Abstract
Inflammatory myofibroblastic tumor (IMT) is a rare neoplasm with intermediate biological potential. The stomach is an uncommon site for IMTs, and gastric IMTs are typically confined to the submucosal layer. This report presents a rare case of a 29-year-old woman with a gastric IMT located in the gastric fundus, exhibiting local invasion into the esophagus and spleen.
Introduction
Inflammatory myofibroblastic tumor (IMT) is classified as a tumor with intermediate malignant potential. 1 Gastric IMTs (G-IMTs) are rare, typically confined to the submucosal layer, and primarily associated with local recurrence; metastasis is uncommon.2,3 In this paper, we report a case of G-IMT with invasion of both the esophagus and spleen and review the relevant literature on IMT.
Case report
This study adheres to the CARE guidelines. 4 All patient details were de-identified, and patient consent was obtained for treatment. In November 2020, a 30-year-old woman presented to Shenzhen Second People’s Hospital with a 1-year history of recurrent melena accompanied by nausea and vomiting. A prior endoscopy had identified a gastric fundus lesion, and biopsy results indicated chronic non-atrophic gastritis with lymphoid hyperplasia. On admission, the abdominal examination revealed no significant abnormalities, although the patient exhibited mild anemia (hemoglobin: 108.0 g/L). Endoscopic ultrasonography detected a 4.0-cm hypoechoic mass in the gastric fundus and a 6.9- × 5.3-mm lymph node outside the gastric wall. The preliminary diagnosis was a gastrointestinal stromal tumor. Contrast-enhanced computed tomography (CT) revealed irregular thickening of the esophagus, cardia, and gastric fundus, with inhomogeneous enhancement and multiple enlarged lymph nodes. Double contrast-enhanced ultrasonography identified hypoechoic areas in the posterior wall of the gastric fundus, cardia, and abdominal esophagus, with rapid contrast agent filling and delayed clearance, favoring a diagnosis of gastric lymphoma. Although the diagnosis remained inconclusive, the gastric mass was considered likely malignant, prompting laparoscopic resection (Figure 1). Laparoscopy revealed a tumor in the posterior wall of the gastric fundus extending to the cardia, invading the abdominal esophagus, penetrating the serosa, and involving the spleen (Figure 2). An open abdominal approach was ultimately selected for D2 total gastrectomy combined with splenectomy and reconstruction of the digestive tract using Roux-en-Y anastomosis. The surgical specimen underwent pathological examination, and the findings are shown in Figure 3. Microscopic examination revealed spindle cells infiltrating the entire thickness of the gastric wall with mild to moderate atypia, accompanied by significant infiltration of lymphocytes and plasma cells. No tumor metastasis was observed in the lymph nodes. Immunohistochemistry results showed strong positivity for anaplastic lymphoma kinase (ALK), smooth muscle actin, and desmin with negativity for CD117, DOG1, S-100, CD34, and CD21. The final histopathological diagnosis was G-IMT with esophageal and splenic invasion. At the time of this writing, the patient had remained recurrence-free throughout 3 years of follow-up as confirmed by regular CT and ultrasound scans.

Contrast-enhanced computed tomography and ultrasound findings. (a, b) Gastric tumor (blue arrow) and abdominal esophageal thickening (red arrow). (c, d) Endoscopic ultrasonography showing a 4-cm tumor and enlarged lymph nodes (yellow arrow) and (e, f) double contrast-enhanced ultrasonography findings.
