Abstract
Umbilical endometriosis (UE) is a rare condition. We have documented the clinical characteristics, management strategies, and follow-up results for five cases treated at our hospital between 1998 and 2020, with patients aged between 31 and 44 years. Patients typically presented with umbilical swelling. In all cases, surgical removal was effective and no complications were reported. Two patients had concurrent ovarian endometriosis and one adenomyosis. No umbilical recurrence was recorded during follow-up. We believe that these findings offer valuable insights for the management of this patient population. Clinical management strategies for this disease should be tailored to each patient and carried out collaboratively by both general and gynaecological physicians.
Background
Umbilical endometriosis (UE), defined as the presence of ectopic endometrial glandular and stromal cells within the umbilicus, represents an unusual location of endometriosis. 1 Its prevalence is estimated to be 0.5–1% in cases of extragenital endometriosis. 2 UE can develop after laparoscopic or other umbilical surgeries, leading to direct implantation of the endometrial tissue into an umbilical port scar (secondary UE), or it can occur without any prior surgical history in the area (primary UE). 3 This is referred to as Villar’s nodule. 4 UE can affect several structures of the abdominal wall. To remove the endometriotic lesion fully, it is necessary to excise the entire umbilicus and surrounding abdominal wall tissues, followed by reconstruction. 5 Therefore, understanding the lesion’s full extent is essential for proper treatment planning. In many cases, a multidisciplinary approach is helpful, particularly when plastic surgery is involved. Few cases with long-term follow-up data have been documented. In this report, we describe the clinical features and long-term outcomes of five cases of primary UE. We also discuss diagnostic, treatment, and pathogenesis aspects.
Methods
We identified UE cases that had been treated at our hospital between 1998 and 2020. Data, including demographic details, clinical and radiological findings at presentation, descriptions of surgical procedures, and follow-up information, were obtained from patient records. All patients were followed for a period of 4 to 26 years, with the observations ending after the women had reached spontaneous menopause. Throughout the follow-up period, patients were monitored via telephone or outpatient visits. A painful mass on the umbilicus found on physical or imaging examination was defined as a clinical recurrence. The reporting of this study conforms to CARE guidelines. 6 The patients gave their informed consent for the publication of their anonymized data. The case series received approval from the Beijing Chao-Yang Hospital’s Institutional Review Board (2024-department-494).
Case reports
Patient 1
A 31-year-old woman (para 0) presented to our hospital in 2020 with a 2-year history of an umbilical hard nodule and a 12-month history of cyclical pain. She complained of dysmenorrhea (visual analogue scale [VAS] score of 7–8). On examination, a 2.2 cm nodule was observed at the umbilicus. Pelvic ultrasound showed adenomyosis. Her serum CA125 level was normal (26.0 U/ml). Based on the diagnosis, the patient opted for surgical management and decided to proceed with the removal of the mass instead of hormonal treatment. A complete excision of the umbilicus was carried out, followed by reconstruction of the umbilical area. She already had a hormone-releasing intrauterine device (Mirena) in place. No additional therapy was requested, and there have been no reports of recurrence up to four years post-surgery.
Patient 2
A 36-year-old woman (gravida 1, para 1), presented at our centre in 2010 with an 8-month history of cyclic umbilical bleeding correlated with menses. Her previous surgery had included an uneventful Caesarean section and concurrent resection of the right ovarian endometrioma. Examination of the umbilicus showed a 1.0 cm raised, pule lesion at the base of the umbilicus. Pelvic examination showed a right adnexal cystic mass and tenderness in left adnexa. The patient gave consent for the excision of the umbilical lesion and laparoscopic cystectomy, along with pre-operative treatment using a gonadotropin-releasing hormone agonist (GnRH-a). The umbilical lesion was identified as a subcutaneous mass on the umbilicus. Dissection was performed until healthy tissue margins were reached, resulting in the removal of approximately 1.7 × 1.5 × 0.5 cm of tissue. Laparoscopic evaluation showed left ovarian cyst and adhesions of the left ovary to the posterior of uterus. Purple-black endometriosis lesions were identified in the Douglas fossa. A cystectomy was performed laparoscopically, and the umbilicus was repaired in two layers suture. The histopathology of the umbilical nodule showed endometrial glands and stroma.
Patient 3
A 36-year-old woman (para 1) presented to our hospital in 2004 with a 13-month history of an umbilical palpable nodule. On examination, a 1-cm nodule was observed at the umbilicus. Pelvic ultrasound showed no evidence of endometriosis in the pelvis. Given the diagnosis, the patient did not wish to undergo hormonal therapy. A total excision of the umbilicus, followed by an umbilical reconstruction, was performed. No therapeutic intervention was required for up to 20 years following the procedure.
