Abstract
Objective
To evaluate the efficacy of the combined application of platelet-rich plasma (PRP) and negative pressure wound therapy (NPWT) in cavitary infected wounds, in addition to the parameter settings of NPWT.
Methods
The outcomes of patients with cavitary wounds who received debridement with either PRP plus NPWT (PRP group) or NPWT alone (controls) as part of their treatment protocol were retrospectively compared by examining preoperative and postoperative data on high-sensitivity C-reactive protein (hs-CRP), erythrocyte sedimentation rate (ESR), white blood cell (WBC) count, neutrophil percentage, bacterial cultures, pain (measured using visual analogue scale [VAS] scores), postoperative hospitalization duration, and effective hospitalization days.
Results
A total of 24 patients were included (11 who received PRP plus NPWT and 13 who received NPWT alone). Postoperative hs-CRP levels, ESR, VAS scores, and effective hospitalization days were found to be significantly reduced in the PRP group versus controls. Moreover, the PRP group exhibited a significantly elevated rate of bacterial culture conversion at postoperative day 3.
Conclusions
The integration of surgical debridement with PRP and NPWT in the management of infected cavitary wounds may expedite infection containment and bacterial eradication, stimulate granulation tissue formation, and facilitate cavity closure, offering a novel and uncomplicated approach to wound healing.
Keywords
Introduction
Numerous factors, such as diseases, trauma, decreased immune function, and other triggers, may compromise the integrity of the skin barrier, allowing pathogenic bacteria to invade the epidermis, dermis, and subcutaneous tissue, resulting in the development of skin and soft tissue infections (SSTIs). 1 Severe SSTIs may be accompanied by cutaneous and subcutaneous defects, potentially resulting in bone and joint exposure and subsequent osteomyelitis. Managing cavitary wounds can be difficult due to their high risk of infection. Upon hospital admission, the initial intervention for this group of patients consists of thorough debridement and application of negative pressure therapy. Further treatment includes surgeries to remove dead space and close the wound.2,3 Soft tissue grafting is necessary for cavitary wounds, but it can cause damage to the donor site, and the technical requirements and risks involved in the procedure make it difficult to manage wounds in primary care hospitals.
Platelet‐rich plasma (PRP) has demonstrated efficacy in various medical fields, including aesthetic plastic surgery, orthopaedics, and dentistry, and is considered a promising therapy in regenerative medicine due to its platelet-rich composition that releases growth factors for cell growth and tissue repair. 4 Additionally, PRP contains anti-inflammatory cytokines that may reduce pain by modulating inflammatory responses. Chemokines from PRP help fight infections by attracting platelets and leukocytes. 5 Negative pressure wound therapy (NPWT) is commonly used in different types of wounds to remove exudate, stimulate healing, and promote tissue growth.6,7
The concurrent utilization of PRP and NPWT in infected cavitary wounds is infrequently documented in the literature, with a lack of standardized criteria for its specific application and timing in clinical practice. The aim of the present study was to examine the clinical outcomes of combining PRP with NPWT to treat infected cavitary wounds, while also proposing empirical guidelines for the application of PRP and optimal negative pressure settings for NPWT in such wounds.
Patients and methods
Study population
Patients who presented with infected cavitary wounds necessitating surgical intervention, and who were evaluated at the First Affiliated Hospital of Wannan Medical College, Wuhu, China between June 2022 and December 2023, were sequentially enrolled into this retrospective study. Patients were categorized into one of two study groups based on whether they received NPWT without PRP (control group, enrolled between June 2022 and December 2022) or PRP in addition to NPWT (PRP group, enrolled between December 2022 and December 2023).
