Abstract
Background
This study aimed to elucidate the distinctions between the expression levels of angiotensin-converting enzyme (ACE) and ACE2, as well as their genetic polymorphisms, in patients with acute aortic dissection (AD) and coronary heart disease (CHD).
Methods
A cohort of 86 patients was enrolled, comprising 34 individuals with acute AD (encompassing Stanford types A and B), 18 with ascending aortic aneurysm, 21 with CHD, and 13 healthy controls. Aortic tissue samples were procured from 44 patients during surgical interventions.
Results
Statistically significant differences were observed in ACE and ACE2 expression levels among the ascending aortic aneurysm, CHD, and control groups (p < 0.05). However, the expression of ACE messenger ribonucleic acid (mRNA) in the aortic wall was significantly higher in the AD group than in the ascending aortic aneurysm and CHD groups (p < 0.05). Additionally, the expression of ACE2 mRNA and the ACE/ACE2 ratio in the aortic wall were significantly different in the AD group compared with the ascending aortic aneurysm and CHD groups (p < 0.05).
Conclusion
Plasma ACE levels, and the gene expressions of ACE and ACE2, are markedly reduced in patients with acute AD. The observed imbalance in ACE and ACE2 expressions may play a pivotal role in the pathogenesis of AD.
Keywords
Introduction
Aortic dissection (AD) represents a critical and life-threatening cardiovascular condition, characterized by a spectrum of associated complications. This pathology arises from the degeneration or cystic necrosis of the aortic media, precipitating an intimal tear through which blood penetrates into the arterial wall, thereby creating a false lumen. Death rates in the early stages can reach up to 50%, even when the best medical care is provided. Depending on the particular subtype of AD, different treatment strategies might be employed. 1 The rate of acute AD, which is 4.4 cases per 100,000 person-years, has remained fairly constant from 1995 to 2015, indicating no significant change in the prevalence of this condition since the 1980s. 2 This stability contrasts with other research that has noted a rising trend in the occurrence of aortic aneurysms. 3 The male-to-female ratio of affected individuals is approximately 2-5:1. It is noteworthy that over 50% of patients presenting with acute AD succumb within the initial 48 h after onset, with AD rupture being the predominant cause of mortality. Established risk factors implicated in the etiology of AD encompass hypertension, atherosclerosis, dyslipidemia, tobacco use, genetic predispositions, and traumatic incidents.
Moreover, the risk of AD significantly increases to 31% when the size of a thoracic aortic aneurysm reaches 6.0 cm in diameter. 4 Although the overall prevalence is not exceedingly high, there has been a noticeable increase in recent years, as documented. 5 AD, when it occurs, is associated with a high mortality rate and represents a critical clinical emergency that severely threatens the patient’s life. The aforementioned risk factors also play a role in the development of AD, with aortic aneurysms being one such factor. However, hypertension, atherosclerosis and aneurysms are commonly seen in clinical settings, and most do not lead to AD, hinting at a genetic predisposition in the disease’s presentation. 6 It is hypothesized that differential gene expression may underlie the development of AD in certain individuals. The renin-angiotensin system (RAS) is a pivotal endocrine regulatory mechanism that governs the progression of hypertension and atherosclerosis, as well as the retention of water and sodium. 7 Angiotensin-converting enzyme (ACE) and ACE2 are integral components within the RAS, with previous research indicating that the RAS modulates the production of angiotensin II and its receptor primarily through ACE, operating along the ACE-angiotensin II-AT1 axis. However, the discovery of ACE2, angiotensin (1–7) (Ang 1–7), and its receptor MAS has introduced the concept of an alternative regulatory pathway within the RAS, namely the ACE2-Ang (1–7)-MAS axis, 8 which is considered antagonistic to the former. Current research both domestically and internationally has identified associations between ACE and ACE2 and cardiovascular diseases such as hypertension, coronary artery disease, heart failure, ventricular hypertrophy, diabetes, and metabolic syndrome. 8 However, investigations into the relationship between these enzymes, their genetic underpinnings and AD remain scarce, with the bulk of research focusing on hypertension, myocardial ischemia, and heart failure. 9 There is a dearth of research concerning the genetic polymorphisms of ACE and ACE2, and their gene expression patterns in the context of AD and aneurysm. This study aimed to elucidate the potential role of ACE, ACE2, and their genetic constituents in the etiology and progression of AD and aneurysm.
Method
This retrospective study was stratified into three distinct phases: quantification of plasma ACE levels; genotypic analysis of ACE and ACE2 genes in blood samples; and assessment of ACE and ACE2 gene messenger ribonucleic acid (mRNA) expression within various tissues. Participants were categorized into four cohorts: the AD group, the ascending aortic aneurysm group, the coronary heart disease (CHD) group, and a control group comprising individuals without cardiovascular pathologies, and we have de-identified all patient details. A total of 86 blood samples were procured for analysis, encompassing 34 subjects diagnosed with acute AD (within a two-week period after onset, encompassing Stanford types A and B), 18 with aortic aneurysms, 21 with CHD, and 13 controls. Exclusions from the study included individuals under treatment with ACE inhibitors or angiotensin receptor blockers and those with Marfan syndrome. Demographically, the AD group comprised 27 males and seven females, with a hypertension prevalence of 73.5% (25 patients) and a diabetes mellitus prevalence of 5.9% (2 patients). The aortic aneurysm group included 14 males and four females, with hypertension affecting 44.4% (8 patients) and diabetes mellitus affecting 11.1% (2 patients). The CHD group was composed of 19 males and two females, with hypertension present in 57.1% (12 patients) and diabetes mellitus in 23.8% (5 patients). The control group consisted of 10 males and three females, all of whom were free from hypertension, diabetes, and any cardiovascular, endocrine, or renal diseases. We conducted this study in accordance with the Helsinki Declaration of 1975 as revised in 2013. The reporting of this study conforms to STROBE guidelines. 10
Detection of ACE and ACE2 gene polymorphisms
The identification of ACE and ACE2 gene polymorphisms was conducted as follows: all enrolled subjects were required to provide 5 mL of venous blood sample in the morning of the second day post-admission, under fasting conditions. The samples were collected in ethylenediaminetetraacetic acid tubes and subsequently stored at −70°C for subsequent genetic polymorphism analysis. Concurrently, 2 mL of venous blood sample was obtained for the determination of plasma ACE concentration.
