Abstract
Giant cell arteritis and subacute thyroiditis are often regarded as clinically distinct diseases with differing pathogenesis. We report a rare case of coexisting giant cell arteritis and subacute thyroiditis that were both successfully managed using glucocorticoid therapy. Ultrasound plays a vital role in the diagnosis and treatment of these diseases. A review of the literature revealed that cases of giant cell arteritis complicated by subacute thyroiditis are very rare, and their pathogenesis and association are still unclear. We summarized the clinical features of giant cell arteritis complicated with subacute thyroiditis and found that none of the patients had symptoms associated with polymyalgia rheumatica. Subacute thyroiditis could be misdiagnosed as giant cell arteritis, and ultrasound is a useful tool for distinguishing the two diseases. Glucocorticoids are effective in the active period in patients with giant cell arteritis complicated with subacute thyroiditis. This case report would encourage clinicians to pay more attention to this phenomenon.
Keywords
Introduction
Giant cell arteritis (GCA) is one of the most common primary systemic vasculitis in people older than 50 years. GCA mainly involves large-sized and medium-sized arteries and has a variety of clinical manifestations, such as headache, visual impairment, sudden loss of vision and jaw claudication, among others.1,2 Subacute thyroiditis, also known as subacute granulomatous inflammation or De Quervain thyroiditis, is one of the most common causes of thyroid pain and is usually characterized by symptoms such as low-grade fever, fatigue and sore throat. 3 GCA and subacute thyroiditis are often viewed as clinically distinct diseases with differing pathogenesis. Herein, we report the case of a patient with GCA and subacute thyroiditis in whom the administration of glucocorticoids relieved the GCA and subacute thyroiditis.
Case presentation
A 65-year-old Chinese man was admitted to our hospital (a tertiary public hospital) with a 1-month history of a gradually worsening headache and dizziness. The headache was localized to the bilateral temples, forehead and occipital region and was associated with nausea and fatigue. The patient denied having flu-like symptoms or thyroiditis symptoms (e.g. diaphoresis) before the initiation of his presenting symptoms. Two weeks after the onset of headaches, he experienced pain in the left side of the neck that was exacerbated by swallowing. He visited our neurology outpatient clinic and denied having fever, vertigo, diplopia, proximal muscle pain or stiffness and palpitations. He was prescribed antibiotics for 4 days, but the symptoms persisted. He had loss of appetite and weight loss of 5 kg within 2 months. The patient had no comorbidities other than gout but was allergic to allopurinol. He was a nonsmoker and did not consume alcohol. His family history was unremarkable. On admission, his vital signs were as follows: body temperature, 37.3°C; pulse rate, 98 beats/min; respiratory rate, 20 breaths/min; and blood pressure, 98/58 mm Hg. The numeric rating scale score for bilateral temple tenderness was 5. A nodular lesion measuring approximately 2 cm in diameter was palpable in the right lobe of thyroid. There were no other significant findings on physical examination. Laboratory results at admission are as follows: erythrocyte sedimentation rate (ESR), 113 mm/h (normal range: 0–15 mm/h); C-reactive protein (CRP) level, 120 mg/L (normal range: 0–8 mg/L); T3, 10.33 ng/mL (normal range: 0.58–1.59 ng/mL); FT3, 6.27 pg/mL (normal range: 1.71–3.71 pg/mL); T4, 10.33 μg/dL (normal range: 4.87–11.72 μg/dL); FT4, 2.88 ng/dL (normal range: 0.70–1.48 ng/dL); and TSH, 0.01 mIU/L (normal range: 0.35–4.94 mIU/L). Antinuclear antibody, rheumatoid factor and antineutrophil cytoplasmic antibody test results were negative. Serum protein electrophoresis was within normal limits, while serologic tests for Epstein–Barr virus, cytomegalovirus, hepatitis B and C viruses and T-spot showed negative results. A cranial computed tomography (CT) scan did not reveal any significant abnormalities. Cranial magnetic resonance imaging (MRI) revealed multiple small ischemic lesions in the bilateral frontoparietal white matter. The patient underwent lumbar puncture; cytologic evaluation of the cerebrospinal fluid (CSF) revealed normal findings. CSF analysis also demonstrated a negative Pandy reaction, normal concentrations of chloride, glucose and microproteins, and negative results for Gram, ink and acid-fast staining. CSF culture yielded no microorganisms.
