Abstract
Atrial myxoma is the most common primary benign tumor of the heart, with over 75% occurring in the left atrium. The migration of myxoma cells can lead to rare neurological complications such as intracranial aneurysms, embolic strokes, and arteriovenous malformations. The most common neurological manifestations in such patients include seizures, nonspecific headaches, and cerebrovascular events. Herein, we present a rare case of a middle-aged man in his early 50s with metastatic myxomatous multiple fusiform aneurysms, who initially presented with the clinical manifestation of intracerebral hemorrhage. The patient underwent atrial myxoma resection, whereas the intracranial multiple fusiform aneurysms were managed conservatively. Four years later, digital subtraction angiography revealed progressive enlargement of the aneurysm in the A3 segment of the left anterior cerebral artery. We present the pathological report of the atrial myxoma and imaging findings showing the changes in the aneurysms, along with a review and summary of the relevant literature. This report offers new insights into treatment options for cases of atrial myxoma complicated with intracranial multiple aneurysms. This is the first reported case of a male patient in China presenting with intracerebral hemorrhage as the initial manifestation of atrial myxoma combined with multiple fusiform aneurysms.
Keywords
Background
Atrial myxoma is the most common primary benign tumor of the heart in adults, accounting for 30%–50% of all primary cardiac tumors, with an annual incidence of approximately 0.5–1.0 case per million people. 1 It primarily affects individuals aged 30–60 years, with a higher prevalence among women.2,3 Patients with atrial myxoma usually have a good prognosis after complete resection of myxoma, with a 20-year survival rate of approximately 85%. However, due to the migratory nature of myxoma cells, this condition may lead to the formation of lesions in migrated sites.3,4 The migration of myxoma cells to the central nervous system often results in complications such as intracranial aneurysms, embolic strokes, and arteriovenous malformations.5,6 Myxoma-related aneurysms are typically multiple and mostly fusiform in nature. According to a recent literature review, a total of 55 cases of atrial myxoma complicated with multiple intracranial aneurysms have been reported, most of which were reported in women. Among these 55 cases, only eight men presented with cerebrovascular events, and only seven cases involved aneurysms in the anterior cerebral artery (ACA) and middle cerebral artery (MCA). 7 Herein, we present the case of a man with metastatic multiple fusiform intracranial aneurysms involved in the ACA and MCA associated with atrial myxoma, who initially presented with intracerebral hemorrhage. We assessed the status of the aneurysms after 4 years of conservative treatment. Furthermore, we discussed the therapeutic options for patients with metastatic multiple fusiform intracranial aneurysms associated with atrial myxoma, drawing on a literature review.
Case report
A middle-aged man in his early 50s was admitted to Union Hospital affiliated with Tongji Medical College of Huazhong University of Science and Technology in October 2020 due to “left upper and lower extremity weakness for 1 day.” Upon admission, he was in a state of confusion, with a blood pressure level of 180/102 mmHg, and had no seizure. He had a Glasgow Coma Scale (GCS) score of 12/15 and a 10-year history of primary hypertension, with the highest systolic blood pressure level reaching 190 mmHg. The muscle strength of his left extremity was grade 0, and that of his right extremity was grade IV+. Cranial computed tomography (CT) at the time of admission indicated a hemorrhage in the patient’s right parietal lobe (Figure 1). He underwent minimally invasive drilling and hematoma drainage surgery under local anesthesia during hospitalization, and relevant examinations were performed. Computed tomography angiography (CTA) revealed segmental dilation in the A3 segment of the left ACA and its branch arteries, with lengths of approximately 2.2 cm and 9.0 mm, respectively, and a maximum width of 4 mm. Multiple small fusiform aneurysms were visible in the parietal–occipital branches of the bilateral middle cerebral arteries, with the largest aneurysm measuring approximately 6.4 × 3.0 mm. Furthermore, CTA revealed the formation of calcified plaques in the intracranial segments of the bilateral internal carotid arteries, accompanied with mild luminal