During surgical procedures, arterial wall tissue samples were harvested and immediately immersed in liquid nitrogen. These samples were then transferred to a −70°C refrigerator for preservation, pending mRNA detection. Thoracotomy was performed in 12 cases of AD, 16 cases of aneurysm, and 16 cases of CHD, allowing for the procurement of aortic wall tissue. The retrieval of aortic wall tissue was not feasible in all patients for several reasons: nine patients in the AD group underwent interventional treatment, eight patients received non-surgical management, and five patients succumbed prior to surgery; in the aneurysm group, two patients were managed non-surgically; and in the CHD group, two patients were treated without surgery, whereas three cases were excluded due to the insufficient quantity of aortic wall tissue available for analysis.
Detection of the expression of ACE and ACE2 mRNA in the tissues
Total RNA extraction from the tissue samples was facilitated through the utilization of the Trizol reagent method. The isolated total RNA samples underwent reverse transcription to generate complementary DNA (cDNA), a process catalyzed by reverse transcriptase. For the amplification of cDNA, we employed Bio-Rad’s iScript™ cDNA Synthesis Kit on a polymerase chain reaction (PCR) apparatus. Following the amplification process, the cDNA samples were archived at −20°C in a refrigerated environment for future application in real-time PCR analysis. 11
The sequences of the primers specific for the ACE, ACE2, and β-actin genes, as well as the PCR amplification products, are delineated in Table 1. These primers are integral to the accurate and specific detection of the target genes within the samples. This primer probe amplifies a 108 bp region of the mRNA and was designed in the same region as the probe that was used in the Northern blot experiments previously described. 12
ACE, ACE2, and β- primer sequences of actin gene and polymerase chain reaction amplification products.

ACE: angiotensin-converting enzyme.
Statistical methods
The data obtained from the study were presented as mean values ± standard deviation. For statistical analysis, we employed SPSS version 25. The chi-square test was used to assess the differences in gender, history of hypertension, and history of diabetes among the various groups. When the frequency count for any group was less than 5, the Fisher’s exact test was applied instead to maintain statistical rigor. To compare the serum ACE concentration, ACE mRNA expression, and ACE2 mRNA expression across the different groups, we conducted a one-way analysis of variance (ANOVA). The differences in the distribution of ACE and ACE2 genotypes among the groups were evaluated using the chi-square test. In instances where the frequency count for a particular genotype was less than 5, the Fisher’s exact test was again employed to ensure the accuracy of the statistical inference. A p value of less than 0.05 was established as the threshold for statistical significance, indicating that any observed differences were not likely due to random chance and were therefore considered meaningful in the context of the study.
Results
The results of gender, hypertension, and diabetes frequency were compared
The chi-square test results indicated no significant differences among the groups with respect to gender composition (p = 0.669), history of hypertension (p = 0.107), and history of diabetes (p = 0.143). These p values are all greater than the conventional threshold of 0.05, suggesting that the observed variations in these variables across the groups are not statistically significant. Consequently, these demographic factors can be considered relatively balanced among the study cohorts, which strengthen the validity of subsequent analyses that focus on the primary outcomes of interest.
Comparison of results for plasma ACE concentration
The mean ACE concentrations for each group were as follows: 17.9 ± 7.9 U/L for the AD group, 25.3 ± 9.1 U/L for the aneurysm group, 33.5 ± 8.1 U/L for the CHD group, and 38.4 ± 4.8 U/L for the control group. The statistical significance of the differences in ACE concentrations among these groups is detailed in Table 2.
Comparison results of plasma angiotensin-converting enzyme concentration.
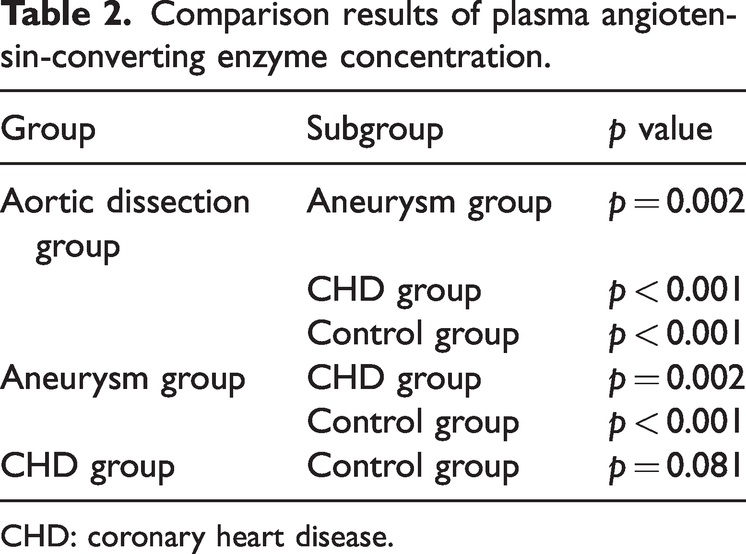
CHD: coronary heart disease.
