Abstract
Liver injury is an increasingly recognized extrapulmonary manifestation of coronavirus disease 2019 (COVID-19), particularly during pregnancy due to overlapping physiological and pathological changes. A 26-year-old primigravida at 24+4 weeks of gestation presented with fever, chills, weakness, and sore throat, who was later confirmed to be severe acute respiratory syndrome coronavirus 2–positive. Laboratory tests revealed lymphopenia, elevated liver enzymes, low platelet count, increased D-dimer level, high total bilirubin level, and elevated lactate dehydrogenase levels. After thorough evaluation, these findings were attributed to COVID-19–associated liver injury. The patient was provided with supportive care and symptomatic treatment, resulting in gradual normalization of liver function. She was discharged at 26 weeks of gestation in stable condition. This case highlights the importance of considering COVID-19–associated liver injury in pregnant patients presenting with hepatic dysfunction, where prompt recognition and conservative management can help achieve favorable maternal outcomes.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has caused approximately 6.7 million deaths worldwide. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes COVID-19 and belongs to the Coronaviridae family, a complex group of single-stranded positive-sense ribonucleic acid viruses. 1 Coronaviruses are commonly associated with mild upper respiratory infections; however, SARS-CoV-2 has shown the ability to infect the lower respiratory tract and cause severe end-organ damage, including liver injury, due to systemic infection and inflammation. Reports indicate that aminotransferase levels exceed five times the normal value in up to 25% of patients. In severe cases of COVID-19 pneumonia, liver enzymes can rise to thousands (U/L), with total bilirubin and alkaline phosphatase levels rising in 1%–53% and 0.3%–80% of patients, respectively. 2 These observations have prompted interest in the extrapulmonary manifestations of COVID-19.
COVID-19–associated liver injury (COVALI) refers to any abnormal liver function test observed in individuals who test positive for SARS-CoV-2.3,4 Unlike other forms of transaminitis, COVALI lacks distinct or specific diagnostic criteria, making it more challenging to collect and analyze data from clinical studies. 5 This challenge becomes particularly evident when applying available data to underrepresented groups in the literature, such as pregnant women.
Although the clinical characteristics of COVID-19 during pregnancy are well documented, there is limited information on the course of liver injury. Between 13% and 42% of COVID-19–positive pregnant patients develop COVALI, an aspartate aminotransferase (AST)–predominant transaminitis in obstetric cases.6,7
Pregnant patients with COVID-19 have a higher prevalence of COVALI, more severe liver enzyme elevations, and a greater likelihood of developing severe disease than nonpregnant patients, indicating that COVID-19 increases the risk of liver injury during pregnancy. 8 Liver injury associated with COVID-19 is associated with worse clinical outcomes and higher mortality rates in pregnant women. Studies have indicated that in COVID-19–positive pregnant women, acute liver injury increases the risk of critical illness during hospitalization by 3.5 times and also raises the mortality rate.8,9
However, many aspects of how COVID-19 affects pregnant women and the specific clinical and laboratory changes remain unclear. These findings can become particularly challenging when they significantly overlap known pregnancy complications such as hemolysis, elevated liver enzymes, and low platelet count (HELLP) syndrome. To contribute to this growing area of research, we present the case of a pregnant patient with confirmed COVID-19 and laboratory results consistent with COVALI. With this case, we aimed to provide insights and encourage further investigation into the relationship of COVID-19 with liver injury during pregnancy.
Case presentation
Chief complaints
A 26-year-old primigravida at 24+4 weeks of gestation presented with a 2-day history of fever, chills, weakness, and sore throat. She was diagnosed with SARS-CoV-2 infection.
History of present illness
A 26-year-old primigravida (g2p0, induced abortion) at 24+4 weeks of gestation presented with complaints of fever, chills, weakness, and sore throat that had started 2 days previously. The oropharyngeal swab sample obtained for the COVID-19 polymerase chain reaction (PCR) tested positive. SARS-CoV-2 infection was diagnosed. The patient was admitted to Wuhan Jinyintan Hospital (Wuhan, Hubei) in November 2022.
