated transcriptional inhibition, resulting in persistently high expression of E6/E7. In approximately 65% of BC cases, the S249C mutation of the FGFR3 gene (a typical APOBEC characteristic mutation) accounts for 60% of all FGFR3 mutations, suggesting that APOBEC activity plays a core role in the occurrence of urothelial carcinoma. At present, the quantitative comparison of APOBEC mutation characteristics in HPV+ and HPV− BC remains limited. A study on a small sample showed that the frequency of ApOBEC-related C→T mutations in HPV+ BC was 1.8 times higher than that in the HPV− group (p = 0.03); however, a larger-scale cohort verification is needed. In addition, LCR hypomethylation is associated with the mutagenicity of APOBEC3B (C→T mutation), increasing the genomic mutation burden.8,28 Meanwhile, integration events induce methylation reprogramming of host adjacent gene promoters. For instance, in prostate cancer, hypermethylation of RASSF1A activates the PI3K/AKT pathway, and in BC, methylation markers such as SOX1 and ZNF154 become independent prognostic indicators. 29 It needs to be highlighted that HPV integration shapes tumor biological behavior and clinical outcomes through multi-dimensional mechanisms.
HPV and p16
p16 plays an important role in clinical pathological diagnosis. Under normal circumstances, p16 regulates Rb protein through a negative feedback mechanism to maintain cell cycle balance. However, HPV infection disrupts Rb function, leading to a significant upregulation of p16 expression. Consequently, p16 expression can serve as a surrogate marker for detecting HPV infection. 14
Studies investigating the co-occurrence of HPV DNA and p16 overexpression found a detection rate of 94% for p16-positive BC cases and 8.8% for urothelial carcinoma (UC) with squamous differentiation. In a study of 35 patients with an HPV prevalence of 17.1%, high p16 overexpression was observed in 45.7%. A meta-analysis of 37 studies, including 2246 BC cases, focused on the relationship between p16 and BC prognosis or clinicopathology. The analysis suggested that p16 downregulation occurs in BC. 30 Thus, the association among p16, BC, and HPV remains less clear than the direct association between HPV and BC.
Effect of HPV on the tumor microenvironment (TME)
HPV infection can create a TME favorable to cancer progression by immune evasion, suppressing antitumor immune responses and upregulating immunosuppressive factors, promoting tumor progression. For example, the E5 protein of HPV can interfere with antigen presentation by MHC class I molecules, hindering CTL attacks and helping the virus avoid immune surveillance. Additionally, the E6 and E7 proteins of high-risk HPV, such as HPV16, reduce type I IFN levels and induce an immune tolerance phase through inflammatory cytokines in the absence of co-stimulatory signals, potentially promoting the TME within tumors. 31 HPV utilizes these strategies to evade effective immune responses, ultimately ensuring the long-term persistence of HPV infection and driving tumor development. Furthermore, the diversity of urinary tract anatomy and function creates a unique microenvironment for HPV. In the bladder, the rapid renewal of transitional epithelium may promote the persistent presence of HPV, while urine exposure limits local immune surveillance. 32 In the prostate, the abundant androgen environment upregulates the expression of HPV E5, which may promote the downregulation of MHC-I and enhance immune evasion. In addition to MHC-1 molecules, the abnormal expression levels of immune checkpoint molecules such as programmed death-ligand 1 (PD-L1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), and lymphocyte activation gene 3 (LAG-3) in HPV-positive urinary system tumors deserve attention. HPV infection can induce the overexpression of PD-L1 through the E6/E7 protein activation pathway and inhibit T cell activity. Studies have shown that PD-L1 expression exists in 68% (113/165) of urological tumors. 33 CTLA-4 antibody can activate T cells and reverse immunosuppression. New treatment options may be related to these markers, but immunotherapy may be heterogeneous due to HPV subtypes. In the future, it is necessary to combine biomarkers and single-cell sequencing to screen the beneficiary population. Renal medullary hypoxia may impair the fidelity of HPV replication, which explains the rarity of integration events in RCC. 4 In summary, HPV orchestrates tumor progression through multifaceted immune evasion in the TME, while urinary tract-specific anatomical and microenvironmental features further modulate HPV persistence and oncogenicity. Clarifying these interactions may inform HPV-targeted immunotherapies for urological cancers.
Noncoding RNAs and post-transcriptional regulation
HPV infection can indeed affect the expression of mirco-RNA (miRNA) in tumor cells, thereby leading to different molecular pathways. High-risk HPV abnormally regulates the host miRNA profile to promote tumorigenesis. It mainly includes miR-21, miR-155, and miR-203. These oncogenic miRNAs are significantly upregulated in HPV-positive BC and penile cancer. miR-21 inhibits apoptosis by targeting PTEN and PDCD4, while miR-155 enhances immune evasion by down-regulating SOCS1 and SHIP1, thereby activating the STAT3/NF-κB pathway. 34 Therefore, analysis of miRNA characteristics in HPV-positive tumors may hold diagnostic significance. For example, in BC, the overexpression of miR-21 is associated with advanced tumor stage and low survival rate. Furthermore, HPV viral proteins E6/E7 can induce the expression of long noncoding RNAs (lncRNAs) (such as HOTAIR/MALAT1) related to promoting metastasis and drug resistance; HOTAIR induces histone H3K27 trimethylation by recruiting the PRC2 complex, while MALAT1 relifies the inhibition of ZEB1 by adsorbing the miR-200 family through sponges, enhances the cell invasion ability, and mediates chemotherapy resistance by activating the PI3K/AKT pathway at the same time. 35 These lncRNAs work synergistically with the miRNAs regulated by HPV to drive abnormal cell cycles and immune escape.
