Abstract
This study aimed to identify risk factors, evaluate management strategies, and assess visual outcomes of suprachoroidal hemorrhage during cataract surgery through a retrospective analysis of five clinical cases and a literature review. We analyzed five cases of suprachoroidal hemorrhage occurring during phacoemulsification cataract surgery with intraocular lens implantation or intrascleral fixation of intraocular lens haptics at our institution (January 2013–September 2024). Among 48,725 cataract surgeries, five cases of suprachoroidal hemorrhage were identified (incidence: 0.01%). Key risk factors included preoperative angle-closure glaucoma (two cases), prior cataract surgery with vitrectomy (two cases), and advanced age (one case, mid-70s). Intraoperative suprachoroidal hemorrhage occurred in two cases (one with posterior capsular rupture), whereas the condition developed postoperatively in three cases. Management strategies comprised pars plana vitrectomy with scleral drainage (three cases), vitrectomy combined with intravitreal triamcinolone injection (one case), and conservative treatment (one case). The mean preoperative visual acuity improved significantly from 2.22 LogMAR to 0.6 LogMAR at the final follow-up (P = 0.007). These findings suggest that, in addition to the previously reported major risk factors for suprachoroidal hemorrhage, such as advanced age, glaucoma, and multiple surgeries, secondary intraocular lens implantation is a major risk factor. Comprehensive preoperative evaluation and meticulous control of intraoperative intraocular pressure fluctuations are essential to mitigate suprachoroidal hemorrhage risks and optimize surgical outcomes.
Keywords
Introduction
Suprachoroidal hemorrhage (SCH) is a rare but potentially severe complication, frequently associated with intraocular surgeries and rarely caused by trauma or occurring spontaneously. Based on the timing of occurrence (i.e. during or after surgery), SCH is classified into acute expulsive SCH and delayed SCH. 1 The prevailing hypothesis is that SCH occurs due to fluctuations in intraocular pressure (IOP) levels leading to the rupture of the long and short ciliary arteries, resulting in the rapid accumulation of blood in the suprachoroidal space. 2 This blood accumulation can cause the lens–iris diaphragm to shift forward, thereby disrupting the circulation of aqueous humor. This phenomenon is typically manifested as a significant increase in IOP, shallowing of the anterior chamber, severe ocular pain, and a rapid decline in vision. Fortunately, with the implementation of phacoemulsification and microincision techniques, the incidence of SCH associated with cataract surgery has reduced to 0.013%–0.5%. 3 Nevertheless, the risk factors for SCH and optimal treatment strategies remain controversial. This case series presents an analysis of five cases of SCH related to phacoemulsification cataract surgery.
Case report
The reporting of this study conforms to the Case Report (CARE) guidelines. 4 We have de-identified all patient details. This retrospective analysis was conducted among 48,725 patients with cataract who were treated at Hainan Eye Hospital, Zhongshan Ophthalmic Center, Haikou, China, between January 2013 and September 2024. This study was conducted in accordance with the principles set forth in the Declaration of Helsinki. The Institutional Review Board and Ethics Committee determined that formal approval was not necessary for this study. We obtained all patients’ consent to treatment. As part of the standard protocol, all patients undergoing surgery were evaluated preoperatively for a range of ocular parameters, including IOP, visual acuity, axial length, crystalline status, indications for surgery, and history of ophthalmic surgery. All best-corrected visual acuity (BCVA) data were converted to LogMAR equivalents of Snellen acuity for analysis. As BCVA values were too poor to be quantified on the Snellen chart, they were converted into LogMAR values according to the study by Roberts et al., 5 wherein light perception was assigned a value of 2.8 LogMAR, hand movement 2.7 LogMAR, and counting fingers 2.6 LogMAR. A comparison of the mean visual acuity outcomes was performed using the paired t-test. A significance level of P < 0.05 was used. Additionally, any systemic diseases and the use of oral medications were recorded. Patients who developed SCH underwent B-scan during the postoperative period for a minimum of 2 weeks or until the SCH had abated. The visual acuity, IOP, size of SCH, and the status of any retinal and vitreous inclusions were assessed. Treatment options included immediate topical and systemic medications to control IOP as well as local and systemic oral steroids. The option of surgery is available when necessary.