This retrospective study was conducted as a controlled investigation, with all treatment interventions performed exclusively at the First Affiliated Hospital of Wannan Medical College. Written informed consent to study participation was obtained from each patient and all patient details were de-identified. In addition, participants in the PRP group provided informed consent to the collection of blood components and subsequent PRP treatment. The study adhered to the principles outlined in the Helsinki Declaration of 1975 as revised in 2013, and the study was approved by the ethics committee of the First Affiliated Hospital of Wannan Medical College (2022 Ethical Review No. 48). The reporting of this study conforms to ‘Minimum Information for Studies Evaluating Biologics in Orthopaedics’ (MIBO) PRP guidelines. 8
Inclusion criteria
Patients who met the following criteria were included in the study: SSTI grade II and above; 1 stage 3 and above of the 2019 International Guidelines for the Prevention and Treatment of Pressure Ulcers; 9 positive bacterial culture; elevated infection index on haematological examination; and absence of contraindications to surgery.
Exclusion criteria
Patients who met the following criteria were excluded from the study: combination of other systemic diseases; patients with platelet count <100 × 109/L and/or haemoglobin <100 g/L; malignant ulcers; and/or severe necrosis of limbs deemed non-preservable.
Platelet-rich plasma collection and preparation
Eligible patients, excluding those with contraindications to blood transfusion and collection, underwent blood component collection using a fully automated blood component separator (Fresenius Kabi, Lake Zurich, IL, USA) under the supervision of a full-time physician in the hospital's transfusion department. A qualified medical professional (SW) within the Department of Burn and Plastic Surgery was tasked with overseeing the prevention and management of adverse reactions during the collection process. The wound size and the patient's body mass index (BMI) was used to determine the quantity of PRP collected for 3–6 applications, typically about 30 ml per use. Following collection, the PRP was processed, cleansed, and divided into 30 ml units by the transfusion department before being stored under refrigeration. Before activating the PRP, an activator was created by combining thrombin and calcium gluconate injection in a ratio of 1000 UL: 10 ml. Subsequently, an activated PRP gel was prepared by adding 1: 10 drops of the activator to the collected PRP. After approximately 1–2 min, the gel solidified and was ready for application to the wound. Prior to application, a small amount of the precipitated supernatant was injected intermittently at the base of the wound in a punctiform manner using a syringe (Figure 1).

Platelet-rich plasma (PRP) collection and preparation for treatment of infected cavitary wound: (a) PRP collection at bedside by allied health staff; (b) PRP collection and dispensing completed; (c) preparation of items before PRP activation for use; and (d) PRP after activation with thrombin and calcium gluconate injection.
Wound management and surgical procedure
Wound secretions were obtained from all patients upon admission and analysed to assess the presence of infection. Thorough debridement procedures were carried out, including removal of necrotic tissue, and scraping of surface bacterial biofilm and aged granulation.
In the PRP group, the supernatant of the gel was uniformly injected into the base and surrounding deeper layers of the wound, followed by the application of the PRP gel over the wound, particularly in regions with inadequate granulation coverage. Subsequently, a negative pressure foam sponge (Suzhou And Science & Technology Development Corp., Suzhou, China) was applied after a waiting period of approximately 5–10 min, and the wound was sealed with a compatible semi-permeable membrane. Negative pressure was turned off for the first 2 h following surgery, then was switched to a low-pressure (40–60 mmHg) intermittent suction mode until 4 h postoperatively, when it was switched to a normal-pressure (100–120 mmHg) intermittent suction mode (representative Figures 2 and 3). The control group received negative pressure foam sponges immediately after full debridement, with the negative pressure adjustment protocol mirroring that of the PRP group. Partial removal of negative pressure occurred every 3 days for wound assessment, secretion collection, and examination. Based on the wound's status, the decision was made to either perform debridement and apply PRP, or to replace the negative pressure sponge and repeat the aforementioned steps until the wound was filled with new granulation tissue and met the criteria for surgical repair. For patients with exposed tendons or bones, physiological saline drops were used to keep the local wound moist during negative pressure therapy. Subsequently, the wound was closed through direct suturing or skin grafting. Concurrently, systemic administration of sensitive antibiotics was initiated based on the findings of bacterial culture and drug susceptibility testing.