Patient 4
A 37-year-old woman (gravida 1, para 1) presented to our hospital in 1999 with a 6-month history of a painful, relatively large umbilical nodule. No other pelvic lesions were observed. Her serum CA125 level was normal (26.8 U/ml). The surgically excised lesion demonstrated endometrial glands and stroma. No recurrence was reported until the patient reached the age of 48 years, when menopause occurred.
Patient 5
A 44-year-old woman (gravida 2, para 1) presented to our hospital in 1998 with a 7-month history of cyclical umbilical bleeding. On examination, a bluish 1.5-cm nodule at the umbilicus was noted and bloody discharge spilled from a small opening at the surface. She refused hormonal treatment. A total excision of the umbilicus, followed by an umbilical reconstruction, was performed. Histological results indicated endometriosis with positive staining for CD10, oestrogen receptors (ER) and progesterone receptors (PR). The patient underwent a hysterectomy and right ovarian cystectomy for multiple myomas and ovarian endometriomas three months later, and no recurrence has been reported 26 years after the surgery.
Summary of findings
Details of the five cases are shown in Table 1. None of the patients had a history of umbilical surgery. Three patients presented with cyclic umbilical bleeding that coincided with their menstrual cycles and were admitted under the care of gynaecologists. Two patients presented with cyclic umbilical swelling related to their menstrual cycles, for six and seven months, respectively, and were admitted under the care of general surgeons. Two patients had concurrent ovarian endometriosis and one had adenomyosis.
Clinical characteristics of the five cases with umbilical endometriosis.
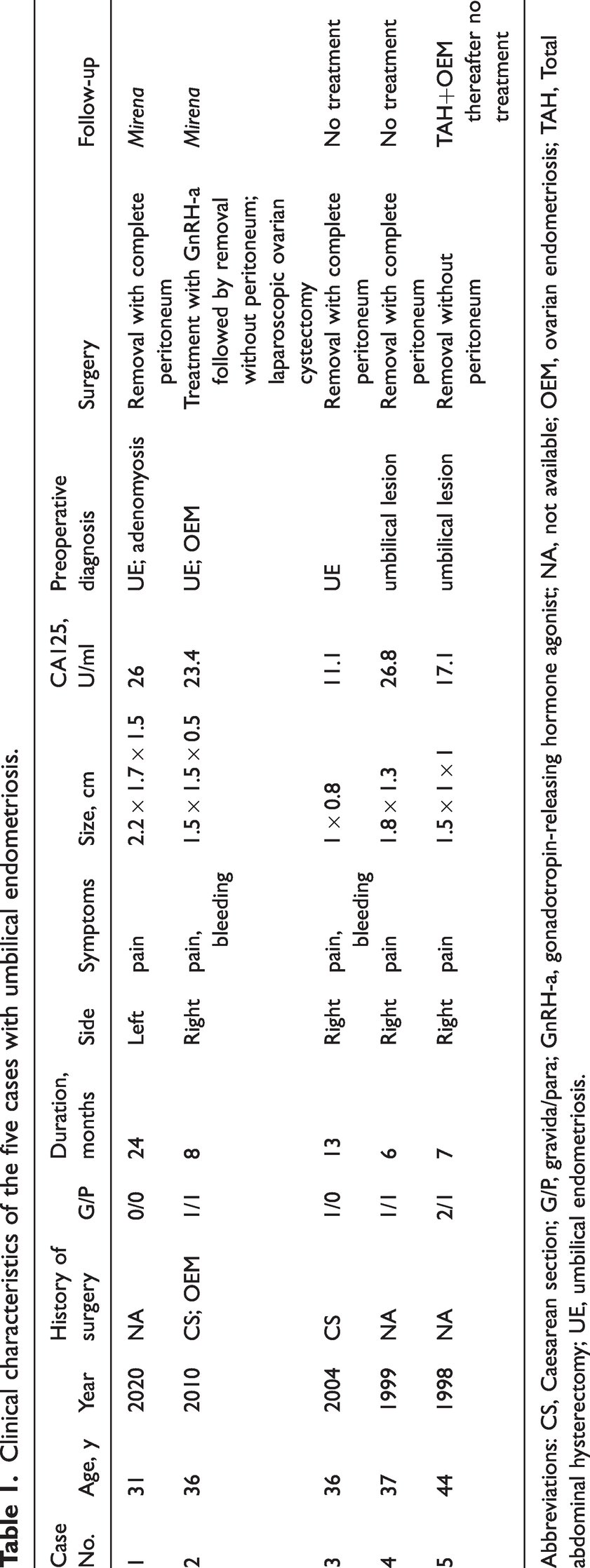
Abbreviations: CS, Caesarean section; G/P, gravida/para; GnRH-a, gonadotropin-releasing hormone agonist; NA, not available; OEM, ovarian endometriosis; TAH, Total abdominal hysterectomy; UE, umbilical endometriosis.