Of the 48,725 cases observed during the study period, SCH was detected in five cases, indicating an incidence rate of 0.01%. Detailed patient information is provided in Table 1. Cases 1 and 3 had undergone cataract extraction and intraocular lens (IOL) implantation several years ago, along with vitrectomy for dislocation of the IOL into the vitreous cavity 1 month ago. Cases 2 and 3 had combined angle-closure glaucoma, and the IOP was normalized with topical medication. Case 5 represents a patient in the mid-70s with age-related cataract. The mean age and mean IOP of the patients were 62.4 ± 10.9 years and 16.2 ± 2.14 mmHg, respectively. None of the patients were taking oral anticoagulants or had other serious systemic diseases.
Basic characteristics of the patients.
BCVA: best-corrected visual acuity; IOP: intraocular pressure; M: male; F: female; CE: cataract extraction; IOL: intraocular lens; P + I: Phacoemulsification with primary IOL implantation; CF: counting finger; HM: hand motion; SCH: suprachoroidal hemorrhage.
SCH occurred at different stages of surgery: (a) in Case 2, after completion of phacoemulsification; (b) in Case 4, during anterior vitrectomy due to posterior capsule rupture; (c) in Case 3, on the first postoperative day; and (d) in Cases 1 and 5, on the second postoperative day. Objective findings included loss of red reflex on the fundus, flattening of the anterior chamber, and a sudden rise in IOP, with B-scan revealing a dome-shaped bulge behind the membrane and low reflex echoes. Subjectively, the patient reported a sudden onset of ocular pain. Vital signs showed that the pain was associated with increased heart rate and blood pressure. The ophthalmologist stopped the procedure immediately after detecting the SCH and confirmed the self-sealing nature of the small incision before completing the procedure. Continuous B-scan monitoring was performed; if the thrombus failed to dissolve 1–2 weeks postoperatively, vitrectomy and scleral drainage were performed.
Due to the limited extent of the bleeding, Case 1 was treated with 0.03% tobramycin and dexamethasone ophthalmic solution (Alcon, Fort Worth, Texas, USA) (4 times a day for 2 weeks) and systemic oral steroids and prednisone tablets (Qilu Pharmaceutical, Hainan, China) (5 mg/10 kg body weight, once a day for 1 week). After 2 weeks, the bleeding was completely resolved, and the BCVA was 20/40. Case 2 underwent IOL implantation on postoperative day 5 and vitrectomy for vitreous opacification on postoperative day 30. Case 3 showed no signs of shrinkage or absorption of SCH on postoperative day 7 (Figure 1); therefore, vitrectomy and scleral drainage were performed. Cases 4 and 5 showed partial absorption of the hemorrhage on postoperative day 14, but residual hemorrhage still persisted; thus, vitrectomy and scleral drainage were performed. After drainage, the retinal and choroidal membranes were flattened. There was no evidence of cystoid macular edema or choroidal detachment at follow-up. The clinical status of all patients remained stable at each scheduled follow-up visit (weekly for the first month, with a mean follow-up of 6 months). The preoperative mean BCVA was 2.22 ± 1.02 LogMAR. The postoperative mean BCVA was 0.6 ± 0.29 LogMAR. The preoperative BCVA and BCVA at the last follow-up of the patients showed significant improvement (P = 0.007).
