Abstract
Objective
To evaluate the efficacy and adverse effects of phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis.
Methods
Electronic searches were conducted based on PubMed, Cochrane Library, EMBASE, and Web of Science using Medical Subject Headings terms and relevant keywords. Using response rates as the primary outcome and adverse effects as the secondary outcome, statistical analysis was performed via Stata 15.1 SE, with Cochrane Q and
Results
The analysis included six randomized controlled trials involving 4681 children with atopic dermatitis. Phosphodiesterase 4 inhibitors were associated with significantly higher response rates than vehicle controls (odds ratio = 2.01, 95% confidence interval: 1.58 to 2.56, p < 0.001,
Conclusion
Our meta-analysis revealed that children with atopic dermatitis treated with phosphodiesterase 4 inhibitors experienced higher remission rates and comparable adverse effect rates compared with vehicle controls. Currently, crisaborole appears to be the optimal choice for balancing safety and efficacy. As research in this area is still in its early stages, further high-quality trials are essential for establishing standards of clinical care.
INPLASY registration number: INPLASY202520121.
Keywords
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disease that may be accompanied with an increased risk of skin and systemic infections. 1 AD affects approximately 20% of children and 3% of adults. 2 A study showed that approximately 85% of AD occurs in children, with 30% of the patients continuing to be affected into adulthood. 3 The incidence of AD is continuously rising worldwide, especially in urbanized countries in northern latitudes. 4 Moreover, AD imposes considerable economic and social burdens on individuals, families, and public health systems. A study in the United States revealed that AD leads to healthcare costs of $4.2 billion per year, with personal healthcare costs being 28.3%–67.9% higher for people with AD than for those without AD. 5 The high disease burden of AD appears to stem primarily from limited treatment options.
Based on clinical guidelines for AD, there is currently no ideal standard care plan for AD, and treatments tailored to individual needs is the most optimal option. 6 Topical intervention, as a first-line treatment option, is currently the main treatment approach for AD. However, the potential local and systemic side effects of the existing topical agents limit their clinical use. 7 Topical calcineurin inhibitors, as second-line anti-inflammatory drugs for AD, work on the principle of inhibiting calcineurin-dependent T cell activation, reducing epidermal dendritic cell activation, and inhibiting mast cell activation, thereby achieving long-term maintenance and specific site applications. 8 However, the black box warning—topical use of calcineurin inhibitors may lead to malignancy—has reduced patient adherence to treatment. 9 Therefore, there is an urgent need for new local therapies to improve the current high-risk status of AD treatment.
Phosphodiesterase 4 (PDE4) inhibitors, a new class of nonsteroidal anti-inflammatory drugs, are one of the most promising new drugs in current AD and psoriasis treatment. By degrading cyclic adenosine phosphate, PDE4 acts as a crucial regulator of inflammatory cytokine production in AD.10,11 PDE4 inhibitors regulate several aspects of the allergy/inflammation process: (a) inhibiting the migration of skin dendritic cells, which is accompanied with the inhibition of MMP-9 activity in the epidermis and dermis and (b) impairing cytokine secretion (tumor necrosis factor α, interleukin (IL)-1β, and IL-12) in human and mouse dendritic cells.12,13 Meanwhile, in vitro experiments have confirmed the inhibitory effect of PDE4 inhibitors on T cell activation,14,15 which can reduce Th1 and Th2 cytokines in vitro14,15 and in the skin of inflammatory mice.16,17 The secretion of proinflammatory mediators by activated keratinocytes is also affected by PDE4 inhibitors. 