To determine the statistical significance, a one-way ANOVA was likely performed, followed by post-hoc tests to identify which specific group comparisons were significantly different. The results of these tests are presented in Table 2, showing the p values for each pairwise comparison. If any of these p values are less than 0.05, it would indicate a statistically significant difference in ACE concentration between the corresponding groups.
Comparison of ACE gene I/D polymorphism
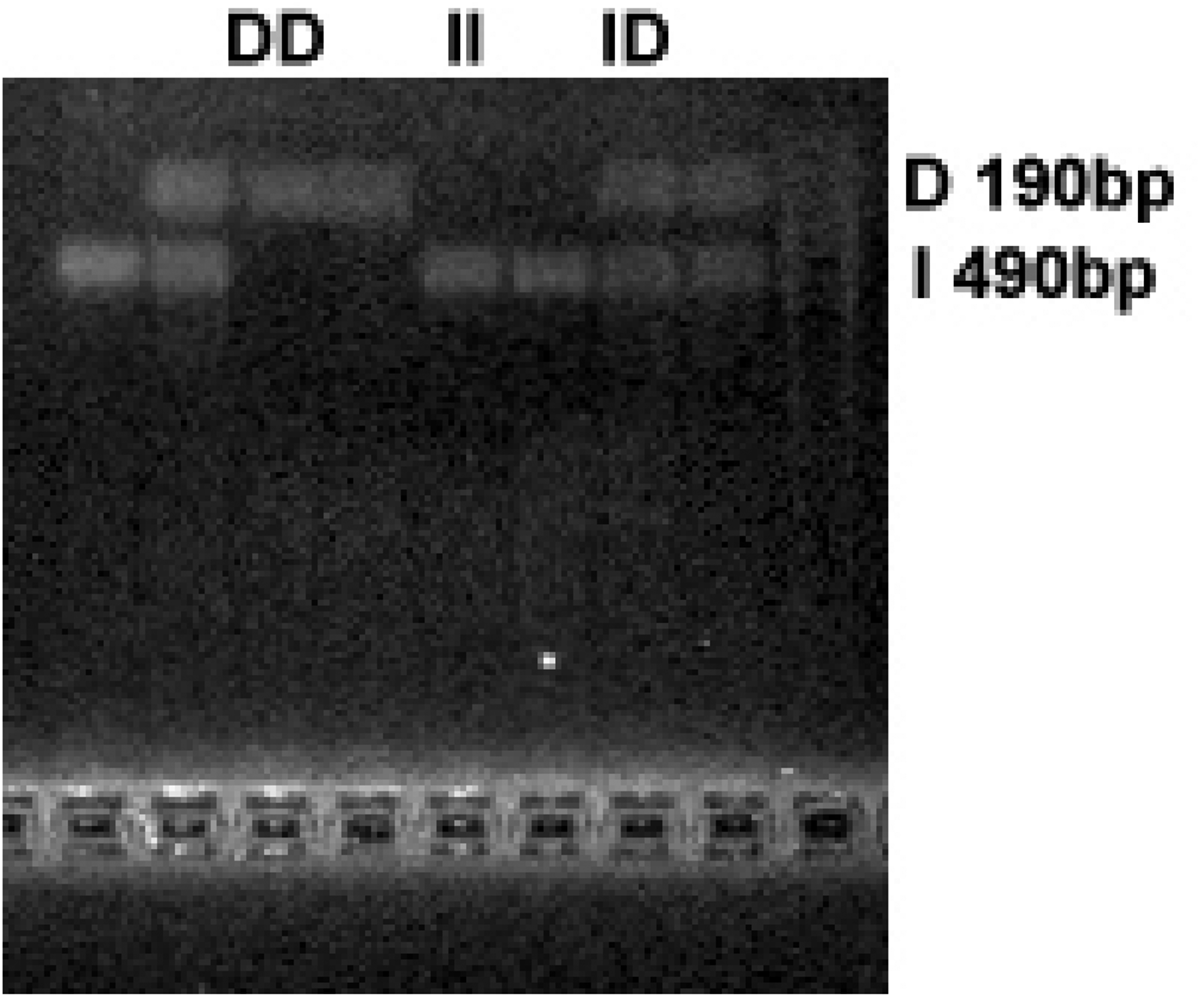
The PCR electrophoresis of the ACE gene yielded three distinct genotypes:
The DD genotype, characterized by the presence of a 190 base pair (bp) band on electrophoresis. The II genotype, indicated by a 490 bp band on electrophoresis. The ID genotype, which shows both a 190 bp and a 490 bp band on electrophoresis, as depicted in Figure 1.
ACE gene I/D polymorphism test. ACE: angiotensin-converting enzyme.
The distribution of these genotypes and their corresponding allele frequencies across the different groups are presented in Table 3. Statistical analysis revealed no significant differences in genotype distribution between the AD group, the aneurysm group, the CHD group, and the control group. The chi-square test statistic was 2.926 with a corresponding p value of 0.843, which is greater than the significance level of 0.05, indicating that the observed distribution of genotypes is not statistically different among the groups. Similarly, there was no significant difference in allele frequencies between the groups, as determined by the chi-square test statistic of 1.348 and a p value of 0.725, which also exceeds the significance level of 0.05. This suggests that the allele frequencies of the ACE gene are similarly distributed across the study cohorts, and genetic variation in the ACE gene, as measured by these alleles, is not associated with the presence of AD, aneurysm, or CHD in this study population.
Genotype and allele distribution of ACE gene I/D polymorphism.

AD: aortic dissection; CHD: coronary heart disease.
Genotype frequency X2 = 2.926, p = 0.843; Allele frequency X2 = 1.348, p = 0.725.