In the beginning, we performed extensive tests, including blood routine, liver function, renal function, CRP\ESR, etc., in order to evaluate the overall condition of the patient. For headache and neck pain, we first screened for the possibility of infection such as viral meningitis, tuberculous meningitis, etc., improved the pathogen detection, and screened for organic diseases of the brain. Brain CT and MRI examination were completed. We found no evidence of infection in the patient. We discussed the abnormal findings on MRI with a neurologist and agreed that the lesion was mild and did not explain the patient’s symptoms. Finally, to test for thyroiditis and GCA, we performed in-depth thyroid B-mode ultrasound (US) and vascular B-mode US, respectively. The temporal vessels revealed coarse and thickened (0.05 cm) intima of both temporal arteries and a halo in the transverse plane (Figure 1(a)). A Doppler US of other vessels revealed a coarse intima of both carotid arteries; however, there was no increase in the intima-media thickness and no stenosis. The lower extremity arteries, aortic arch and abdominal aorta also exhibited a coarse intima and atherosclerotic plaque. The iliac and renal arteries appeared normal. A Doppler US of the thyroid gland revealed a hypoechoic and diffusely heterogeneous thyroid with increased blood flow. A nodule with regular margins was evident in the upper pole of the left lobe of thyroid (Figure 1(b)). Moreover, a thyroid scan revealed decreased uptake of technetium-99m pertechnetate, consistent with subacute thyroiditis. Consequently, we made a diagnosis of GCA accompanied by subacute thyroiditis. Given the features of the bilateral temporal arteritis revealed by US, we recommended a temporal artery biopsy; however, the patient declined. Due to suspicion of GCA, steroid treatment was commenced immediately. The patient received 40 mg of methylprednisolone daily. The symptoms rapidly subsided, and the patient improved and was discharged after 7 days. At the 8-month follow-up, he was asymptomatic on 4 mg daily of methylprednisolone. A repeat US revealed coarse intima of both temporal arteries, but the intimal thickness had not increased. The other vessels had not been affected by treatment (Figure 2(a)). Moreover, B-mode US of the long-axis section of the left lobe of thyroid revealed that the low and uneven echoes had become moderate (Figure 2(b)). The thyroid volume reduced; however, the blood supply was normal. The CRP level decreased to 1.1 mg/L, with an ESR of 7 mm/h.

(a) Before treatment, the wall of the left temporal artery of the patient has obvious thickening, and the cross-section of the blood vessel shows a hypoechoic halo and (b) the left thyroid parenchyma of the patient is diffusely reduced and uneven, and the parenchyma is rich in blood supply.

(a) The patient's left temporal artery trunk cross-section can be seen by ultrasound, the wall is thin (arrow) and the original halo sign disappeared and (b) the long-axis section of the left lobe of thyroid was seen through ultrasound, showing that the original obviously low and uneven echo changed to moderate echo.
Discussion
This is a rare case of concomitant GCA and subacute thyroiditis. Because some subacute thyroiditis is self-limited, it is easy to be missed by clinicians. The pathogenesis and association between GCA and subacute thyroiditis are still unclear. Studies have found that patients with GCA have a higher incidence of hypothyroidism compared with the control group. 4 Milioniz et al. 5 suggested that GCA may involve the thyroid artery, leading to the occurrence of subacute thyroiditis; however, they did not provide substantial evidence to support this explanation in their report. GCA and subacute thyroiditis may be caused by virus-induced changes in the autoantigens in the arterial wall and thyroid gland, along with subsequent induced immune responses that result in clinical symptoms. Because of the recent serologic evidence of infection, the patient was not available for testing, so this assumption remained speculative. In our case, we examined for respiratory pathogens and performed CSF tests but found no evidence of infection. Additionally, B-mode US found no evidence of thyroid artery involvement. Therefore, the pathogenesis of GCA with subacute thyroiditis remains unknown.
We summarized the characteristics of GCA complicated with subacute thyroiditis reported so far (Table 1).5–7 Almost all patients had neck pain in the course of the disease, which was easy to ignore because it was similar to the characteristics of GCA itself. We also found reports of cases that presented only with subacute thyroiditis but were misdiagnosed as GCA. 6 This further indicates that the symptoms of the two diseases can be easily confused. However, US plays a major role in distinguishing these two conditions, with a high value in the diagnosis of GCA. Studies have reported that the specificity of US in the diagnosis of vasculitis is between 90% and 100%. The advantages of US in GCA over other imaging modalities are its availability, safety and tolerability. 8 However, this does not mean that temporal artery US can replace temporal artery biopsy, which is a limitation of our case. US is also useful in the diagnosis of subacute thyroiditis. Furthermore, a thorough comparison of available literature5,7,9 revealed that all four reported cases of patients with GCA combined with subclinical thyroiditis had intracranial vascular involvement, and none of the four patients showed signs of peripheral vascular involvement, such as intermittent claudication of limbs. Similarly, none of the four patients had symptoms associated with polymyalgia rheumatica. We found that during the active phase of the disease, patients with GCA complicated by subacute thyroiditis are responsive to glucocorticoid therapy. Therefore, in terms of treatment, these patients are not different from patients with only GCA. Unfortunately, subacute thyroiditis is often overlooked or missed by clinicians.
Clinical features of giant cell arteritis complicated with subacute thyroiditis.