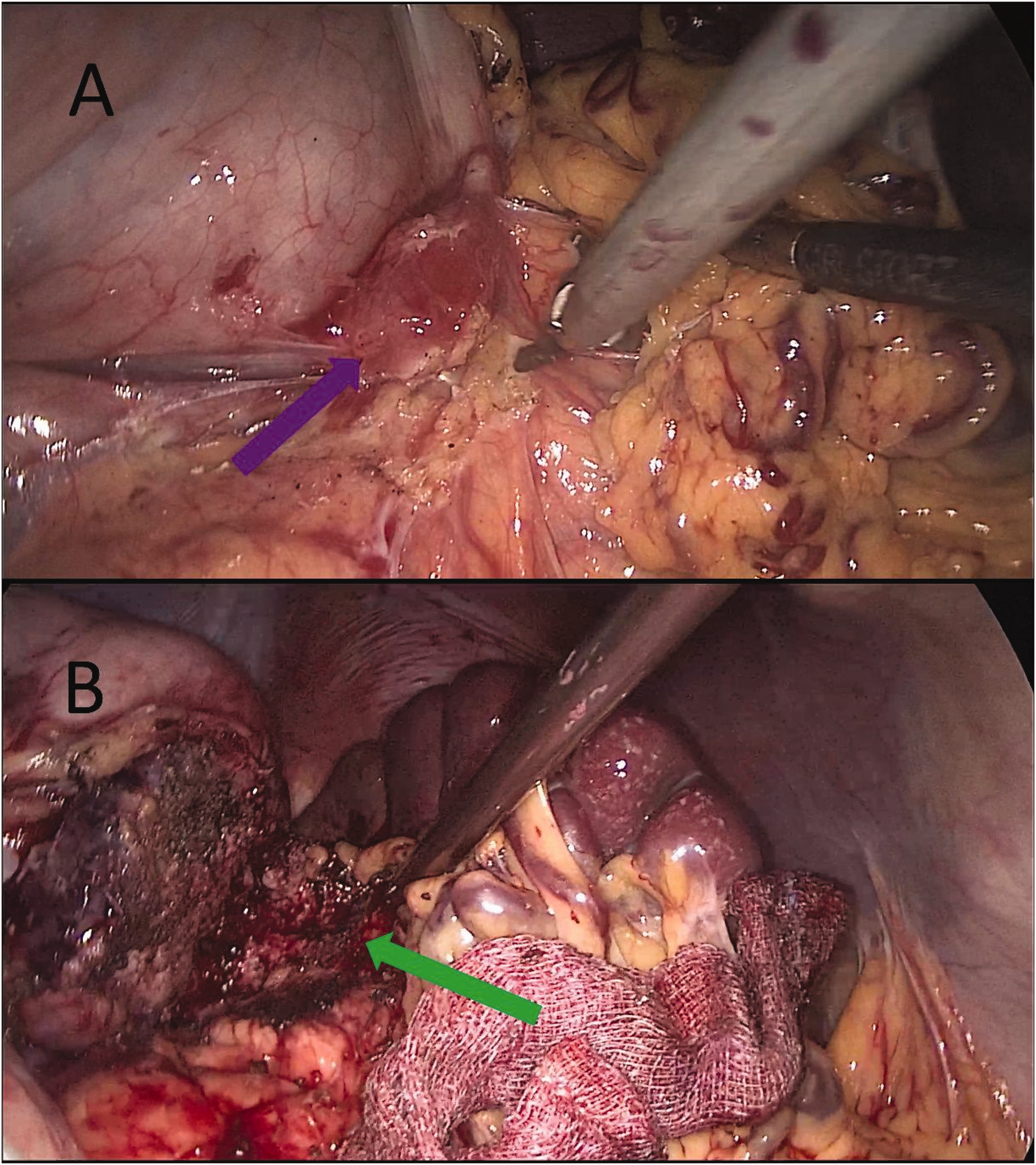
Laparoscopic examination findings. (a) Tumor on the posterior wall of the gastric fundus penetrating the serosa (purple arrow) and (b) tumor infiltrating the spleen (green arrow).

Pathological and immunohistochemical results. (a) Pathology showing spindle cells infiltrating the entire thickness of the gastric wall with mild to moderate atypia and significant infiltration of lymphocytes and plasma cells. No tumor metastasis observed in the lymph nodes. Immunohistochemical results: positive for (b) ALK, (c) desmin, and (d) smooth muscle actin; negative for (e) CD21, (f) DOG1, (g) CD117, (h) S-100, and (i) CD34.
Discussion
IMT is a rare neoplasm that was first identified in the lung in 1939. 5 Initially, it was considered a rare form of inflammatory pseudotumor. 6 In 2013, the World Health Organization classified IMT as a tumor with indeterminate malignant potential. 7 The etiology of IMT is multifactorial, potentially involving infections, surgical or traumatic events, autoimmune disorders, and genetic factors such as ALK mutations. 3
Preoperative diagnosis of IMT is challenging because of the lack of specific clinical manifestations and imaging features.2,8 On CT imaging, G-IMT may appear as a well-defined, enhancing submucosal tumor or as an irregularly enhancing thickening of the gastric wall, often mimicking gastrointestinal stromal tumors, leiomyomas, or gastric carcinomas. 9 The submucosal location of these tumors frequently results in insufficient depth of endoscopic biopsy specimens, making definitive diagnosis difficult. 9 Preoperative needle biopsy and intraoperative frozen sections are also often inconclusive. 10 A retrospective analysis of 41 G-IMT cases revealed that only 2 patients received a confirmed diagnosis through preoperative biopsy. 3 In our case, endoscopic ultrasonography and double contrast-enhanced ultrasonography provided conflicting diagnoses, while a biopsy performed during gastroscopy at an external hospital failed to yield a definitive diagnosis.