Comparison results of ACE2 gene (A8790G) polymorphism
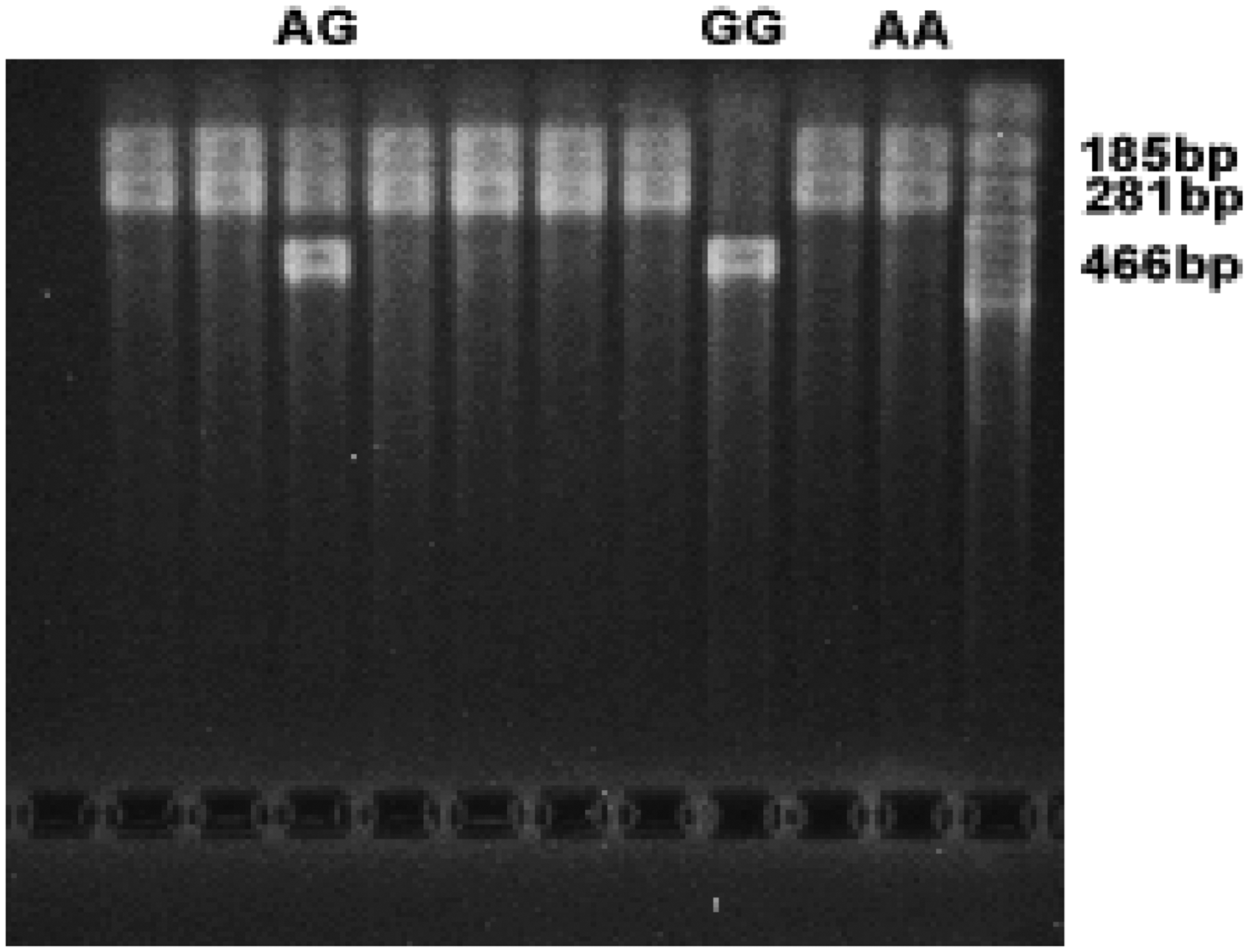
The PCR products of the ACE2 gene, specifically the A8790G polymorphism, can be digested by the Alu I restriction endonuclease to reveal three genotypes:
The AA genotype, which results in two bands of 281 bp and 185 bp upon electrophoresis. The GG genotype, which produces a single band of 466 bp upon electrophoresis. The AG genotype, which shows three bands of 466 bp, 281 bp, and 185 bp upon electrophoresis, as illustrated in Figure 2.
Identification of ACE2 gene polymorphism. ACE: angiotensin-converting enzyme.
Given that the ACE2 gene is situated on the X chromosome, the genotypic analysis for females can encompass AA, GG, and AG, whereas males can only exhibit the alleles A and G. Consequently, it is essential to analyze the results for males and females separately. The distribution of genotypes and alleles among female participants is detailed in Table 4. Statistical analysis indicated no significant differences in genotype distribution among the female AD group, aneurysm group, CHD group, and control group, with a chi-square test statistic of 3.181 and a p value of 1.000. Similarly, there were no significant differences in allele frequency, with a chi-square test statistic of 0.577 and a p value of 1.000. The distribution of alleles among male participants is presented in Table 5. The allele frequency was also not statistically significant, with a chi-square test statistic of 0.776 and a p value of 0.875. Furthermore, the overall distribution of A/G allele frequencies across each group was not statistically significant, as indicated by a chi-square test statistic of 2.602 and a p value of 0.462. This suggests that the ACE2 A8790G polymorphism does not significantly differ among the groups studied and may not be associated with the conditions under investigation in this population.
Distribution of ACE2 genotype and allele (female).
