Abstract
The most effective treatment for graft infection is still debated, and the success rate of current treatments is low. We herein report the results of surgical treatment and follow-up of a case of infection acquired during carotid stenting with the aim of exploring the most effective treatments for graft infection. We retrospectively analyzed a patient who was admitted in September 2019. This patient underwent debridement, autologous saphenous vein replacement of the common carotid to internal carotid artery, external carotid artery suturing, and continuous negative-pressure wound therapy for carotid stent infection. Ten days after carotid artery revascularization, the growth of granulation tissue in the incision was good, and we decided to suture the neck incision. Five days after removing the stitches, grade A healing was noted. Furthermore, the carotid artery and autologous vein grafts were unobstructed as shown by carotid artery computed tomography angiography reexamination. The patient was monitored for 8 months with no new neurological symptoms and good healing of the incision. Effective treatment of vascular graft infection includes debridement and removal of the infected graft, autologous vein graft revascularization, and negative-pressure wound therapy combined with antibiotic therapy.
Keywords
Introduction
The application of vascular grafts has been revolutionary for the treatment of peripheral vascular and aortic diseases, and grafts have become indispensable in vascular surgery. However, with the extensive application of vascular grafts, graft infections have become a major problem for vascular surgeons. Graft infection is characterized by arterial wall destruction, thrombosis, septic embolism, pseudoaneurysm formation, arterial rupture, and hemorrhage. 1 Carotid stent infection is much rarer. Son et al. 2 reported that only 2 of 77 cases of graft infection were associated with carotid stenting. No standardized treatment has been established for vascular graft infection. Treatment of carotid artery graft infection is very dangerous, and the patient is always at risk of perioperative large-area cerebral infarction. We herein report the successful diagnosis and surgical treatment of a case of carotid artery stent infection at China-Japan Union Hospital Jilin University.
Case report
A 51-year-old man with neck trauma underwent debridement, implantation of a carotid artery covered stent (8 × 60 mm, Fluency Plus; C. R. Bard, Murray Hill, NJ, USA), and thrombectomy of the middle cerebral artery at a local hospital in October 2018 (Figure 1). After emergency admission, three-dimensional angiography of the carotid artery was performed. A dissecting aneurysm was present at the end of the left common carotid artery and the beginning of the left internal carotid artery. The blood flow toward the brain was significantly decreased, but the rate at which it was decreased was unknown. Compression of the left common carotid artery for angiography of the right internal carotid artery and left vertebral artery revealed poor compensation of the anterior and posterior communicating arteries. General anesthesia was administered and debridement was conducted to remove the foreign bodies. A wood splinter was located between the carotid artery and the internal jugular vein. The dissecting aneurysm ruptured and started bleeding during the process of tissue separation. A common carotid artery stent was therefore implanted into the femoral artery. A cerebral protection device was not used. The patient took aspirin (100 mg) and clopidogrel (75 mg) orally after the operation. Thrombosis of the left middle cerebral artery was found in the second intracranial angiography examination and was considered to have been caused by exfoliation of the thrombus during the dissection. Thrombectomy of the middle cerebral artery was therefore performed. The lumen of the middle cerebral artery was unobstructed as shown by angiography. After the above-described operation, debridement of the wound was continued, and the wood splinter was removed. Postoperatively, brain magnetic resonance imaging indicated previous hemorrhage and the formation of a softening lesion in the left basal ganglia. In February 2019, the incision was debrided and re-sutured because of infection (Figure 2). However, the incision did not heal completely after this operation. This was an extremely dangerous situation because of the risk of carotid blowout; the carotid arterial flow was protected only by a thin intimal layer within the stent and was accompanied by infection.