The diagnosis of IMT is primarily based on microscopic observation and immunohistochemical analysis of postoperative gross specimens. 11 Microscopically, IMT is characterized by the proliferation of mildly atypical myofibroblasts arranged within a mucoid-to-collagenous matrix, infiltrated by plasma cells, lymphocytes, and eosinophils.1,8 Immunohistochemistry of IMT typically shows positive staining for one or more of the following markers: ALK, smooth muscle actin, vimentin, desmin, and calponin.2,8,12,13 More than 50% of patients with IMT exhibit ALK gene rearrangements on the short arm of chromosome 2 in the p21-p23 region.2,8,14 ALK positivity is a favorable prognostic indicator,2,12,15 whereas ALK-negative IMTs are associated with a higher risk of distant metastases. 15
The majority of G-IMTs are localized to the submucosal layer, most commonly found in the prepyloric region, gastric body, and antrum, with metastasis being a rare occurrence.2,8 Reports of G-IMTs with local invasion into other organs, such as the spleen, esophagus, liver, pancreas, diaphragm, and lung, are uncommon.3,16 Lazure et al. 17 reported two pediatric cases, one with G-IMT invading the spleen and the other with G-IMT invading the lower esophagus and extending into the mediastinum. Kim et al. 18 described a young adult with G-IMT and peritoneal dissemination, along with its imaging manifestations. Cho et al. 19 described a case of G-IMT presenting with fever, invading the pancreas, and encircling the splenic vessels. He et al. 16 reported G-IMT invading the spleen and diaphragm, necessitating resection of the tumor, spleen, and part of the diaphragm. Chen et al. 20 described extensive IMT involving the greater curvature of the stomach and upper spleen, with the patient achieving a cure following total gastrectomy and splenectomy. Prillwitz et al. 21 reported a pediatric case of G-IMT invading the left hepatic lobe, left diaphragm, and esophageal lymph nodes, treated with extended resection. Billingsley et al. 22 presented a case of a young patient with G-IMT involving the esophagus who achieved complete tumor regression after 10 months of crizotinib treatment. To the best of our knowledge, this study represents the first report of G-IMT simultaneously invading the esophagus and spleen.
Currently, there are no established treatment protocols for IMT. The optimal treatment remains complete surgical resection of the tumor.1,12,23 Incomplete resection is the most common factor contributing to tumor recurrence. To achieve curative resection (R0), the preferred approaches are total surgical excision or wide local excision, both of which involve removing the tumor along with its surrounding healthy tissue. 8 For patients with inoperable tumors or distant metastases, pharmacological intervention serves as an alternative treatment option. The efficacy of anti-inflammatory drugs, including corticosteroids and nonsteroidal anti-inflammatory drugs, remains debated within the medical community. 1 Anthracycline-based or alkylating agent-based chemotherapy regimens may offer benefits for some patients with IMT.1,8 Clinical trials have shown that targeted ALK inhibitors, such as crizotinib, provide therapeutic benefits for patients with ALK-positive IMT.1,8 For ALK-negative IMT, immunotherapy is emerging as a potentially effective treatment option. 8
Conclusion
We have presented a rare case of G-IMT with local invasion of the esophagus and spleen. The rarity of IMT contributes to the ongoing lack of consensus on its diagnosis and treatment. Currently, there are no specific clinical symptoms, laboratory findings, or imaging characteristics unique to G-IMT. Diagnosis relies heavily on histological examination and immunohistochemistry. Enhancing the preoperative diagnostic rate of IMT through novel approaches is crucial because it significantly influences treatment planning. Achieving adequate surgical margins is essential for curative treatment. However, for patients who are not surgical candidates or those with metastatic disease, a clear consensus on pharmacotherapy regimens—including drug types, combination strategies, treatment durations, and dosages—is lacking. There is an urgent need for multifaceted, large-scale clinical trials to address these gaps. Additionally, developing neoadjuvant drug therapy regimens for patients with resectable IMT could improve resection rates, preserve organ function, reduce recurrence and metastasis risks, and enhance overall prognosis. Despite the low incidence of IMT, international clinical trial collaborations and systematic registration of IMT diagnosis and treatment processes by individual physicians can significantly enhance the efficiency of establishing standardized diagnostic and treatment protocols.
Footnotes
Authors contribution
L.Z.: Conceptualization, investigation, writing – original draft, writing – review and editing. Z.Y. and H.D.: Data curation. H.L.: Writing – review and editing. Y.Q.: Conceptualization, investigation, writing – review and editing.
Consent statement
All identifying details of the patient have been removed, thus waiving the need for informed consent for publication.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author (Ying Qin). Data that could compromise the privacy of the patient are not publicly available.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Institutional review board approval was not required because of the retrospective nature of the study.
Funding
None.