18 Thus, reduced secretion of chemokines leads to a subsequent influx of inflammatory cells (macrophages, T cells, and neutrophils), resulting in a diminished inflammatory response in allergic skin diseases.16,19,20 The oral PDE4 inhibitor apremiast has been approved for treating moderate-to-severe plaque psoriasis and psoriatic arthritis; however, dose titration is required to avoid gastrointestinal side effects (nausea and diarrhea) due to the inhibitory effect of PDE4 in nontarget tissues.21,22 In contrast, topical PDE4 inhibitor formulations may address the need for targeted inhibition of inflammation in skin diseases while avoiding unwanted side effects. There are currently three common PDE4 inhibitors: (a) crisaborole, approved by the US Food and Drug Administration and the European Medicines Agency for treating patients with AD aged over 2 years 23 ; (b) difamilast (also known as MM36 or OPA-15406), a new selective PDE4 inhibitor that has shown significant efficacy and safety in mild-to-moderate AD in phase II trials among children and adult patients in the United States and Japan24,25; and (c) topical E6005, which has been shown to improve topical rashes and pruritus in children and adults with AD without any serious side effects, although the number of relevant studies is limited. 26
Although previous studies have evaluated the use of different types and concentrations of PDE4 inhibitors to treat patients with AD across different demographics, its effectiveness remains controversial. A randomized controlled trial (RCT) published in 2016 found no significant difference in reduced severity scores between the PDE4 inhibitor E6005 and loaded therapy 27 ; however, a large-sample static global evaluation clinical trial showed a higher success rate in patients treated with crisaborole than in those treated with loaded therapy. 28 This meta-analysis aimed to quantitatively synthesize the current evidence on the efficacy and safety of local PDE4 inhibitors in the treatment of children with mild-to-moderate AD, hoping to organize the current situation and provide more details for future clinical decision-making.
Methods
Literature search
Using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines,29,30 potential article search strategy was designed according to the PICOS (P: participants, I: intervention, C: control, O: outcome, S: study design) principle. Until July 2023, articles published in PubMed, EMBASE, Web of Science, and the Cochrane Library databases were searched, using the Medical Subject Headings terms “Phosphodiesterase 4 Inhibitors” and “Dermatitis, Atopic” (Table S1). Two researchers conducted the literature search and selection independently. Any disagreements in the process were resolved through discussion or access by the third researcher. We did not prospectively register this trial, but we have currently registered it retrospectively at INPLASY (registration number INPLASY202520121).
Eligibility criteria
Articles were selected using the following inclusion criteria: (a) studies targeting patients with AD aged below 18 years; (b) those involving an intervention group that received any type of topical PDE4 treatment; (c) those involving a control group that received a placebo, conventional treatment, or any other anti-AD interventions; (d) study endpoints included the treatment response rate and adverse effect (AE); (e) studies involving RCTs; and (f) studies with full text available.
The exclusion criteria were as follows: (a) studies using combined interventions (e.g. PDE4 + conventional treatment vs. conventional treatment); (b) reviews, animal trials, single-arm studies, chapters in handbooks, case reports, dissertations, editorials, and conference papers; (c) studies without international peer review; and (d) studies with duplicate data.
Although systematic reviews and meta-analyses do not necessitate ethical approval, we ensured that only original studies that adhere to ethical standards were included.
Data extraction
Data extraction was conducted by two independent investigators. A third independent reviewer intervened to resolve disagreements. Data included the authors of the articles, publication year, study design, study location, sample size, female proportion, mean age, diagnosis criteria, intervention, control, treatment duration, and study outcomes. The primary outcome was the response rate of PDE4 treatment, and the secondary outcome was the AE. Response rate was defined as the number of people responding to PDE4 therapy, as reflected in the clinical scales or as provided directly in the original text. AEs were defined as any observable poor clinical prognosis following PDE4 treatment. Disagreements were resolved by discussion among all authors.
Quality assessment
The Cochrane risk of bias tool 31 was used to assess the risk of bias and the quality of evidence. The quality assessment was conducted by two independent researchers. Risk of bias was assessed across five different areas, with the results expressed as “low risk of bias,” “some risk,” or “high risk of bias.” Evaluations in each area influence the overall bias risk judgment.