Impact of HPV on the prognosis of urological tumors
HPV-driven genomic instability (via APOBEC upregulation) and immune evasion correlate with aggressive tumor phenotypes. In BC, HPV positivity is associated with higher tumor grades and recurrence rates (47.3% recurrence in HPV+ cohorts vs. 36.8% recurrence in HPV− cohorts). 36 High-risk HPV subtypes (e.g. HPV16/18) further exacerbate poor prognosis through sustained oncoprotein expression (E6/E7), which disrupts cell cycle control and amplifies signaling pathways, such as the PI3K/AKT pathway. 37 However, conflicting studies report no significant association of HPV-prognosis in UC, potentially due to viral load heterogeneity or undetected immune-modulating cofactors. 38
Current discrepancies (68.3% studies link HPV to poor survival vs. 25% showing neutrality) likely stem from variable detection methods and tissue-specific HPV oncogenicity. 36 Emerging evidence underscores HPV’s dual role; while promoting genomic instability, it may enhance immunotherapy responsiveness in PD-L1+ tumors. 31 Future prospective trials are needed to standardize HPV subtyping, integrate multi-omics profiling (e.g. immune microenvironment analysis), and validate prophylactic/therapeutic vaccines targeting E6/E7.
Prevention and prediction of HPV
Detection of HPV infection is crucial for the prevention and prediction of urological tumors. HPV antibodies and vaccines are key factors in this context. Together, these factors form a comprehensive strategy for effective prevention and prediction of urological tumors.
Notably, HPV vaccination has shown significant effectiveness in preventing HPV-associated cancers. However, research on its role in preventing urological tumors remains insufficient. This section will focus on the applications of HPV antibodies and vaccines in urological tumors, providing references for future research. Although HPV has a clear role (50% attributable risk) in penile cancer, clinical guidelines for other urinary system tumors (such as bladder and prostate tumors) do not recommend routine HPV testing because there is currently a lack of standardized testing protocols. Furthermore, compared with the established biomarkers (such as PSA, NMP22), the cost-effectiveness of HPV testing remains poor.
HPV antibody testing in urological tumors
HPV antibody testing plays a critical role in the context of urological tumors. Testing of HPV antibodies in patient serum enables the evaluation of the presence of HPV infection and its correlation with urological tumors. This has significant reference value for early diagnosis and the development of personalized treatment plans. A study investigating the cross-protection of a bivalent vaccine against nononcogenic viruses in 84 participants showed that 58.3% (49/84) of them were unvaccinated and HPV DNA-positive, while 41.6% (35/84) were vaccinated and HPV DNA-negative. HPV-VLP-ELISA tests on these 84 participants showed that 41% (13/32) had high serum IgG antibody concentrations for HPV16, 66% (2/3) for HPV18, and 83% (5/6) for HPV6. The IgG detection rate was 44% (14/32), and the serum IgA antibody positivity rate for HPV16 was 31.2% (10/32); the positivity rate for HPV6 and HPV72 was 50%. The study revealed that IgG antibodies were more prevalent in the vaccinated group, while higher IgA antibody levels were more commonly detected in the unvaccinated group. 39 This suggests that the presence of antibodies can effectively reduce HPV infection risk and provide protection for relevant populations. Notably, the E6 and E7 antigens are considered ideal tumor antigens for eliminating HPV-associated precancerous lesions and cancers. However, Shibata et al.’s study on the role of E6 and E7 proteins in treating HPV-associated malignancies yielded unsatisfactory results, with 74% (14/19) of tumor patients dying within 22 months. 40 Therefore, treatment strategies for HPV-associated precancerous lesions and cancers require further research and improvement.