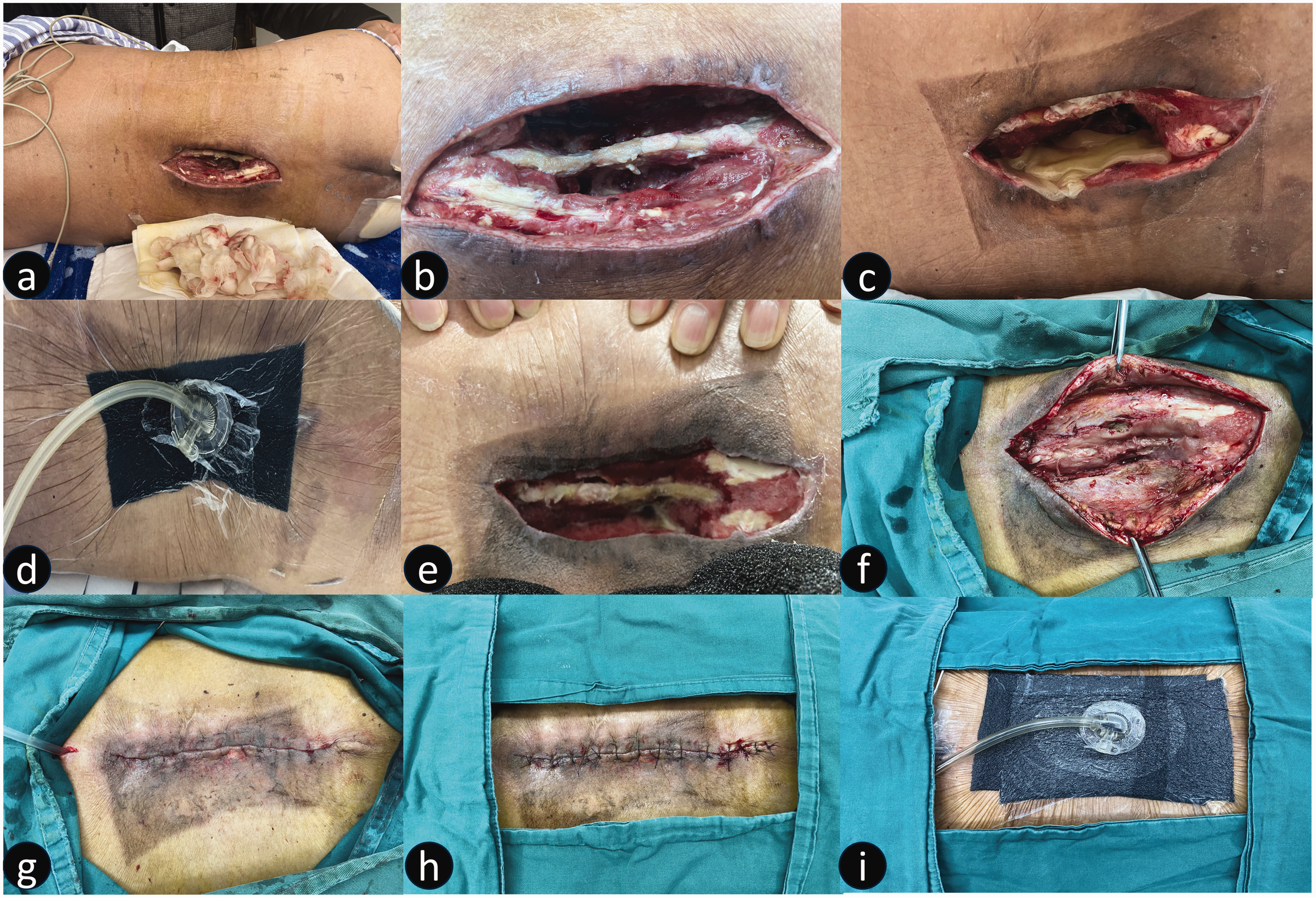
Representative images from a 45-year-old male patient with a burst vertebral fracture from a fall, who had undergone surgery at a local hospital and who’s incision had become infected 1 week later, leading to an abscess cavity, showing: (a) infected incision at 1 month following lumbar spine surgery, at which time the patient was hospitalized; (b) a large cavity with soft tissue defect and exposed spine observed on day 3 of hospital admission, after the first debridement; (c) immediate placement of platelet-rich plasma; (d) installation of negative pressure device; (e) removal of negative pressure device on day 7 of admission; (f) repeat debridement day 11 of admission showing closure of the cavity; and (g–i) closure of the wound with layered reduction sutures and installation of a negative pressure device for adequate drainage and reduction.