History of past illness
The patient has no history of chronic illnesses such as hypertension, cardiovascular disease, hepatitis, or tuberculosis. She denied any history of trauma or blood transfusion and did not have any known drug or food allergies. She had received 2 doses of the COVID-19 vaccine. She had a medical history of appendicitis. She was gravida 2, para 0, with one previous induced abortion. Her last menstrual period was on 14 June 2022.
Personal and family history
The patient did not smoke or consume alcohol. No significant family medical history was reported.
Physical examination
The patient was alert and in generally good condition. Her respiratory rate was 20 breaths per minute, blood pressure was 106/71 mmHg, oxygen-free saturation was 98%, heart rate was 120 bpm, and body temperature was 36.5°C. Her breathing was unlabored, lips were not cyanotic, and no jaundice was observed on the skin or sclera. No enlargement of superficial lymph nodes was noted. The skull was of normal shape, and the neck was supple. Conjunctivae were not congested, and the pharynx shows no signs of hyperemia. Cardiac and pulmonary auscultation revealed no abnormalities. The abdomen was distended but soft, with no tenderness or rebound pain on palpation. There was no uterine activity. The fetal heart rate was 138 beats per minute on auscultation. The liver and spleen were not palpable below the costal margin. No edema is present in the lower extremities. Range of motion in all four limbs was normal.
Laboratory examinations
The oropharyngeal swab sample obtained for COVID-19 PCR tested positive. Initial laboratory examinations of the patient on admission revealed lymphopenia, elevated liver enzyme levels (alanine transaminase (ALT), 558 U/L; AST, 523 U/L), low platelet count, high D-dimer level, high total bilirubin level, and high lactate dehydrogenase level; hemoglobin and kidney function tests were normal (Table 1). Gynecological examination by the obstetrician showed no uterine tenderness, no foul-smelling discharge, and no amniotic discharge in speculum examination and ponding test. Ultrasound findings (head presentation, fetal heart rate: 148 beats per minute, amniotic fluid volume, biparietal diameter, placenta anterior) were normal. In addition, the hepatitis markers studied and human immunodeficiency virus test were negative; cardiac markers (troponin I, creatine kinase MB (CKMB)) and renal function parameters (urea, creatinine) were also normal. On the second day of hospitalization, liver enzymes were elevated 2-fold (ALT, 1321 U/L; AST, 1020 U/L).
Laboratory findings of the patient.
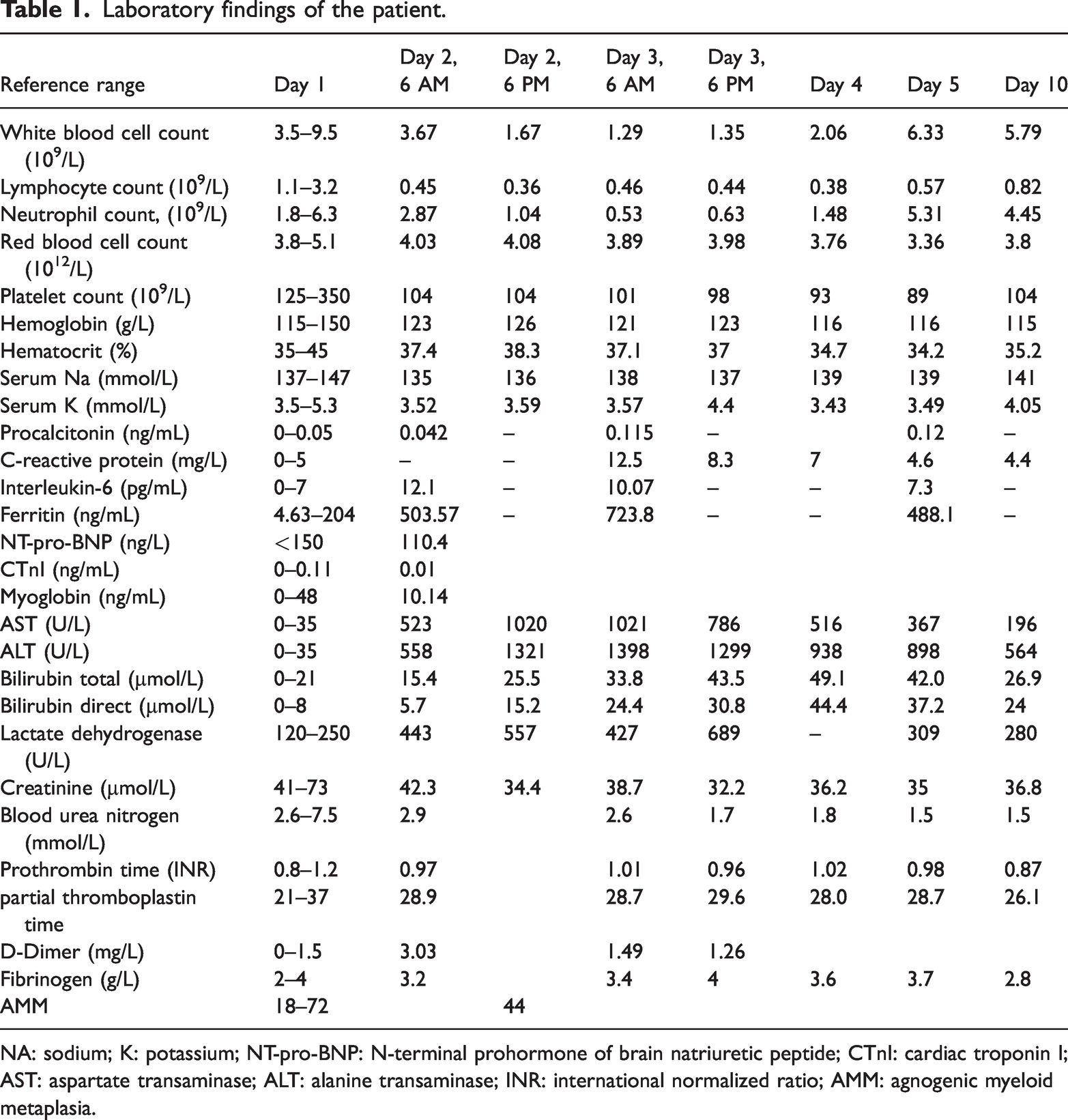