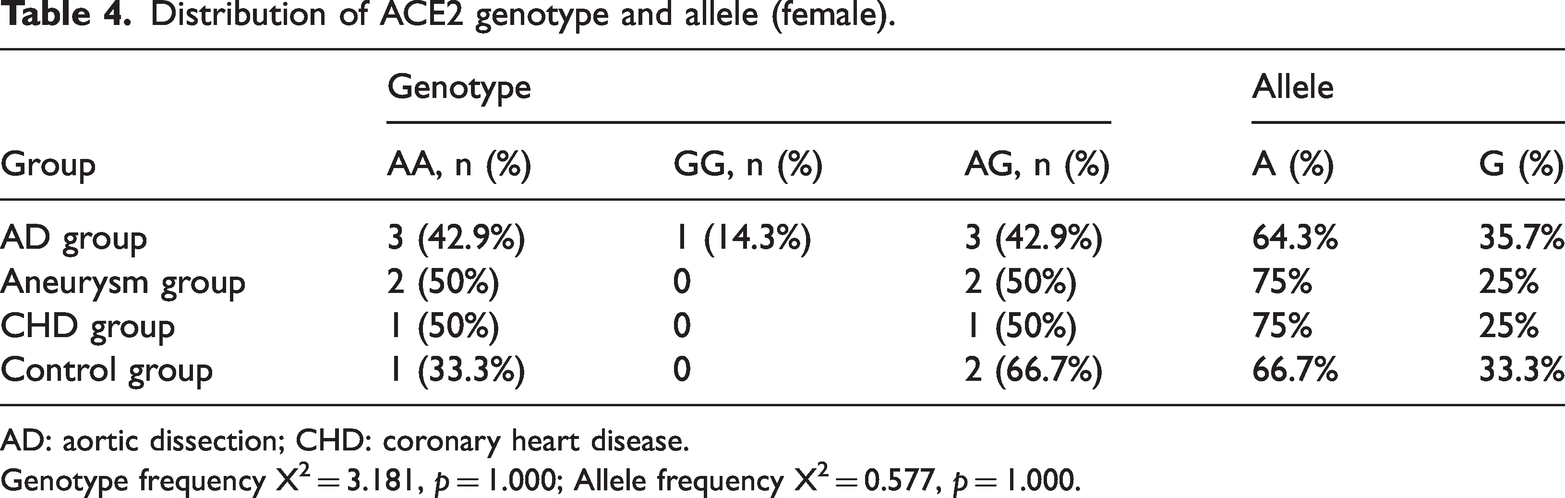
AD: aortic dissection; CHD: coronary heart disease.
Genotype frequency X2 = 3.181, p = 1.000; Allele frequency X2 = 0.577, p = 1.000.
Allele distribution of ACE2 gene (male).

AD: aortic dissection; CHD: coronary heart disease.
Allele frequency X2 = 0.776, p = 0.875.
Comparisons of ACE and ACE2 mRNA expressions in ascending aortic wall
The amplification curves, dissolution curves, and standard curves for both ACE and ACE2 are depicted in Figures 3 and 4, respectively. The melting curves exhibit a single peak without any additional heteropeaks, which signifies that the primers used have high specificity and that no nonspecific amplification has occurred.

Amplification curve, dissolution curve, and standard curve of angiotensin-converting enzyme (ACE). (a) ACE Amplification curve, (b) ACE dissolution curve, and (c) ACE standard curve.
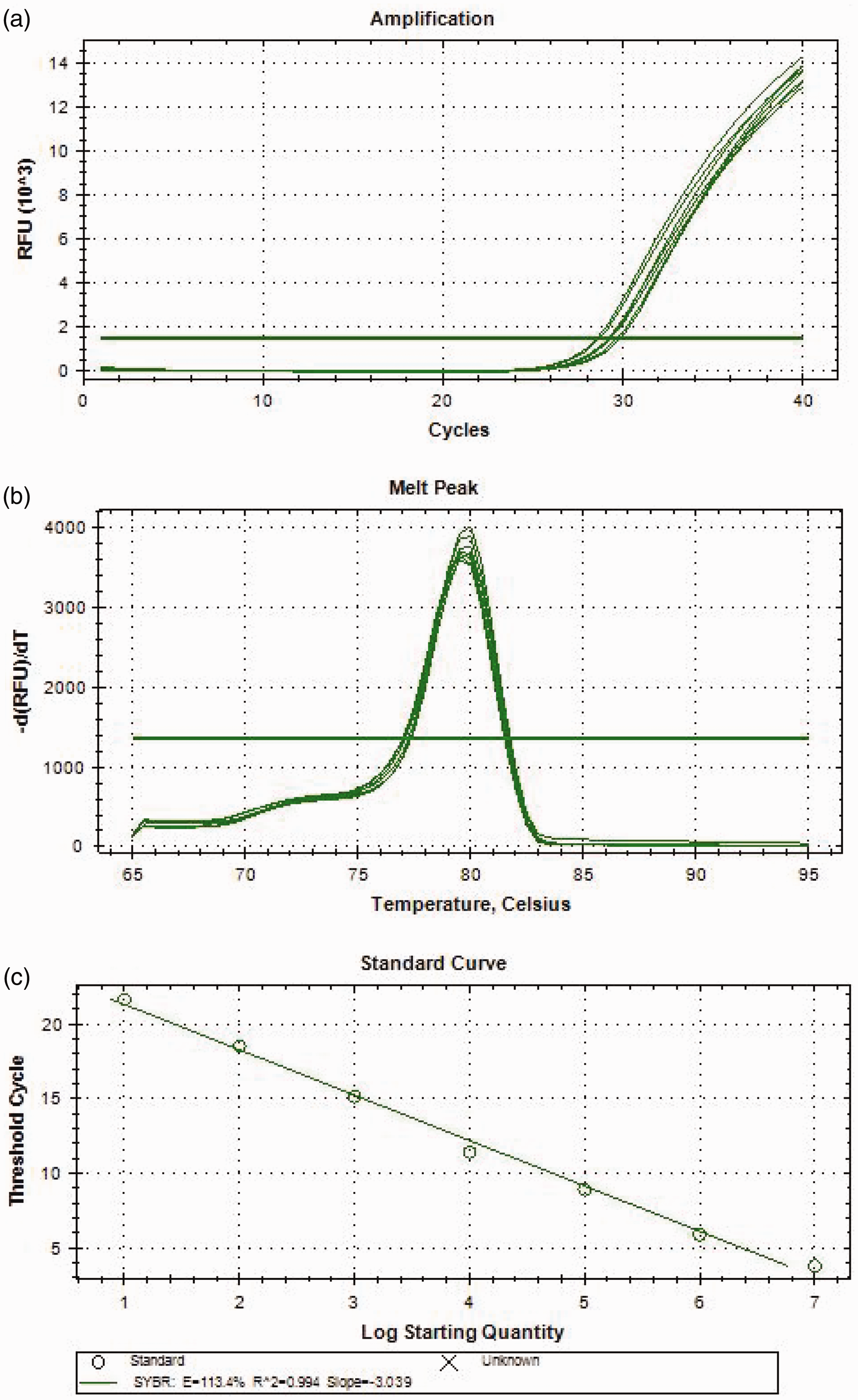
Amplification curve, dissolution curve, and standard curve of angiotensin-converting enzyme 2 (ACE2). (a) ACE2 Amplification curve, (b) ACE2 dissolution curve, and (c) ACE2 standard curve.