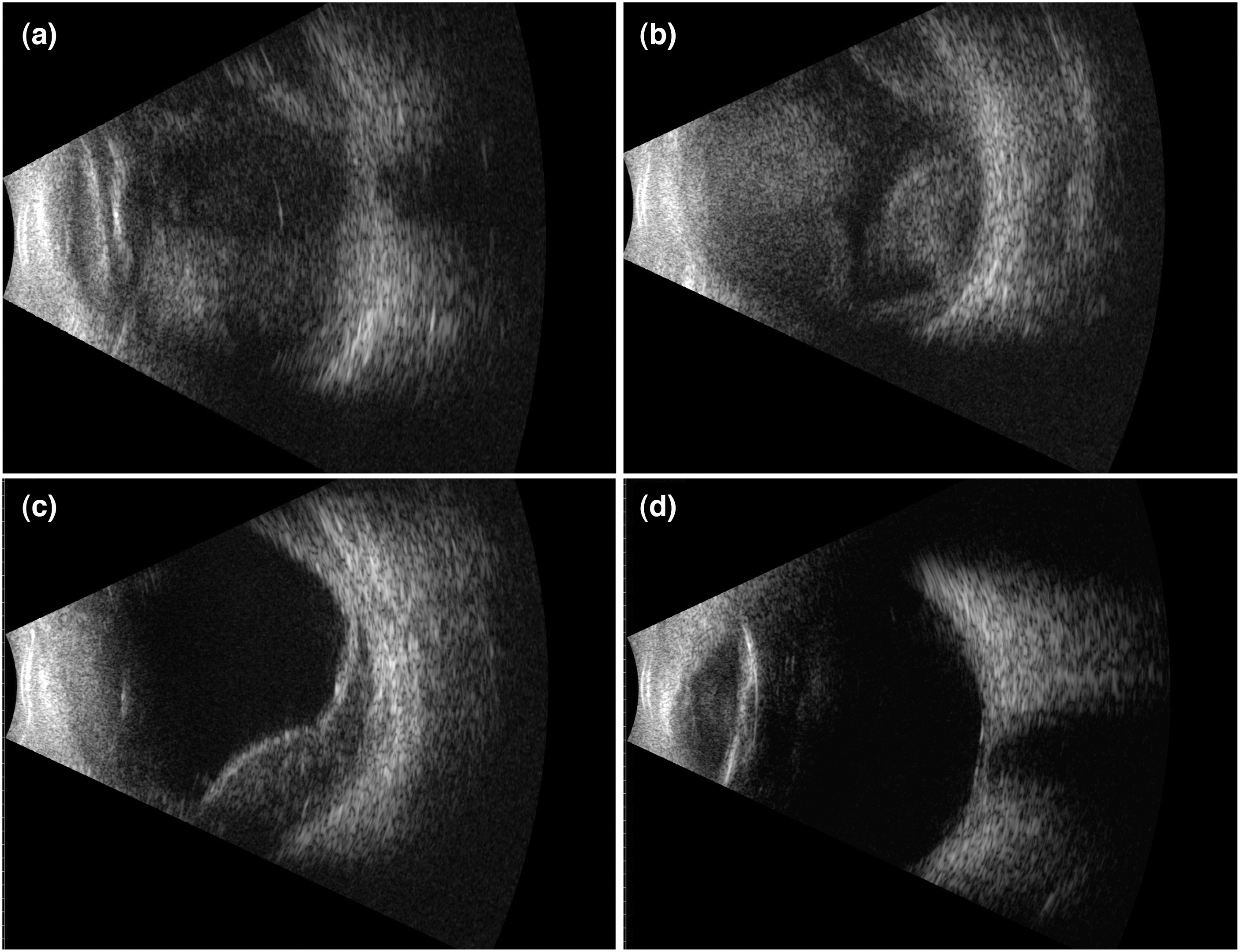
B-mode ultrasound imaging indicates the recovery process after SCH through vitrectomy combined with scleral drainage. (a) On the first day after surgery, B-scan revealed a strong echo area in the vitreous cavity, which could not distinguish retinal elevations from choroidal elevations. (b) On the seventh day after surgery, no significant absorption of the hemorrhage in the vitreous cavity was observed, and multiple focal elevations of the retina and choroid were visible. (c) On the seventh day after surgery, vitrectomy was performed in combination with scleral drainage, resulting in a large amount of blood flowing out of the sclera. On the first day after the surgery, B-scan revealed that the vitreous hemorrhage was cleared. Localized hypoechoic areas beneath the retina and choroid that have not been fully absorbed were still visible and (d) three weeks after surgery, the residual subretinal and choroidal hemorrhages were completely absorbed. SCH: suprachoroidal hemorrhage.