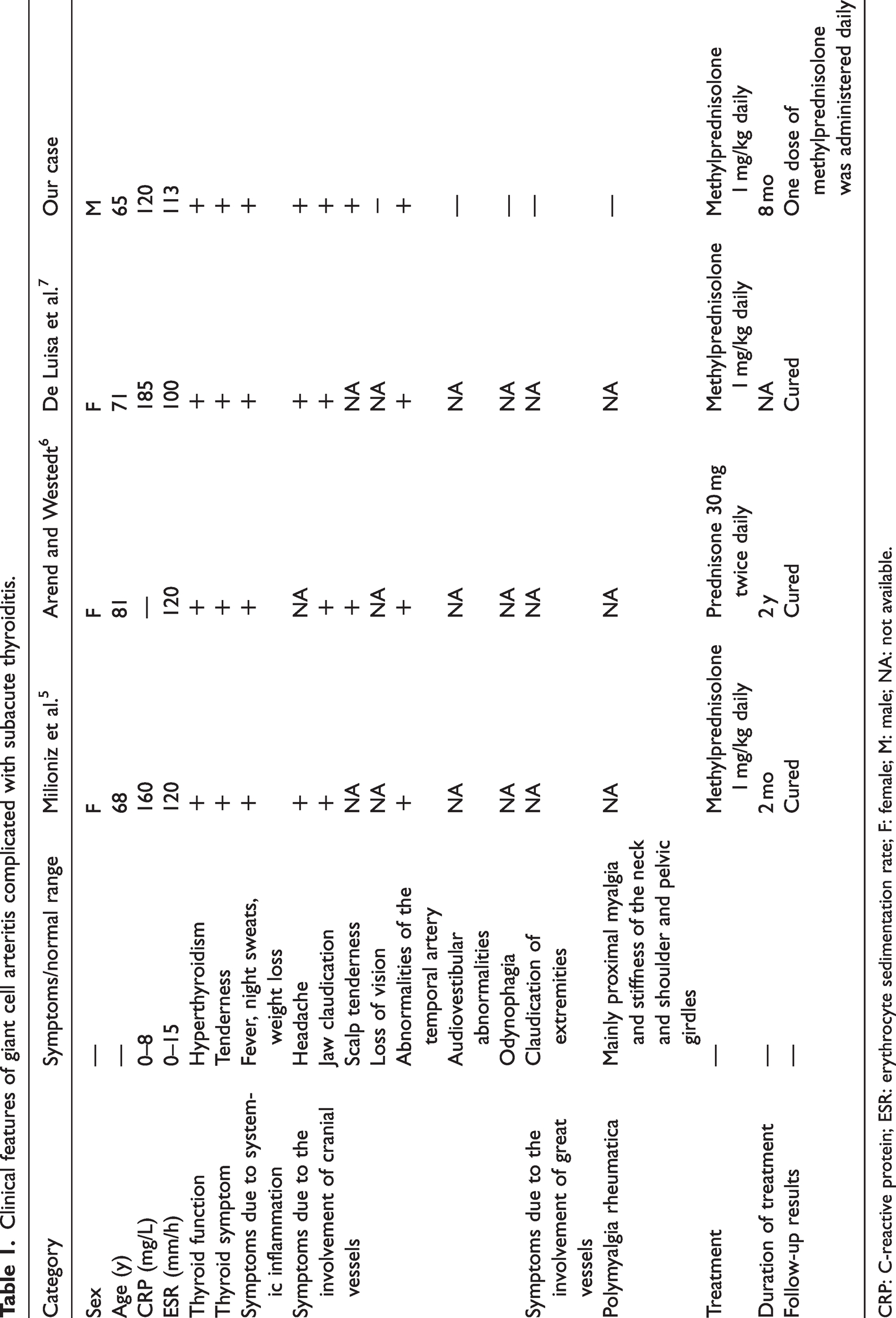
CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; F: female; M: male; NA: not available.
Conclusions
Subacute thyroiditis may be missed because most patient-related symptoms can be quickly relieved after glucocorticoid treatment. Since such cases are rarely reported, it is unclear whether their occurrence is truly a low-probability event or missed by clinicians in clinical settings. B-mode US plays an important role in the differential diagnosis and follow-up of patients with GCA complicated with subacute thyroiditis. We hope that through such case reports, more clinicians will pay close attention to this phenomenon.
Footnotes
Acknowledgements
The authors thank the patient for their commitment to this research.
Author contributions
All authors of this article were actively involved in the diagnosis and treatment process, follow-up process and case analysis. Division of labor and cooperation were implemented for discussion; thus, all authors contributed considerably to the work. All authors agreed on the journal to which the manuscript was submitted and are responsible for all aspects of the manuscript.
Consent for publication
Informed consent has been obtained from the patient for the publication of the case report.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Grant No. 2019ZD043 from the Health Commission of Zhejiang Province and Grant No. 2017ZYC-A19 from Zhejiang Medical Association.