Representative images from a 19-year-old male patient with depression and multiple vertebral fractures due to jumping from height, who was found to have a large pus cavity 2 weeks after lumbar surgery, showing: (a) internal fixation device exposed after initial debridement and negative pressure therapy placement; (b–e) placement of platelet-rich plasma (PRP) and negative pressure therapy devices after clearance of obvious infection (negative pressure was not switched on during the early postoperative period; (f–g) significantly reduced cavity, second placement of PRP and negative pressure device; (h and i) further reduction of the cavity, tissue essentially covering the internal fixation, and third placement of PRP and negative pressure devices; (j) the cavity largely filled with granulation prior to the final surgery; (k) the wound closed with layered reduction sutures and placement of subcutaneous drains; and (l) the healed incision at 2 weeks after surgery. This patient was not included in the study analysis due to associated exclusion criteria.
Observation metrics
Partial blood indices, including high-sensitivity C-reactive protein (hs-CRP), erythrocyte sedimentation rate (ESR), white blood cell (WBC) count, and % neutrophils, were utilized upon admission and at day 7 following initial debridement to monitor the patient's infection control; the patient's daily pain level was measured with a visual analogue scale (VAS); bacterial cultures were recorded at admission, and at day 3 and 7 following initial debridement, in addition to the wound bacterial culture conversion rate ([number of existing negative cases – number of original negative cases]/[number of all cases – number of original negative cases] × 100%); the effective duration of hospitalization (days of patient admission from date of first debridement to date of discharge), and the duration of last postoperative hospitalization (days from date of last reconstructive surgery to date of discharge) were recorded.
Statistical analyses
Data were analysed using IBM SPSS software, version 22.0 (IBM Corp., Armonk, NY, USA). Normally distributed data are presented as mean ± SD and were compared using Student’s
Results
A total of 24 patients (14 male and 10 female) with a mean age of 55.78 ± 13.56 years (range, 22–76 years) were included in the study. Among the cases studied, there were eight patients with acute post-traumatic infected wounds, six patients with post-traumatic chronic ulcerative wounds, six patients with post-surgical incision non-healing wounds, and four patients with pressure ulcer wounds. Patients were categorized into either the PRP group (
There were no statistically significant between-group differences in baseline data at admission. Bacterial culture result ratios for the PRP: control group at hospital admission included three cases of
Comparison of baseline information between 24 patients with infected cavitary wound who received either platelet-rich plasma (PRP) in addition to negative pressure wound therapy (PRP group) or negative pressure wound therapy without PRP (control group).

Data presented as mean ± SD or
BMI, body mass index; SSTI, skin and soft tissue infection.
No statistically significant between-group differences were shown in haematological parameters at admission, but they all showed varying decreases after 7 days following initial debridement (Table 2). Postoperative changes in hs-CRP and ESR were significantly different between the PRP and control groups.
Comparison of haematological parameters before and at 7 days after initial debridement, between 24 patients with infected cavitary wound who received either platelet-rich plasma (PRP) in addition to negative pressure wound therapy (PRP group) or negative pressure wound therapy without PRP (control group).

Data presented as mean ± SD.
WBC, white blood cell count; hs-CRP, high-sensitivity C-reactive protein; ESR, erythrocyte sedimentation rate; Pre, preoperative; Post, 7 days after initial debridement.
