Abstract
Abdominal aortic aneurysms are the most common types of aneurysms worldwide, whereas aneurysms in the blood vessels of the abdominal organs (splanchnic aneurysms) are rare. Few studies have reported cases of two giant aneurysms occurring simultaneously. Presented here is a case of a 67-year-old man who began to experience periumbilical pain with a diagnosis of a giant abdominal aortic aneurysm and a hepatic artery aneurysm. He was successfully treated using a staged endovascular stent grafting procedure. Postoperatively, the patient’s abdominal pain was resolved, and he was prescribed medication for hypertension control, lipid control, heart rate control, antiplatelet therapy, and smoking cessation, which he took regularly. Follow-up evaluations were performed at 3, 6, and 9 months after surgery, and no late complications were observed. This case report suggests that when two giant aneurysms are diagnosed simultaneously, risk of rupture, surgical interventions, and postoperative complications are significantly increased. The choice of a treatment strategy is complex and has a high degree of individual specificity.
Introduction
An abdominal aortic aneurysm (AAA) is an irreversible, pathologically local dilation of the abdominal aorta with a diameter of 3 cm or more. AAA has a high incidence of more than 200,000 cases per year worldwide. AAA rupture has a very high mortality rate and is listed as one of the top ten causes of death in the global age group of 55–74 years. 1 Splanchnic aneurysm refers to the aneurysmal dilation of the arteries and their branches in the abdominal organs, and its incidence is very low. Hepatic artery aneurysms (HAAs) are the second most common type of splanchnic aneurysms, with an incidence of only 0.4%. 2 Currently, few studies have reported cases of two giant aneurysms occurring simultaneously, especially HAA, which are rare and complex, and no consensus exists on the treatment strategy. This case report describes the course of treatment for a patient diagnosed with macro-AAA and HAA with clinical symptoms.
Case report
A 67-year-old man with recurrent periumbilical pain, lasting minutes to hours for a week, was referred to our hospital for further treatment after an abdominal ultrasound performed at another hospital suggested mixed echoes of the abdominal aorta (81 × 67 mm). His medical history included hypertension and smoking for 10 years. Physical examination revealed a flat abdomen, mild tenderness around the umbilicus, and a palpable pulsatile mass in the left side of the periumbilical region. Computed tomography angiography (CTA) of the aorta revealed marked fusiform dilation of the middle and lower parts of the abdominal aorta with mural thrombosis measuring approximately 80 × 80 × 101 mm. There was dilation of the bilateral iliac artery aneurysms (BIAAs), and the diameters of the involved arterial segments were 22.8 mm and 12.3 mm, while the lengths were 160 mm and 163 mm, respectively. Additionally, the hepatic artery propria revealed a large sac-like protrusion with adherent thrombosis, with a diameter of approximately 76 mm, and the affected arterial segment measured 6.2 mm in diameter and 26.4 mm in length. The bilateral common iliac artery revealed an aneurysm-like dilation of approximately 24 mm at its widest point (Figure 1). Blood samples did not reveal any obvious abnormalities. The patient was diagnosed with an AAA, HAA, bilateral common iliac artery aneurysm, and grade 2 hypertension (very high risk). Staged endovascular stent graft implantation treatment was decided.
