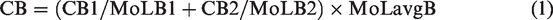Abstract
Objective
The objective of this study was to determine the relationship between the plasma polymyxin B concentration and renal function in elderly patients.
Methods
This prospective, case-control, observational study included elderly patients who received polymyxin B therapy and were divided into an acute kidney injury (AKI) group and a non-AKI group based on their renal function. We monitored the pharmacokinetics and pharmacodynamics of polymyxin B, including the minimum plasma concentration (Cmin), mean blood drug concentration at steady state (Css,avg), and area under the concentration-time curve across 24 h at steady state (AUCss,24h) in both study groups. The plasma polymyxin concentration was determined using high-performance liquid chromatography-tandem mass spectrometry.
Results
The loading doses, Cmin, Css,avg, and AUCss,24h were significantly higher in the AKI group than in the non-AKI group (p < 0.05). Receiver-operating characteristic curve analysis showed that the optimal cutoff values for predicting AKI were 2.94 mg/L for Cmin, 4.14 mg/L for Css,avg, and 99.35 mg·h/L for AUCss,24h, with corresponding sensitivities and specificities ranging from 78.57% to 82.14%.
Conclusion
Monitoring plasma polymyxin B concentrations is essential in elderly patients. Keeping the Cmin below 2.94 mg/L, the Css,avg below 4.14 mg/L, and the AUCss,24h below 99.35 mg h/L may help prevent AKI in this population.
Keywords
Introduction
Systemic infections caused by carbapenem-resistant gram-negative bacteria carry a high risk of mortality and morbidity and are a public health problem worldwide. 1 The prevalence of multidrug-resistant and pan-drug-resistant gram-negative bacteria poses a challenge in terms of anti-infective therapy as a result of the slow development of newer antimicrobial agents. 2 Polymyxins, including polymyxin B (PB), have re-emerged as important agents in combating these infections, despite their known adverse effects from historical use. 3 PB undergoes clearance through pathways that are primarily nonrenal. However, there has been an increasing number of reports of PB-related acute kidney injury (PB-AKI) among critically ill patients over the past few years.4,5
PB is a fermented mixture produced by the polymyxin-like bacterium Bacillus sphaericus, which consists of more than 30 components. The two main components in commercial formulations are polymyxins B1 and B2 (which constitute more than 80% of total PB), and their blood concentrations are usually determined using the Therapeutic Drug Monitoring method. 6 Owing to the narrow therapeutic window of polymyxins, it almost overlaps with the threshold concentration for nephrotoxicity.7–9 Studies have shown that total, loading, and maintenance PB doses >150 mg/d are independent risk factors for PB-AKI; however, clinical data on whether and how these doses are related to the pharmacokinetics of PB, especially in elderly patients, are very limited.10,11
The World Health Organization defines “elderly” as those aged 65 years or older. Alongside global climate change, the other major shift happening globally is population aging. This demographic transition is taking place in nearly every country, with its effects being particularly noticeable in regions such as Europe and the United States. As societies continue to age, the increasing strain placed on healthcare systems will be a critical concern for policymakers and health professionals in the foreseeable future. 12
With advancing age, there is a natural decline in an individual’s physiological reserve across various organ systems. This reduction in functional capacity means that medications can have a disproportionately greater impact on the health of older adults than on their younger counterparts. 13 In the clinical management of critically ill elderly patients, owing to their specific physiology, factors such as reduced hepatic and renal function and reduced plasma protein binding significantly affect the metabolism and clearance of drugs.14,15 Accumulation of drugs in the body may increase the risk of drug-related adverse events. This phenomenon highlights the need for close monitoring when medication is administered in older patients.
Materials and methods
Study design and population
The study follows the tenets of the Declaration of Helsinki, was performed at Zhejiang Hospital, and had a single-center prospective design. The study protocol was approved by the Zhejiang Hospital Ethics Committee, and informed consent was obtained from either the patient directly or a family member. All patient information has been deidentified to ensure patient confidentiality and prevent any form of identification. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 16
The study population consisted of critically ill elderly patients who received PB injections for confirmed or suspected bacterial infections between 1 September, 2022 and 1 September, 2023. The study inclusion criteria were age >65 years and administration of PB intravenously for more than 3 days. Patients with underlying renal failure or pre-existing AKI and those receiving renal replacement therapy or extracorporeal membrane oxygenation were excluded.