No statistically significant between-group differences were revealed regarding VAS scores and bacterial distribution at admission (Table 3). The postoperative VAS scores and extent of score reduction was significantly different between the PRP and control groups. The duration necessary for VAS scores to decrease to 5 or below was notably shorter in the PRP group compared with the control group (
Comparison of other results before and at 3 and 7 days after initial debridement, between 24 patients with infected cavitary wound who received either platelet-rich plasma (PRP) in addition to negative pressure wound therapy (PRP group) or negative pressure wound therapy without PRP (control group).
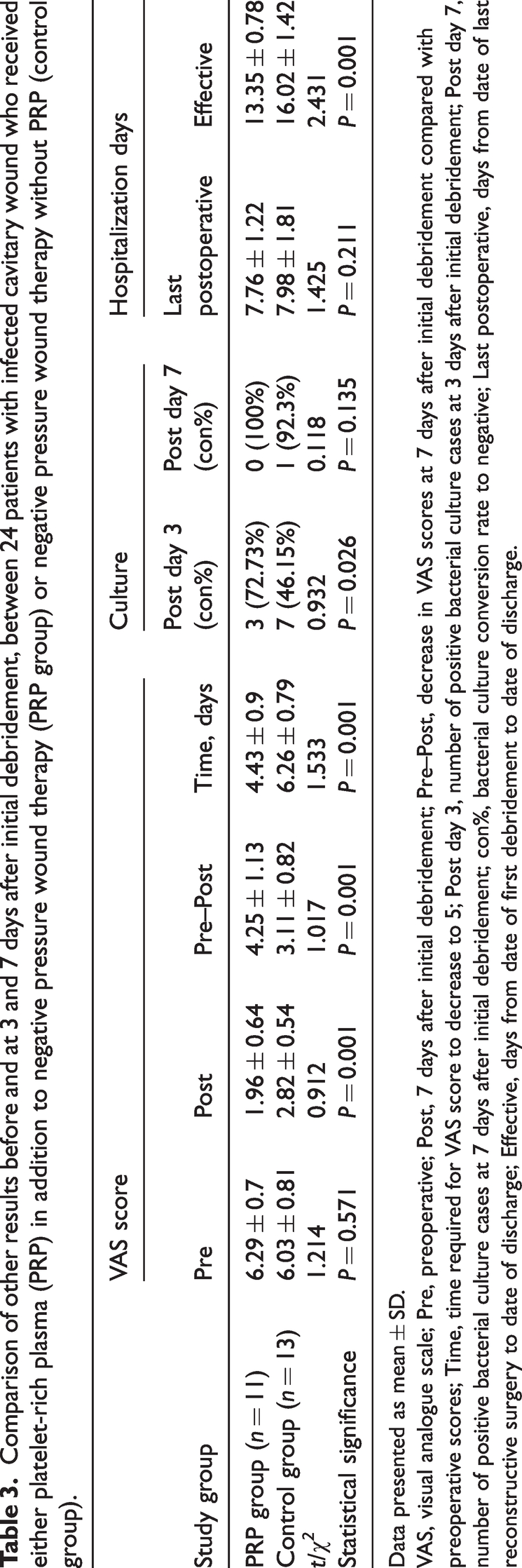
Data presented as mean ± SD.
VAS, visual analogue scale; Pre, preoperative; Post, 7 days after initial debridement; Pre–Post, decrease in VAS scores at 7 days after initial debridement compared with preoperative scores; Time, time required for VAS score to decrease to 5; Post day 3, number of positive bacterial culture cases at 3 days after initial debridement; Post day 7, number of positive bacterial culture cases at 7 days after initial debridement; con%, bacterial culture conversion rate to negative; Last postoperative, days from date of last reconstructive surgery to date of discharge; Effective, days from date of first debridement to date of discharge.