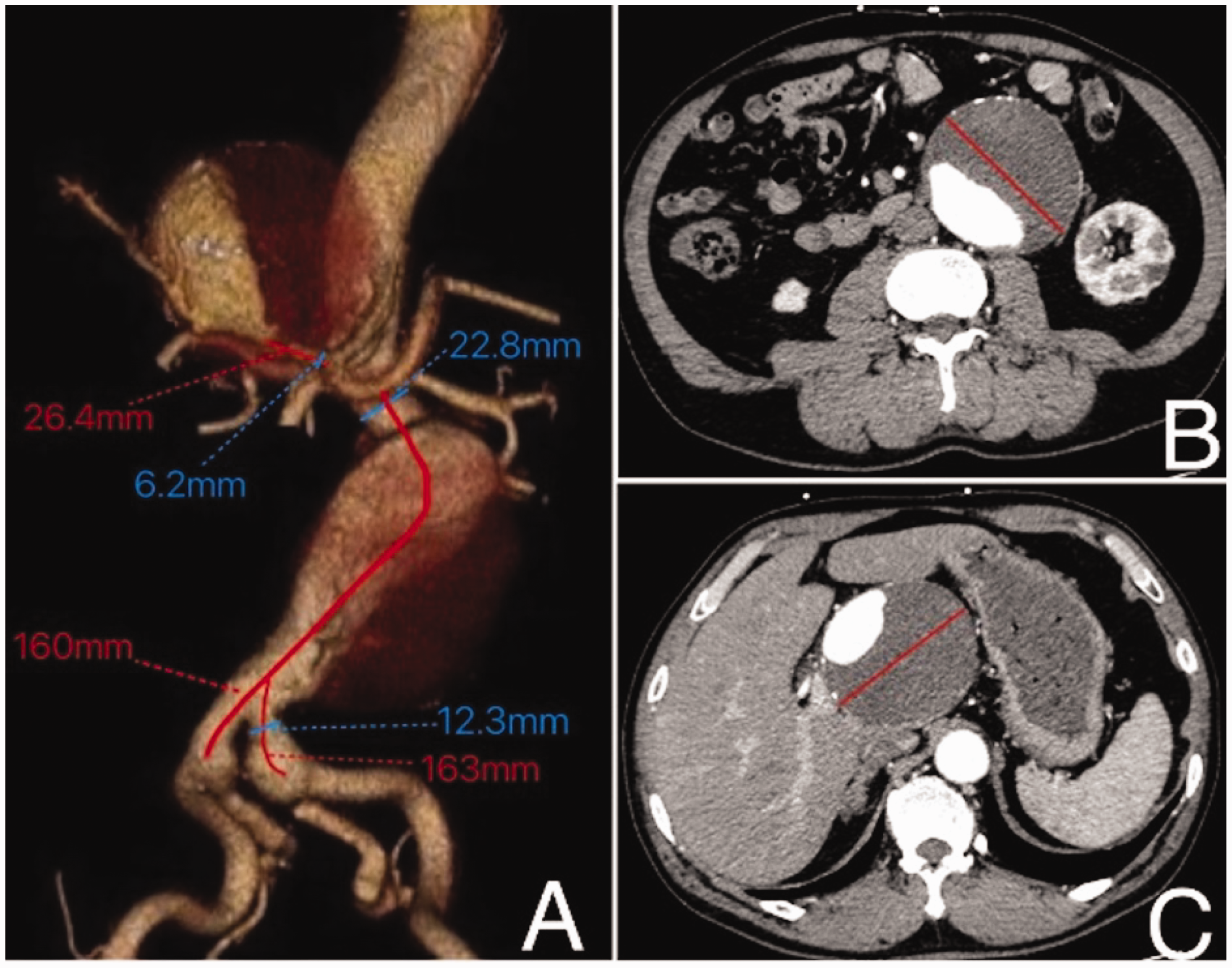
(a) Preoperative computed tomography (CT) angiography of the aorta revealed cystic dilation of the hepatic artery aneurysm (HAA) originating from the hepatic artery propria. The affected arterial segment measured 6.2 mm in diameter and 26.4 mm in length. Abdominal aortic aneurysm (AAA) displayed fusiform dilation, with dilation of the bilateral iliac artery aneurysm, and the diameters of the involved arterial segments were 22.8 mm and 12.3 mm, and the lengths were 160 mm and 163 mm. (b) Abdominal CT scan showed that the maximum diameter of the AAA was 80 mm, the aneurysm cavity had wall thrombosis, and the annular calcification lesion was continuously interrupted on the tube wall and (c) CT of the abdomen revealed that the maximum diameter of the HAA was 76 mm, and the aneurysm cavity was a thrombosis attached to the wall.
The first-stage surgery was performed using local anaesthesia involving abdominal aortogram and renal arteriography to identify AAA and BIAA (Figure 2(a)). GORE® EXCLUDER® AAA Endoprosthesis (RLT281218, 28.5 mm × 40 mm × 12 mm ×18 cm) stent was delivered through the right femoral artery along the 18-F DRYSEAL sheath under the support of a 0.035 × 260 cm stiffer exchange guide-wire. The stent was released proximally midway below the opening of the left renal artery in the abdominal aorta. Another GORE® EXCLUDER® AAA Endoprosthesis (PLC231200, 16 mm × 120 mm × 23 mm) stent was delivered through the left femoral artery along the 14-F DRYSEAL sheath under the support of a 0.035 × 260 cm stiffened exchange guide-wire released from the abdominal aorta to the left iliac artery. The GORE®) EXCLUDER® AAA Endoprosthesis (RLT281218, 28.5 mm ×40 mm × 12 mm × 18 cm) stent was released again to the right iliac artery segment. Repeated pigtail imaging revealed that the AAA and BIAA were completely covered, the stent was properly adherent, and the blood flow was smooth (Figure 2(b)). Postoperatively, the patient’s abdominal distension and pain resolved, and he was treated with antihypertensives, antiplatelets, ventricular rate control, and lipid regulation, and advised about smoking cessation.

