Abstract
Localized scleroderma, or morphea, is a chronic fibrosing skin condition with limited therapeutic options, particularly for patients who are elderly, immunocompromized, or intolerant to systemic immunosuppressants. We present three adult patients with severe, treatment-refractory localized scleroderma who demonstrated rapid and substantial clinical improvement following initiation of topical roflumilast 0.3% cream under occlusion. All patients had failed or declined systemic therapy. Within 6–12 weeks, each showed marked reductions in inflammatory activity, pain, and skin induration, as measured by validated clinical scores (Localized Scleroderma Activity Index, Physician’s Global Assessment of Activity/Damage). No adverse events were reported. Roflumilast, a potent phosphodiesterase-4 inhibitor with anti-inflammatory and antifibrotic properties, may offer a well-tolerated, nonimmunosuppressive treatment option in morphea. This is the first reported case series using topical roflumilast in localized scleroderma and supports further investigation of phosphodiesterase-4 inhibition as a therapeutic strategy in fibrosing skin disease.
Introduction
Localized scleroderma (LS), or morphea, is a rare immune-mediated skin disease characterized by chronic inflammation, dermal fibrosis, and vascular dysfunction. 1 It presents in several subtypes—including circumscribed, linear, generalized, and pansclerotic—with deeper or widespread forms carrying risk of permanent tissue atrophy, joint contractures, and functional impairment. 2
Management depends on subtype, depth, and disease activity. First-line therapies for active, moderate-to-severe LS typically include systemic immunosuppressants such as methotrexate, with or without systemic corticosteroids, which have the strongest evidence for disease control. 1 Topical agents and phototherapy are generally reserved for superficial, limited disease, or used as adjuncts. 1 However, systemic therapies are often contraindicated or poorly tolerated in older, frail, or cancer-affected patients, highlighting the need for safer, effective alternatives. 3
Phosphodiesterase-4 (PDE4) inhibitors have demonstrated anti-inflammatory and antifibrotic activity in several dermatologic conditions through suppression of cytokine signaling and fibroblast activation. 4 Roflumilast, a potent topical PDE4 inhibitor approved for psoriasis, has shown strong local tolerability and rapid clinical efficacy. 4 Although oral apremilast has been reported in small case series for LS, topical PDE4 inhibition has not yet been explored. 5
We report three patients with severe LS—including generalized and checkpoint-inhibitor–associated subtypes—who experienced rapid and sustained improvement with topical roflumilast 0.3% under occlusion. This case series represents the first documented use of topical PDE4 inhibition in LS.
Case presentations
Patient 1
A 73-year-old woman with metastatic nonsmall cell lung cancer on pemetrexed, carboplatin, and pembrolizumab developed progressive violaceous erythema and induration of the lower limbs approximately 6 months after initiating immune checkpoint blockade. At presentation, she was wheelchair-bound due to severe pain (10/10) and stiffness, with a Localized Scleroderma Activity Index (LoSAI) of 16, Physician’s Global Assessment of Activity (PGA-A) of 90, and PGA of Damage (PGA-D) of 10 (Figure 1(a)). She had previously failed ultrapotent topical corticosteroids. A biopsy was performed to exclude eosinophilic fasciitis. Due to pancytopenia, hepatic dysfunction, and advanced malignancy, systemic immunosuppressants were contraindicated. She was initiated on roflumilast 0.3% cream under occlusion once daily, in combination with ultrapotent topical steroids while applications for off-label intravenous immunoglobulin, extracorporeal photopheresis, and tocilizumab were submitted. Upon follow-up 6 weeks later, she reported complete pain resolution and improved ambulation. Clinical scores improved markedly: LoSAI 6, PGA-A 10, PGA-D 0 (Figure 1(b)). She remained on topical therapy alone with sustained control despite ongoing immunotherapy.