The expression levels of ACE in the different groups are as follows:
AD group: 0.2265 ± 0.3783 Aneurysm group: 0.9926 ± 0.9615 CHD group: 7.0857 ± 7.6929
Statistical analysis revealed significant differences in ACE expression among the groups, as detailed in Table 6.
Comparison of ACE mRNA expression in ascending aortic wall.

AD: aortic dissection; CHD: coronary heart disease; mRNA: messenger ribonucleic acid.
The expression levels of ACE2 in the different groups are:
AD group: 0.7662 ± 0.8586 Aneurysm group: 8.0054 ± 7.4411 CHD group: 9.6127 ± 11.5426
The results of the statistical analysis for ACE2 expression are presented in Table 7.
Comparison of ACE2 mRNA expression in ascending aortic wall.

AD: aortic dissection; CHD: coronary heart disease; mRNA: messenger ribonucleic acid.
The expression levels of the ACE/ACE2 ratio in the different groups are:
AD group: 0.4138 ± 0.4486 Aneurysm group: 0.2241 ± 0.3388 CHD group: 0.8111 ± 0.2563
The statistical analysis results for the ACE/ACE2 ratio are shown in Table 8.
Results of ACE/ACE2 ratio in ascending aortic wall.
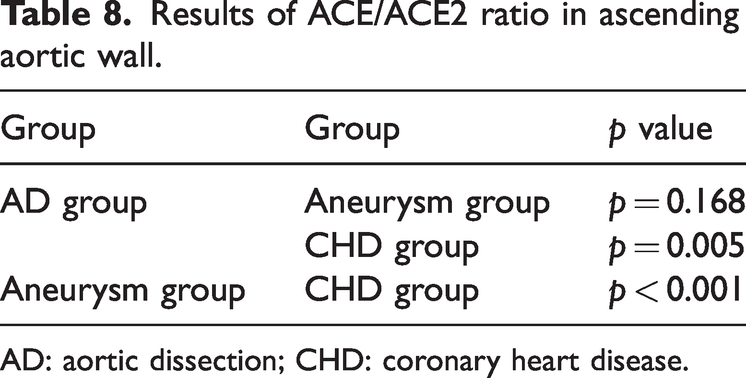
AD: aortic dissection; CHD: coronary heart disease.
These data suggest that there are notable variations in the expressions of ACE and ACE2, as well as their ratio, across the groups studied. The specific statistical tests and p values provided in Tables 6–8 would indicate which group comparisons are significantly different from one another, helping to elucidate the potential roles of these enzymes in the pathophysiology of AD, aneurysm, and CHD.
Discussion
ACE is a zinc-requiring metallopeptidase that is predominantly expressed within the vascular system. It is notably present in tissues such as the lung, kidney, small intestine, brain, heart, adrenal gland, ileum, jejunum, and duodenum. The primary function of ACE is to catalyze the conversion of the inactive decapeptide angiotensin I (Ang I) into the potent vasoconstrictor angiotensin II (Ang II). Ang II exerts its physiological effects by binding to the angiotensin II type 1 receptor (AT1), which leads to vasoconstriction and promotes the secretion of aldosterone. This, in turn, contributes to an increase in blood pressure. Additionally, ACE has the capability to hydrolyze and inactivate bradykinin, a peptide that, when active, can induce vasodilation and has anti-inflammatory properties. 13
Bradykinin can release endothelial cytokines such as no and prostaglandins to relax blood vessels and inhibit the proliferation of vascular smooth muscle cells. In addition, ACE can hydrolyze many other vasoactive peptides, such as substance P, enkephalin, neurotensin, etc.
14
Studies have shown that ACE level is mostly regulated by ACE gene polymorphism, whereas ACE activity is increased in patients with the D allele; the increase in ACE activity leads to the increase of Ang II activity, the transformation from AT1 to AT2 and the production of aldosterone.