stenosis (Figure 2). During hospitalization, transthoracic echocardiographic examination revealed a slightly hyperechoic mass attached to the interatrial septum near the entrance of the inferior vena cava in the left atrium, with a size of approximately 6.8 × 3.6 cm and a stalk width of approximately 2.6 cm. The mass swung between the left atrium and the mitral valve orifice within the cardiac cycle, and the blood flow through the mitral valve orifice was slightly accelerated during diastole. The diagnosis was a left atrial myxoma. The patient was transferred to the cardiac surgery department for surgical treatment. Coronary artery CTA revealed a low-density mass shadow in the left atrium, with a size of approximately 51.3 × 39.3 ×33.7 mm. During diastole, the mass could pass through the valve orifice into the left ventricle. This mass was considered a myxoma. Under general anesthesia, a left atrial myxoma resection was performed. The findings of intraoperative echocardiography were consistent with those of preoperative echocardiography and coronary artery CTA. During surgery, two irregularly shaped, grayish-red tissues were resected, with a combined size of approximately 7.5 × 5.0 × 3.0 cm. Both tissues exhibited pedunculated structures, and their surfaces were cauliflower-like with a mucinous texture. Postoperative pathology confirmed the diagnosis of myxoma (Figure 3). Postoperative transesophageal echocardiographic examination showed no obvious abnormal echoes in the left atrium. The patient recovered well after surgery and was left with residual weakness in the left extremity. He was discharged, with no follow-up visits at the hospital during the 4-year post-discharge period. Four years later, he was admitted to our department for a follow-up examination with a diagnosis of “multiple intracranial aneurysms.” He had a history of hypertension, smoking, and alcohol consumption. The patient was conscious, with a GCS score of 15/15. His left extremity movement was restricted. Muscle strength grading in all four extremities was as follows: (a) left upper extremity, grade IV; (b) left lower extremity, grade IV; (c) right upper extremity, grade V; and (d) right lower extremity, grade V, with normal muscle tone. Cranial CT indicated a hypodense area in the right temporal–parietal lobe (Figure 4). Digital subtraction angiography (DSA) revealed tortuosity of the bilateral carotid and vertebral arteries, indicating atherosclerosis. DSA images showed a dissecting aneurysm in the left ACA (A2–A3 segment) and bilateral MCA (M4 segment). A localized fusiform aneurysmal dilation was observed in the A3 segment of the left ACA, with a size of approximately 28.3 × 4.5 mm on the right side and 15.4 ×3.6 mm on the left side. Additionally, two isolated small strip-like fusiform dilations were observed at the distal end of the M4 segment of the left MCA, with sizes of approximately 6.5 × 2.7 and 4.1 × 2.1 mm, respectively (Figure 5). Multiple intracranial arterial atherosclerotic changes were observed. Due to limited treatment options and unclear efficacy of the current relevant treatment plans, the patient opted for conservative treatment after a comprehensive evaluation of surgical options and associated risks and was discharged from the hospital. The patient will return to our hospital within 6 months for a follow-up cerebral vascular examination.
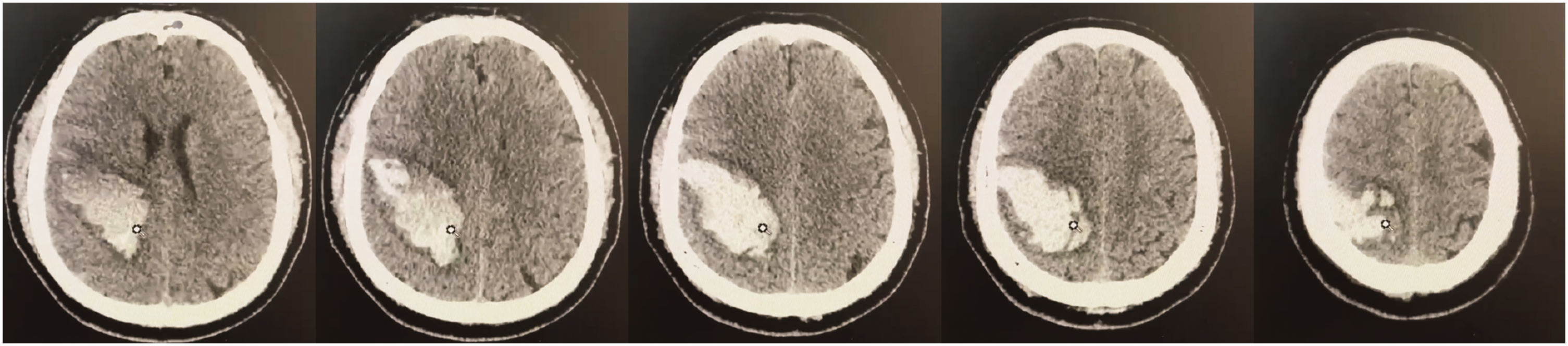
Cranial computed tomography at the time of admission indicated a hemorrhage in the patient’s right parietal lobe.

Computed tomography angiography demonstrating multiple intracranial aneurysms and the formation of calcified plaques in the intracranial segments of the bilateral internal carotid arteries, accompanied with mild luminal stenosis.

Confirmation of the diagnosis of atrial myxoma via histopathological findings, demonstrating myxoid matrix and scattered tumor cells.

Cranial computed tomography 4 years after intracerebral hemorrhage suggesting a hypodense area in the right temporal–parietal lobe.

Digital subtraction angiography showing a dissecting aneurysm in the left anterior cerebral artery (A2–A3 segment) and bilateral middle cerebral artery (M4 segment). Aneurysms were circled in red.
The reporting of this study conforms to the Case Report (CARE) guidelines. 8 Written informed consent was obtained from the patient for publication of his anonymized data. This report was approved by the clinical ethics committee of Central Theater Command General Hospital of the People’s Liberation Army (NO.[2024]077-01).