Two lesions presented as raised, reddish, firm, painful irreducible swelling in the umbilicus (Figure 1(a)). The other three lesions presented as normal, firm, irreducible lesions at the base of the umbilicus (Figure 1(b)). Ultrasonography showed a solid nodule under the umbilical skin with poor lower peritoneal integrity in two patients, and a solid nodule under the umbilical skin with good lower peritoneal integrity in three patients. In one patient, magnetic resonance imaging (MRI) showed a normal uterus and ovaries, with no signs of adnexal masses. However, the MRI showed enhancement at the umbilicus along with an underlying soft tissue abnormality (Figures 1(c) and (d)).
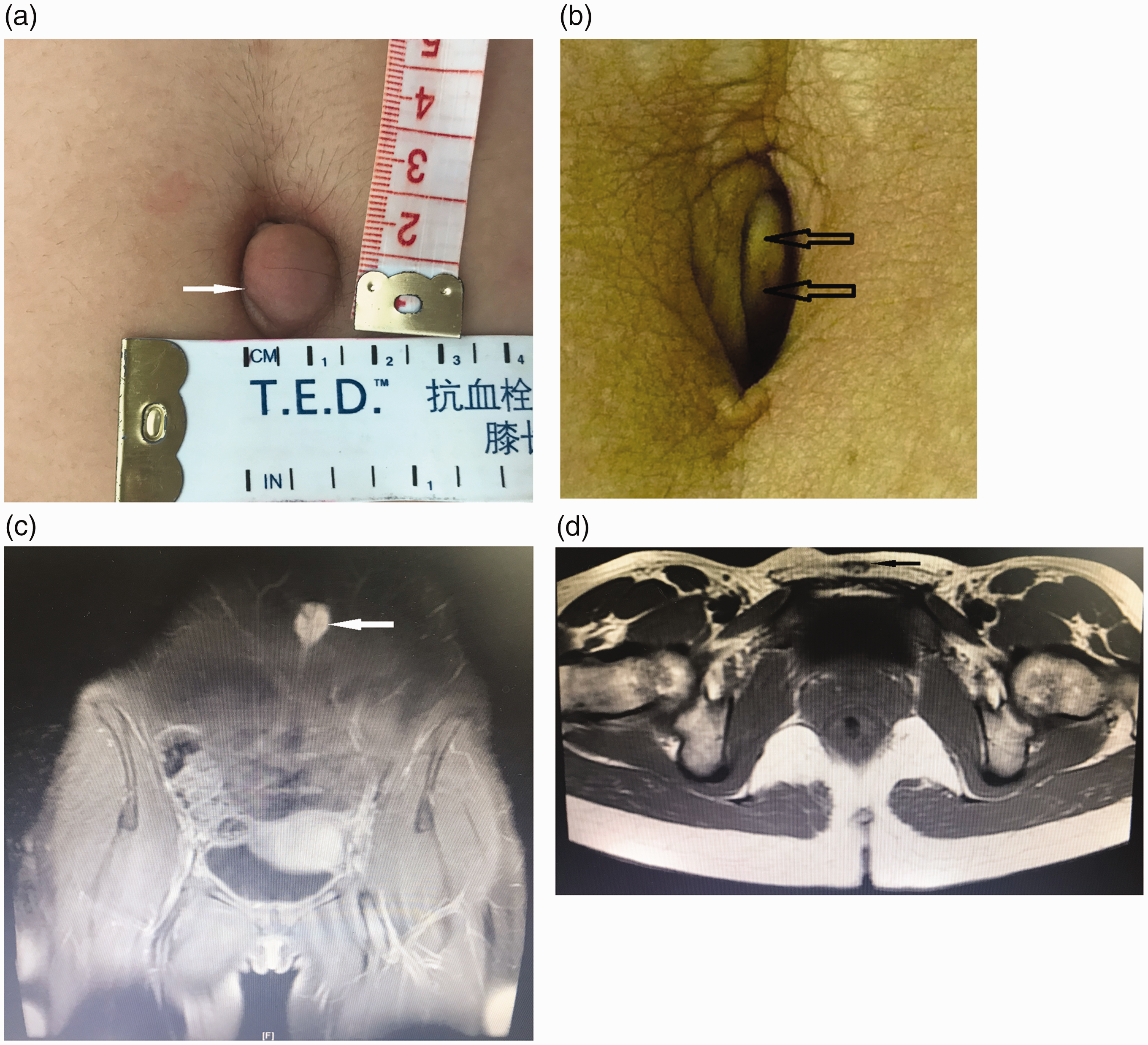
(a) A raised, reddish, firm, painful, ovoid nodule measuring 2.0 × 1.5 cm is seen nested within the umbilicus. (b) A normal, firm, and irreducible lesion at the base of the umbilicus and (c and d) magnetic resonance imaging (MRI) scans showed enhancement at the umbilicus (white arrow) along with an underlying soft tissue abnormality.