(a) Aortogram showing abdominal aortic aneurysm (AAA) fusiform dilation below the level of the renal artery and aneurysmal dilation of the bilateral iliac arteries. (b) After releasing the stent, the AAA and bilateral iliac artery aneurysm were well insulated, no signs of endoleak were observed, and both renal arteries were visible. (c) Angiography of the coeliac trunk shows cystic dilation of the hepatic artery aneurysm (HAA) and (d) after releasing the stent (white arrow), the celiac trunk (yellow arrow), splenic artery (blue arrow), and distal circulation were unobstructed, the HAA was well isolated, and no signs of endoleak were observed.
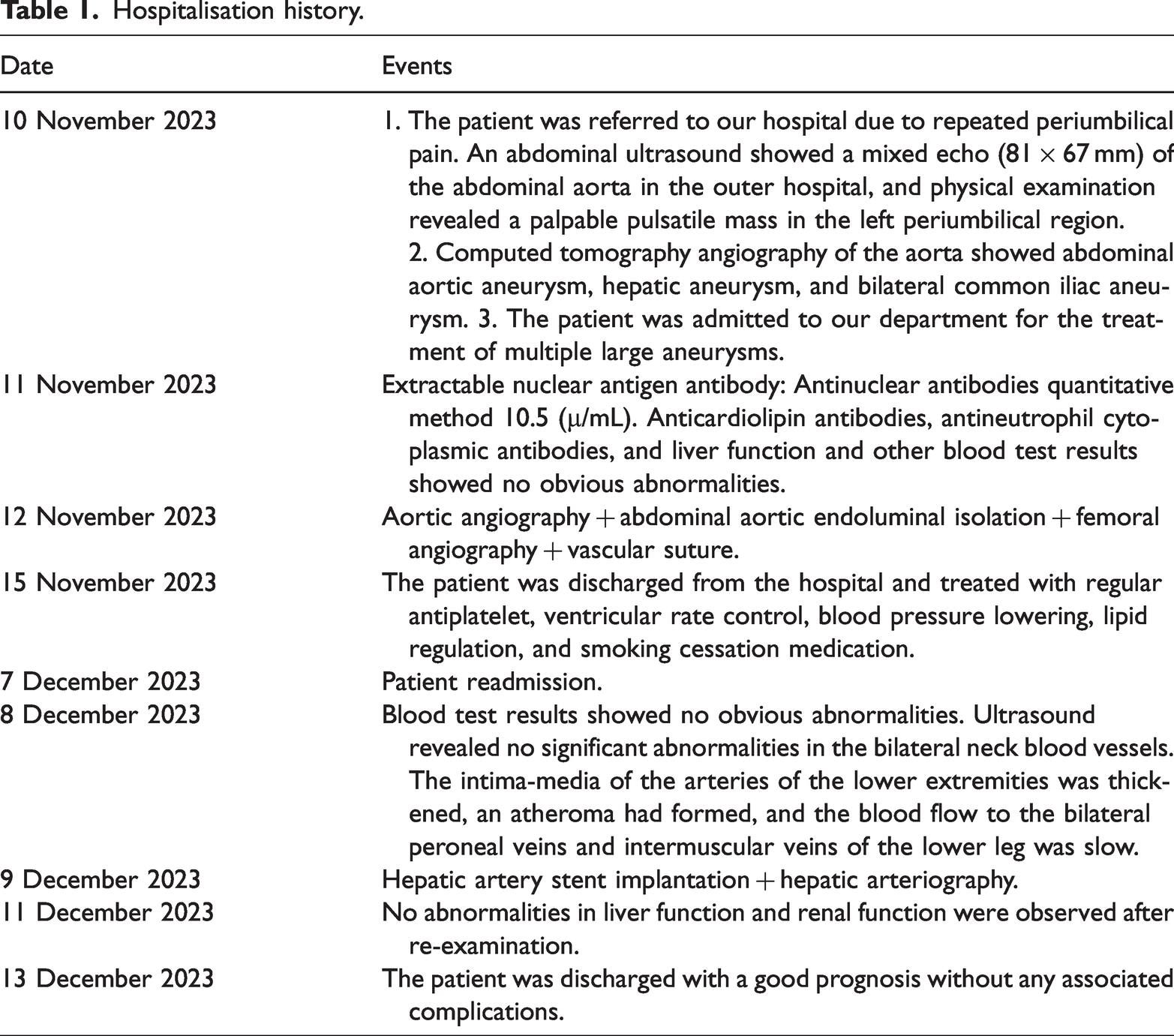
After an interval of 1 month, the second-stage surgery isolated the HAA. The left brachial artery was punctured under local anaesthesia, and an 8-F sheath was inserted. Initial attempts to pass a loach guide-wire through the left subclavian to the thoracic aorta using an MP1 contrast tube were unsuccessful. The passing of the Tig tube also failed; however, the loach guide-wire was successfully passed through the distal thoracic aorta under the reinforced support of the SIM1 contrast tube. An unsuccessful attempt was made to advance a 0.035 ×260 cm stiffened exchange guide-wire through the tortuosity of the thoracic aorta. The guide-wire was then replaced with an elongated and stiff guide-wire, which successfully passed through the tortuosity of the thoracic aorta to reach the abdominal aortic stent graft. The 8-F MP guide catheter was successfully transported to the opening of the abdominal trunk under the reinforced support of the MP1 contrast tube. Hepatic arteriography revealed that the HAA was located between the splenic and right hepatic arteries (Figure 2(c)). A Corsair microcatheter was then fed, and a Runthrough guide-wire was delivered along the microcatheter to the distal end of the right hepatic artery, which was replaced with an EV3 peripheral microcatheter to confirm that the guide-wire was located in the true lumen of the blood vessel. The V18 guide-wire was delivered along the EV3 microcatheter to the distal end of the right hepatic artery, after which the MP guide-wire was replaced with an AB46 sheath, and a heparin-coated intravascular stent graft (VBH091002, 9 mm × 10 cm ×120 cm) was delivered to the distal splenic artery for release. Reimaging revealed complete coverage of the HAA with no residual shunt, and the splenic artery opening was unaffected, with smooth blood flow (Figure 2(d)). Postoperatively, antiplatelet, lipid-lowering, ventricular rate-control, and antihypertensive drugs were continued. The important time node events during the patient’s hospital stay are shown in Table 1.