Statistical analysis
All analyses were performed using Stata 15.1 SE version (StataCorp; Texas, USA). Odds ratios (ORs) and their corresponding 95% confidence intervals (CIs) were used to compare the outcomes. To ensure consistency, changes (endpoints, baseline values) in all study endpoints were calculated for pooled analysis. Cochran’ s Q-test and
Results
Study selection and study characteristics
The study selection process is presented in Figure 1. The initial search yielded 1733 studies with 687 duplicate records. After reviewing the titles and abstracts, 458 records were excluded due to the absence of relevant information, and 1 record was excluded after reviewing the full text of 7 articles. Finally, six studies27,32–36 were included in this meta-analysis.

Flowchart of the study selection.
Table 1 presents the characteristics of the included studies. Overall, 4681 patients with AD were included in this meta-analysis, with 3030 patients in the PDE4 groups and 1651 in control groups. According to five single-center and one multicenter studies, five countries were included in this meta-analysis, including America, Germany, Japan, Canada, and Australia. All included studies were RCTs with vehicle controls. Three studies selected crisaborole for PDE4 treatment, two studies selected OPA-15406 (0.3% or 1%), and one study selected E6005 (0.2% or 0.05%). The Hanifin and Rajka criteria 37 were selected by four included studies as the diagnosis criteria for AD. Two studies used the Japanese Dermatological Association’s criteria. 38 The mean age of the included patients with AD was 13.5 years. The mean female proportion in PDE4 groups was 53.74%. The PDE4 treatment duration ranged from 2 weeks to 1 month.
Characteristics of the included studies.

Study quality
Figure S1 presents the results of the risk of bias assessment. Five of the six included RCTs were evaluated as having low potential bias.32–36 The main considerations for the remaining study 27 were deviations from the intended intervention and potential bias in the measurement of the outcomes.
Response rate
Six studies with eight trials have examined the response rate of PDE4 in patients with AD. Meta-analysis showed that PDE4 produces 2.01 times higher response rate than vehicle controls (OR = 2.01, 95% CI: 1.58 to 2.56, p < 0.001,