NA: sodium; K: potassium; NT-pro-BNP: N-terminal prohormone of brain natriuretic peptide; CTnI: cardiac troponin I; AST: aspartate transaminase; ALT: alanine transaminase; INR: international normalized ratio; AMM: agnogenic myeloid metaplasia.
The maximum alanine aminotransferase reached 1419 U/L on the night of hospital day 2.
As per the suggestion from a multidisciplinary consultation, anti-ds DNA studied for differential diagnosis of autoimmune or viral hepatitis, lupus anticoagulant/confirmation test, anti-nuclear antibody, and anti-cardiolipin immunoglobulin G (IgG) tests were negative on the third day of hospitalization. In addition to these, thyroid function test, blood ammonia test, urine culture showed normal results.
Imaging examinations
After a multidisciplinary consultation, the patient accepted the tomography recommendation, and there was no obvious pulmonary infection on thorax computed tomography. Abdominal echocardiography showed normal results (liver size and parenchymal echo were normal), and there was no manifestation of acute fatty liver in pregnancy.
Multidisciplinary expert consultation
After 2 days of hospitalization, the patient’s liver injury progressively worsened, and the maximum alanine aminotransferase reached 1419 U/L on the night of hospital day 2. Therefore, a multidisciplinary team discussion was held with members from the Infectious Diseases Department, Obstetrics Department, Neonatal Department and Department of Critical Care Medicine. Obstetricians considered that in patients with acute liver damage and thrombocytopenia, the possibility of conditions such as HELLP syndrome and acute fatty liver during pregnancy should be considered. The infectious disease team found that liver function was severely abnormal. Further, the team concluded that since the patient had no history of chronic hepatitis, acute hepatitis should be considered, and the causes of hepatitis should first consider the damage of liver function caused by novel coronavirus. At the same time, autoimmune hepatitis and other conditions related to viral hepatitis (hepatitis A virus, hepatitis E virus, cytomegalovirus (CMV), Epstein-Barr virus (EBV)) should be excluded. The Department of Critical Care Medicine considered that if the disease progresses further, the patient might suffer from liver failure, systemic organ failure, disseminated intravascular coagulation, fetal death in the uterus, and in serious cases, it might endanger the life of the mother and child. However, the neonatologist considered that the gestational age of the patient was only 24+4 weeks. Therefore, if the pregnancy would be terminated at that stage, the fetus would not survive.