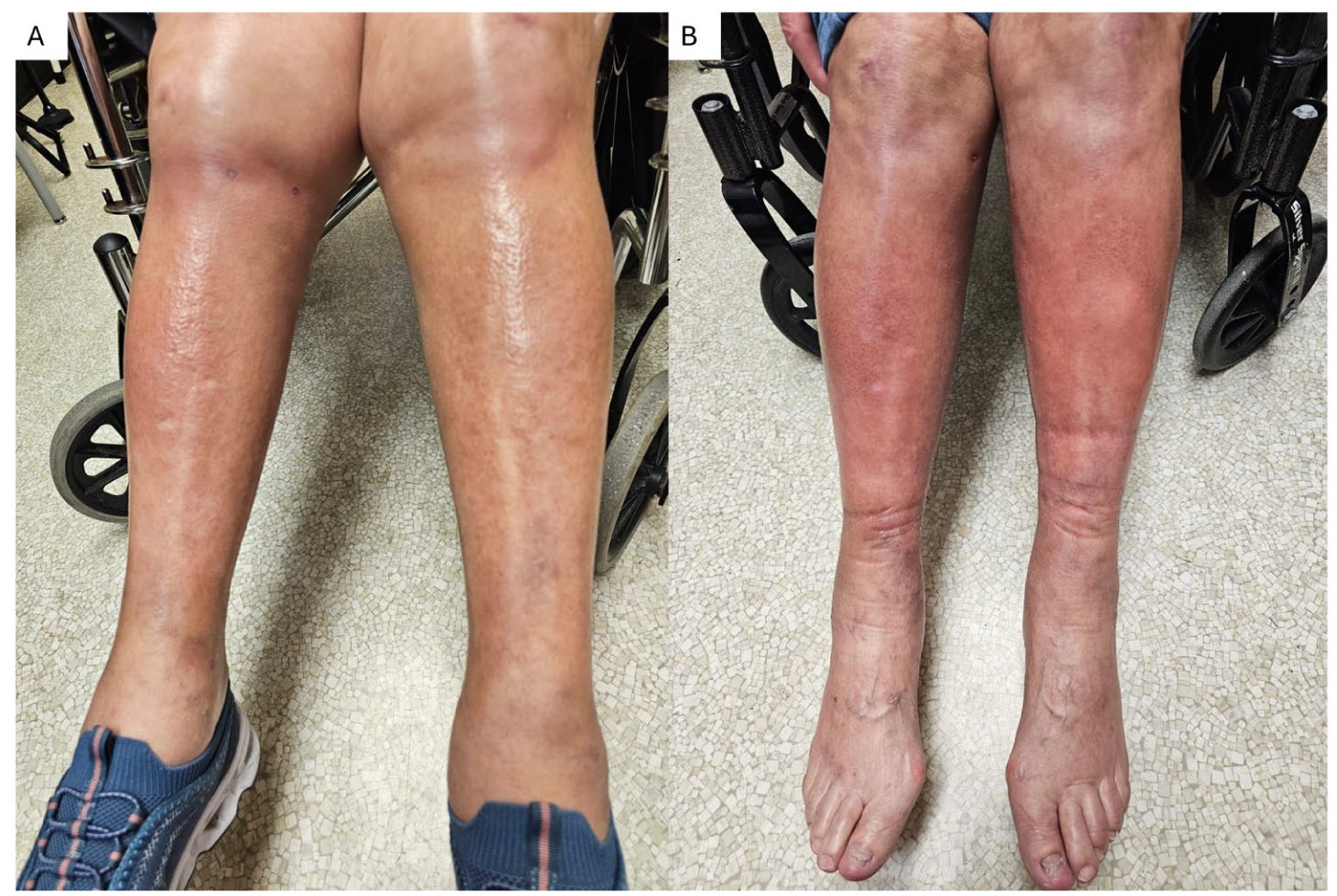
Clinical improvement of localized scleroderma in patient 1 following topical roflumilast therapy. (a) At baseline, before initiation of treatment, firm induration is seen extending from the knees to the ankles with violaceous erythema at edges, consistent with active generalized symmetric LS. (b) Six weeks after starting topical roflumilast 0.3% under occlusion, substantial clinical improvement is observed, with no new/expanded plaques, resolution of lilac halo and reduction of fibrosis.
Patient 2
An 86-year-old woman with a history of breast and thyroid cancer presented with generalized symmetric LS involving the upper and lower extremities (ankles to knees, wrists to elbows) and had previously failed narrow band ultraviolet radiation (NB-UVB) in combination to ultrapotent topical steroids. Baseline assessments revealed LoSAI 32, localized scleroderma skin damage index (LoSDI) 0, PGA-A 50, PGA-D 0, pain 9/10, and itch 6/10. She declined systemic therapy and hence, topical roflumilast 0.3% cream under occlusion was added to her treatment regimen. One month later, she reported improved mobility and symptom control, with LoSAI reduced to 10, LoSDI to 8, PGA-A to 30, and PGA-D to 40. Further clinical improvement was observed at 4 months (LoSAI 2, LoSDI 2, PGA-A 10, PGA-D 10) and 7 months follow-up (LoSAI 0, LoSDI 4, PGA-A 0, PGA-D 2). One year later, she transitioned to oral roflumilast 500 µg daily monotherapy due to coverage constraints, with maintained disease control at 18-months follow-up.