According to the Chinese Expert Consensus on Clinical Application of Polymyxin, 17 the loading dose of PB was 2.0–2.5 mg/kg with a 1-h infusion time, which was followed by a maintenance dose of 2.5–3.0 mg/kg/d administered after 12–24 h in two divided doses, with a continuous infusion for more than 1 h.
Definitions
Efficacy
Efficacy was classified as follows: cure (infection is controlled, and all inflammatory indices have returned to normal); improvement (inflammatory indices and body temperature decreased with improvement in clinical manifestations, such as decreased ventilator parameters and improved vital signs); or ineffective (no improvement in inflammatory indices or clinical manifestations). Both cure and improvement were considered effective.
Renal function
Patients with a glomerular filtration rate of <60 mL/min at baseline before infection were defined as having chronic renal insufficiency. PB-AKI was diagnosed if the serum creatinine level increased by more than 50% from baseline. AKI was divided into stages I, II, and III in accordance with the Kidney Disease: Improving Global Outcomes guidelines. 18 Recovery of renal function was defined as the recovery of creatinine to normal levels.
Clinical characteristics
Information on clinical characteristics was collected, including the Acute Physiology and Chronic Health Evaluation II score, underlying diseases, comorbidities, laboratory indices, and dosage of PB, including loading dose, maintenance dose, and number of days of administration.
Plasma PB concentrations
Two blood samples were collected from each patient after at least six injections of PB to determine the steady-state concentration (Css): blood sample A was collected within 0.5 h after the end of the intravenous infusion of PB for determination of the peak concentration (Cmax), and blood sample B was the predose concentration (Cmin) collected immediately after the start of dosing. Samples were collected and centrifuged at 3000 rpm for 10 min for analysis. After centrifugation, the upper plasma layer was stored at –20°C. Plasma PB concentrations were determined by high-performance liquid chromatography-tandem mass spectrometry. 19
Calculation of plasma PB concentration
After determining the concentrations of polymyxins B1 and B2, the concentration of PB was calculated and reported according to the Therapeutic Drug Monitoring guideline. The calculation was based on the molar concentration of PB and its components according to the following equation:
Statistical analysis
Normally distributed measurement data are expressed as the mean ± SD (x ± s), and a t test was used for comparisons between the groups. Counting data were analyzed using the Chi-square test or Fisher exact test. Receiver-operating characteristic (ROC) curve analysis was used to determine the sensitivity and specificity of the Cmax, Cmin, and estimated AUCss,24h for the prediction of AKI. The statistical analysis was performed using SPSS version 20.0 (IBM Corp, Armonk, NY, USA), and the statistical graphs were created using GraphPad Prism 9 software (GraphPad Software Inc, San Diego, CA, USA). A p value of <0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of the AKI and non-AKI groups
A total of 105 critically ill patients who received PB were screened for eligibility to participate in the study. After the exclusion of 33 patients with underlying renal insufficiency, 10 received renal replacement therapy within 3 days of injection of PB, and four received PB injections for <3 days, 58 patients were enrolled in the study (AKI group, n = 28; non-AKI group, n = 30). The demographic and clinical characteristics of the two groups are shown in Table 1. The mean age was slightly but significantly higher in the AKI group than in the non-AKI group (86.93 ± 9.022 years vs 81.10 ± 10.02 years, p < 0.05), as were the Acute Physiology and Chronic Health Evaluation II score (22.14 ± 4.735 vs 19.70 ± 4.036, p < 0.05), the numbers of patients with combined respiratory failure and with shock (both p < 0.05) and the mortality rate (p < 0.05).
Clinical characteristics in the AKI and non-AKI groups.

AKI: acute kidney injury; ALT: alanine aminotransferase; APACHE: Acute Physiology and Chronic Health Evaluation; AST: aspartate transaminase; BUN: blood urea nitrogen; Ccr: creatinine clearance; CPR: cardiopulmonary resuscitation; CRP: C-reactive protein; PCT: procalcitonin; Scr: serum creatinine; TB: total bilirubin; WBC: white blood cell.
PB dosing in the AKI and non-AKI groups
The loading dose was significantly higher in the AKI group than in the non-AKI group (p < 0.05). The maintenance and total doses were slightly but not significantly higher in the AKI group, as was the number of days of treatment with PB (Table 2).