Hospitalisation history.
At the third and sixth months of postoperative telephonic follow-ups, the patient did not complain of any discomfort. At the outpatient 9-month follow-up, the liver and kidney functions were normal, aortic CTA confirmed that the stent was completely patent with no endoleak, and no delayed complications were observed (Figure 3).

Nine months postoperatively, computed tomography angiography of the aorta showed that the stent was not ectopic. The abdominal aortic aneurysm, hepatic artery aneurysm, and bilateral iliac artery aneurysm were well isolated, the stent was patent, and there was no endoleakage.
All patient details were anonymized, and written informed consent was obtained prior to the commencement of this report. This study was approved by the Ethics Committee of the Guangdong Pharmaceutical University (No. 2024-71). This case report has been written in accordance with the CARE guidelines. 3
Discussion
The decision to perform surgical treatment for AAA warrants a comprehensive evaluation of factors such as the aneurysm diameter, growth rate, symptoms, embolic events, survival expectancy, surgical risk, age, and concomitant diseases. Among these, the AAA diameter is the primary factor determining the need for surgical intervention. Based on data from an abdominal aorta diameter survey in the Chinese population, the recommended surgical indications include an AAA diameter > 5.0 cm for males and 4.5 cm > for females. According to the guidelines on the progress of diagnosis and treatment of splanchnic aneurysms in China, surgical intervention are recommended for HAA with a diameter of > 2.5 cm, an annual growth of more than 0.5 cm, and for all intrahepatic aneurysms. In this case, the patient had a maximum diameter of 80 mm for the AAA, 76 mm for the HAA, and 24 mm for the bilateral common iliac aneurysms. The aneurysms were large, with obvious symptoms of abdominal distension and pain, a history of hypertension, and risk factors for long-term smoking. Given the high risk of aneurysm rupture that could not be ruled out, active surgical treatment was deemed necessary.