One patient received pre-operative medical treatment with GnRH-a for three months, while the other four patients declined pre-operative medical treatment. Three of the umbilical lesions involved the entire umbilicus, down to the level of the fascia. In these cases, dissection was performed, resulting in the removal of approximately 4 × 5 × 3 cm, 3 × 4 cm, and 3 × 3 cm of tissue, along with the complete peritoneum. (Figure 2(a)).

(a) Excised lesion with complete peritoneum following omphalectomy (umbilectomy) and (b) excised lesion without peritoneum.
Two of the umbilical lesions were subcutaneous. In these cases, dissection resulted in approximately 1.7 × 1.5 × 0.5 cm, 1.5 × 1.5 × 1.0 cm of tissue without the peritoneum (Figure 2(b)). The umbilicus healed well after the operation, and there was no evidence of recurrence during follow-up. Three patients were diagnosed with adenomyosis 2, 3, and 2 years after surgery, respectively. Two of these patients were treated with a levonorgestrel-releasing intrauterine system. (Mirena).
Histopathological analysis showed skin tissue with dense fibrosis. There were several glandular structures lined with simple columnar epithelium, and surrounded by stroma, which resembled endometrial stroma (Figure 3(a)). Lesions were diagnosed histologically. Results showed that the glandular and stromal cells were ER and PR positive, the stromal was CD10 positive, and the epithelium was PAX8 positive (Figures 3(b)–(e)).

(a) Haematoxylin and eosin (H&E) staining of the cutaneous part of the umbilical lesion showing endometriotic epithelial (black arrow) and stromal cells (white arrow) near the epidermis and (b–e) expression pattern of oestrogen receptors (ER), progesterone receptors (PR), CD10 and PAX8, respectively. ER and PR were positive in glandular and stroma, and CD10 was positive in stromal cells, but not in epithelial cells. PAX8 was positive in epithelium, but not in stromal cells.
Discussion
The five cases reported here suggest that while UE is rare, primary UE occurred relatively frequently among these cases. Additionally, UE was found to occur concurrently with ovarian endometriosis in two cases and with adenomyosis in one case. Surgical intervention, involving local excision of the nodule or complete removal of the umbilicus, is the primary treatment for UE and has been associated with a low recurrence rate during long-term follow-up. No cases of malignant transformation were identified. A comprehensive examination was conducted to assess the expression of PAX8, CD10, ER, and PR in the five UE cases, making this the first report to thoroughly examine immunostaining in UE.
In our report, 60% patients had no history of abdominal surgery. These findings are consistent with two previous studies, one indicating that 67% of patients had no history of abdominal surgery, 7 and another showing that 73% had no such history. 8 The mechanism behind primary UE may differ from that of other types of abdominal wall endometriosis, which are closely linked to surgical history and scars, particularly from Caesarean sections. 9 Two (40%) of our cases had undergone a Caesarean section and one of these women had also undergone laparoscopic surgery for ovarian endometriosis. Primary UE may be associated with the migration of endometrial cells to the umbilicus via the abdominal cavity, the lymphatic system, or through embryonic remnants in the umbilical fold, such as the urachus and/or the umbilical vessels. 10 We comprehensively examined the lymphatic vessels in the resected specimens of our cases, and found no endometrial-like tissue. Based on a case report and immunohistochemical analysis, it was suggested by some authors that UE lesions may originate from reactivated multipotent cells. 11 Due to the limited number of cases in our report, we were unable to determine its exact pathogenesis. However, distinguishing between primary and secondary UE is crucial for a better understanding of the pathogenesis of this condition.