Use of polymyxin B/pharmacokinetics and pharmacodynamics of polymyxin B in the AKI and non-AKI groups.

AKI: acute kidney injury; AUCss,24h: area under the concentration–time curve across 24 h at steady state; Cmax: maximum plasma concentration; Cmin: minimum plasma concentration; Css,avg: average steady-state concentration.
Pharmacokinetics and pharmacodynamics of PB in the AKI and non-AKI groups
Cmin, Css,avg, and AUCss,24h values were significantly higher in the AKI group than in the non-AKI group (p < 0.05), and the Cmax level was similar (Figure 1 and Table 2).

Pharmacokinetics and pharmacodynamics of polymyxin B in the acute kidney injury (AKI) and non-AKI groups.
Receiver-operating characteristic curve analysis of the ability of pharmacokinetics and pharmacodynamics of PB to predict AKI
The sensitivity and specificity of the pharmacokinetics and pharmacodynamics of PB for the prediction of PB-AKI were obtained from the ROC curves. The areas under the Cmin, Css,avg, and AUCss,24h curves were 0.786, 0.713, and 0.713, respectively (Figure 2). The cutoff value for Cmin was 2.94 mg/L (sensitivity 78.57%, specificity 80.00%, and 95% Confidence interval [CI]: 0.661–0.910), for Css,avg was 4.14 mg/L (sensitivity 82.14, specificity 53.33%, and 95% CI: 0.579–0.847), and for AUCss,24h was 99.35 mg.h/L (sensitivity 82.14%, specificity 53.33%, and 95% CI: 0.579–0.847).

Receiver-operating characteristic curve analysis of the ability of the pharmacokinetics and pharmacodynamics of polymyxin B to predict AKI. AKI: acute kidney injury; AUCss,24h: area under the concentration-time curve across 24 h in steady state; Cmin: minimum plasma concentration; Css,avg: average steady-state concentration.
Efficacy of PB and the prognosis in the AKI and non-AKI groups
There was no statistically significant difference in the efficacy rate between the AKI group and the non-AKI group (57.14% vs 66.67%; p > 0.05; Table 3). The mortality rate was significantly higher in the AKI group (57.14% vs 20.00%, p < 0.05).
Efficacy of polymyxin B and the prognosis in the AKI and non-AKI groups.

AKI: acute kidney injury.
Discussion
This pharmacokinetic study has made a significant contribution to our understanding of how to optimize the use of PB in critically ill elderly patients. The current guidelines recommend the use of the standard dosing regimen for PB in individuals with renal insufficiency. 21 However, some clinical studies have found that patients with PB-AKI have a higher AUCss,24h,22,23 and others have reported a higher baseline serum creatinine level or lower baseline glomerular filtration rate in these patients in comparison to those who do not have PB-associated nephrotoxicity.23–25 These findings suggest a heightened susceptibility to the accumulation of PB in patients with renal insufficiency that warrants careful consideration and potential dose adjustments to mitigate the risk of nephrotoxicity. However, these observations have been made primarily in nonelderly populations, highlighting the need for further investigations focusing specifically on the elderly demographic. We monitored and analyzed the plasma concentrations in both groups, and the results indicated that Cmin, Css,avg, and AUCss,24h were markedly higher in the AKI group than in the non-AKI group, whereas the Cmax levels were similar. PB-related renal injury may be associated with the Cmin, Css,avg, and AUCss,24h. Wang et al. 26 reported that the mean AUCss,24h for PB was 68.63 ± 43.43 mg·h/L in their non-AKI group and 93.04 ± 59.52 mg·h/L in their AKI group; both these values are lower than those in our study, in which the mean AUCss,24h for PB was 99.58 ± 21.88 mg·h/L in the non-AKI group and 117.5 ± 23.23 mg·h/L in the AKI group (Figure 1 and Table 2). This finding is most likely attributable to the fact that drug metabolism and clearance are significantly reduced in elderly patients because of their gradual decline in renal function, resulting in a generally high plasma PB concentration.