Application of vaccines in urological tumors
HPV is a major factor in the development of related malignancies, and HPV vaccination is critical for alleviating the burden of these diseases by enabling primary prevention of HPV-related cancers. 13 However, current research is primarily focused on cervical cancer, with insufficient studies on the use of HPV vaccines for preventing urological tumors. Currently, there are 5 vaccines targeting common high-risk HPV types: Cervarix (bivalent), Gardasil (quadrivalent), Gardasil9 (nonavalent), Cecolin (bivalent), and Silgard (quadrivalent). 41 Matti Lehtinen and his team conducted an 11-year long-term follow-up study involving 3341 women vaccinated with either bivalent or quadrivalent vaccines and compared them to a control group of 16,526 unvaccinated women. During the follow-up period, no HPV-related cancers were detected in the vaccinated group, whereas 15 cases were identified in the unvaccinated group. 42 Additionally, a randomized placebo-controlled study on males aged 16–26 years showed a significant reduction in the incidence of genital warts after 9.5 years of follow-up. In a population of homosexual males, the occurrence of HPV6-, HPV11-, HPV16-, or HPV18-associated anal intraepithelial neoplasia or anal cancer was also significantly reduced (20.5 vs. 906.2/10,000 person-years). 43 These findings suggest that HPV vaccines can lower the incidence of HPV-related cancers, highlighting the need for further research on the vaccine’s effectiveness in preventing other cancer types, such as urological tumors. Additionally, vaccination strategies and the selection of target populations require further study and discussion.
Application of immunotherapy
Immunotherapy, particularly cell therapy including dendritic cell (DC) vaccines, has become an important approach for cancer treatment. DC vaccines, which can be loaded with tumor-specific antigens, aim to trigger strong antitumor immune responses. Researchers are particularly focused on developing DC vaccines that target HPV-specific antigens, such as E6 and E7 oncoproteins, which are crucial in the carcinogenesis of HPV-related cancers, including cervical, penile, and oropharyngeal cancers. 44 For example, a study led by Zhenyu Ding investigated the use of a personalized neoantigen-loaded autologous DC vaccine (Neo-DCVac) for patients with advanced lung cancer. The trial involved 12 patients, with a total of 85 doses of the vaccine being administered. While the primary focus of the study was lung cancer, it is part of the broader trend of personalized immunotherapies targeting specific tumor antigens, which may also be applied to HPV-associated cancers. In this trial, all participants (100%) experienced mild grade 1 injection-site reactions, with a few instances of neutropenia and rash. The vaccine demonstrated an objective response rate of 25%, and a disease control rate of 75%. 45 These findings indicate the potential of DC vaccines as a therapeutic strategy for cancer immunotherapy, which could be extended to HPV-related malignancies (Table 2).
Compounds and drugs targeting HPV-mediated pathways in urological tumors.

HPV: human papillomavirus; EGCG: epigallocatechin gallate; NF-κB: nuclear factor kappa B; MDM2: mouse double minute 2.
Conclusion and outlook
The role of HPV in urological tumors has gained significant attention, with accumulating evidence linking high-risk HPV subtypes (e.g. HPV16/18) to bladder, penile, and upper urinary tract cancers. Mechanistically, HPV-driven oncogenesis primarily involves E6- and E7-mediated inactivation of tumor suppressors p53 and Rb, disrupting cell cycle regulation and promoting genomic instability. While the association between HPV and certain urological malignancies (e.g. prostate cancer) remains debatable, emerging epidemiological and molecular studies underscore HPV as a key contributor to tumor initiation and progression. Notably, HPV-associated tumors exhibit unique molecular signatures, including APOBEC-mediated mutagenesis and immune evasion via PD-L1 upregulation, which correlate with aggressive phenotypes and poor prognosis.
To address existing controversies and knowledge gaps, future research should prioritize large-scale, multicenter studies with standardized HPV detection protocols. Innovative technologies such as droplet digital PCR for noninvasive urine-based HPV DNA quantification and patient-derived three-dimensional organoid models hold promise for elucidating viral integration dynamics and therapeutic responses. Expanding HPV vaccination strategies beyond cervical cancer to include urological malignancies—particularly in high-risk populations—could mitigate disease burden. Additionally, environmental cofactors such as smoking and exposure to chemical carcinogens synergize with HPV by exacerbating DNA damage and fostering immunosuppressive microenvironments, highlighting the need for holistic risk assessment frameworks.
This review acknowledges limitations inherent to narrative synthesis, including potential selection bias and exclusion of studies from non-English literature. Future systematic reviews and prospective trials are essential to validate findings and refine clinical guidelines. By unraveling HPV’s multifaceted role in urological carcinogenesis, researchers can advance targeted therapies, optimize early detection, and ultimately improve patient outcomes.
Footnotes
Acknowledgements
We sincerely thank all colleagues who contributed their time and expertise to this research. Our gratitude also extends to the editors and anonymous reviewers for their insightful feedback and constructive suggestions, which were invaluable in enhancing the quality of this manuscript.
Author contributions
H. H. contributed toward writing and review and editing of the manuscript. S. F. contributed toward writing and review and editing of the manuscript as well as conceptualization of the study and validation of data. Y. L. contributed toward writing and review and editing of the manuscript as well as supervision of the study. All authors agreed with the published version of the manuscript.
Data availability statement
All data utilized in this review are sourced from publicly available literature, accessible through the PubMed and Web of Science databases. All references cited in the text have been included in the reference list. This study did not create any new datasets.
Declaration of conflicting interests
The authors confirm that this review was conducted without any commercial or financial relationships that could be perceived as a potential conflict of interest.
Funding
No specific funding was received for this work.