Patient 3
A 61-year-old man with obesity, hypertension, and hypothyroidism presented with a 2-year history of a well-demarcated, thickened violaceous plaque over the lower abdomen. He had a biopsy-confirmed LS circumscribed subtype and had previously discontinued hydroxychloroquine and cyclosporine due to side effects. At presentation LoSAI was 8, LoSDI 6, PGA-A 80, PGA-D 10, pain (6/10) and itch 10/10 (Figure 2(a)). He was initiated on once-daily roflumilast 0.3% cream without occlusion in combination with halobetasol propionate 0.01% cream. At 3-week follow-up, ~70% clinical improvement was noted (LoSAI 2, LoSDI 6, PGA-A 20, PGA-D 30), with complete resolution of pain and near-complete itch relief (pain 0/10, itch 2/10; Figure 2(b)). At 12 months follow-up, LS plaque was nearly resolved.

Clinical improvement of localized scleroderma in patient 3 following topical roflumilast therapy. (a) At baseline, an ivory indurated plaque with significant erythema is observed on the lower abdomen, consistent with active circumscribed LS. The patient also developped type 4 hypersensitivity to hydroxychroquine explaining the surroing erythema and desquamation. (b) Three weeks following daily topical roflumilast, halobethasol cream and hydroxychroloquine discontinuation there is a complete resolution of erythema, improved subcutaneous atrophy, residual superficial dermal fibrosis manifesting as an ivory plaque.
Discussion
This is the first case series documenting topical roflumilast 0.3% use in LS, including checkpoint inhibitor-associated and treatment-refractory forms. All three patients experienced rapid and sustained improvement in inflammation, pain, and functional outcomes as measured by validated scoring tools. Importantly, all patients avoided systemic immunosuppression altogether, highlighting the potential role of topical PDE4 inhibition as a safe and effective therapeutic strategy.
In two patients, roflumilast was applied under occlusion (wet wraps for 1 hour) and used in combination with ultrapotent topical corticosteroids and/or NB-UVB phototherapy—treatments that had previously failed when used alone. In the third patient, roflumilast was applied without occlusion due to anatomic localization in a body fold, but still combined with a superpotent corticosteroid. These observations suggest roflumilast may serve best as adjunctive therapy in inflammatory LS.
LS pathogenesis includes lymphocytic infiltration, fibroblast activation, and matrix deposition, leading to sclerosis and potential disability. 1 Aberrant immune pathways—including Th2 and Th17 skewing, interferon signaling, and immunosenescence—are central to disease persistence. 1 PDE4 is highly expressed in activated immune cells and dermal fibroblasts, and its inhibition suppresses cytokine release (e.g., TNF-α, IL-6, IL-13) and fibroblast activation. 6
Roflumilast is a second-generation PDE4 inhibitor with 25–300× greater potency than apremilast. 4 It has demonstrated rapid onset of action and a favorable safety profile in a variety of dermatologic conditions. 4 Apremilast, which has also been used off-label in LS, is well tolerated but limited by cost and systemic delivery. 5 However, its efficacy is likely confined to early inflammatory or anatomically superficial disease. In more extensive, deep, or long-standing fibrotic LS, topical delivery may not achieve adequate tissue penetration or clinical impact. Accordingly, patient selection is critical, and lesion depth, extent, and subtype should guide treatment planning. Hence, topical roflumilast should not replace standard immunomodulatory therapy in patients at risk of disfigurement or disability. Rather, it may serve as a valuable adjunct in selected patients or clinical scenarios where systemic treatment is contraindicated, declined, or poorly tolerated.
Limitations of this report include small sample size, short-to-intermediate follow-up, and lack of histopathologic correlation or biomarker analyses. However, the consistent and meaningful clinical improvements highlight roflumilast’s therapeutic potential.
In summary, topical roflumilast 0.3% cream was effective, well tolerated, and clinically meaningful in three patients with severe or treatment-limited LS. These findings support its further investigation as a targeted, steroid-sparing therapy in fibrosing dermatoses.
Footnotes
Ethical considerations
No ethics approval was required for this case report.
Consent to Participate
The patients provided written consent for the case report and images to be submitted to this journal.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EN has served as a speaker, consultant, investigator, or advisory board member for AbbVie, Apogee, Arcutis, Bausch Health, BioJamp, Boehringer Ingelheim International, Bristol Myers Squibb, Celltrion, Eli Lilly, Galderma, Innovaderm, Janssen, LEO Pharma, Medexus, Novartis Pharmaceuticals, Organon, Pfizer, Sanofi Genzyme, Searchlight Pharma, Sun Pharmaceuticals, and UCB. EN has received honoraria, consulting fees, and investigator-initiated research funding. Other authors have nothing to declare.