15
Ang II can lead to the production of superoxide anion (
ACE1 and its counterpart ACE2 are recognized as regulators of blood pressure that maintain a balance through the production of vasoactive peptides within the RAS. Disruptions in the equilibrium of tissue ACE/ACE2 levels may stem from functional genetic variations in these genes, leading to blood pressure fluctuations. ACE2 deficiency is associated with increased blood pressure, whereas higher ACE2 levels are protective against hypertension. 18 A number of studies have shown that genetic variations in ACE genes, such as the I/D polymorphism (rs4646994), G2350A (rs4343), and others (rs4341, rs4344, rs4359, rs2106809), are linked to the development of hypertension. 18 Various gene families have been associated with cardiovascular diseases, and genetic diversity is prevalent among the Jordanian population. It has also been suggested that several single nucleotide polymorphisms could affect the response to cardiovascular drugs, including warfarin. 2 In Latin America, the incidence of hypertension is on the rise, influenced by factors such as an aging population, a diet resembling that of Western countries, and potential genetic predispositions related to the ACE gene. Understanding the genetic and molecular underpinnings of the disease, its dietary associations, and their interactions is crucial for tailoring treatments to enhance efficacy and minimize side effects. 19 However, the link between ACE gene polymorphisms and hypertension remains a subject of debate due to the influence of ethnic differences and geographical characteristics. Based on the above mechanism, many studies suggest that the ACE gene I/D polymorphism is closely related to cardiovascular disease. Although the experiment found that the ACE activity in cardiovascular diseases such as heart failure, hypertension, and angina pectoris increased significantly in the disease group in this experiment, the detection of peripheral venous blood ace concentration showed contradictory results. 20 The concentration of ACE in AD group and aneurysm group was significantly lower than that in aneurysm group (p < 0.05); there was no significant change in ACE concentration in CHD group and control group. This change was consistent with the expression of ACE mRNA in the wall of ascending aorta. The expression of ACE mRNA in AD group was the lowest, followed by aneurysm group and CHD group, and there was a significant difference between the two groups (p < 0.05). Our observation did not retrieve other similar research reports. We speculate that the causes of this phenomenon may be as follows: (1) the body changes after AD or aneurysm led to the decrease of ACE. ACE can be transformed into Ang II by acting on Ang I and Ang II can also regulate ACE. At present, studies have shown that Ang II can regulate ACE through AT1-ERK (extracellular regulated signal)/p38 MAP (mitogen-activated protein) kinase and downregulate ACE2. 15 Hence, is there any other negative regulatory mechanism, that is, the formation of inflammatory factors and atherosclerosis, or the corresponding changes in the body after the occurrence of dissection and aneurysm, so as to reduce ACE (2) the decrease of ACE leads to AD or aneurysm. ACE produces vasoconstriction after converting Ang I into Ang II. When ACE is excessively reduced, the vasoconstriction decreases, which may lead to the change of aortic wall tension, which may lead to the occurrence of AD or aneurysm to a certain extent or the appropriate expression of ACE and which may play a role in maintaining aortic wall tension. However, the number of cases in this experiment is relatively small, and it is necessary to further expand the sample to study whether this relationship exists. In addition, the patients in the AD group in this experiment are all acute AD. It is unknown whether the patients with chronic AD also have a reduction in ACE concentration and the degree of reduction, which needs further experimental verification. In the past, it was thought that there was only ACE-Ang II -AT1 axis in RAS system, but in 2000, Donoghue et al. 12 and Tipnis et al. 21 confirmed that there was another homologous substance of ACE, namely ACE2. It is found that there is another branch system of RAS system: ACE2 Ang (1–7)-MAS axis. Like ACE, ACE2 is also a zinc-containing metalloproteinase. Research shows that 42% of the metal catalytic region of ACE2 is consistent with ACE, and the gene encoding ACE2 contains 18 exons, which is similar to the gene encoding ACE with 17 exons. ACE2 is not widely distributed in the human body as ACE. It is mainly distributed in heart, kidney, testis, lung, artery, vein, digestive tract, brain, and other tissues, with the largest distribution in ileum and more in cardiac vascular endothelial cells and kidney tissues. 14 ACE2 can decompose Ang II into Ang (l–7). In addition, ACE2 can also directly act on Ang I to decompose it into Ang (l–9), and Ang (l–9) can be decomposed into Ang (l–7) under the action of ACE. At the same time, the efficiency of ACE2 decomposition of Ang II is 400 times that of Ang I. 22 The physiological functions of Ang (1–7) are mainly to relax blood vessels, reduce blood pressure, improve coronary perfusion and endothelial function, inhibit cardiomyocyte hypertrophy, inhibit the proliferation of myocardial fibroblasts and also inhibit vascular smooth muscle cells. In vitro experiments also confirmed that ACE2 can decompose other vasoactive peptide terminal amino acid residues, including motor vasopressin and de arginine bradykinin. 23 ACE2 can also catalyze the decomposition of the activities of apelin-13 and apelin-16. Experiments have confirmed that apelin-13 can cause the contraction of isolated coronary arteries. 24 At the same time, ACE2 also has the function of stimulating the body to produce vasodilator substances such as nitric oxide, prostaglandin, and bradykinin. Because the physiological function of ACE2 is opposite to that of ACE, many studies at home and abroad have suggested that ACE2 can stabilize atherosclerotic plaque, reduce platelet adhesion, antithrombotic, reduce inflammatory reaction, promote myocardial remodeling, and reduce blood pressure.25,26 Therefore, the current literature generally believes that ACE2 plays a protective role in cardiovascular diseases. In this experiment, the expression of ACE2 mRNA in the wall of ascending aorta in AD group was significantly lower than that in aneurysm group and CHD group, which were 0.7662 ± 0.8586, 8.0054 ± 7.4411 and 9.6127 ± 11.5426, respectively (p < 0.05), but there was no statistical significance in aneurysm group and CHD group (p > 0.05). The results suggest that ACE2 may play a role in the occurrence of AD. When the expression of ACE2 is reduced, the protective effect on cardiovascular system is weakened, making patients prone to AD on the basis of aneurysm or atherosclerosis.