Discussion
After entering the era of phacoemulsification in the field of cataract surgery, the incidence of SCH has considerably reduced, potentially due to the small incision that better maintains IOP stability.3 Moreover, there have been changes in the surgical methods and choices for cataracts. Evidence indicates an increase in the number of patients who have undergone multiple surgeries on the same eye, such as IOL implantation surgery after vitrectomy for cataract surgery and stage II IOL implantation surgery after cataract extraction for IOL dislocation.6 In recent years, with the use of capsular tension rings, patients with IOL dislocation have received effective surgical treatment in the early stages. However, over time, there has been an increase in the number of patients who experience dislocation of the IOL along with the capsular bag, leading to its fall into the vitreous cavity. The patients must undergo multiple surgeries, such as vitrectomy combined with stage II IOL implantation, which greatly increases the risk of developing SCH. In this report, we presented five specific cases of SCH and reviewed the associated risk factors identified in the literature over the past decade (Table 2).
Studies on risk factors for SCH in different types of cataract surgery over the past decade.

COVID-19: coronavirus disease 2019; IOL: intraocular lens; SCH: suprachoroidal hemorrhage; SICS: small-incision cataract surgery; ECCE: extracapsular cataract extraction; ICCE: intracapsular cataract extraction; LP: light perception; NLP: no light perception; CF: counting finger; HM: hand motion; PPV: pars plana vitrectomy; PPL: pars plana lensectomy; ROP: retinopathy of prematurity; LA: local anesthesia; HTN: hypertension; DM: diabetes mellitus; rt-PA: recombinant tissue plasminogen activator; SO: silicon oil; AF: atrial fibrillation; AC: anterior chamber.
The symbol “/” indicates not applicable.
“1/2 m counting finger” indicates the ability to count fingers at a distance of 50 cm.
Cases 1 and 3 represented patients with aphakic eyes who underwent a total of three ophthalmic surgeries before the onset of delayed SCH. Previous studies have reported that a history of multiple intraocular surgery is one of the risk factors for the development of SCH. 7 Meanwhile, a retrospective analysis by Chen et al. revealed that patients with aphakia seemed to have a higher postoperative incidence of SCH. This is because the absence of an intact lens–iris diaphragm makes the eye more susceptible to pressure fluctuations. 8 We propose that secondary IOL implantation is also a risk factor for SCH. This procedure often involves additional surgical manipulation and may be performed in patients with pre-existing ocular conditions or complications, further increasing the risk of hemorrhage. Cases 2 and 4 had a history of glaucoma diagnosed prior to surgery and developed acute expulsive SCH. It has been reported that any intraocular surgery is potentially linked to the development of SCH, with glaucoma surgery exhibiting the highest risk. 9 The main reason for this is the large preoperative and postoperative IOP difference, which may lead to choroidal effusion and subsequent vascular stretching and rupture. 10 Patients with cataract who have a history of glaucoma face the same risk during surgery.
The association between minimally invasive glaucoma surgery (MIGS) and SCH in combined cataract–MIGS procedures presents an ongoing clinical concern. MIGS—characterized by an ab interno approach, minimal tissue disruption, and rapid postoperative recovery—has been increasingly integrated with cataract surgery to manage concurrent glaucoma and lens opacification. Although MIGS devices (e.g. iStent, Hydrus Microstent®, and Kahook Dual Blade®) demonstrate lower complication rates than traditional trabeculectomy, perioperative factors may modulate SCH risks. For example, transient postoperative hypotony following MIGS, although uncommon, may increase the risk of choroidal effusion or hemorrhage, especially in eyes with high-risk ocular profiles (e.g. elevated preoperative IOP or compromised vascular integrity). 11 Okubo et al. 12 further analyzed early identification and management of suprachoroidal effusion in combined surgeries. Current evidence suggests that SCH incidence in standalone MIGS remains low; however, its risk may increase incrementally in complex multiprocedural interventions (e.g. combined cataract–MIGS with IOL exchange), necessitating large-scale prospective cohorts to delineate risk stratification.