Forest plot of the response rates.

Sensitivity analysis of the response rates.
Subgroup analysis, which evaluated various PDE4 inhibitors and treatment durations, demonstrated that both covariates reduced the heterogeneity of the overall analysis to varying degrees. Furthermore, the findings from each subgroup were consistent with the results of the original pooled analysis (E6005: OR = 3.67, 95% CI: 1.12 to 12.05, n = 1; OPA-15406: OR = 3.31, 95% CI: 2.17 to 5.05,
AE incidence
Four studies with seven trials reported the AE incidence after treatment. Compared with vehicle controls, no significant difference was observed in the AE incidences in PDE4 groups (OR = 1.07; 95% CI: 0.87 to 1.30, p = 0.536,

Forest plot of adverse effects.
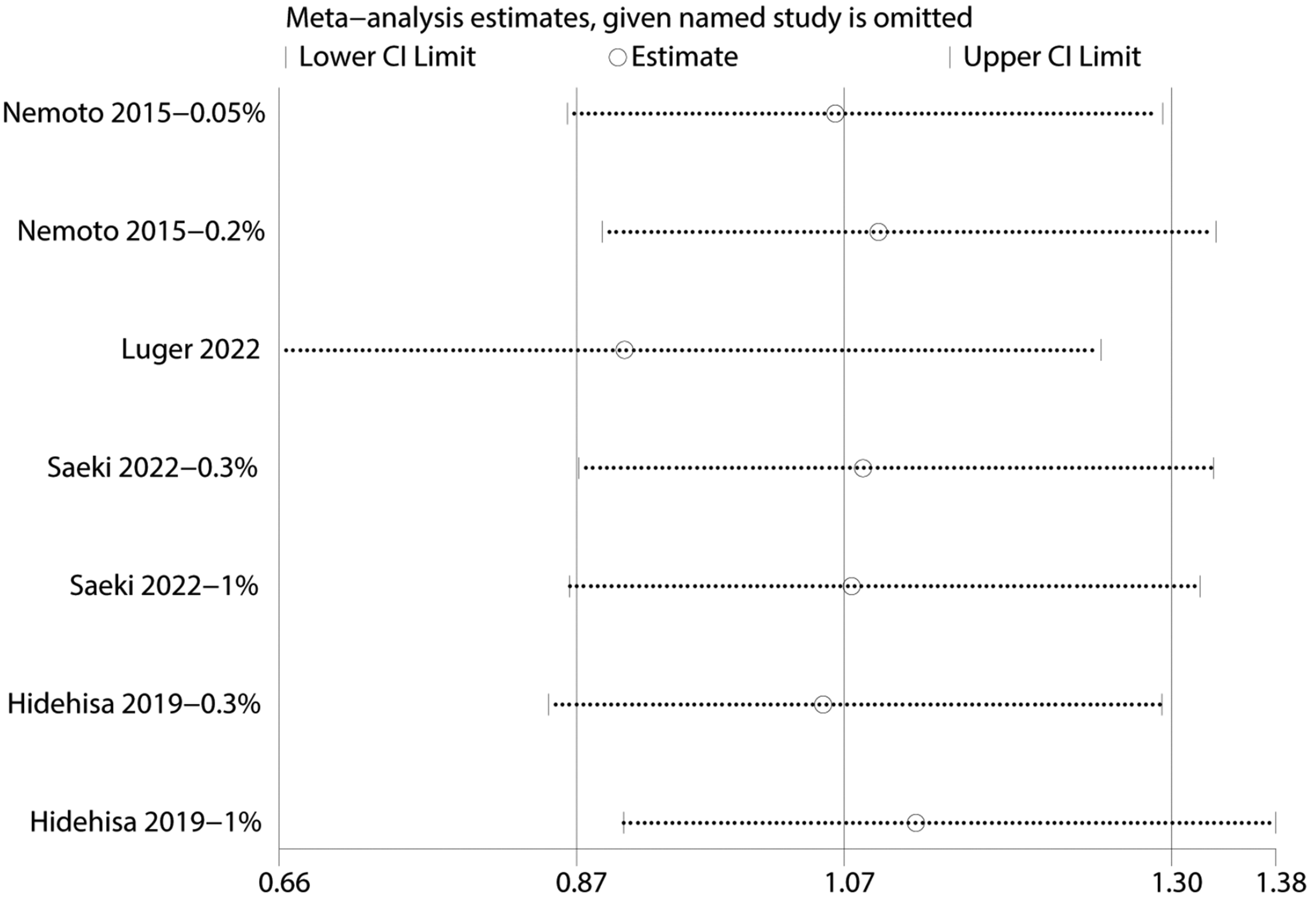
Sensitivity analysis of adverse effects.
Subgroup analysis showed that the pooled results from certain PDE4 inhibitors and treatment duration groups could further reduce inter-study heterogeneity compared with the original pooled analysis (OPA-15406: OR = 0.94, 95% CI: 0.67 to 1.31,
Publication bias and sensitivity analysis
The results of sensitivity analyses showed that removing any of the studies did not affect the results of this study (Figures 3 and 5, Supplementary Materials).
The funnel plot showed that most of the trials fell on the right side of the combined effect axis (Figure S4A-S4B). Egger’s test further confirmed the existence of publication bias (p = 0.047) (Supplementary Materials). The trimming and padding of the response rates showed no reversal of the pooled results after filling in the four potentially omitted studies. The results of the heterogeneity test after the trim-and-fill method were consistent with and slightly lower than the original meta-analysis results (Q = 30.284, p = 0.001; OR = 1.656, 95% CI: 1.273 to 2.154, p = 0.000; Figure S5). The results of the trim-and-fill method showed that the existing publication bias had a limited effect on the direction of the combined analysis results. Considering the current research landscape in this field and the information obtained from the literature review, we believe that the existing publication bias is likely attributable to a lack of uniform definitions and measurement criteria within this research area.