Combined with the expression of ACE mRNA in the ascending aortic wall, the expression of ACE in AD group and aneurysm group decreased, suggesting that the two groups may have similar pathogenesis. However, the expression of ACE2 in AD group was significantly lower than that in aneurysm group, which further confirmed the protective effect of ACE2 in the occurrence of AD. However, limited to the selection of cases, there is no expression result of ACE and ACE2 in the ascending aortic wall of normal people in this experiment, and the expression amount of ACE and ACE2 in normal people is still unknown. In addition, some studies have shown that the imbalance of the ratio of ACE and ACE2 is related to the occurrence of cardiovascular diseases.27–29 In this experiment, we also observed the difference in the ratio of ACE and ACE2 between each group. The results show that the ratio of ACE/ACE2 in AD group, aneurysm group, and CHD group is 0.4138 ± 0.4486, 0.2241 ± 0.3388, and 0.8111 ± 0.2563, respectively. Statistical analysis showed that there was a significant difference in the proportion of AD group, aneurysm group and CHD group (p < 0.05), suggesting that the proportion of ACE and ACE2 may be related to the occurrence of different diseases. Although the proportion of AD group and aneurysm group was not statistically significant, it can be seen that the proportion of AD group was higher than that of aneurysm group; hence, it was not statistically significant, it may be that the number of cases is too small. Further experiments can be carried out to increase the number of samples for observation and analysis. At present, there are few studies on the relationship between ACE gene polymorphism and AD and aneurysm, and the results are inconsistent.30–32 Some studies show that ACE gene is related to AD, whereas others show no obvious connection.33,34 The relationship between ACE2 gene polymorphism and AD and aneurysm has not been reported. The I/D polymorphism of ACE gene and A8790G polymorphism of ACE2 gene were detected in this experiment. The results showed that there was no significant correlation between genotype or allele and AD, aneurysm, and CHD (p > 0.05). The possible reasons for this result are the small number of samples and the differences between regions and races.
The exploration of molecular mechanisms underlying this condition is instrumental in elucidating its pathophysiology. Clifton’s seminal work in 1977 35 established the genetic basis of transmission for this disease. Subsequent studies have identified autoimmune disorders with a genetic component as a significant risk factor for abdominal aortic aneurysm. 36 The multi-drug resistance (MDR) gene complex is implicated in drug resistance, with the MDR-1 gene being particularly noteworthy. This gene encodes for the P-glycoprotein (P-gp), which is crucial for the active transport of a variety of substrates. The role of this gene extends to the detoxification process, nutrient absorption, ion, and peptide transport and cellular signal transduction. A single nucleotide polymorphism in the 26th exon of the MDR-1 gene has been shown to reduce the expression of P-gp, an ATP-dependent transport protein. 37 Moreover, research has demonstrated a positive correlation between cholesterol ester levels and MDR-1 gene expression, which is associated with increased cellular proliferation. Additionally, the presence of MDR-1 has been documented in pathologies that impact the vessel wall, such as atherosclerosis.
Conclusion
In the context of acute AD, plasma ACE concentrations and the gene expression levels of both ACE and ACE2 are markedly diminished. This reduction in expression suggests that the imbalance between ACE and ACE2 may contribute to the pathogenic mechanisms underlying AD. Specifically, the mRNA expression levels of ACE in the ascending aortic wall were found to be lower in both the AD and aneurysm groups when compared with the CHD group. Similarly, the mRNA expression of ACE2 in the ascending aortic wall was notably lower in the AD group than in both the aneurysm and CHD groups.
Despite these differences in gene expression, there were no significant differences in the distribution of the ACE gene I/D polymorphism among the AD, aneurysm, and CHD groups. The same lack of a significant difference was observed in the A8790G polymorphism of the ACE2 gene across these groups. These findings imply that although the expression levels of ACE and ACE2 are altered in AD, genetic polymorphisms in these genes may not be the primary drivers of the disease. Further research with larger sample sizes and broader genetic analyses may be necessary to elucidate the complex interplay between ACE, ACE2, and the development of AD.
Limitations
The study examined the expression levels and genetic polymorphisms of ACE and ACE2 in patients with acute AD and CHD has several limitations. First, the small sample size of 86 patients may restrict the generalizability of the findings and affect statistical power. The research was also limited to acute AD, excluding chronic cases, and only specific genetic polymorphisms were analyzed, potentially overlooking other variants that could influence disease risk. Additionally, the study lacked a normal control group for gene expression analysis and may be subject to regional and ethnic biases, limiting the applicability of the results to other populations. The exclusion of patients on certain medications and those with Marfan syndrome further narrows the scope of the study. Lastly, the study did not explore the functional implications of the genetic variations on protein function or disease pathogenesis, focusing instead on genetic polymorphisms and gene expression alone.
Abbreviations
angiotensin-converting enzyme aortic dissection one-way analysis of variance coronary heart disease matrix metalloproteinase messenger ribonucleic acid polymerase chain reaction renin-angiotensin system
Footnotes
Acknowledgements
All authors should be considered as first co-authors.
Author contributions
YL: concept/design, data analysis/interpretation, drafting article, critical revision of article, approval of article, statistics, and data collection.
ZX: concept/design, data analysis/interpretation, drafting article, critical revision of article, approval of article, statistics, and data collection.
Availability of data and materials
Not applicable. Please contact author for data requests.
Consent for publication
Not applicable.
Declaration of conflicting interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The protocol was approved by the Chengdu Second People’s Hospital, Clinical Research Ethics Committee, and all subjects provided informed consent.
Funding
Not applicable.