Research has revealed that intraoperative fluctuations in IOP are one of the main risk factors for SCH. In recent years, femtosecond laser-assisted phacoemulsification has gradually gained wider attention. Multiple docking of the vacuum aspirator in femtosecond laser-assisted phacoemulsification leads to excessive fluctuations in IOP compared with conventional phacoemulsification, constituting a new risk factor for SCH. 13 The most recent data from the Royal College of Ophthalmologists' National Ophthalmology Database 14 showed that the incidence of acute expulsive SCH was 0.03%, with risk factors including posterior capsular rupture, elevated IOP, glaucoma, and advanced age, especially >90 years of age.
Apart from these intraoperative risk factors, several ocular risk factors that increase the incidence of SCH include ocular hypotony, uncontrolled high IOP, elevated extra-scleral vascular pressure in carotid–cavernous sinus fistulas, 15 hard nucleus cataract, vitreous loss, longer axial length of the eye, aphakia, prior SCH in the presenting or other eye, choroidal lesions, intraocular malignancy, age-related macular degeneration, 16 and uveitis or past trauma. 17 Systemic risk factors also include elevated intraoperative pulse rate and blood pressure, atherosclerosis, elevated venous pressure during the Valsalva maneuver, hemodialysis, liver disease, diabetes mellitus, chronic hypercapnia, hematological malignancy, erythrocytosis, thrombocytopenia, 18 and coronavirus disease 2019. 19 There is no evidence that anticoagulation therapy is a risk factor for acute SCH in cataract surgery. 20
In addition to the identified risk factors, the choice of anesthesia may influence SCH outcomes. Local anesthesia, although commonly used in cataract surgery, has been associated with challenges in managing severe intraoperative hemorrhage due to limited control over systemic blood pressure and patient movement. 21 Okubo et al. 12 reported cases of suprachoroidal effusion occurring during cataract surgery under local anesthesia. Conversely, general anesthesia allows for hemodynamic stability and eliminates intraoperative patient motion, potentially reducing mechanical stress on ocular structures. Further studies are needed to evaluate whether anesthesia selection (e.g. prioritizing general anesthesia for high-risk patients with glaucoma or advanced age) can mitigate SCH incidence.
If a suspected acute SCH is detected intraoperatively, the surgery should be immediately terminated and the incision should be closed. If the wound cannot be closed effectively, suture should be performed rapidly to avoid drainage of intraocular materials. The prognosis of SCH is mostly poor. Fan et al. 22 conducted a single-center retrospective study and revealed that observation remains a reasonable management strategy for non-appositional SCH. Although a case report by Pakravan et al. 23 concluded that surgical drainage immediately after the diagnosis of SCH may provide a better visual prognosis, we recommend drainage within 1–2 weeks after the onset of SCH to allow for blood liquefaction. A daily B-scan was also performed to detect the extent of SCH for enlargement or accumulation of maculae as a means of determining the optimal time for surgical treatment. In Case 3, surgical intervention was performed after confirming no sign of absorption of the clot on consecutive B-scans up to postoperative day 7. Other indications for surgery include patients presenting with uncontrolled elevated IOP, severe pain, flat anterior chamber, retinal detachment, appositional SCH, prolapse of intraocular contents, retinal or vitreous incarceration, macular involvement, and enlarged SCH.