Discussion
This systematic review and meta-analysis evaluated recent RCTs on the response rate and AE incidence of PDE4, compared with vehicle controls, in children with mild-to-moderate AD. The results of our study demonstrated higher response rates of all three PDE4 inhibitors and the same AE incidence rate compared with vehicle controls. Meanwhile, after considering the number and quality of supporting studies, the response rate appeared to be the highest in children treated with 4 weeks of OPA-15406 treatment. However, from the perspective of safety, crisaborole is still the best choice for current pediatric AD treatment. Although the observed publication bias in the reporting of response rates did not impact the overall pooled results, the limited number of studies and the high heterogeneity among them suggest that these pooled analyses should be interpreted with caution. Additional high-quality studies are necessary to establish a standard of clinical care in this area. Although our study is the first known review of PD4 therapy in pediatric patients with AD, the results of our analysis align with previous studies in adult patients with AD. Compared with vehicle controls, higher response rates and similar safety profiles of PDE4 inhibitors were observed in adults, but this beneficial effect was noted only with the PDE4 inhibitors crisaborole and AN2898. 39 Another meta-analysis of nonsteroidal roflumilast cream (0.15%) indicated that a significantly higher number of children, adolescents, and adults reached the primary endpoint of achieving at least a grade 2 improvement on the Validated Investigator Global Assessment compared with the solution cream group. 40
Studies in the 1980s have shown that increased PDE activity is associated with inflammatory dysregulation in patients with AD.41–43 Further speculation about genetic abnormalities was raised after the detection of increased PDE activity in cord blood cells of newborns with atopic parents. 44 Since then, multiple in vitro experiments have shown that the production of IL-10, IL-4, and prostaglandin E2 in atopic leukocytes is reduced after exposure to high concentrations of PDE inhibitors, 41 which primarily promote the expansion, maturation, and functional activation of inflammatory cells. These inflammatory cells include eosinophils, allergen-specific B cells, and mast cells. If not inhibited, these activated B cells can promote the recombination of immunoglobulin genes in a sequential manner to produce allergen-specific immunoglobulin E antibodies, which will activate mast cells, basophils, and eosinophils to mediate allergic inflammation via FcϵRI. 45 Subsequent in vivo experiments have confirmed that inhibition of PDE4 activity can directly affect IL-4 production or Th2 cell balance by reducing the release of cytokines and mediators. 41 The abovementioned clinical anti-inflammatory effects are the basic principles for the development and application of PDE4 inhibitors.
Crisaborole is the first PDE4 inhibitor on the market that has been shown to improve disease severity in patients with AD. Although its response rate advantage, compared with vehicle controls in this study and a previous study, 46 is inferior to other PDE4 inhibitors, its good safety supports its marketing. Most of the crisaborole-related AEs observed in previous studies were mild-to-moderate, including application site reactions such as pain and dermatitis.47–50 Although the incidence of these AEs was higher in the treatment group than in the drug-loaded control group, most patients experienced symptom relief within 1 day. 28 Another safety and tolerability review with a follow-up of 12 months found that AD attacks (3.1%), application site pain (2.3%), and infection (1%) were the main AEs observed after crisaborole treatment. 51 The proportion of patients reporting at least one treatment-related AE in previous phase III and expansion trials was 65%. Therefore, given the importance of crisaborole in improving disease severity (including pruritus, erythematosis, exudation, abrasion, induration/plaque, and lichenization) 28 and safety in pediatric patients, it remains the ideal choice for mild-to-moderate AD.
Unlike crisaborole, 0.3% and 1% OPA-15406 demonstrated excellent treatment response rates in the current study as well as a previous network meta-analysis. 46 Two newly published phase III controlled trials showed that 0.3% and 1% difamilast ointment outperformed vehicle controls in twice-daily treatment for 4 weeks in Japanese children and adults with AD.33,52 The success rate of AD treatment at week 4 was as high as 44.6% in the 0.3% difamilast group. However, in terms of safety, the Japanese phase III trial showed that treatment-emergent adverse event (TEAE) occurred in 33% of patients in the 0.3% difamilast group, 34% in the 1% difamilast group, and 34% in the vehicle control group. In the 0.3% difamilast group, one patient developed a severe TEAE with worsening AD. 33 As safety and tolerability are critical factors, especially for pediatric patients, it is important to choose a drug with higher safety. Although the results of the published studies evaluating difamilast will be available, further RCT results with head-to-head comparisons with crisaborole or other AD-related inhibitors are required to demonstrate that difamilast can be used as a new treatment option for pediatric patients with AD.