Discussion
Trauma and medically induced injuries are the predominant aetiologies of infected wound development, potentially disrupting the wound healing cascade and resulting in the formation of recalcitrant chronic wounds. Infection plays a pivotal role in this process, particularly in the context of cavitary wounds arising from soft tissue deficiency, which frequently exhibit localized infection. 10 Local treatment should include mechanical debridement in addition to conventional systemic anti-infective therapy. Negative pressure therapy is deemed essential for its ability to enhance fluid balance in wounds, although its efficacy in controlling infection is constrained and occasionally detrimental. 7 Soft tissue padding and flap coverage are frequently employed for the ultimate closure of cavitary wounds.2,3 The combined utilization of PRP and NPWT has the potential to alter the conventional approaches to wound management.
Principle of action and application of platelet-rich plasma
Previous studies have shown the effectiveness of PRP in facilitating tissue repair.4,5 Reduced growth factors in wounds may slow healing, but PRP can release a variety of growth factors, including transforming growth factor-β, basic fibroblast growth factor, and platelet-derived growth factor, as exogenous growth factors to directly replenish the traumatised tissue cells, thereby accelerating fibroblast proliferation and differentiation, endothelial cell migration, stimulating angiogenesis, and stabilizing the structure of neovascularization. In addition, PRP may work with fibronectin to boost tissue inhibitor of metalloproteinase-1 levels and block matrix metalloproteinase-1, aiding in wound healing.11,12 Platelets in PRP can directly act on bacteria by promoting the release of reactive oxygen species and indirectly enhance the antimicrobial effect of leukocytes through the release of peptides and cytokines. Leukocytes in PRP can be activated by platelet factors to release myeloperoxidase and kill bacteria. Furthermore, PRP contains other bacteriostatic components, including human beta-defensin 2, stromal cell-derived factor 1α/CXC motif chemokine 12, and interleukin (IL)-6. However, their exact actions are not fully understood.13–17
Clinical studies show that PRP reduces infection rates in surgical wounds, but it is not recommended for infected wounds.18–20 All cavitary wounds in the present study were infected, and as demonstrated by the results, postoperative infection rates were decreased in patients who received conventional debridement and NPWT alone or with the addition of PRP, and the PRP group displayed significantly lower ESR and hs-CRP levels post-surgery. Bacterial culture results at 3 and 7 days showed effective control of wound infection in both groups, with the PRP group having a higher rate of conversion to negative bacterial culture on day 3. Notably, two patients with drug-resistant bacteria in the PRP group saw their bacterial cultures improve after debridement, NPWT, and PRP treatment. This suggests PRP may have an inhibitory effect on drug-resistant bacteria, though the role of restricted antibiotics is not ruled out.21–23 The bacteriostatic profile of PRP remains uncertain, with varying levels of bacterial inhibition against different strains. Production of PRP is limited by equipment availability and human intervention, and research teams may have conflicting results.24–26 The present study found that PRP may be effective in inhibiting common gram-positive cocci and gram-negative bacilli, as part of a combined treatment protocol. Due to the limited number of cases, it was only speculated that PRP may be effective against multi-resistant bacteria. However, timing of the final surgery was also based on bacterial culture results, and the PRP group had a shorter hospital stay compared with the control group.
Platelet-rich plasma was found to have pain-relieving effects in the present study, with postoperative pain scores decreasing significantly more than the control group. In addition, the PRP group exhibited a shorter time for VAS to fall below 5 points compared with the control group, and this may have been due to the PRP regulating IL-17/IL-1β levels and 5-HT release, which can affect pain signaling.27,28
Platelet-rich plasma forms a gel shortly after activation, which contracts and precipitates liquid components. This process may be related to the contraction after activation of platelet-rich fibrin, which also contains growth factors. Growth factors are released more quickly from PRP when activated by thrombin and calcium, with activators increasing within 2 hours and calcium-dependent factors released continuously for 24 hours.29–30
Based on the above release characteristics of PRP, the present authors have developed an appropriate surgical procedure. First, after the necessary debridement, the PRP gel is prepared followed by a 2-min wait. The supernatant precipitated from the gel is then extracted and injected into the base and surrounding area of the wound in a uniform spot so that the active substance can be released into the deeper layer of the wound; secondly, the moulded PRP gel is laid flat on the wound or cavity, making sure that the gel and the wound are in full contact, so that the active ingredients can play a sustained role in the anti-inflammatory and anti-infective effect in the superficial layer of the wound; finally, the negative pressure drainage device is installed.