Surgical treatment can include either drainage surgery alone or in conjunction with vitreoretinal surgery to address the associated problems; it has demonstrated efficacy in our case series and the literature. Drainage procedures include conventional radial sclerotomies and transconjunctival trocar cannula systems with 23G or 25G systems. 24 The 23G or 25G systems can be inserted at a location 10–15 mm posterior to the limbus, allowing for more passive drainage of the posterior part of the SCH. However, it is slower than radial sclerotomies and cannot drain clotted blood. 25 To prevent irreversible optic nerve damage due to prolonged hemorrhage and secondary high IOP, tissue plasminogen activator can be injected directly into the suprachoroidal space to promote early liquefaction and drainage of the clot, a treatment that has demonstrated positive visual outcomes. 26 Akram et al. 27 described a massive SCH following left-eye phacoemulsification, managed via a two-stage approach: high-dose recombinant tissue plasminogen activator (rt-PA; 100 μg/0.1 mL) injected into the hemorrhage quadrant for clot lysis, followed by inferior sclerostomy to drain suprachoroidal blood. This combined strategy synergized pharmacological and mechanical interventions, suggesting improved visual outcomes with reduced complications. Significant variability exists in rt-PA dosing; for example, Fei et al. 28 used 10 μg/0.2 mL per quadrant on postoperative day 5 prior to vitrectomy. Although current evidence supports the safety of surgery integrated with rt-PA, multicenter trials are needed to standardize the protocols.
An important aspect of SCH is its impact on visual prognosis, which is considerably influenced by the anatomic location of the hemorrhage. 29 The location of the SCH can dictate the extent of damage to surrounding ocular structures and the potential for recovery. For example, when the hemorrhage involves the posterior segment, it can directly damage the retina and macula, leading to more severe visual impairment. Conversely, if the hemorrhage is located anteriorly and does not involve the retina, it remains confined to the anterior segment without retinal involvement; the visual prognosis may be more favorable, as the retina may remain intact. The extent of vision loss associated with SCH also depends on the volume of the hemorrhage and speed of its accumulation. Rapidly accumulating blood can exert pressure on the retina and other intraocular structures, leading to more significant and immediate visual consequences. Conversely, a smaller, slower-developing hemorrhage may allow for some degree of compensation and recovery, depending on the patient’s baseline ocular status and the presence of other risk factors.
Moreover, the timing of surgical intervention critically determines the final visual outcome. Early recognition and management of SCH can mitigate the risk of permanent damage. Surgical drainage of the hemorrhage may be indicated, especially if it threatens the integrity of the retina or if the patient experiences significant vision loss. The choice of surgical technique and the timing of intervention can further influence the prognosis, highlighting the need for an individualized management strategy guided by case-specific factors. Meanwhile, as reported in this case series, conservative treatment with local and systemic steroids is essential. Systemic steroids have been found to be beneficial in controlling inflammation and aiding clot lysis. Additionally, factors such as systemic health status, age, and ocular comorbidities may considerably influence the prognosis. Patients with advanced age or systemic comorbidities often exhibit reduced reparative potential, which may lead to a poorer post-SCH visual prognosis. 30
However, this study has the limitations of a small number of SCH cases and a short follow-up period. Therefore, a multicenter study with a larger sample size should be conducted in the future to draw definitive conclusions.
In conclusion, this study revealed that patients with advanced age, glaucoma, and multiple surgeries, particularly secondary IOL implantation, were more likely to develop SCH. Many patients achieved good surgical results through vitrectomy combined with scleral drainage. These risk factors need to be brought to the attention of ophthalmologists, who should be aware of the need to adequately assess the patient’s general condition preoperatively, avoid fluctuating IOP during the surgery, and provide timely and appropriate treatment to save the patient’s vision after the surgery.
Footnotes
Acknowledgements
None.
Author contributions
Rubing Liu and BeilingTan are joint first authors. Rubing Liu, BeilingTan, and Mingbing Zeng conceived the study and contributed to the study design. Rubing Liu, BeilingTan, Han Wang, Zebin Li, Furong Luo, and Jifa Kuang conducted the statistical analyses. Rubing Liu, BeilingTan, Han Wang, Zebin Li, Furong Luo, Jifa Kuang, and Mingbing Zeng interpreted the results and wrote the manuscript. All authors contributed to writing of the manuscript, approved the final version, and had final responsibility for the decision to submit for publication. The corresponding author attests that all listed authors meet the authorship criteria.
Availability of data and materials
All data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This study was supported, in parts, by an unrestricted grant from the National Natural Science Foundation of China (No:82060176) and Natural Science Foundation of Hainan Province (No: 822MS190).