Day of neck trauma. The first debridement, carotid stenting, and thrombectomy of the middle cerebral artery were performed on this day.
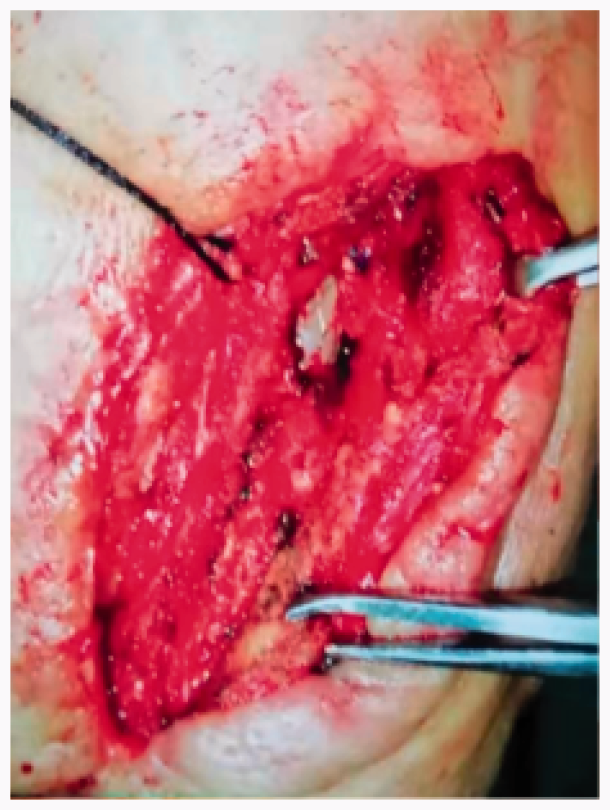
Second debridement. The covered stent was partially exposed.
In September 2019, the patient was admitted to the China-Japan Union Hospital of Jilin University because the incision had not healed completely after 7 months. Specialized physical examinations revealed a sinus tract with pus overflowing on the left side of the neck accompanied by patchy redness and tenderness around the previous incision. Bilateral carotid pulses were palpable, and the right and left upper limb muscle strength was Lovett muscle strength grade IV and V, respectively. Biochemical examination revealed an erythrocyte sedimentation rate of 28 mm/hour and C-reactive protein concentration of 35.4 mg/L. Culture of the secretions revealed methicillin-resistant Staphylococcus aureus (MRSA) infection. The MRSA was sensitive to moxifloxacin and levofloxacin. Through the purulent sinus, we could vaguely see the carotid artery stent and obtained the pus from the deep tissue. We believed that the results of the MRSA culture were clinically significant. Ultrasound examination showed unobstructed blood flow in the carotid stents. Computed tomography angiography (CTA) indicated that the blood circulation in the carotid artery stent was smooth with intimal hyperplasia, the internal carotid artery was unobstructed, and the external carotid artery was completely occluded (Figure 3). The patient was young, had no underlying disease, had experienced a long disease course, and strongly wished for surgical treatment. We decided upon the surgical plan after a discussion with several professors.

Computed tomography angiography findings. The carotid artery stent and internal carotid artery were patent, the external carotid artery was completely occluded, and intimal hyperplasia was evident in the stent.
Moxifloxacin (250 mL: 0.4 g, 1/day) was given intravenously and low-molecular-weight heparin (4100 IU) was given subcutaneously in accordance with the patient’s drug sensitivity results after hospitalization. Moxifloxacin (250 mL: 0.4 g) was intravenously administered 30 minutes before the operation. We monitored the brain activity with intraoperative electroencephalography without assessing the tolerance of the internal carotid artery occlusion before surgery. The supine position was adopted, the shoulders were raised, and the head was tilted back to the uninjured side. The length of the incision along the anterior edge of the sternocleidomastoid muscle and the original incision was about 15 cm. The inflammatory reaction within the incision and the tissue adhesion were severe. The inflammatory reaction around the anchorage area of the stent was also severe. The stent completely covered the opening of the external carotid artery. The proximal end of the common carotid artery was carefully dissected, and blocking tape was added. The internal and external carotid arteries were dissected, although they were difficult to distinguish because of severe inflammation and adhesion. The appropriate length of the saphenous vein was then obtained. After attaining sufficient blood supply to the brain, the distal internal and external carotid arteries were blocked off and the infected stent was removed. The external carotid artery was completely occluded. The internal carotid artery was anastomosed in an end-to-end manner with the saphenous vein. After restoring the blood supply of the internal carotid artery for 5 minutes, the common carotid artery was anastomosed in an end-to-side manner with the great saphenous vein, and the stump of the common carotid artery was sutured (Figure 4). We then intermittently sutured the surrounding tissue and loosely wrapped the transplanted great saphenous vein and the anastomosis at both ends. We did not cover it with muscle. Finally, we covered the wound with a negative-pressure device, which was not in direct contact with the bypass. The operation lasted 2.5 hours, and the intraoperative blood loss was 600 mL. The patient was administered 3 units of type A-positive blood and 200 mL of ordinary frozen plasma. No neurological complications occurred after extubation.

First operation. The infected stent was removed and the carotid artery was reconstructed.
On the day after the operation, the patient was given an intravenous injection of dexamethasone (5 mg), and mannitol (250 mL: 50 g) was given intravenously at a volume of 125 mL every 12 hours for 4 days. Moxifloxacin (250 mL: 0.4g) was given intravenously once a day for 10 days, low-molecular-weight heparin (4100 IU) was given subcutaneously once a day for 10 days, and levofloxacin sodium chloride injection (250 mL: 0.5 g) was used to rinse the negative-pressure suction device once a day. The drainage fluid was not turbid. The granulation tissue grew well after 10 days of carotid artery reconstruction; therefore, we closed the incision (Figure 5). CTA re-examination showed that the autogenous vein was unblocked. After discharge, the incision was followed up for 1 month and showed grade A healing (Figure 6). Telephone follow-up was conducted regularly for 1 month postoperatively. At the time of this writing, the patient was in good condition without obvious neurological symptoms.

Second operation. Good granulation tissue growth was present, and the incision was sutured.