Initial findings led to a preliminary diagnosis of COVALI, with differential considerations, including HELLP syndrome, acute fatty liver of pregnancy, viral hepatitis, and thrombocytopenia associated with pregnancy at 24+4 weeks of gestation. Finally, we decided to focus on improving the patient’s liver function, attempt to normalize all vital parameters and continue to monitor the relevant indicators to consider pregnancy termination in case of further deterioration.
Final diagnosis
We established a diagnosis of COVID-19 and COVALI at 24+4 weeks of gestation as well as pregnancy complicated with thrombocytopenia.
Treatment
The patient underwent hepatoprotective treatments, including reduced glutathione level, 1200 mg qd ivdrip, compound glycyrrhizin 3 tablets tid po, adenosylmethionine 1 g tid po, and polyene phosphatidylcholine 465 mg qd ivdrip to support liver function and treat jaundice. Despite these interventions, the liver injury continued to worsen.
After the multidisciplinary consultation, intensive hepatoprotective therapy and hepatocyte regeneration therapy (ursodeoxycholic acid 150 mg tid po) were initiated as per the recommendation of the internal medicine physicians. Informed consent for treatment and data publication was obtained from the patient.
Outcome and follow-up
Following 3 days of liver protection treatment, the patient’s liver function began to improve, with AST and ALT levels decreasing to 196 and 564 U/L, respectively. At the time of discharge, these levels had dropped to <100 U/L. She was successfully discharged on the day 10. The patient was monitored with weekly follow-up visits for the first month and monthly thereafter. Follow-up blood tests confirmed normalization of liver function by the third week after hospital discharge. At 38 weeks and 6 days, she delivered a healthy male infant via vaginal delivery, with an Apgar score of 9–10. Both prenatal and postpartum examinations showed normal liver function, and no abnormalities were observed during the 42-day postpartum follow-up.
The reporting of this study conforms to the Case Report (CARE) guidelines. 10
Discussion
We report the case of a patient with severe COVID-19 during the second trimester of pregnancy, resulting in acute liver injury and thrombocytopenia. While the patient initially presented with symptoms consistent with a respiratory infection, laboratory findings on admission indicated liver dysfunction. The rapidly worsening condition and decreasing platelet count initially necessitated obstetric consultations that suggested pregnancy termination to avoid potential complications such as HELLP syndrome and other COVID-19–related issues. However, the patient’s liver function gradually improved after she was administered hepatoprotective therapy and hepatocyte regeneration treatment, as recommended by an internist. Further tests ruled out other liver diseases and pregnancy-related hypertension, excluding diagnoses such as HELLP syndrome and acute fatty liver of pregnancy (AFLP), thus supporting the diagnosis of COVALI.
In this case, the patient’s condition improved with respect to both liver injury and thrombocytopenia with conservative management for COVID-19, without the need for early delivery, as is typically required in perinatal liver diseases. This report contributes to the growing body of evidence raising concerns about the potential adverse maternal outcomes of COVID-19 during pregnancy.