AAAs and splanchnic aneurysms are often asymptomatic and difficult to diagnose until rupture, causing abdominal pain and hypovolemic shock. The patient had recurrent abdominal pain, and ultrasonography at other hospitals revealed an AAA, which made people aware of AAA rupture. However, the patient’s vital signs were stable, and aortic CTA revealed no aneurysm rupture or bleeding. The prevailing insight in national and international academic communities is that the use of endovascular aneurysm repair (EVAR) significantly reduces the 30-day postoperative mortality and complication rates for abdominal aortic and thoracic aneurysms. 4 EVAR should be the preferred option, especially in emergency situations, owing to its less invasive nature, faster access, and quicker bleeding control. 5 According to the latest edition of the American Society for Vascular Surgery guidelines, endoluminal therapy is the preferred treatment for all visceral aneurysms that meet the anatomical indications, except for renal aneurysms. This choice is not limited by patient age or clinical risk factors. Open surgery is characterised by its invasive nature, excess bleeding, and postoperative infection, which requires high cardiopulmonary function in patients; however, it has shown its advantages in reducing the reintervention rate. With advances in interventional technology and equipment, the morphological range of aneurysms suitable for endovascular intervention is gradually expanding and its long-term efficacy is gradually improving. The data showed no differences in the long-term outcomes between endovascular intervention and open repair for unruptured aneurysms. First, both of the patient’s aneurysms were unruptured, with no invasion of other organs in the abdominal cavity and no signs of systemic infection; the erythrocyte sedimentation rate and C-reactive protein levels were normal, rheumatologic immunity indices were negative, and enhanced computed tomography (CT) did not suggest retroperitoneal fibrosis. Therefore, inflammatory and mycotic aneurysms were excluded. Second, the morphology of the two aneurysms exhibited simple anatomical structures without tortuosity or dissection, and the shape of the aneurysm neck was similar to that of straight pipes, providing relatively suitable conditions for endovascular repair. Based on the patient’s preference, we decided to perform endovascular intervention.
The hepatic artery plays a vital role in the blood supply to the liver, and embolisation within the critical hepatic artery is generally avoided to prevent hepatic ischaemia, infarction, or gangrenous cholecystitis. 6 The large size of the HAA and AAA in this patient presented challenges. Simple coil embolisation treatment is characterised by prolonged procedural time, difficult execution, high risk of aneurysm cavity rupture and bleeding, a large number of coils required for dense embolisation, and high cost of operation and equipment such as the postoperative CT scan artefact. Additionally, embolisation may lead to the development of large parenchymal lesions, which have the potential to compress the biliary tract and duodenum, leading to serious complications such as jaundice, gastrointestinal obstruction, and even duodenal fistula.7,8 Performance differences exist among different spring coils, and effective and durable vascular occlusion is a major factor considering the selection of spring coils. The curvature of the blood vessels at the AAA and HAA was minimal, and there were sufficient stent-anchoring areas on both sides of the aneurysm. The use of stent graft endovascular isolation effectively blocks the direct impact of blood flow on the aneurysm wall, stabilises the aneurysm by thrombosis and fibrosis, and prevents the risk of distal embolism caused by dislodgment of the thrombus in the aneurysm lumen. Compared to coil embolisation, stent graft endovascular isolation is more convenient to operate and more economical and feasible in terms of operation time and cost, making it an ideal choice for patients.