Principle of action and application of negative pressure wound therapy
The long history of NPWT and associated research shows it can stimulate tissue growth, remove secretions, reduce oedema, and restore wound fluid balance.6,7 However, there is limited information on the setup methods and modes of negative pressure drainage devices combined with PRP.
In the present authors’ experience, negative pressure devices are placed over the wound and PRP, and sealed with a semi-permeable membrane. The mainframe's negative pressure was kept switched off for 2 hours postoperatively, then switched on in intermittent mode at 40–60 mmHg. After 4 hours, the pressure was adjusted to 100–120 mmHg. Saline drops were administered to some patients with severe infection or exposed tendon and bone the day after to help remove dead tissue and keep the wound moist. This was necessary because the PRP gel did not provide enough moisture as it broke down. The early low negative pressure intermittent drainage pattern reduces the amount of liquid material precipitated from the gel that is drained, minimizing loss while providing adequate mechanical stimulation signals to the wound.
The present negative pressure mode facilitates optimal contact between PRP and the wound during the initial 2-hour period of peak growth factor release. This is especially crucial in the management of cavitary wounds characterized by a large surface area. Tissue volume deficiency and wound coverage represent significant challenges in the reconstruction of cavitary wounds. Conventionally, addressing cavernous wounds necessitates the use of complex flaps or free flaps for filling and coverage, raising questions about the cost-effectiveness of such approaches. Most wound problems can be resolved through the growth of adequate granulation tissue and satisfactory filling of the wound with simple skin grafts or local flaps, unless the wounds are located in functional areas or require reconstruction of certain functions.
The synergistic effect of PRP and NPWT expedites tissue regeneration and wound cleansing, leading to a streamlined closure process involving techniques such as skin grafting or direct suturing. Potential harm to the donor site may be mitigated or disregarded altogether. This innovation has the potential to streamline wound care in primary healthcare settings and alleviate the strain on healthcare professionals by reducing the level of surgical expertise needed.
Limitations
The results of the present study may be limited by several factors. The study only looked at infected cavitary wounds, which may skew the results due to the small sample size. More samples are needed to fully understand the effectiveness of PRP and bacterial inhibition. The efficacy of a single dose of PRP, timing of negative pressure application, and parameter settings are based on clinical experience and further theoretical studies are needed to determine optimal treatment measures. Additionally, cavitary wounds often occur secondary to trauma or pressure ulcers. The baseline conditions of these patients may vary, and clinical data may be influenced by numerous confounding factors, which are issues that need to be considered in subsequent research.
Conclusions
The results of the present study suggest that PRP combined with NPWT may help heal cavitary wounds by reducing inflammation, promoting tissue growth, and creating a conducive environment for healing. The integration of PRP with NPWT may decrease the technical obstacles associated with surgical procedures, reduce healthcare expenses, enhance patient satisfaction, and mitigate harm to donor sites, potentially altering the conventional approaches to wound healing.
Footnotes
Acknowledgments
The authors are grateful to the patients and their families who participated in this study, as well as the staff who were not at the centre. We thank Dr Tao Yuqi of the Blood Transfusion Department of the First Affiliated Hospital of Wannan Medical College for her work in venous blood collection.
Author contributions
Kun Qian and Wen-Xiu Bao participated in study conception and design, the acquisition, analysis and interpretation of data, and writing of this article. Shuai Wang participated in the analysis and interpretation of data, and the writing of this article. Na Zuo and Wei Ding participated in study conception and design, the acquisition, analysis and interpretation of data, and the writing and revising of this article. All authors read and approved the final manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The Authors declare that there are no conflicts of interest.
Funding
This work was supported by the Construction of Standardized Treatment System for Acute Wounds in Southern Anhui Province, China (No. 662202204007).