Compared with the abovementioned two PDE4 inhibitors, the number of studies related to E6005 is limited, with only one trial 27 conducted in children. The results indicated that topical E6005 was safe and well-tolerated for 2 weeks, with no AE in the skin. The decrease in AD severity scores in patients treated with 0.2% E6005 was greater than that in the vehicle group (−45.94% vs. −32.26%), but the difference was not statistically significant. E6005 ointment was well-tolerated at 0.05% and 0.2% concentrations. There were no serious TEAEs. Overall, 20% of the participants in the vehicle group, 30% of the participants in the 0.05% group, and 3.1% of the participants in the 0.2% group reported at least one TEAE. There were also some limitations to the study that may limit the strength of the findings. Due to the different colors of various PDE4 ointment, these studies were not conducted in a double-blind manner. Thus, further studies are needed to provide more clinical evidence of the efficacy and safety of E6005 in the treatment of mild-to-moderate AD in children.
Limitations
This systematic review and meta-analysis had some limitations. Limited databases and publishing languages were included in our search strategy. This may be one of the reasons for the small number of included studies, resulting in only one study involving response rate analysis of crisaborole and E6005 subgroups. Second, due to the limited number of studies, only the number of respondents provided in the original text can be used as an indicator to measure the response rate. However, the different response rate assessment criteria used in the original text will inevitably become a potential source of heterogeneity. Most importantly, this study assessed the effect of treatment on disease severity using a simple measure of response rate, which is not comprehensive. For example, the Scoring Atopic Dermatitis and the Eczema Area and Severity Index, which considers objective signs and subjective experiences of patients, is a more reliable measurement tool for evaluating the severity of AD compared with response rate.
Conclusions
This meta-analysis revealed higher response rates and the same AE incidence rate of all three types of PDE4 inhibitors compared with vehicle controls. After considering the number and quality of supporting studies, the response rate appeared to be the highest in children treated with 4 weeks of OPA-15406 treatment. However, from the perspective of safety, crisaborole is still the best choice for current pediatric AD treatment. The limited cumulative evidence regarding the efficacy and safety of PDE4 inhibitors for the treatment of mild-to-moderate AD in children, alongside significant heterogeneity and publication bias among studies, necessitates a cautious interpretation of the results from combined analyses. High-quality clinical trials are essential for obtaining more meaningful clinical insights in this field.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251333654 - Supplemental material for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251333654 for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis by Long Wang, Lequ Zeng, Yuyu Wu, Min Zhong, Lizhen Zhang and Chen Li in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251333654 - Supplemental material for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605251333654 for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis by Long Wang, Lequ Zeng, Yuyu Wu, Min Zhong, Lizhen Zhang and Chen Li in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251333654 - Supplemental material for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis
Supplemental material, sj-pdf-3-imr-10.1177_03000605251333654 for Effectiveness and safety of topical phosphodiesterase 4 inhibitors in children with mild-to-moderate atopic dermatitis: A systematic review and meta-analysis by Long Wang, Lequ Zeng, Yuyu Wu, Min Zhong, Lizhen Zhang and Chen Li in Journal of International Medical Research
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
Conception and design: Long Wang, Chen Li; Administrative support: Lequ Zeng; Provision of study materials or patients: Lizhen Zhang; Collection and assembly of data: Yuyu Wu; Data analysis and interpretation: Min Zhong; Manuscript writing: all authors; Final approval of manuscript: all authors.
Consent for publication
Not applicable.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Funding
Not applicable.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