After the decision to use an endovascular stent graft, one should consider the potential risk of aneurysm rupture, patient’s ability to tolerate the surgery, and skill and experience of the surgical team when choosing between concurrent or staged surgery. In 2021, the vascular surgery team at the West China Hospital conducted an updated meta-analysis of contemporaneous and staged hybrid surgeries for the treatment of thoracoabdominal aortic aneurysms. 9 A total of 1,725 patients with thoracoabdominal aortic aneurysms were included in the study. They reported technical success rates of 100% for contemporaneous surgery and 96% for staged surgery. The main reason for technical failure in staged surgery was a ruptured aortic aneurysm in patients awaiting implantation of the second stent. Staged surgery was associated with lower 30-day mortality, fewer major cardiovascular events, and fewer intestinal complications than contemporaneous surgery. Therefore, staged surgery is considered a better option for patients who are relatively stable, have a low risk of aneurysm rupture, or are in poor overall condition. The patient was compliant, and to avoid increased surgical risk, trauma, and complications, a more robust and safe staged surgical strategy was adopted based on the strict regulation of blood pressure, antiplatelet aggregation, ventricular rate control, lipid-lowering therapy, and smoking cessation. However, deciding whether to treat the HAA or AAA first posed a considerable challenge, and regardless of which one we chose to treat first, there was a risk that either aneurysm could rupture at any time. AAAs usually grow faster and are larger in diameter than HAAs. Moreover, the continuous interruption of the calcified tube wall of the AAA ring, coupled with the thinning of the appendage thrombus layer and symptoms of abdominal pain, suggest a higher risk of rupture. Studies have estimated the annual risk of rupture based on the diameter of the AAA, with a rupture rate of 75–95% when the AAA reaches ≥70 mm in diameter. Once an AAA ruptures, the mortality rate can be as high as 90%. In contrast, HAA has a rupture rate of approximately 44%, 10 with a post-rupture mortality rate of 40%. 11 Given the abundant collateral circulation in the liver, the risk of hepatic necrosis is relatively low, even after HAA rupture. Therefore, prioritising the treatment of AAA ensures the safety of patients and creates better conditions and opportunities for subsequent HAA treatment.
The patient had bilateral iliac artery involvement in the AAA and the distal end of the stent graft extended to the left and right iliac arteries when the AAA was isolated. GORE® EXCLUDER® AAA Endoprosthesis (RLT281218) with 25% oversize and GORE® EXCLUDER® AAA Endoprosthesis (PLC231200) with 30% oversize were selected for Phase I surgery. GORE® has a relatively complete abdominal aorta and iliac artery reconstruction system, which has been widely used in clinical practice. Its safety and effectiveness have been well recognised, and it can provide good anchoring and isolation effects. For the second staged procedure, the hepatic artery propria was curved, and the heparin-coated endovascular overlay stent system (VBH091002W), with better pliability and 48% oversize, was selected for its superior radial support. It provides better resistance to torsion and stretching, adapting to the lesion with a large angle of manoeuvrability, and the bioactive heparin on the surface of the stent graft can effectively prevent thrombosis while ensuring the patency of blood vessels.
In conclusion, cases of concomitant giant AAA and HAA are extremely rare, significantly increasing the complexity of the disease and the difficulty of treatment. The choice of an optimal treatment approach is highly demanding due to variations in the lesion types, extent, and different vessels involved in aneurysms. The surgical approach, choice of stent, and timing of surgery should be highly tailored to the individual patient’s anatomy and disease characteristics. Detailed preoperative imaging evaluation and accurate stent design are critical to achieving surgical success. Long-term postoperative follow-up is essential to assess stent location, splanchnic artery patency, and timely identification of potential complications.
Footnotes
Acknowledgements
We are very grateful to the patient’s family for allowing us to publish the complete record of the patient’s diagnosis and treatment.
Author contributions
Chunyan ZHU collected the patient’s history and information and was a major contributor to writing and revising the manuscript. Tong ZHOU, Tudi LI, and Rong CHEN analysed the characteristics of the aneurysm and explained the choice of surgical plan and surgical procedure. Zhihuan ZENG contributed to the final review of the manuscript. All authors have read and approved the final manuscript.
Data availability
The datasets generated and analysed during the current study are available in the Guangdong Pharmaceutical University repository.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Ethics statement
This research was approved by the Ethics Committee of The First Affiliated Hospital of Guangdong Pharmaceutical University. The study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. Participation in this study was voluntary. Anyone may withdraw at any time without penalty or loss of benefits to which we are entitled. Written informed consent was obtained from the patient for publication of this case.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Guangdong Medical Research Foundation (No. B2024095), Traditional Chinese Medicine Bureau of Guangdong Province Research Foundation (No. 20221229), Guangzhou Science and Technology Plan Project (No. 2025A03J3718), and “Flagship” Hospital Project of Chinese and Western Medicine Collaboration (No. ZXYXTQJ202301).