A systematic review of 179 published article reported that the mean (SD) age of diagnosis of UE was approximately 37.8 (6.8) years. 12 Symptoms uniquely associated with this condition include: umbilical discharge; bleeding; swelling; pain; location of pathologies related to the embryology. 12 Asymptomatic primary UE is extremely unusal. 13 The list of potential diagnoses for UE includes non-cancerous conditions such as haemangioma, umbilical hernia, sebaceous cyst, granuloma, lipoma, abscess, and urachal anomaly, as well as malignant conditions like melanoma, sarcoma, adenocarcinoma, and lymphoma. 14 Several studies have highlighted the challenges in diagnosing primary UE. For example, imaging methods such as ultrasonography, computerized tomography (CT) scans, and MRI exhibit limited sensitivity and specificity, and no technique has yet been able to identify pathognomonic findings consistently. 15 However, imaging is helpful in providing information about nodule size and excluding the possibility of a hernia. 16 Indeed, MRI is able to define the size and position of the lesions and can help rule out any intraabdominal extension. The occurrence of concurrent types of endometrioses has been explored in several studies,7,17,18 with one rare case reporting a patient with inguinal, umbilical, and pelvic endometriosis. 19 According to one study, concurrent or prior peritoneal endometriosis was observed in 86% of cases with UE. 20 Given the development and anatomy of the umbilicus and groin, it has been suggested that UE should be considered a distinct subgroup of abdominal wall endometriosis. This could not only enhance the understanding of the pathogenesis of UE but also improve the clinical management of patients within this group.
Surgery is commonly indicated as a treatment, with local excision of the nodule or removal of the whole umbilicus being the most common approach. In addition, laparoscopy is indicated for the diagnosis and treatment of pelvic endometriosis. 21 Surgical treatment is indicated based on factors such as lesion size, recurrence after previous excision, and symptoms of pelvic pain. To minimize the risk of local recurrence, the preferred approach is total removal of the umbilicus (complete resection). We also stress the significance of ensuring complete resection, which includes the fascia and peritoneum. Medical therapy has been used in a limited number of cases, though the results have been mixed. 22 To our knowledge, preoperative medical treatment for umbilical endometriosis has been documented in only five cases: one treated with combined oral contraceptives, two with GnRH agonists, and two with oral progestogens. 12 In that study, symptoms improved in 60% (3/5) of cases following medical treatment. In the largest and most comprehensive study on UE, which examined the efficacy of hormonal therapy and postoperative recurrence rates, only a small number of patients (4/96) received hormone therapy alone. As a result, no definitive conclusions could be made regarding the effectiveness of hormonal therapy in this condition. 7
Detailed histopathological findings in UE are essential not only for reaching a definitive diagnosis but also for clarifying its origin and pathogenesis. While CD10 and PAX8 staining are commonly used in pathology, their application in gynaecology is less frequent. Given that endometrial glands can be present in the oedematous stroma, CD10 staining may prove diagnostically useful in this context. 23 PAX8 is a lineage marker for Mullerian lineage epithelial cells, which includes normal/nonneoplastic Mullerian epithelial cells and Mullerian primary and metastatic tumour cells. 24 PAX8 may also be a useful sensitive marker for endometriotic epithelial cells. 25 It is important to note that UE retains the expression of this lineage marker, similar to ovarian endometriomas, despite occurring at different sites and potentially having distinct causes and developmental mechanisms.
To the best of our knowledge, this is the largest study on UE conducted at a single institution. We believe that these findings will offer valuable insights for the management of this patient population. While these case histories have provided clinical findings and a pathognomonic history of UE, our study has limitations, including its retrospective nature and being conducted at a single centre. Further, large scale studies are required to validate our findings. Diagnosis of UE is often made at the time of surgery. We cannot definitively conclude whether expectant or medical management is effective for UE. Healthcare providers should be aware of the possibility of UE, so they can consider surgical intervention at the same time as any gynaecological procedure if needed.
In conclusion, most cases of UE are primary rather than secondary, and pelvic endometriosis can coexist as a comorbidity with UE. Clinical management strategies for this disease should be tailored to each patient and carried out collaboratively by both general and gynaecological physicians. It is recommended that patients be followed up by a gynaecologist to monitor any intra-abdominal disease and provide guidance on fertility concerns.
Footnotes
Acknowledgements
The authors wish to thank Dr Jinhua Leng who gave her useful information as well as for her assistance in the writing the text.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was partly supported by National key R&D Program of China Number: 2017YFC1001204 (Grant-in-Aid for Scientific Research for CHD Liu).