Discussion
Herein, we report the first case of a male patient in China presenting with intracerebral hemorrhage as the initial clinical manifestation of atrial myxoma combined with multiple fusiform aneurysms. Specifically, this case involved aneurysms of the ACA and MCA. The patient was initially admitted to the hospital due to intracerebral hemorrhage as the primary clinical manifestation. Subsequent examinations revealed that the patient had an atrial myxoma accompanied with multiple intracranial fusiform aneurysms. During hospitalization, he underwent resection of the atrial myxoma, whereas the intracranial multiple fusiform aneurysms were managed conservatively with follow-up observations. Four years later, DSA revealed a progressive increase in the size of the aneurysm in the A3 segment of the left ACA. Based on this case, we conducted a literature review of myxomatous metastatic aneurysms from the perspectives of pathogenesis, early diagnosis, and treatment options.
Myxoma cells are believed to originate from pluripotent mesenchymal cells. 9 Although myxomas are typically benign tumors, they often exhibit malignant phenotypes because of their fragile or villous characteristic. 10 Detached particles from some myxomas can metastasize to other locations, causing lesions. Moreover, these particles may cause embolic occlusion of intracranial vessels, damage the vessel walls, penetrate the injured cerebral vascular endothelium, and proliferate within the intimal layer of the vessel wall, resulting in aneurysmal dilation. 5 This theory is supported by histopathological studies, which have demonstrated the presence of myxoma cells within the lumen of resected cerebral aneurysm tissues. 11
Although surgical resection of atrial myxomas can reduce the risk of subsequent vascular embolism, it does not eliminate the risk of delayed cerebral aneurysm formation. This is similar to the mechanism of intracranial mycotic aneurysm formation caused by septic emboli, where mycotic aneurysms typically develop at intracranial arterial branches following vascular wall damage. 12 In addition, polycystic kidney disease and hypertension can contribute to the development of multiple cerebral aneurysms. 13 Furthermore, heparinization during atrial myxoma resection may lead to vascular endothelial injury, and further research is warranted to determine whether this phenomenon exacerbates the progression of multiple fusiform intracranial aneurysms. 14
For patients with cardiac myxoma, we need to conduct a comprehensive examination and follow-up for a long time postoperatively to monitor the possible occurrence of cerebral aneurysms and other complications. Similarly, for patients with multiple aneurysms, embolism, metastatic lesions, and long-term heart burden, we should be alert to the possibility of atrial myxoma for timely detection and surgical treatment.5,7
Due to the rarity of atrial myxoma, the optimal treatment method has not yet been determined. Conservative treatment is adopted in approximately 60% of the cases, and regular follow-up observations have found that myxomatous aneurysms can remain static for over 15 years. 15 Among patients undergoing conservative treatment, 80% of aneurysms remain stable or even shrink, whereas 20% of aneurysms show enlargement. 16 For continuously growing aneurysms, chemotherapy can be administered, and combination therapy with whole-brain radiotherapy or local radiotherapy followed by chemotherapy has been reported to exert positive effects. 9 Frameless stereotactic radiosurgery is another alternative with minimal invasiveness and low toxicity. 9 Given the multiple and fusiform characteristics of cerebral myxomatous aneurysms, surgical clipping and embolization are generally not considered. In a previous study, some large aneurysms were treated with open craniotomy aneurysm clipping. 17 For patients treated with endovascular coil embolization, there is a possibility of further aneurysm enlargement. 18 Vascular bypass combined with aneurysm isolation is also an optional surgical treatment. 19 Furthermore, for continuously growing or ruptured aneurysms in which the parent artery supplies noncritical functional areas, parent artery occlusion can be considered. 20 Further observation and large-scale studies are needed to validate the short- and long-term efficacy of different treatment options. 7 In the current case of cerebral myxomatous aneurysm, the patient received conservative treatment, and a progressive increase was observed in the size of the aneurysm in the A3 segment of the left ACA. Therefore, further research and exploration are required to establish standardized treatment protocols for such patients.
Conclusion
Herein, we reported a very rare case of metastatic multiple fusiform aneurysms caused by atrial myxoma and offered new insights into treatment options for cases of atrial myxoma complicated with intracranial multiple aneurysms.
Footnotes
Acknowledgements
The authors would like to thank the patient for letting us publish his case and all the contributors for their input and work.
Author contributions
Conception and design: HH and YGJ; Acquisition of the materials and data: AXF, HYF, WK, ZJ, and LK; Drafting of the manuscript: XM and WY; Critical revision of the manuscript for important intellectual content: HH and YGJ. All authors were involved in the clinical care of the patient. All authors read and approved the final manuscript.
Data availability statement
The data used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was supported by Key Discipline Construction of Central Theater General Hospital–Neurosurgery Project (1749).