Our study included critically ill patients older than 65 years of age. According to our findings, the mean patient age was slightly older in the AKI group than in the non-AKI group, suggesting that the risk of kidney injury increases with age. Previous studies have not shown an age-related rate of PB clarity in critically ill patients.27–29 However, the findings of a recent population pharmacokinetics study by Liang et al. 30 seem to support our view. In that study, age had a significant effect on the clearance of PB in critically ill patients with infection, which may reflect the fact that age can affect the functioning of various organs in the body. Furthermore, respiratory failure, shock, and mortality were more common in patients with AKI than in those without AKI. These results suggest that critically ill elderly patients on antibiotic therapy, especially polymyxins, are at higher risk of complications because of weakened renal metabolism and excretion, further underscoring the vulnerability of elderly patients when using polymyxin therapy.
The loading dose was significantly higher in our AKI group than in our non-AKI group (p < 0.05), whereas the maintenance and total doses were slightly but not significantly higher in the AKI group. Notably, the treatment duration was slightly longer in the AKI group, but again, the difference was not statistically significant. The occurrence of PB-AKI may have been related to the loading dose. However, very few studies have reported on the safety (or efficacy) of loading doses of polymyxins, and their conclusions have been inconsistent.31,32 To date, no large-scale clinical study has investigated the correlation between the loading dose of polymyxin and PB-AKI in elderly patients. The findings of the present study suggest that rational adjustment of the loading dose is important to reduce the incidence of AKI in elderly patients treated with polymyxin owing to reduced renal clearance.
Finally, we analyzed the efficacy and prognosis of PB. Although the blood concentration index was markedly higher in the AKI group than in the non-AKI group, there was no advantage in terms of treatment efficacy, and the mortality rate was higher in the AKI group than in the non-AKI group. In a multicenter study, the mortality rate of patients with PB-associated AKI was 17% higher than that of patients without AKI. 33 In our study, the mortality rate of patients with AKI was 37.14% higher than that of those without AKI and was significantly higher than in the abovementioned study probably because our patients were older, and the prognosis of AKI was extremely poor in these cases.
Our study has some inherent limitations. First, the modest sample size restricts the generalizability of our findings. A larger study cohort would bolster the statistical power and validity of our conclusions. Second, the heterogeneity of critically ill patients introduces additional complexity. Unlike the general patient population, these patients have a multitude of factors that can significantly impact drug metabolism, distribution, and clearance. Therefore, the applicability of our results to the broader patient population remains uncertain and requires further investigation. However, in the current social context, there is a significant trend of population aging, leading to a continuous increase in the number of geriatric critical care cases. Critically ill elderly patients will be a challenge for healthcare in the future.
Conclusion
Monitoring of plasma concentrations is important in critically ill elderly patients who are treated with PB. By ensuring that the Cmin, Css,avg, and AUCss,24h remain below respective thresholds of 2.94, 4.14, and 99.35 mg·h/L, it may be possible to decrease the risk of AKI. This approach allows healthcare providers to optimize antibiotic therapy while minimizing potential nephrotoxicity, which is particularly important in this vulnerable patient population. Regular monitoring enables timely adjustments to dosage regimens, thereby promoting both effective and safe treatment.
Abbreviations
acute kidney injury Acute Physiology and Chronic Health Evaluation II blood urea nitrogen creatinine clearance C-reactive protein maximum plasma concentration minimum plasma concentration average steady-state concentration Kidney Disease: Improving Global Outcomes high-performance liquid chromatography-tandem mass spectrometry polymyxin B procalcitonin Risk, Injury, Failure, Loss, End-Stage Society of Critical Care Medicine Society of Infectious Diseases Pharmacists therapeutic drug monitoring World Health Organization extensively drug resistant
Footnotes
Acknowledgements
We thank Liwen Bianji (Edanz) for editing the English text of a draft of this manuscript.
Author contributions
CYZ, YHY, and WMC contributed to data acquisition, analysis, and interpretation. CYZ and KLC contributed to manuscript preparation. YHY, FFY, and CY performed the experiments. SJG supervised the research and revised the manuscript. YHY designed the research. All authors approved the final manuscript.
Consent for publication
All authors have consented to the publication of the present manuscript.
Data Availability
The datasets used during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Ethics approval and consent to participate
This study was approved by the Ethics Committees of the Zhejiang Hospital (2022-49K) and informed consent was obtained from either the patient directly or a family member.
Funding
This study was supported by the Zhejiang Medical and Health Science and Technology project (Grant No. 2023KY433).