COVALI is more likely to occur in patients with severe COVID-19. A prospective cohort study found that during hospitalization, the liver enzyme levels of 87.5% of pregnant patients with severe COVID-19 pneumonia became abnormal although their baseline levels had been normal. In addition,, pregnant patients with liver injury experienced more severe disease, with two-thirds of this cohort ultimately succumbing to COVID-19–related lung complications.11,12 Liver injury is a rare but potentially serious complication in pregnancy. The differential diagnosis for hepatic dysfunction in pregnant women includes pregnancy-related liver diseases, such as pre-eclampsia/eclampsia, HELLP syndrome, acute fatty liver of pregnancy, and intrahepatic cholestasis, as well as autoimmune, viral, and nonalcoholic steatohepatitis. Given the high mortality rate associated with perinatal liver disorders, early delivery is often necessary to ensure the health survival of the mother. 13 Given its potential to impact decisions regarding delivery, COVALI during pregnancy holds significant clinical importance. 14
Most extrapulmonary injuries caused by COVID-19 during pregnancy, including COVALI, are associated with endothelial damage, inflammation, and abnormal coagulation. Preeclampsia is more common in pregnant individuals with COVID-19 than in those without COVID-19, suggesting that the pathophysiology of COVALI may mirror or exacerbate that of obstetric hepatobiliary disorders.15,16 Alpha-1-antitrypsin, an enzyme that can help prevent SARS-CoV-2 infection and protect endothelial cells from oxidative stress during pregnancy, may be linked to the increased risk. The levels of this enzyme are reduced in pregnant patients with pre-eclampsia.17,18 The upregulation of ACE-2, which is highly expressed in the placenta and contributes to blood pressure regulation by controlling systemic vascular resistance, may also contribute to the risk. 19 This suggests that the enhanced ACE-2 activity in the endothelium of pregnant women makes the placenta a potential target for COVID-19. Despite their similarities, COVID-19 and HELLP syndrome can be distinguished using several criteria. In HELLP syndrome, chemical analysis shows hemolysis, low levels of placental growth factor (PlGF), and a high sFlt-1/PlGF ratio, regardless of the presence of clinical symptoms, such as proteinuria and hypertension. 11 Although the patient presented with thrombocytopenia and elevated liver enzymes, there was no evidence of microvascular hemolysis, and the platelet count remained stable over time.
AFLP is a rare but life-threatening condition that typically occurs in the third trimester. It is thought that preeclampsia, HELLP syndrome, thrombocytopenic purpura, and AFLP may be part of the same disease spectrum. 20 AFLP can be challenging to diagnose due to its varied clinical presentations and non-specific symptoms. It shares many features with conditions such as HELLP syndrome, viral hepatitis, and obstetric cholestasis. Therefore, ruling out other causes remains the most effective diagnostic approach. In contrast to HELLP syndrome, AFLP is associated with more significant coagulation issues and liver enzyme abnormalities. However, our patient did not exhibit key symptoms such as pruritus, digestive issues (vomiting, abdominal pain, polydipsia/polyuria), hypoglycemia, hyperuricemia, elevated blood ammonia, leukocytosis, coagulation dysfunction, or abnormal ultrasound findings.
In addition to severe conditions such as HELLP syndrome and AFLP, it is important to consider liver damage caused by immune factors, such as autoimmune hepatitis, as well as viral hepatitis from other infections, such as EBV and CMV infections. These conditions should be ruled out using virological testing. On the third day of illness, our patient underwent the necessary tests, all of which showed negative results. Successful outcomes in such cases require a multidisciplinary approach for managing and treating severe COVID-19 during pregnancy. Pregnant women should be regarded as a particularly vulnerable population in the context of COVID-19 and given special care to prevent complications.
Conclusion
This report describes the case of a 26-year-old primigravida who presented with respiratory symptoms and acute severe liver injury, later confirmed to be caused by the novel coronavirus. While hepatic damage is common in pregnant women infected with COVID-19, it rarely progresses to acute hepatitis. The disease progression in this case aligned with the typical course of Omicron variant infection, suggesting that liver protection treatments for COVALI can be effective, especially in pregnancy. We hypothesize that early identification of COVID-19 in pregnant women could help clinicians initiate timely treatment, potentially preventing complications such as HELLP syndrome and COVALI.
Footnotes
Acknowledgements
The authors thank the research staff for the recruitment of participants and retrieval of data from the medical records. We also thank all study participants.
Author contributions
All authors contributed to the study conception and design. Nan Yu collected and analyzed the data. Nan Yu and Yiyao Huang wrote the first draft of the manuscript. Ling Feng and Qian Liu revised the manuscript. All authors reviewed and approved the final manuscript.
Data availability statement
The data supporting the findings of this study are available within the article.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This study was supported by the Natural Science Foundation of Xinjiang Uygur Autonomous Region (No. 2021D01F20).
Informed consent statement
This study has been approved by the Ethics Committee, and patient consent was obtained for the publication of this case report.