Computed tomography angiography findings. The autogenous vein was unblocked, and the incision achieved grade A healing. The patient was followed up for 1 month after discharge.
The patient provided oral authorization for participation in this case study and publication of the details of his case. We have de-identified all patient details. The requirement for ethics approval was waived because this article does not contain information that can be used to identify the patient and because he agreed to publication.
Discussion
About 80% of the pathogenic microorganisms involved in vascular graft infections are Staphylococcus aureus and coagulase-negative Staphylococcus. The risk factors for vascular graft infection include patient-related factors (periodontal disease, nasal Staphylococcus aureus colonization, postoperative bacteremia and stent characteristics, diabetes or postoperative hyperglycemia, obesity, renal failure, liver disease, malnutrition, and immunosuppression) as well as operation-related factors (intraoperative violation of sterility, a graft in the groin, prolonged operation time, repeated vascular surgery, and lack of perioperative antibiotic administration).3–5 The cause of the stent infection in this case was considered to be that the open neck injury was a contaminated wound and that the stent was partially exposed after two operations (Figure 2).
Vascular graft infection can be divided into Szilagyi grade I, II, and III. Szilagyi grade I infection is limited to the skin, Szilagyi grade II infection is limited to the skin and subcutaneous tissue, and Szilagyi grade III infection involves the graft. 6 These different levels of infection involve different treatments. Generally, the treatments can be divided into antibiotic and surgical treatments. The overall treatment goal is to eradicate the infection and maintain the blood supply. Szilagyi grade I and II infections are easier to control, and conservative treatment can be considered. For Szilagyi grade II infection, extra attention should be invested to assess whether the infection has progressed to grade III. No clear guidelines have been established for antibiotic treatment, and intravenous administration of antibiotics for 4 to 6 weeks is generally considered necessary. Our patient was considered to have Szilagyi grade III infection. Surgical treatment combined with antibiotics was effective. The patient was given intravenous antibiotics for 3 weeks in hospital. Considering that the patient’s infection was eliminated and the autologous saphenous vein had strong antimicrobial protection, no oral antibiotics were given after discharge.
Surgical intervention is required for a clearly diagnosed grade III infection when sepsis, anastomotic rupture, aortoenteric fistula, and MRSA infection occurs. Conservative treatments are less likely to succeed for graft infection. It was previously recommended that anatomic bypass reconstruction be performed in the uninfected area. The disadvantages of this are a low patency rate, high disability rate, and risk of anastomotic rupture. The methods of in situ circulation reconstruction include partial graft removal and reconstruction with cryopreserved homografts, fresh arterial allografts, autologous veins, or antibiotic- or silver-bonded prosthetic grafts.7,8 The reinfection rate of biological materials is currently considered to be low, and autogenous veins are considered the best choice to avoid reinfection. 9
Negative-pressure wound therapy is reportedly an effective auxiliary treatment. Continuous negative-pressure wound therapy can reduce the bacterial content in the wound, accelerate blood flow, and promote the growth of granulation tissue. 10 However, negative-pressure wound therapy is associated with a risk of anastomotic bleeding; thus, low negative pressure can be considered. 11
Patients with limb graft infection are at risk of amputation, whereas patients with carotid artery graft infection are at high risk of cerebral infarction and intracranial infection. This can be disastrous to their health and their family’s wellbeing. If the anchorage zone and the anastomosis of the stent break, massive bleeding within a short period of time can result in a huge hematoma, which may compress the trachea and cause suffocation. In patients with grade I or II infections, the graft can be retained, and local debridement and muscle flap coverage can be used together with antibiotic treatment. If the infection involves the graft (grade III), removing all grafts and reconstructing the arteries may be necessary. The material used to reconstruct the vasculature is usually autologous veins, and the saphenous vein is most frequently used. The infection cure rate is very high, and the risk of reinfection is only 0% to 10%. 4
We achieved a definitive diagnosis of stent infection by combining the results of the culture of the pus secretions, CTA, and ultrasound. The following points were considered. First, the patient had no underlying disease and could tolerate surgery. Second, the patient had undergone two previous neck operations, and the infection was severe because of exposure of the stent to the environment through the sinus tract. Third, long-term stent infections cannot be cured through conservative treatments. Finally, the patient had experienced a long disease course and had a strong desire to eradicate the infection. Therefore, surgical treatment was the best method.
In general, infection of carotid artery stents and vascular grafts is relatively rare. Such infections increase patients’ disability rate and endanger their lives. Perioperative sterility is important to prevent infection. If infection does occur, early identification and treatment can prevent fatal consequences. For Szilagyi grade III infections, removal of the infected graft and autologous venous reconstruction combined with low negative-pressure wound therapy and sensitive antibiotic therapy are effective treatment methods. In addition, individualized treatment plans should be formulated depending upon the grade of the vascular graft infection.
Footnotes
Acknowledgements
We are very grateful to the vascular surgery team of the China-Japan Union Hospital of Jilin University. We also thank Professor Dajun Sun, Director of Vascular Surgery of the China-Japan Union Hospital of Jilin University, and Professor Yue Zhao.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
